Abstract
Introductions:
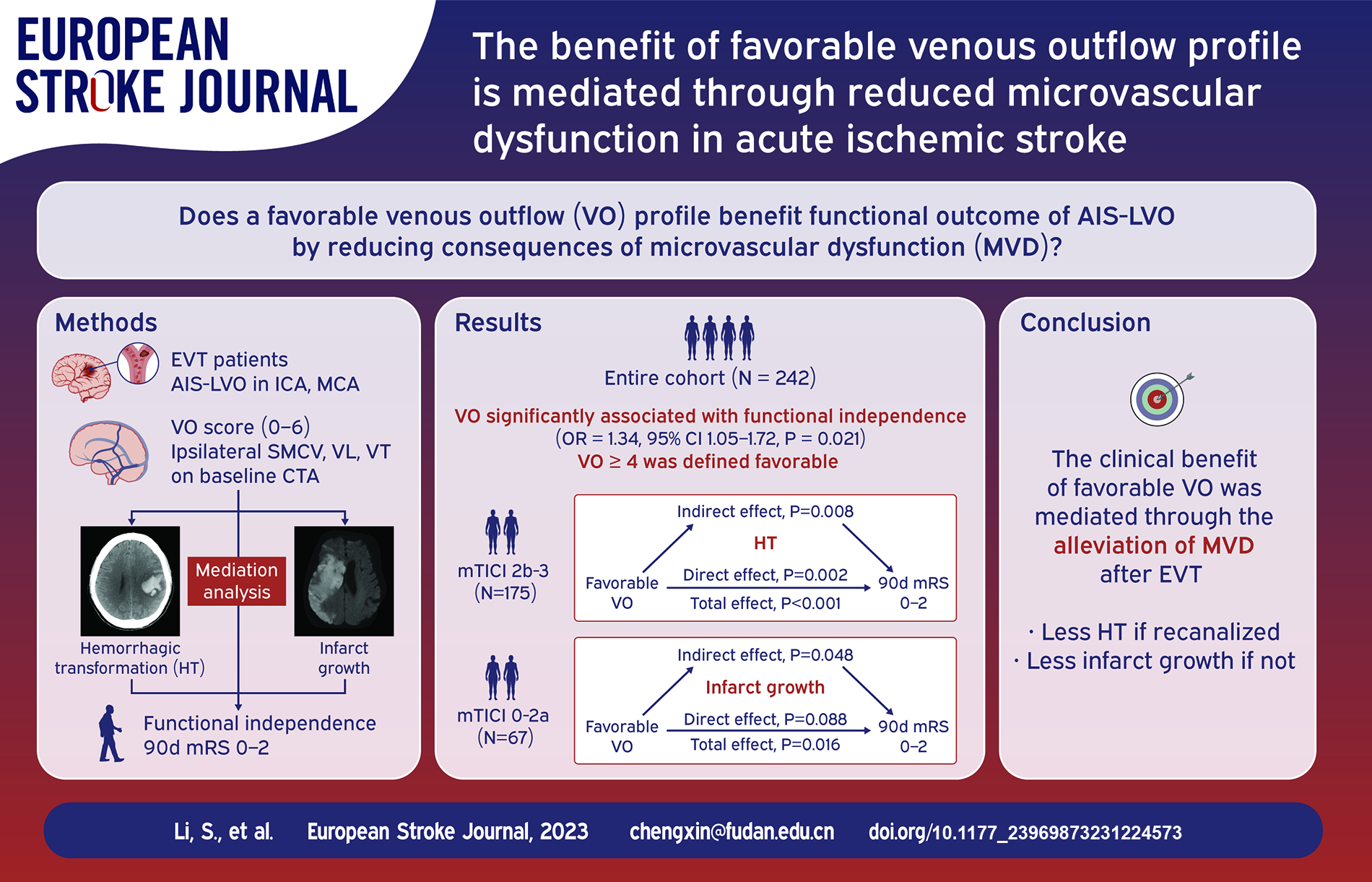
Venous outflow (VO) is emerging as a marker of microvascular integrity in acute ischemic stroke. Using hemorrhagic transformation (HT) and infarct growth as mediators, we tested whether a favorable VO profile benefited functional outcome by reducing consequences of microvascular dysfunction.
Patients and methods:
Patients receiving thrombectomy in three comprehensive stroke centers due to acute anterior circulation occlusion were included. VO was assessed semi-quantitatively by the opacification of ipsilateral vein of Labbé, Trolard and superficial middle cerebral vein. HT was graded on follow-up CT. Infarct growth volume (IGV) was the difference of final infarct volume and baseline core volume. The association of VO and functional independence (90-day modified Rankin Scale ⩽ 2) was examined by logistic regression. Mediation analysis was performed among VO, HT or IGV, and functional outcome in patients with or without recanalization, respectively.
Results:
In 242 patients analyzed, VO was strongly correlated with functional independence and VO ⩾ 4 was defined favorable. In 175 patients recanalized, favorable VO was associated with a reduced risk of HT (OR = 0.82, 95% CI 0.71–0.95, p = 0.008), which accounted for 13.1% of the association between VO and favorable outcome. In 67 patients without recanalization, favorable VO was associated with decreased IGV (β = −0.07, 95% CI −0.11 to −0.02, p = 0.007). The association of favorable VO and functional independence was no longer significant (aOR = 4.84, 95% CI 0.87–38.87, p = 0.089) after including IGV in the model, suggesting a complete mediation.
Discussion and Conclusion:
In patients with acute anterior large vessel occlusion, the clinical benefit of VO may be mediated through reduced microvascular dysfunction.
Introduction
Recent investigations have shed light on the role of venous outflow (VO) in the newly-proposed cerebral collateral cascade (CCC) in patients with acute ischemic stroke due to large vessel occlusion (AIS-LVO).1–4 As it locates downstream to the microcirculation and represents the most distal component of the CCC, VO evaluates the egress of collateral flow through the draining veins and governs local perfusion from the downstream.5,6 Therefore, VO has been regarded as a surrogate marker of the integrity of microvasculature after ischemia.
Microvascular dysfunction (MVD) after ischemia reflects the multifaceted pathophysiology of perfusion and reperfusion deficits, including microvascular obstruction, microvascular injury and inflammation. 7 MVD may manifest itself in catastrophic complications during ischemic stroke. For example, hemorrhagic transformation (HT) shows consequence of severe microvascular injury after reperfusion while infarct progression denotes the result of persistent hypoperfusion in the microcirculation without reperfusion. To date, whether favorable VO benefits the overall outcome of AIS-LVO patients through the alleviation of MVD remains unclear.
To address this issue, we proposed two mediation relationships in the current study. We hypothesized that a favorable VO benefited through a reduced risk of HT in patients with successful recanalization. While in patients with persistent occlusion, a favorable VO benefited through a reduction of infarct growth.
Patients and methods
Study design, patient selection and outcome measurement
This was a multicenter retrospective cohort study of patients undergoing EVT triage in three comprehensive stroke centers from March 2015 to December 2022. The study was approved by institutional medical ethics committee.
Patients were included if they (1) presented within 24 h of last known well; (2) underwent multimodal CT scanning (including non-contrast CT[NCCT], CT angiography [CTA] and CT perfusion [CTP]) upon arriving at the emergency room; (3) presented with an occlusion in the extracranial or intracranial internal carotid artery (ICA), M1 or M2 segment of the middle cerebral artery (MCA); 3) underwent emergent EVT; (4) had interpretable digital subtraction angiography images for recanalization assessment. Exclusion criteria were (1) occlusion of the posterior circulation or bilateral infarction; (2) inadequate CTA imaging timing for evaluation of target veins, incomplete opacification of the dural venous sinuses, or poor quality of CTA source images due to motion artifact; (3) significant hemispheric dominance of the transverse sinus; (4) missing data on modified Rankin Scale (mRS) score at 90 days.
The 90-day mRS was assessed via telephonic interview by trained neurologists who were blinded to the clinical data. Functional independence was defined as mRS 0–2 at 90 days.
Imaging analysis
Baseline CT imaging was performed using 64- or 256-detector scanners. Detailed imaging acquisition protocols were listed in the Supplemental Table 1. Imaging interpretation of baseline CTA and follow-up NCCT was independently performed by two neurologists (XL and WY, with 6 and 8 years of experience respectively) blinded to clinical outcomes. Discrepancies were settled by a consensus read.
Venous outflow was assessed on single-phase CTA source images by grading the contrast opacification of the vein of Labbé (VL), the vein of Trolard (VT) and the superficial middle cerebral vein (SMCV) as follows: 0 = not visible, 1 = moderate opacification, and 2 = full opacification. Each vein was graded separately and the sum of grades in the affected hemisphere defined VO score, which ranged from 0 to 6.
Pial arterial collaterals were evaluated on single-phase CTA maximum intensity projection images using modified Tan (mTan) scale that classified the pial collaterals as “good” if visible in at least 50% of the MCA territory and otherwise “poor.” 8
CTP source images were automatically post-processed with commercial software MIStar (Apollo Medical Imaging Technology, Melbourne, Australia) to generate cerebral blood flow (CBF) and delay time (DT) maps. Hypoperfusion was defined as the volume of tissue with a delay time of ⩾ 3 s [DT ⩾ 3]. 9 The ischemic core was defined as the volume of tissue with a 70% or more reduction in CBF relative to the contralateral hemisphere. Hypoperfusion intensity ratio (HIR) was the ratio of brain volume with delay time of ⩾ 6 s [DT ⩾ 6] over the volume with delay time of ⩾ 2 s [DT ⩾ 2]. 10
Angiographic recanalization status was assessed using modified Thrombolysis in Cerebral Infarction (mTICI) score on final run of angiography. mTICI grading was performed by the interventionist during EVT procedure and double-checked by an experienced neurologist (YL with 10 years of experience) blinded to clinical outcomes. A mTICI score of 2b-3 was defined successful recanalization and mTICI 2c-3 was defined excellent recanalization. 11
Follow-up NCCT was obtained within 48 h post-thrombectomy, accompanied by the evaluation of follow-up NIHSS. HT in our analysis referred to any reperfusion hemorrhagic transformation, asymptomatic or symptomatic. HT was classified and symptomatic intracranial hemorrhage (sICH) was defined according to European Cooperative Acute Stroke Study-II (ECASS-II) criteria. 12
Final infarct volume (FIV) was calculated by semi-automatic segmentation on follow-up diffusion-weighted MRI (b = 1000) acquired at 3–5 days after thrombectomy or on NCCT images acquired after 48 h post-thrombectomy using MIStar. Infarct growth volume (IGV) was defined as the difference between FIV and baseline ischemic core volume.
Statistical analysis
Shapiro-Wilk tests were used to test for normality. Continuous variables were described as median and interquartile range (IQR) if skewed. Categorical variables were reported as counts and percentages. Demographic, baseline clinical and imaging characteristics were compared between patients with or without functional independence using Mann-Whitney U-test for continuous variables and chi-squared test or Fisher’s exact test for categorical variables. Multivariable logistic regression and backward stepwise regression were performed to identify independent factors of 90-day mRS 0–2. Variables with p < 0.05 in univariate analyses or with clinical relevance (i.e. mTan, HIR, site of occlusion and HT) were included in the stepwise regression model. Akaike information criterion was used to optimize model fit versus model complexity. To determine the optimal cutoff value of VO score for outcome prediction, a receiver operating characteristic curve (ROC) analysis was performed and the VO score was dichotomized at the point which maximized the Youden index.
For VO assessment, agreement beyond chance was estimated by using quadratic weighted κ statistics on the full range VO in a random subset of 60 baseline CTA scans. Agreement for dichotomous cutoff values was estimated by using unweighted κ statistics.
For patients with successful recanalization, we hypothesized a mediation where the effect of VO on functional outcome was mediated through a change of HT risk based on the approach described by Baron and Kenny. 13 The association between VO and 90-day mRS (known as the total effect), the association between VO and HT, the association between HT and 90-day mRS adjusted for VO (known as the direct effect) were tested using logistic regression models. Mediation was considered present when all associations were significant. The indirect effect was calculated by the product of the effect size of VO on HT and the effect size of HT on functional outcome. Sensitivity analysis was performed (1) adjusting for age, baseline glucose level, NIHSS and baseline core volume in each model; (2) in a subset of patients with excellent recanalization.
For patients without successful recanalization, an alternative mediation where the effect of VO on functional outcome was mediated through a reduction of infarct growth was tested. IGV was log-transformed before entering regression models. The association between VO and IGV was tested by linear regression. The association between VO and 90-day mRS unadjusted and adjusted for IGV were tested using logistic regression models. Sensitivity analysis was performed adjusting for age, NIHSS and arterial collaterals measured by mTan in each step.
Statistical analyses were performed with IBM SPSS Statistics, version 27 (Armonk, NY) and R studio (version 2022.07.2 Build 576) based on R 4.2.2. Mediation analysis was performed using R package Mediation for estimation of the total effect, the average mediation effect and the average direct effect. Number of simulations was 1000 and nonparametric bootstrapping was used to test the model. A two-tailed p < 0.05 was considered significant.
Results
Of the 463 patients who underwent EVT in our multicenter stroke database, 242 patients met the inclusion criteria (Supplemental Figure 1). One hundred forty-nine (61.6%) patients were male and the median [IQR] age was 70 [61, 78] years. On admission, the median [IQR] NIHSS was 14 [10, 18]. One hundred seventy-five patients (72.3%) achieved successful recanalization after EVT. Among multimodal imaging parameters, robust pial arterial collaterals were found in 171 (70.7%) patients. The median [IQR] VO score at presentation was 3 [2, 4] points. Detailed baseline demographic, clinical and imaging characteristics of all patients are listed in Table 1.
Baseline characteristics in the study cohort and stratified by functional outcome.
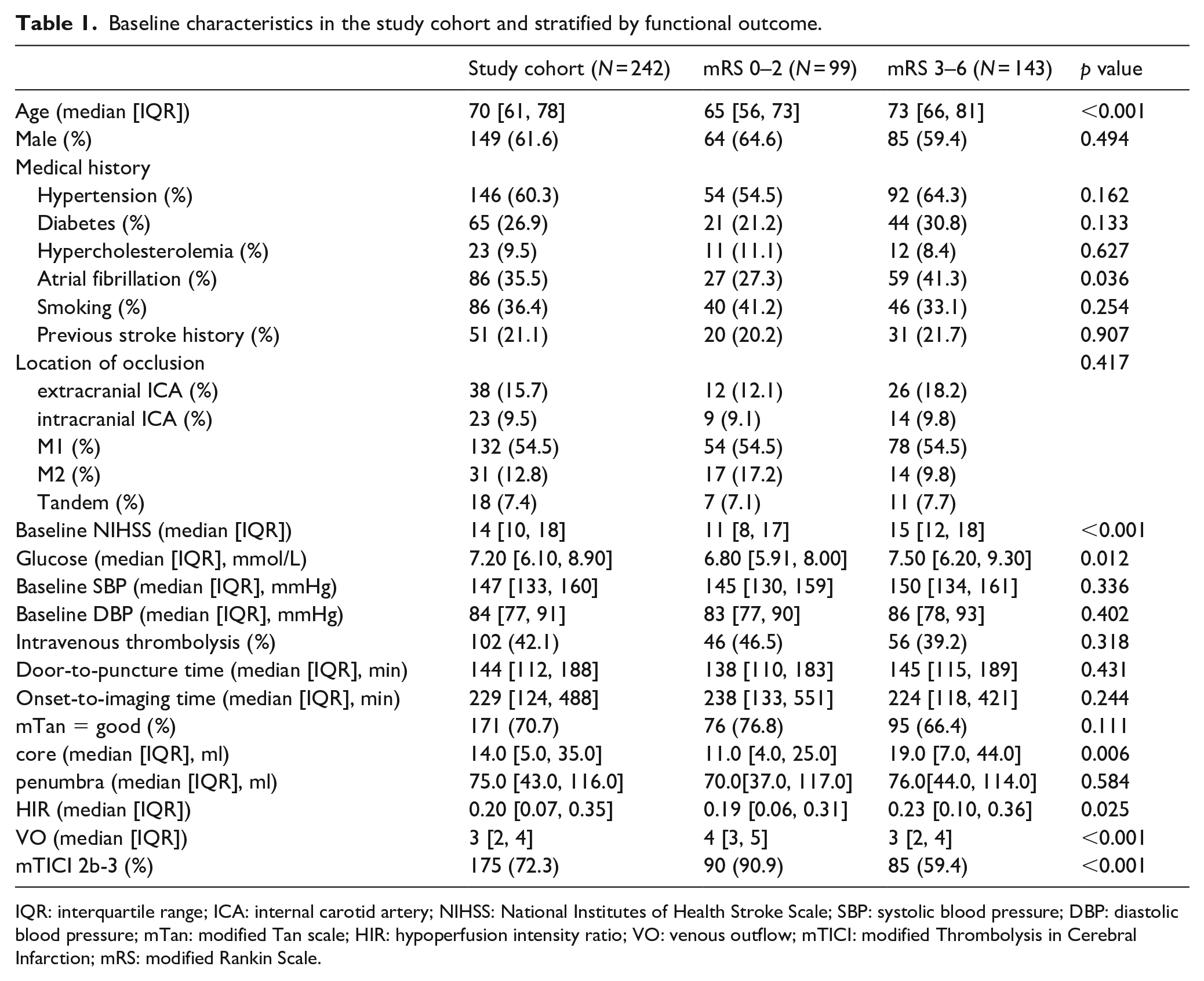
IQR: interquartile range; ICA: internal carotid artery; NIHSS: National Institutes of Health Stroke Scale; SBP: systolic blood pressure; DBP: diastolic blood pressure; mTan: modified Tan scale; HIR: hypoperfusion intensity ratio; VO: venous outflow; mTICI: modified Thrombolysis in Cerebral Infarction; mRS: modified Rankin Scale.
The association between venous outflow and functional outcome
First, we examined whether an independent association existed between baseline VO and functional outcome at 90 days. Patients who achieved a better outcome were significantly younger with less comorbidity of atrial fibrillation, a lower NIHSS and a lower serum glucose level. They demonstrated a significantly smaller core and a lower HIR on admission CTP. Arterial collaterals were similar in the two groups while VO was better in those with a favorable outcome (Table 1). Higher VO score was an independent factor of functional independence (OR = 1.34, 95% CI 1.05–1.72, p = 0.021) after multivariable stepwise logistic regression analysis adjusted for age, baseline serum glucose level, baseline NIHSS, recanalization and presence of any HT (Table 2). The optimal cutoff value of VO score was 3.5 (Figure 1) with a Youden index of 0.37. Therefore, we defined a VO score of ⩾ 4 as favorable VO, which predicted functional independence with an AUC of 0.69 (95% CI 0.63–0.75). Examples of a favorable and poor VO profile were provided in Supplemental Figure 2.
Model parameters for independent factors of 90-day functional independence.
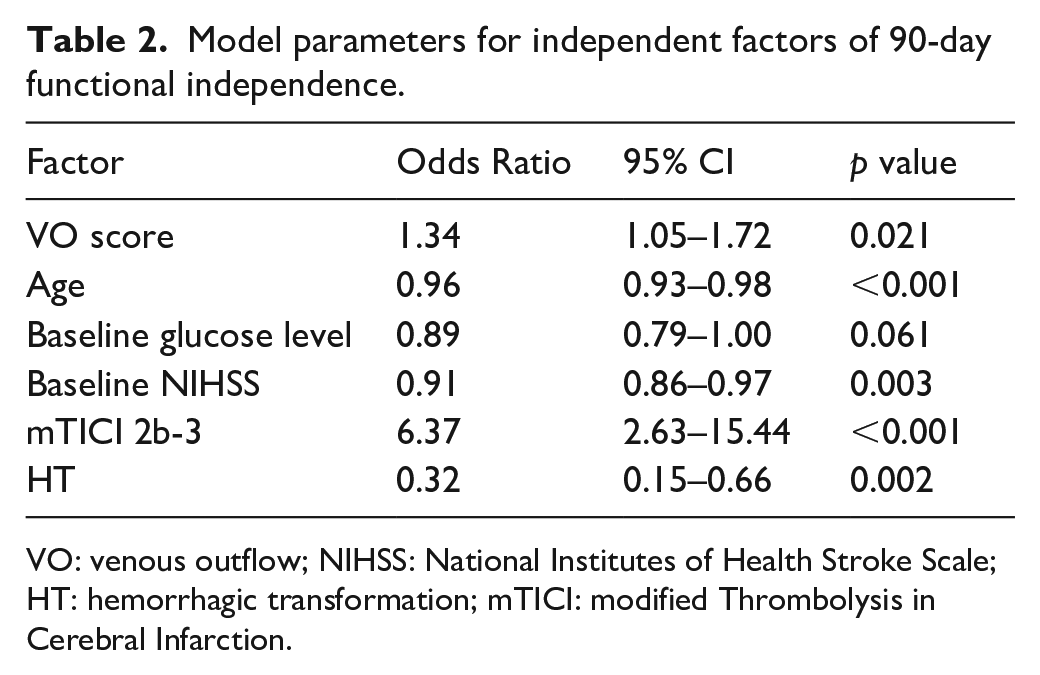
VO: venous outflow; NIHSS: National Institutes of Health Stroke Scale; HT: hemorrhagic transformation; mTICI: modified Thrombolysis in Cerebral Infarction.
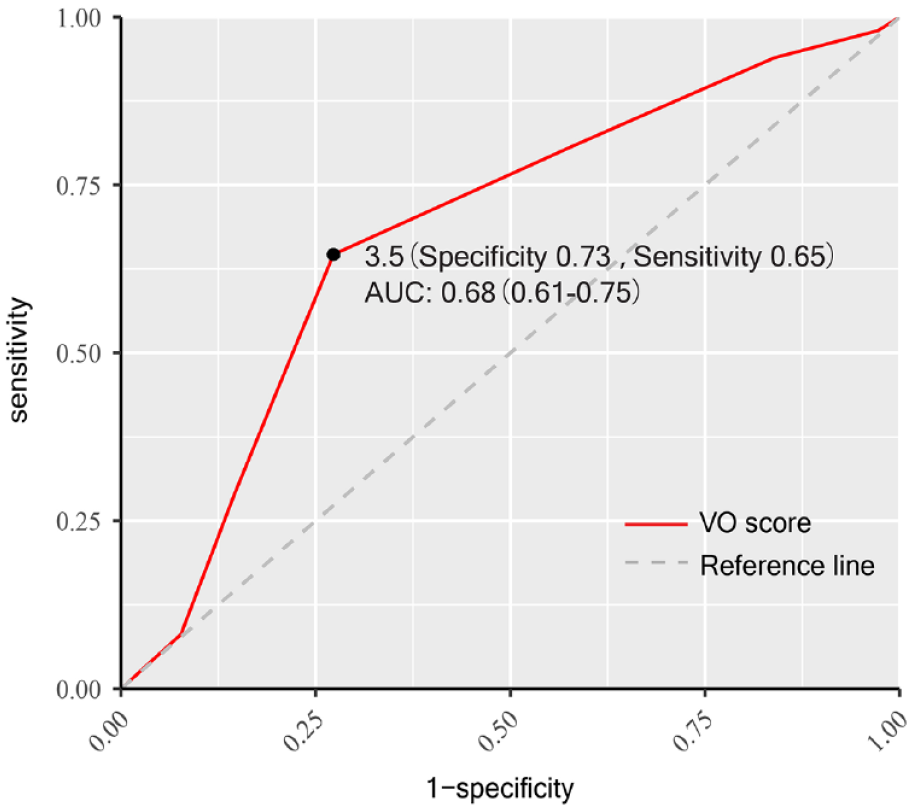
Receiver operating characteristic (ROC) analysis of venous outflow score in predicting 90-day functional independence. mRS: modified Rankin Scale; AUC: area under the curve; VO: venous outflow.
Inter-rater agreement for the full VO score was substantial, with a quadratic weighted κ value of 0.83 (95% CI 0.73–0.92, p < 0.001). Agreement was moderate when VO was graded ⩾ 4 versus < 4, with an unweighted κ value of 0.64 (95% CI 0.44–0.84, p < 0.001). The two readers agreed in 50 of 60 cases.
Mediation of venous outflow on functional outcome by hemorrhagic transformation in patients successfully recanalized
In 175 patients who achieved successful recanalization, the rate and type of all hemorrhagic complications were provided in Supplemental Table 2. A favorable VO was associated with higher odds of functional independence (OR = 3.74, 95% CI 1.97–7.30, p < 0.001) and a lower risk of HT (OR = 0.82, 95% CI 0.71–0.95, p = 0.008) in unadjusted analyses. The association between favorable VO and functional independence remained significant after including HT in the model (aOR = 3.27, 95% CI 1.68–6.48, p < 0.001, Table 3). The reduction of the regression coefficient of VO indicated a partial mediation effect and the indirect effect accounted for approximately 13.1% of the total effect (Figure 2). In the sensitivity analysis including age, baseline glucose level, NIHSS and baseline core volume as covariates, a favorable baseline VO was independently associated with a better outcome (aOR = 3.36, 95% CI 1.59–7.30, p = 0.002) as well as the mediator HT (aOR = 0.86, 95% CI 0.73–1.00, p = 0.050). The indirect effect could explain 12.2% of the association between venous outflow and functional outcome (Figure 2). Analysis in a subset of patients with excellent recanalization (mTICI 2c-3) yielded similar effect, with HT explaining 8.5% of the association of favorable VO with functional outcome. The mediation among VO, sICH and functional independence was also examined in patients with mTICI 2b-3, which showed a partially mediation as well (Supplemental Figure 3).
Mediation analysis regression models.
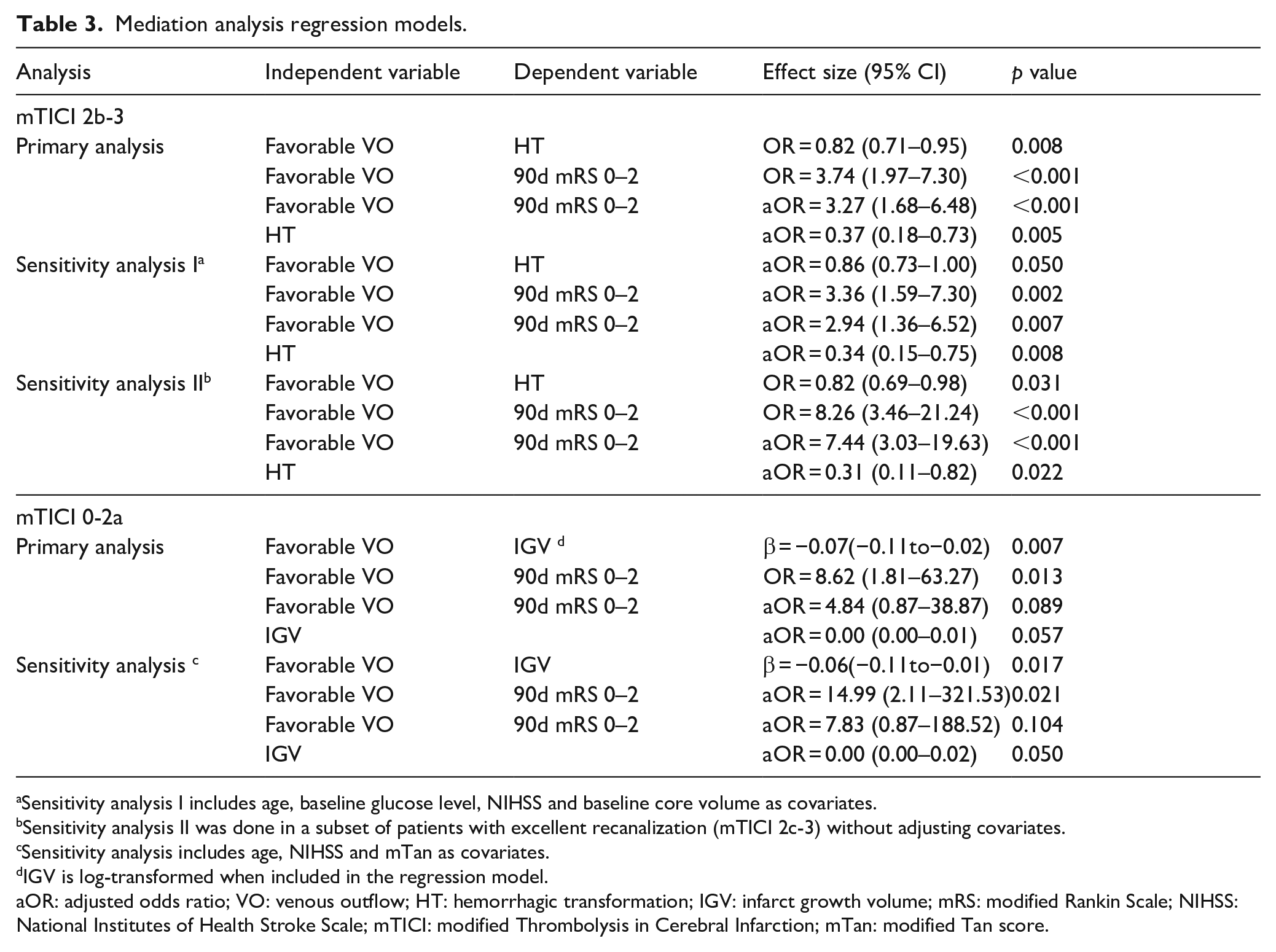
Sensitivity analysis I includes age, baseline glucose level, NIHSS and baseline core volume as covariates.
Sensitivity analysis II was done in a subset of patients with excellent recanalization (mTICI 2c-3) without adjusting covariates.
Sensitivity analysis includes age, NIHSS and mTan as covariates.
IGV is log-transformed when included in the regression model.
aOR: adjusted odds ratio; VO: venous outflow; HT: hemorrhagic transformation; IGV: infarct growth volume; mRS: modified Rankin Scale; NIHSS: National Institutes of Health Stroke Scale; mTICI: modified Thrombolysis in Cerebral Infarction; mTan: modified Tan score.
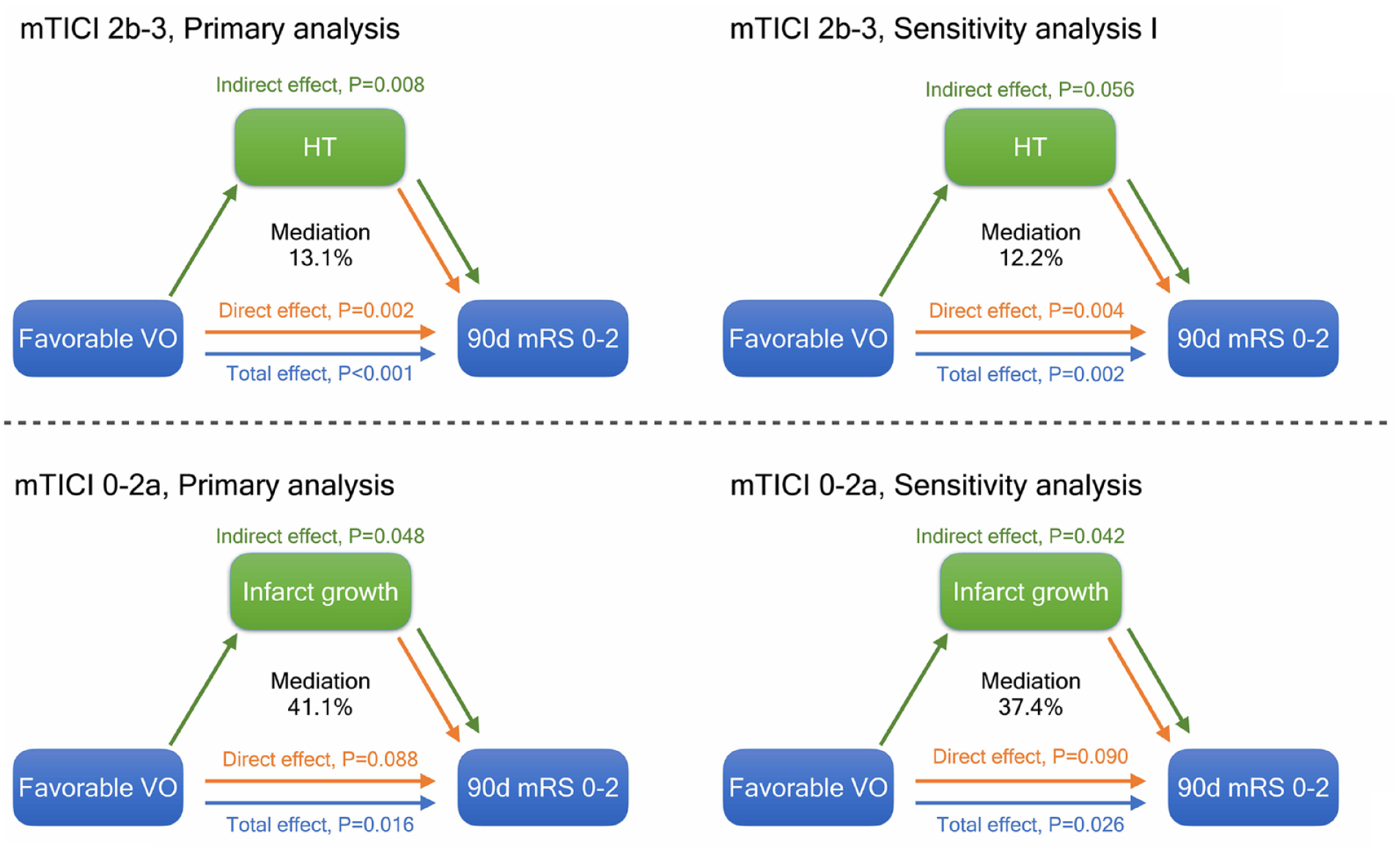
Illustration of the results of mediation analysis. mTICI: modified Thrombolysis in Cerebral Infarction; VO: venous outflow; HT: hemorrhagic transformation; mRS: modified Rankin Scale.
Mediation of venous outflow on functional outcome by infarct growth in patients without recanalization
In 67 patients with failed attempt to recanalize, a favorable VO on admission CTA was associated with increased chance of functional independence (OR = 8.62, 95% CI 1.81–63.27, p = 0.013) and decreased volume of infarct growth (β = −0.07, 95% CI −0.11 to −0.02, p = 0.007). Favorable VO was no longer associated with a better functional outcome (aOR = 4.84, 95% CI 0.87–38.87, p = 0.089) after including log-transformed IGV in the regression model, indicating a complete mediation. Reduced infarct growth explained 41.1% of the association between favorable VO and functional independence (Table 3). The result was consistent in the adjusted model including age, NIHSS and mTan, where the proportion of the association of favorable VO and functional independence explained by reduction in infarct growth was 37.4%, as displayed in Figure 2.
Discussion
This study provided additional evidence for the association between pretreatment venous outflow and clinical outcomes in patients with AIS-LVO. We found that the clinical benefit of favorable VO was mediated by a reduction of infarct growth in patients without successful recanalization and partially mediated by a lower chance of HT in patients successfully recanalized. To our knowledge, it is the first study that investigated in detail how VO profile affected functional outcome stratified by recanalization status.
The association between absent cortical venous opacification and clinical benefit of EVT was first discussed in a post-hoc analysis of the Multicenter Randomized Clinical Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands (MR CLEAN).4,14 Three veins evaluated to represent VO profile of the MCA territory were the SMCV, VL and the sphenoparietal sinus (SPS), composing the cortical vein opacification score (COVES). We chose VL, VT and SMCV because they are the three largest pathways of cortical drainage on the lateral surface and anastomose with one another. While the combination of VL and SMCV being the most common (19.5%) predominant veins depending on neurosurgical findings, codominance of VT and VL, VT and SMCV and all three veins were found in nearly 40% of patients. 15 In this case, the combination of all three veins should cover a more extensive venous drainage from the anterior cerebral circulation, especially the frontal territory draining into the superior sagittal sinus that might be overlooked in COVES. Another important rationale is the drainage pattern of SMCV, which enters predominantly into SPS. 16 The assessment of VT instead of SPS might overcome the potential interrelation between opacification scores in SMCV and SPS. The difference in VO threshold from the aforementioned COVES with respect to functional independence may be explained by assessment of different veins.
Central to our analysis is the use of VO profile as a surrogate marker of the microvascular integrity. From the perspective of the cerebral collateral cascade, VO locates distal to the microcirculation so that the change in VO profile may reflect the robustness of pial arterial collaterals and tissue-level collaterals. Our result supported this idea by showing that VO score was strongly correlated with functional outcome independent of mTan and HIR. In line with previous studies,1–4,17,18 our finding underscores the importance of adequate venous drainage from the hypoperfused tissue as it exhibits the final pathway of the CCC. From a more microscopic point of view, emerging evidence shows that the impairment of VO from the ischemic tissue may reflect microvascular dysfunction after ischemia.6,18,19 It has been observed in ischemic animal models induced by MCA occlusion that platelet-leukocyte-endothelial cell interaction starts from the postcapillary venules. 20 Constrictive lumen narrowing, subsequent neutrophil trapping and increased interstitial pressure further exacerbate the obstruction in the capillary bed.21–23 The combination of these mechanisms possibly limits blood transit through the microcirculation and results in a reduced or even absent filling of the draining veins. To support this speculation, intravenous tPA administration before EVT allows better venous outflow, as it may act not only on the arterial thrombus but also on venule microvascular thrombi. 24 Taken together, a favorable VO profile indicates sufficient microperfusion.
We unraveled two important mediation models explaining the effect of sufficient venous outflow on functional outcome. In patients with angiographic recanalization, we found HT reduction to account for around 13% of the benefit of favorable VO in our model. Despite recanalization, hypoperfusion in the cerebral microcirculation, as indicated by impaired VO, may cause endothelial dysfunction in the capillaries, resulting in the disruption of the blood brain barrier (BBB). In support of this theory, recent studies found a strong negative correlation between regional cerebral blood flow and BBB permeability.25,26 Increased BBB permeability contributes to reperfusion injuries and thus consequents hemorrhagic complications after reperfusion, which is a well-recognized indicator for poor functional outcome.27,28 Therefore, HT is a potential mediator that explains why microvascular dysfunction during ischemia and reperfusion hampered clinical benefit of EVT. Since mTICI = 2b may still cause persistent hypoperfusion within the infarcted area and confound the effect of microvascular failure on functional outcome, sensitivity analysis was conducted in a subgroup of patients with nearly-complete or complete recanalization (mTICI 2c-3) and confirmed a partial mediation.
As indicated in the second mediation model, venous collaterals may grant clinical benefit by reducing infarct growth volume in the absence of reperfusion. It is plausible that persistent occlusion of the major artery leads to constant hypoperfusion in the microcirculation, which causes the ionic pump to fail and induces progressive brain edema. Focal venous compression due to edema may result in cerebral venous steal, 29 which further diverts the collateral flow to the periphery of ischemic region and accelerates the involution of penumbra tissue into ischemic core. Importantly, the finding that VO benefited clinical outcome by preventing lesion expansion was independent of the recruitment of arterial collaterals measured by mTan scale in our study. Arterial collaterals are believed to be determinant in sustaining salvageable brain tissue in early stage of ischemia. However, it may be compromised by increased downstream resistance induced by elevated interstitial pressure after local edema formation. VO may outperform arterial collaterals in determining tissue fate because it measures the blood flow finally exiting from the ischemic lesion, which to a certain degree reflects both the robustness of arterial collaterals and the extent of edema.
There were several limitations to our study. Firstly, the retrospective design and the different veins chosen with established score may introduce bias. The optimal threshold for VO dichotomization needs to be verified in larger prospective cohorts. Secondly, venous drainage of the basal ganglia and striatum into the internal cerebral vein was not examined. 30 Future investigations are required to determine whether the consolidation of deep venous outflow into the current VO score could provide additional information on clinical outcomes of AIS-LVO patients. Thirdly, single-phase CTA was used in this study as previously proposed, 4 which may limit the evaluation of the entire venous phase. The application of multiphase CTA or CTP-derived angiography may better illustrate the intracranial vasculature and diminish the influence of imaging acquisition timing. However, single-phase CTA has been widely available in clinical practice, leading to its applicability. Finally, anatomical variation of cortical veins must also be noted. Cortical veins with relatively limited variation 15 were selected and patients with unilateral predominance of the transverse sinus were excluded to minimize the influence of anatomical variation. Despite possible variations, we have established a scoring system which was highly associated with functional outcome with a reasonable univariate AUC and a satisfactory inter-rater liability.
Conclusion
In conclusion, a favorable venous outflow profile was strongly associated with functional independence in patients with AIS-LVO. Whether they achieved recanalization or not, the benefit of favorable venous outflow was mediated through the alleviation of microvascular dysfunction after treatment, reflected by hemorrhagic transformation if recanalized and less ischemic lesion growth if not. In this regard, VO might be helpful in identifying patients with increased risk of MVD-related complications as a valuable surrogate marker of microperfusion. Future laboratory studies on the dynamics of microcirculation during ischemia and reperfusion are warranted to address the superiority of VO from the perspective of cerebral collateral cascade. Promoting venous outflow might be a promising neuroprotection target.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231224573 – Supplemental material for The benefit of favorable venous outflow profile is mediated through reduced microvascular dysfunction in acute ischemic stroke
Supplemental material, sj-docx-1-eso-10.1177_23969873231224573 for The benefit of favorable venous outflow profile is mediated through reduced microvascular dysfunction in acute ischemic stroke by Siyuan Li, Lan Hong, Wenhao Yang, Xinyu Liu, Yiran Zhang, Yifeng Ling, Zhijiao He, Xinru Wang, Yunhua Yue, Qiang Dong, Feng Wang and Xin Cheng in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873231224573 – Supplemental material for The benefit of favorable venous outflow profile is mediated through reduced microvascular dysfunction in acute ischemic stroke
Supplemental material, sj-docx-2-eso-10.1177_23969873231224573 for The benefit of favorable venous outflow profile is mediated through reduced microvascular dysfunction in acute ischemic stroke by Siyuan Li, Lan Hong, Wenhao Yang, Xinyu Liu, Yiran Zhang, Yifeng Ling, Zhijiao He, Xinru Wang, Yunhua Yue, Qiang Dong, Feng Wang and Xin Cheng in European Stroke Journal
Footnotes
Acknowledgements
None
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by National Natural Science Foundation of China (82271352), Science and Technology Commission of Shanghai Municipality (20Z11900802), and Shanghai Municipal Health Commission (2022XD022).
Ethical approval
Ethical approval for this study was obtained from Huashan Hospital Ethics Committee (KY2022-963).
Informed consent
Informed consent was not sought for the present study because the retrospective nature of the study.
Guarantor
SL and XC.
Contributorship
SL, LH, WY, XL, YZ, YL, ZH, XW and YY participated in data collection. SL analyzed the data, drafted this manuscript, prepared tables and figures. HL, QD, FW and XC critically review and revise the manuscript. XC and QD designed the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
