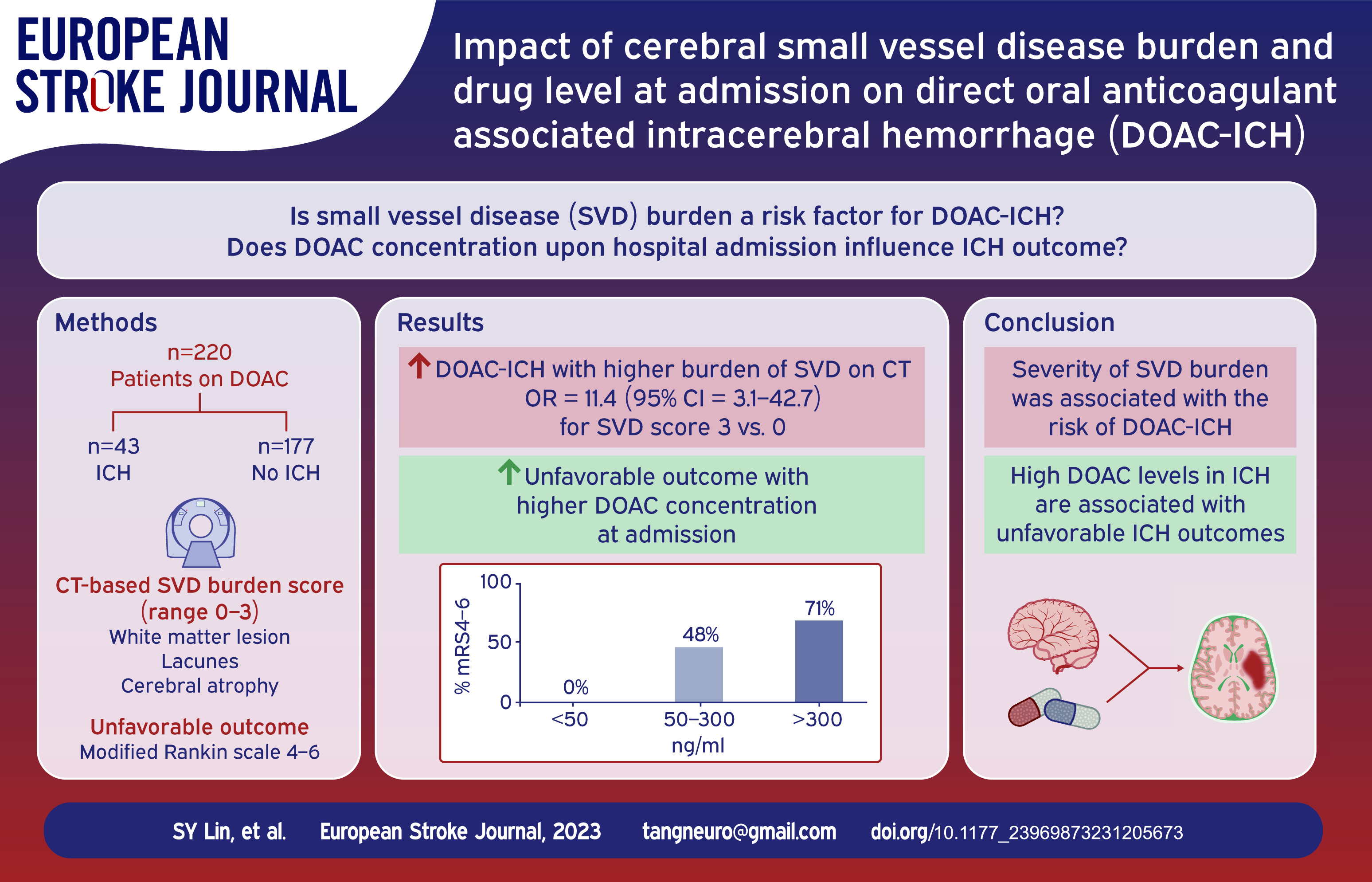
Abstract
Introduction:
Direct oral anticoagulant (DOAC)-associated intracerebral hemorrhage (ICH) is a catastrophic complication. The aim of this study was to investigate the association between computed tomography (CT)-based cerebrovascular small vessel disease (SVD) burden and DOAC-ICH as well as the DOAC concentration upon hospital admission and ICH outcome.
Patients and methods:
The study included two cohorts: (1) DOAC-ICH: patients who suffered from DOAC-ICH and underwent drug level measurements upon admission; (2) DOAC-non-ICH: stable DOAC users who underwent head CT without ICH during treatment. We categorized the DOAC levels of the DOAC-ICH patients as low (<50 ng/mL), medium (50–300 ng/mL), and high (>300 ng/mL). The CT-based SVD burden (including white matter lesions [WML], lacunes, and cerebral atrophy) was evaluated, and SVD scores (range, 0–3) were used to evaluate SVD severity.
Results:
A total of 43 DOAC-ICH patients and 177 DOAC-non-ICH patients were enrolled. DOAC-ICH patients were more likely to have WML, lacunes, or cerebral atrophy compared to DOAC-non-ICH patients. After adjustment, the SVD burden was associated with DOAC-ICH, with a higher risk of more severe SVD (SVD score of 2; odds ratio [OR], 10.3 [3.17, 33.3]; score of 3; OR, 16.8 [4.50, 62.6]). The proportions of patients with high, medium, and low drug levels in the DOAC-ICH group were 16.3%, 55.8%, and 27.9%, respectively. Additionally, the high-level group displayed a larger hematoma size and had worse functional outcomes at 3 months than the other two groups.
Discussion and conclusion:
The severity of SVD burden was associated with DOAC-ICH. Furthermore, high DOAC levels in ICH were associated with unfavorable clinical outcomes. To address the potential selection bias from these two cohorts, a prospective study to investigate the co-contribution of drug levels and SVD to DOAC-ICH is essential.
Introduction
Direct oral anticoagulants (DOAC) are widely used to prevent thromboembolism in patients with atrial fibrillation (AF) and to treat venous thromboembolism (VTE). 1 Compared to vitamin K antagonists, DOAC demonstrates a 50% lower risk of intracerebral hemorrhage (ICH).1,2 Nevertheless, DOAC-associated ICH remains a risk. 3 In addition, prior DOAC therapy, when compared to those not using anticoagulants, is associated with increased in-hospital death, discharge to death, or hospice care after ICH.4,5 Therefore, identifying patients at risk of ICH during DOAC therapy holds significant importance.
The HAS-BLED score is a tool commonly used to evaluate bleeding risk in anticoagulant users. 6 Nevertheless, the HAS-BLED score was developed during the era of vitamin K antagonists. 6 Furthermore, it has limitations in terms of clinical utility, as many risk factors for bleeding are also important risk factors for ischemic stroke or thromboembolism. Additionally, unlike for major bleeding, investigations of the risk factors associated with ICH during anticoagulant therapy have demonstrated that the HAS-BLED score performed poorly in predicting ICH. 7
Cerebral small vessel disease (SVD) is characterized by abnormalities in the small blood vessels of the brain, and ICH is a major cerebrovascular event in patients with SVD.8,9 Previous studies have suggested that the severity of SVD is associated with the occurrence and outcomes of ICH. 9 However, traditional risk scoring tools commonly used for hemorrhage prediction in anticoagulant users, such as the HAS-BLED score, do not consider the SVD burden. 6 Thus, whether the risk of ICH significantly increases in patients with a high SVD burden who are receiving DOAC remains uncertain.
The Clinical Relevance of Microbleeds in Stroke (CROMIS-2) study in the United Kingdom and the Netherlands enrolled patients treated with anticoagulants for secondary stroke prevention. The study found that the severity of SVD burden, as determined by head magnetic resonance imaging (MRI) or computed tomography (CT), could distinguish cases with ICH occurring during anticoagulant therapy.10,11 In 2021, the Microbleeds International Collaborative Network (MICON-ICH) score, developed based on clinical variables, antithrombotic therapy, and cerebral microbleed burden on head MRI, 12 performed better than the HAS-BLED score in identifying patients at risk of intracranial hemorrhage. 12 Nevertheless, while MRI has unique advantages, CT is associated with fewer limitations in examination performance. Furthermore, CT surpasses MRI in terms of utility in healthcare systems where MRI is not available or access to MRI is very limited. Moreover, a previous study reported a strong correlation between CT and MRI in assessing SVD burden. 13
To date, no study has exclusively enrolled DOAC users or discussed the impact of SVD on the development of ICH. The aims of our investigation were as follows: (1) to compare the CT-based SVD burden between patients who experienced DOAC-associated ICH and patients who were on stable DOAC therapy without ICH development in order to identify imaging markers associated with the occurrence of ICH during DOAC therapy; and (2) to explore the association between DOAC levels upon hospital admission and ICH severity or outcomes.
Patients and methods
Study design
This observational study was conducted at the National Taiwan University Hospital (NTUH) between 2016 and 2023. The NTUH International Ethics Committee approved the study protocol. All participants provided informed consent before enrolment in the study.
Participants
The study participants were required to be aged 20 years or older at study recruitment. This investigation consisted of two study groups from two cohorts within the NTUH.
(1) DOAC-ICH (DOAC-associated ICH): This group comprised patients from the DOAC-ED cohort who experienced intracranial hemorrhage. The DOAC-ED cohort prospectively enrolled patients who presented to the emergency department (ED) due to acute stroke, including ischemic stroke (IS), transient ischemic attack (TIA), or intracranial hemorrhage. Head CT data and DOAC levels were collected upon hospital admission. 3 Patients in the DOAC-ICH group were excluded if the ICH was associated with a tumor, any other structural lesion, or an isolated intraventricular hemorrhage.
(2) DOAC-non-ICH: This group included prospectively enrolled patients from the DOAC cohort who were receiving stable DOAC therapy with a steady-state trough level. 14 Patients were included in the DOAC-non-ICH group if they underwent head CT within 1 year before DOAC treatment initiation or during DOAC therapy. Patients were excluded if they experienced spontaneous ICH before DOAC therapy or if spontaneous ICH occurred during DOAC treatment.
Study process
Blood samples were collected from patients in the DOAC-ICH group at the time of ED presentation. The time from the last DOAC dose was recorded. DOAC concentration was measured by ultra-high-performance liquid chromatography with tandem mass spectrometry (UHPLC-MS/MS). The analytical method has been validated and published previously, and the details are provided in the Supplemental Material. 15 The blood samples were sent in batches for concentration measurements, and the results were not available to the researchers at the time of clinical decision making.
Head CT image interpretation
We used a head CT-based grading system to define the SVD burden for all participants. For patients in the DOAC-ICH group, head CT was performed following acute ICH. For patients in the DOAC-non-ICH group, head CT scans acquired within 1 year of or during DOAC therapy were interpreted. These scans were reviewed and graded by a neuroradiologist (YF Chen) who also determined the SVD scores in our previous investigations.13,16
The neuroimaging features of SVD on CT include the severity of the white matter lesions (WML), presence of lacunes, and cerebral atrophy. WML are characterized by ill-defined hypodensity in the periventricular or subcortical areas of the brain. The severities of the WML in the anterior or posterior sections of the brain were graded separately. A score of 0 indicated no WML, 1 indicated the existence of WML restricted to the periventricular region, and 2 indicated that the WML extended into the subcortical areas. Lacunes were defined as ovoid or round cavities with diameters ranging from 3 to 15 mm and radiolucency similar to cerebrospinal fluid. The presence of lacunes was assigned a score of 1, while their absence was graded as 0 points. Cerebral atrophy was assessed at the central (deep nuclei and lateral ventricle level) and cortical (supraventricular or centrum semiovale) regions, with scores of 0 (no atrophy), 1 (moderate), and 2 (severe atrophy). 17
A previously developed CT-based SVD scoring system, which was utilized in our previous studies,13,16 was used in this study. This ensured an appropriate rating system in the presence of ICH. The above three markers were incorporated into the SVD score: 1 point was assigned for severe WML (if the WML scored a grade of 2 in the anterior or posterior brain regions), severe atrophy (if the atrophy scored a grade of 2 in the central or cortical regions), and any lacunes, resulting in a maximum score of 3.18,19 We also calculated the average anterior/posterior WML and central/cortical atrophy scores to analyze the impact of different SVD parameters on ICH during DOAC therapy. The anterior and posterior WML scores were summed (minimum, 0 points; maximum, 4 points) to analyze the impact of the extent of WML on DOAC-associated ICH.
Cerebral edema can interfere with head CT assessments in patients with acute ICH. For patients presenting with supratentorial ICH, the extent of WML and cerebral atrophy was assessed in the hemisphere opposite to the ICH site. For patients with infratentorial ICH, scoring was based on the hemisphere displaying more pronounced WML or atrophy. This method was consistently applied in our previous study. 13
Data acquisition and clinical outcomes
Demographic characteristics, laboratory tests, and comorbid conditions were retrieved from the electronic medical records of all participants. For patients in the DOAC-ICH group, the Glasgow Coma Scale (GCS), initial hematoma location, size, and ICH management details (including the use of reversal agents, prothrombin complex concentrations, and surgical details) were recorded. They were also prospectively followed up to document hematoma expansion and modified Rankin Scale (mRS) scores or record instances of mortality at 3 months.
Hematoma volume was calculated using the ABC/2 formula. 20 Hematoma expansion was defined as an increase of ⩾6 mL in hematoma volume or ⩾33% on the follow-up image (usually obtained 24–48 h after the initial scans). 21 A good functional outcome was defined as an mRS score of ⩽3. Two neurologists (SC Tang and CH Chen) reviewed the head CT images from the DOAC-ICH group to determine the hematoma volume data and evaluate the hematoma expansions.
Impact of DOAC concentration on ICH presentation and outcomes
We categorized the patients into three groups according to their acute DOAC levels: (1) low, <50 ng/mL; (2) medium: 50–300 ng/mL; and (3) high, >300 ng/mL. A lower cutoff value of 50 ng/mL was chosen as it represents a DOAC level with an active pharmacological effect, a threshold that has been validated in previous investigations.3,22,23 Notably, a widely accepted criterion for defining a high DOAC concentration with random sampling is currently unavailable. Therefore, we opted for 300 ng/mL because it approximated the upper limit of the expected peak range (i.e. 383 ng/mL for dabigatran, 343 ng/mL for rivaroxaban, 321 ng/mL for apixaban, and 288 ng/mL for edoxaban). 1
Statistical analysis
To compare between-group differences, we employed the Student’s t-test or Kruskal–Wallis test for continuous variables and the chi-square test or Fisher’s exact test for categorical variables. Univariate analysis, followed by multivariate logistic regression, was performed to investigate the effect of SVD burden on DOAC-associated ICH. Factors with p < 0.1 in the univariable analyses were identified and included in the multivariable logistic regression analysis. Because age and creatinine clearance (CrCL) are important factors affecting SVD,24,25 these were also incorporated in the multivariable model to assess the impact of the SVD score on DOAC-ICH.
Ordinal logistic regression (shift analysis) was used to investigate the effect of acute DOAC use on the mRS score at 3 months. Statistical significance was set as p < 0.05. All statistical analyses were performed using IBM SPSS Statistics version 26.0. (IBM Corp., Armonk, NY, USA).
Results
Basic characteristics, ICH presentation, and outcomes
In 2016 and 2023, we enrolled 48 DOAC-ICH patients and 186 DOAC-non-ICH patients for whom head CT images were available. After applying the exclusion criteria, we included 43 patients (60.5% men) in the DOAC-ICH group and 177 patients (49.7% men) in the DOAC-non-ICH group. In the DOAC-non-ICH group, the reasons for undergoing head CT imaging were stroke-like neurological symptoms in 135 patients (76.3%), falling episodes in 18 patients (10.2%), and other indications in 24 patients (13.6%), such as health examinations, malignancy surveys, or follow-ups for organic brain lesions. The study enrollment process is shown in Figure 1.
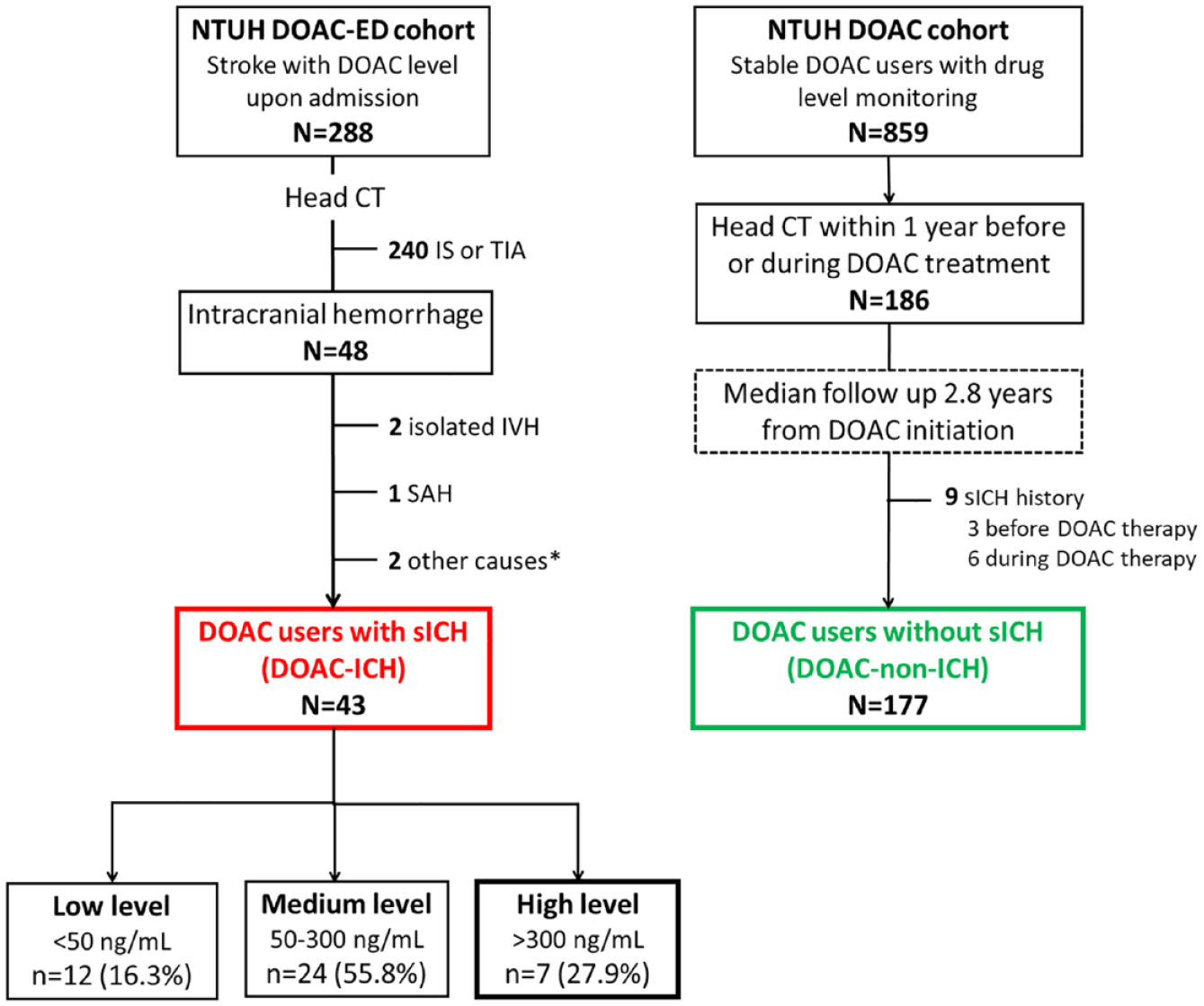
The process of study enrollment.
The demographic characteristics of the two study groups are presented in Table 1. In the DOAC-ICH group, 38 patients (88.4%) used DOAC for AF, 2 used DOAC for deep vein thrombosis, and 3 used DOAC for off-label indications, including the treatment of peripheral arterial occlusive disease (apixaban with an AF regimen; n = 1), prevention of thrombosis due to nephrotic syndrome (n = 1), and management of a mural thrombus in an aortic arch aneurysm (n = 1). All patients in the DOAC-non-ICH group required DOAC for AF. Patients in the DOAC-ICH group were older and had worse renal function than those in the DOAC-non-ICH group. In addition, they were less likely to have AF, diabetes, or a history of IS or TIA; however, they were more likely to have a history of intracranial hemorrhage than those in the DOAC-non-ICH group.
Demographic characteristics across different groups.
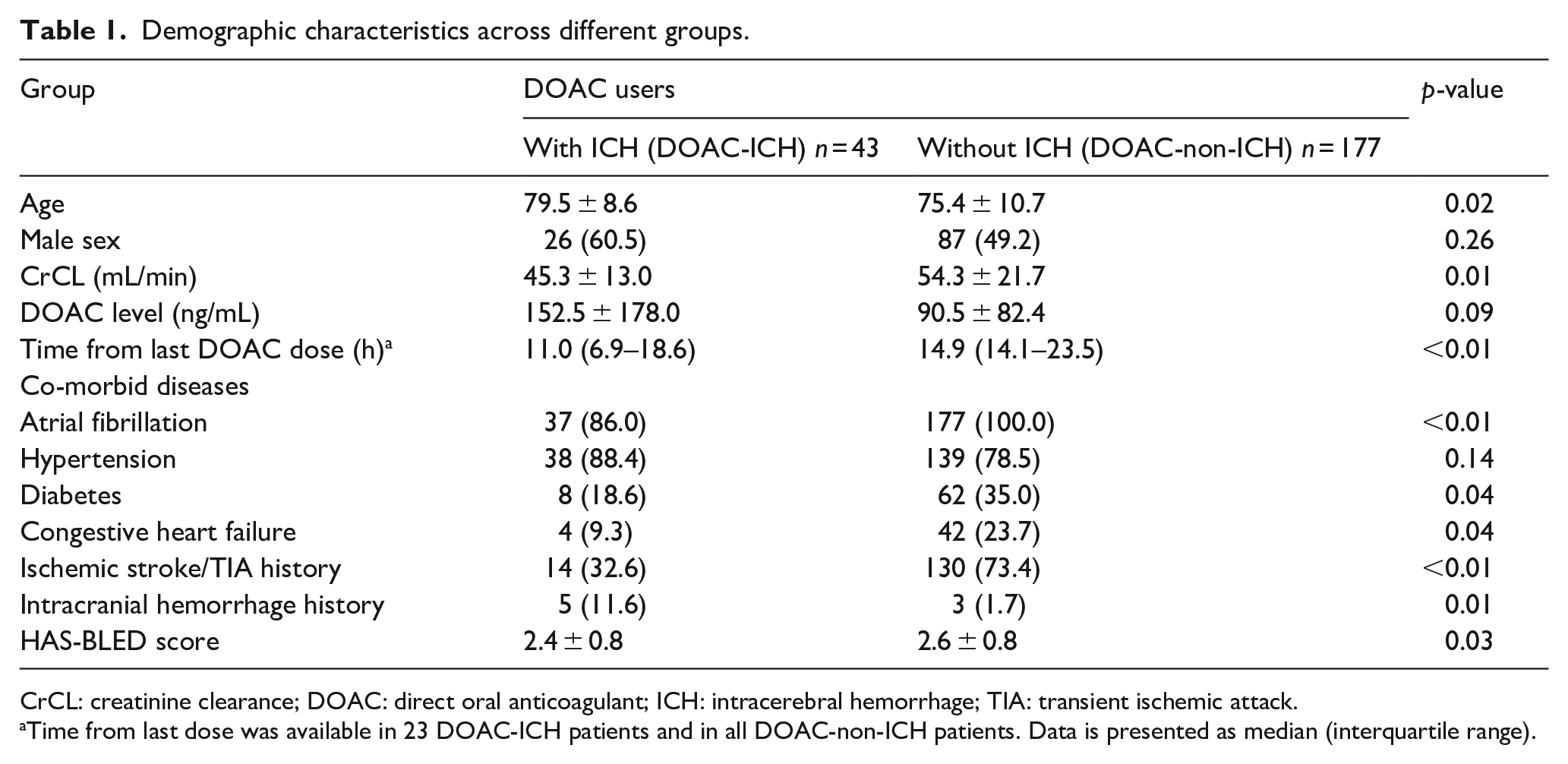
CrCL: creatinine clearance; DOAC: direct oral anticoagulant; ICH: intracerebral hemorrhage; TIA: transient ischemic attack.
Time from last dose was available in 23 DOAC-ICH patients and in all DOAC-non-ICH patients. Data is presented as median (interquartile range).
The average HAS-BLED score was 2.4±0.8 points in the DOAC-ICH group and 2.6±0.8 points in the DOAC-non-ICH group (p = 0.09). Additionally, the DOAC level upon hospital admission was 152.5±178.0 ng/mL for patients in the DOAC-ICH group.
CT-based SVD burden in different study groups
A comparison of the SVD parameters between the two study groups is presented in Figure 2. Patients in the DOAC-ICH group displayed significantly higher SVD scores than those in the DOAC-non-ICH group (1.8 ± 1.0 points vs 0.9 ± 1.0 points, p < 0.01). Regarding different SVD parameters, the proportions of patients with anterior and posterior WML in the DOAC-ICH group were 93.0% and 86.0%, respectively, whereas they were 59.3% and 40.1%, respectively, in the DOAC-non-ICH group (p < 0.01 for both tests). In total, 46.5% of patients in the DOAC-ICH group had lacunes, whereas this percentage was 23.7% in the DOAC-non-ICH (p < 0.01). The proportions of patients with central and cortical atrophy were 95.4% and 100.0%, respectively, in the DOAC-ICH group, and 86.4% and 93.7%, respectively, in the DOAC-non-ICH group (p < 0.01 for both).
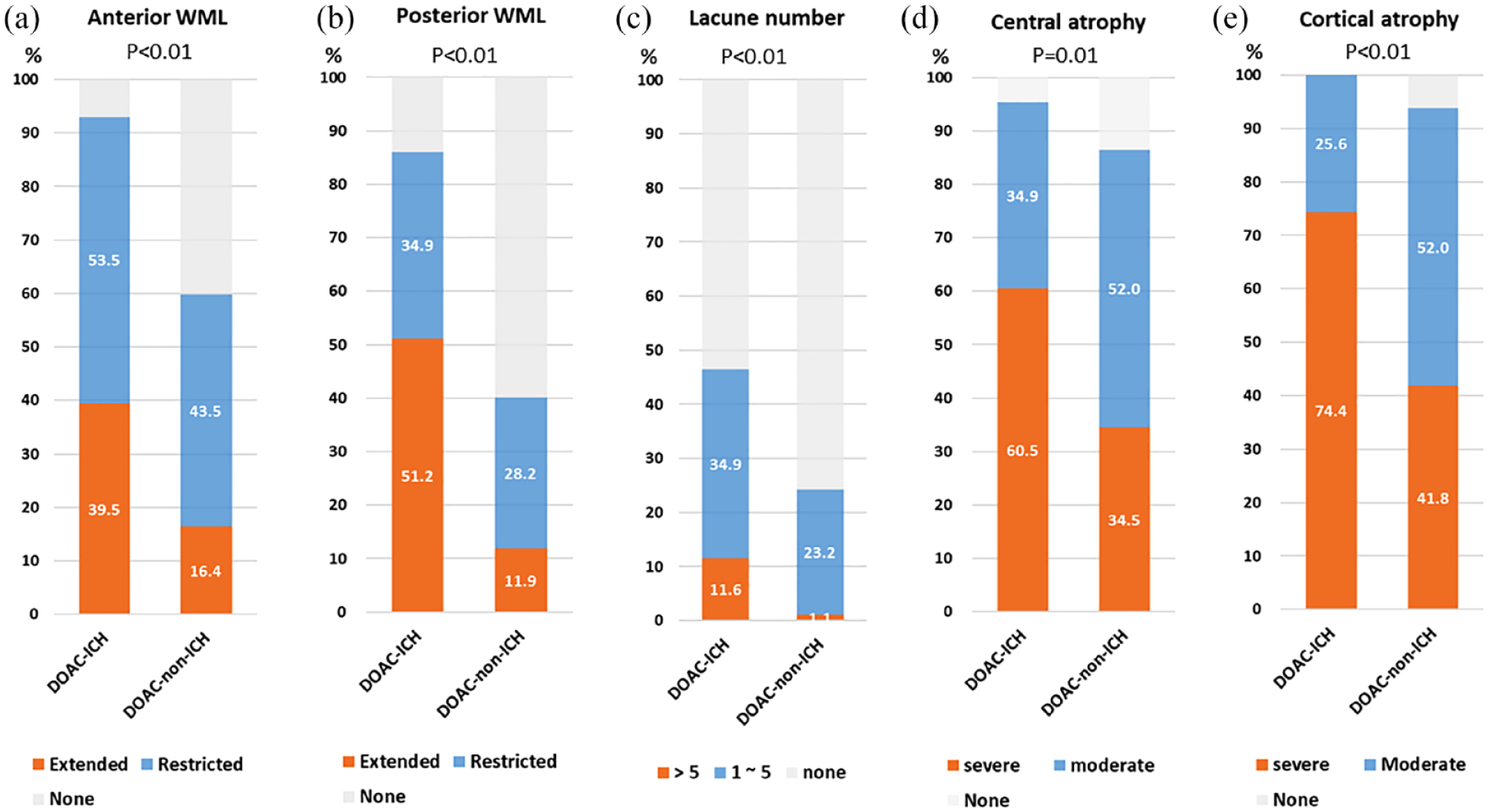
The cerebral small vessel disease burden in different study groups.
Association between CT-based SVD burden and DOAC-associated ICH
The details of the model selection process are presented in Supplemental Table S1. We observed a substantial number of variables that fulfilled the criteria for inclusion in the multivariable analysis. Because of the restricted size of our study population, it was not feasible to incorporate all of them in the multivariable analysis. Consequently, we exclusively incorporated factors with p < 0.01 in the univariable analyses, along with age and CrCL, which are clinically important factors, in the final model. After adjustment, the SVD score was significantly associated with DOAC-ICH, and the risk increased with higher scores (2 points vs 0 points, odds ratio [OR] and 95% confidence interval [CI], 10.3 [3.17, 33.3], p < 0.01; 3 points vs 0 points, OR 16.8 [4.50, 62.6], p < 0.01), as shown in Table 2. We further compared the impact of different SVD parameters and found that WML were independently associated with DOAC-ICH (OR 3.80 [1.40, 10.3], p = 0.01), but lacunes or cerebral atrophy did not show such an association (Table 2). A WML score of 2 or higher was significantly associated with ICH (ORs for scores of 2, 3, and 4 vs 0, 12.1 [2.92, 50.4], 18.1 [3.36, 97.4] and 49.2 [10.2, 236], respectively; Supplemental Table S2).
Impact of small vessel disease burden on intracerebral hemorrhage among direct oral anticoagulant users.
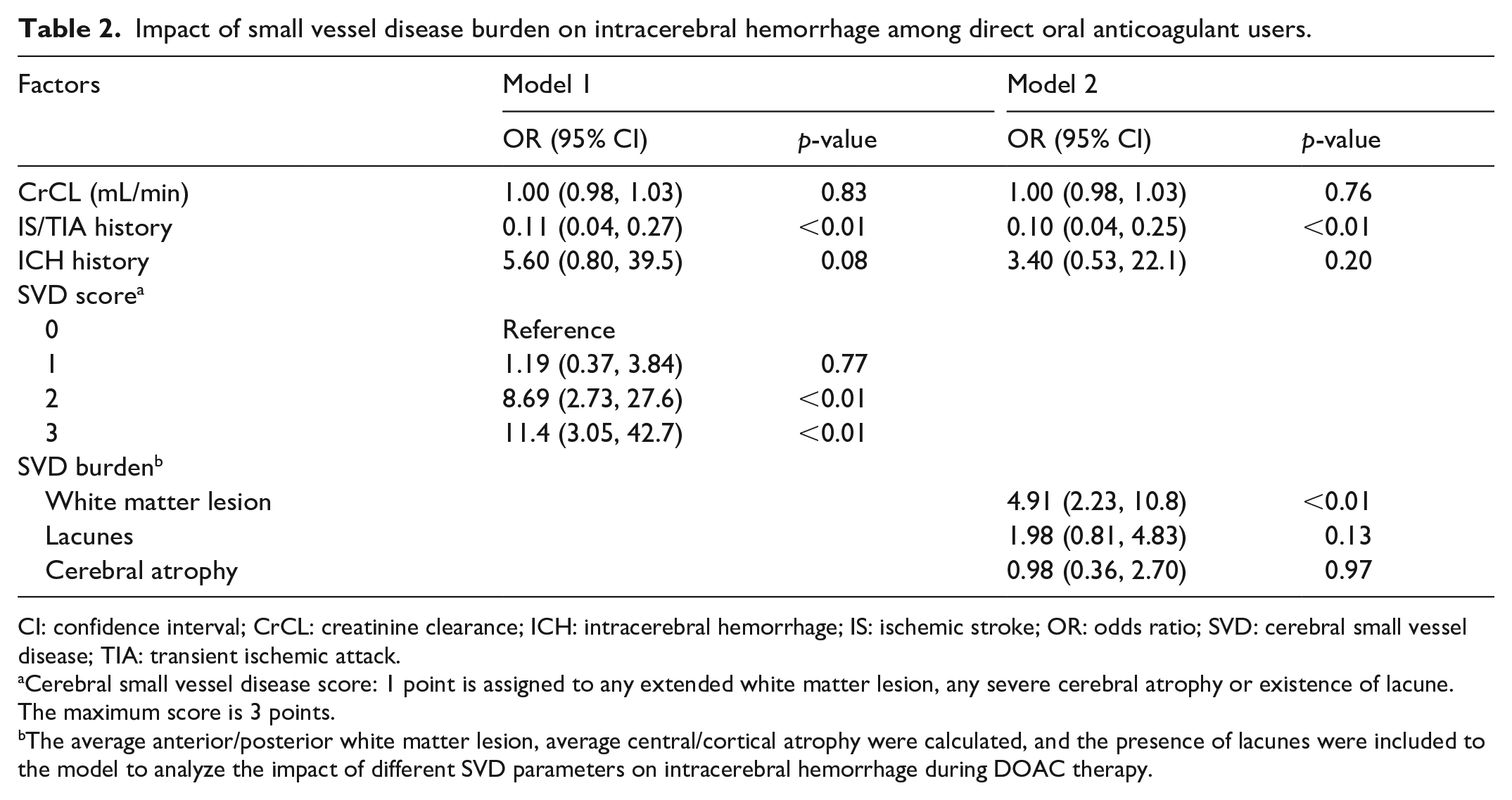
CI: confidence interval; CrCL: creatinine clearance; ICH: intracerebral hemorrhage; IS: ischemic stroke; OR: odds ratio; SVD: cerebral small vessel disease; TIA: transient ischemic attack.
Cerebral small vessel disease score: 1 point is assigned to any extended white matter lesion, any severe cerebral atrophy or existence of lacune. The maximum score is 3 points.
The average anterior/posterior white matter lesion, average central/cortical atrophy were calculated, and the presence of lacunes were included to the model to analyze the impact of different SVD parameters on intracerebral hemorrhage during DOAC therapy.
Effect of DOAC concentration on acute ICH
Among the patients with DOAC-ICH, the distribution of each DOAC before ICH was as follows: dabigatran, 2 patients (4.7%); rivaroxaban, 12 patients (27.9%); apixaban, 19 patients (44.2%); and edoxaban, 10 patients (23.3%). Seven patients (16.3%) had high DOAC levels, 24 (55.8%) had medium DOAC levels, and 12 (27.9 %) patients had low DOAC levels, respectively (Figure 1). A comparison between patients in the different DOAC level groups is shown in Table 3.
Demographic characteristics, small vessel disease burden, and outcome of intracerebral hemorrhage among patients with different acute direct oral anticoagulant level.
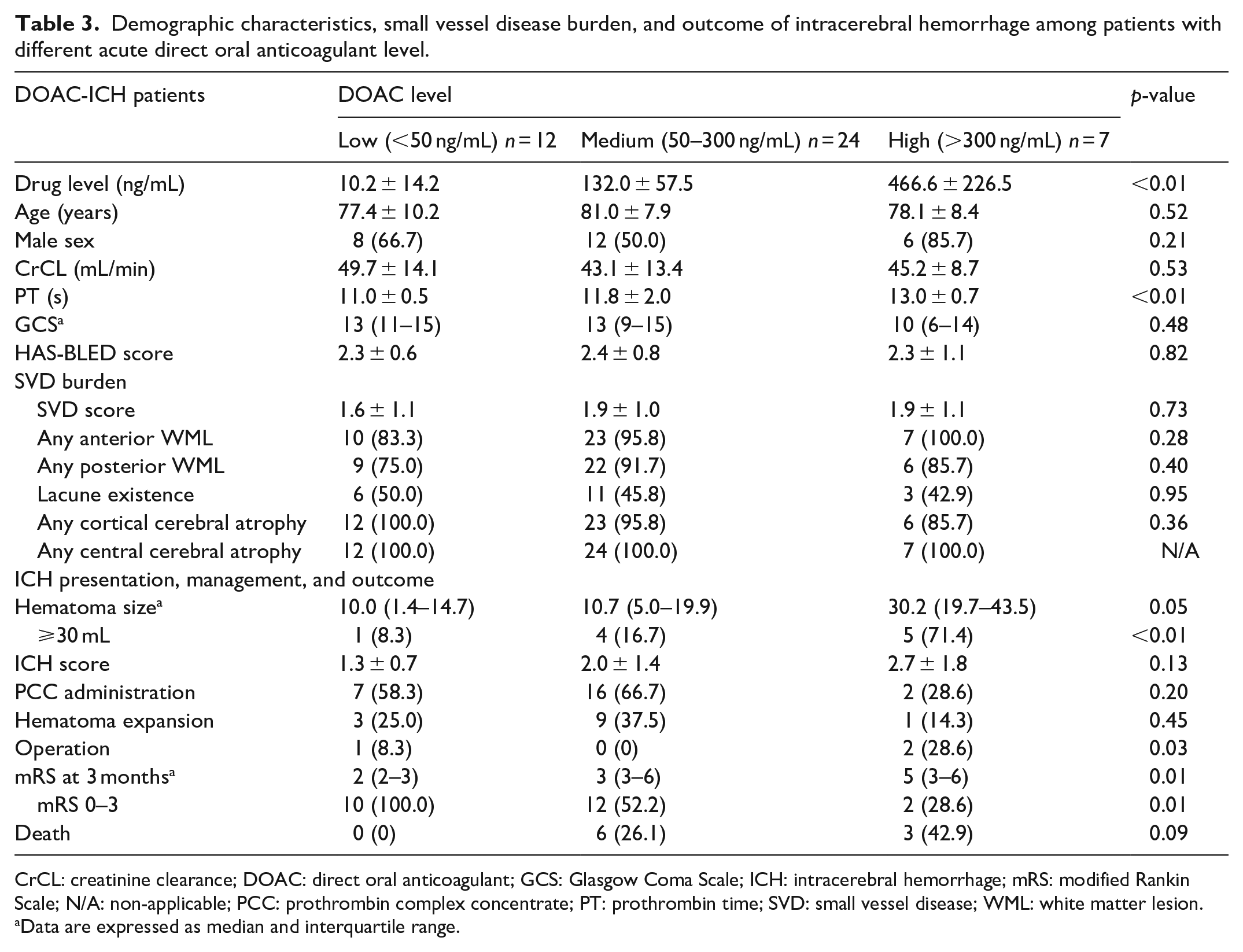
CrCL: creatinine clearance; DOAC: direct oral anticoagulant; GCS: Glasgow Coma Scale; ICH: intracerebral hemorrhage; mRS: modified Rankin Scale; N/A: non-applicable; PCC: prothrombin complex concentrate; PT: prothrombin time; SVD: small vessel disease; WML: white matter lesion.
Data are expressed as median and interquartile range.
Patients with high DOAC levels had prolonged prothrombin times (PT). However, age, sex, renal function, and HAS-BLED scores were comparable across the different drug level groups. The average SVD scores were 1.9 ± 1.1, 1.9 ± 1.0, and 1.6 ± 1.0 points in the high, medium, and low-level groups, respectively (p = 0.73). Additionally, individual SVD parameters, such as anterior/posterior WML, lacunes, and central/cortical atrophy, were all comparable across the three groups (Table 3).
Patients with high DOAC levels had larger hematomas compared to those in the other two groups. In addition, they were more likely to undergo surgery, less likely to have good functional outcomes at 3 months, and displayed a trend of higher mortality at 3 months. In the shift analysis, after adjusting for age and sex, high and medium drug levels, in contrast to low levels, significantly predicted high mRS scores. Moreover, higher drug levels were associated with a greater risk of poor functional outcomes (common OR, 9.75 [1.34, 70.9] for high levels, p = 0.02; 6.96 [1.61, 30.1] for medium levels, p = 0.01, Figure 3).
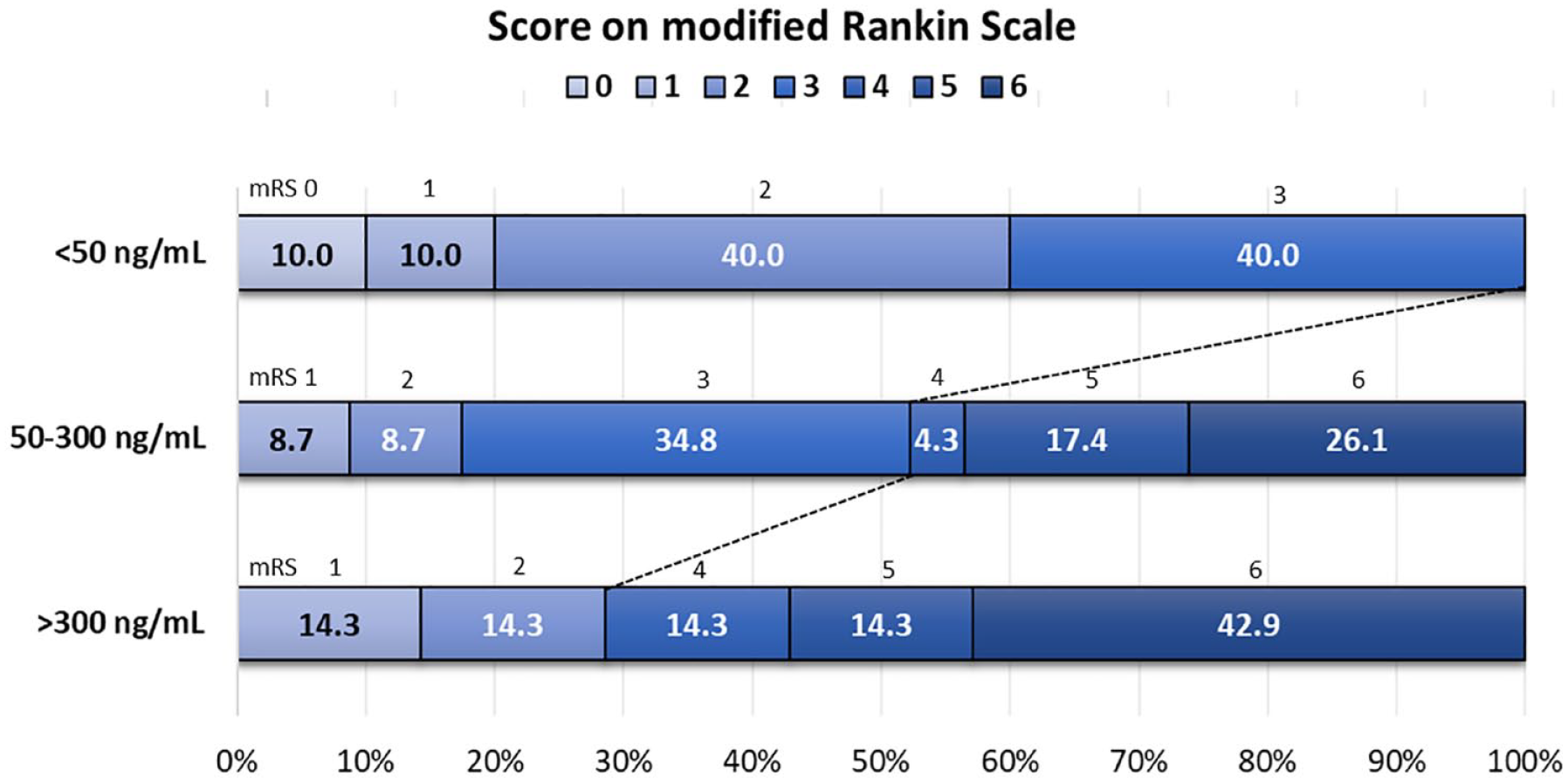
The distribution of modified Rankin Scale among direct oral anticoagulant associated intracerebral hemorrhage (DOAC-ICH) patients with different drug level.
Discussion
This is the first study to report the association between the CT-based SVD burden on DOAC-associated ICH and the impact of DOAC level at hospital admission on ICH outcomes in Asian patients. Our results show that patients with DOAC-ICH had a higher SVD burden than those without ICH. A higher SVD score (particularly a score of ⩾2) was associated with ICH during DOAC therapy. Among the different SVD features, WML, but not lacunes or cerebral atrophy, were more closely associated with the occurrence of ICH. Furthermore, a high drug level on admission is not only associated with a larger hematoma size but is also associated with worse functional outcomes in patients with DOAC-associated ICH.
Our findings emphasize the contribution of SVD burden on ICH occurrence during DOAC therapy, which is in line with the CROMIS-2 study.10,11 The most remarkable difference between our study and the CROMIS-2 trial is that our investigation was limited to DOAC users, whereas the CHROMIS-2 trial analyzed more than 90% of warfarin-associated ICH cases.10,11 As mentioned previously, compared to warfarin, DOAC prominently reduce the risk of ICH. 1 However, DOAC-associated ICH can still manifest. 3 Thus, surveying the imaging-based SVD burden in DOAC users can help identify patients at risk of this devastating side effect during treatment. Paradoxically, we found that a history of IS or TIA was inversely associated with ICH occurrence during DOAC therapy. This finding could be attributed to selection bias since, in most cases, patients in the DOAC-non-ICH group who underwent head CT for the evaluation of stroke-like symptoms exhibited a high frequency of a history of IS or TIA.
Another pivotal aspect of our findings is the connection between high DOAC levels and poor ICH outcomes, which highlights the importance of proper DOAC prescription in stroke prevention. 14 Factors associated with increased DOAC levels have been widely discussed, encompassing dosage regimens, drug interactions, and demographic characteristics such as congestive heart failure and renal function impairment.14,26–30 Furthermore, approximately 28% of DOAC-ICH patients exhibit low DOAC levels upon hospital admission. Therefore, measuring the acute DOAC levels in patients with DOAC-ICH could assist in clinical decisions regarding the use of reversal agents or component therapy (such as prothrombin complex concentrates) when the DOAC level is >50 ng/mL. 31
In our previous investigation, we found that, among stable DOAC users, increased DOAC levels increased the risk of major bleeding. However, there was a temporal trend, although not statistically significant, between drug levels and the occurrence of ICH. This can be attributed to the very low incidence of ICH in our cohort (0.48/1000 person-years). 14 To date, the cutoff DOAC level for predicting the occurrence of ICH remains unclear. Nevertheless, our results showed that among patients with DOAC-ICH, high DOAC levels upon hospital admission were associated with worse ICH outcomes.
Our data suggest that SVD may play an important role in DOAC-ICH. Nevertheless, the existence of an SVD burden should not preclude DOAC therapy. Notably, an increased SVD burden is also associated with an increased IS risk. 10 Among patients with AF who survived antithrombotic-related ICH, retrospective cohort studies have shown that restarting anticoagulant therapy reduces the risk of IS without increasing the ICH risk.32,33 However, the results of a randomized controlled trial were inconclusive. In two studies, re-initiating anticoagulant therapy was associated with a non-significant increase in ICH risk compared to non-users.34,35 However, a trend of fewer symptomatic major vascular events and an increased incidence of hemorrhagic stroke was observed in the SoSTART (Start or Stop Anticoagulant Randomized Trial). 34 Overall, the risks of stroke and mortality after an ICH event are high. 35 Therefore, the careful selection of patients who are suitable for restarting treatment, such as those with a low SVD score, may be essential. In addition, strategies to prevent ICH, such as strict blood pressure control and lifestyle modifications, should also be considered. 31
Another important finding of our investigation was the role of WML in predicting ICH during DOAC therapy. The utilization of CT-based SVD markers, such as WML, lacunes, and cerebral atrophy, is instrumental in assessing SVD burdens. In our study, the severity of WML, but not lacunes or cerebral atrophy, was correlated with DOAC-ICH. This may be attributed to the limitations of CT imaging in accurately detecting and quantifying lacunes, which are more reliably visualized using advanced imaging techniques, such as MRI. 36
The strength of this study is that it is the first to focus specifically on DOAC users to report the association between SVD and ICH occurrence and the impact of drug levels on ICH outcomes. However, this study has the following limitations. First, the participants were enrolled from two cohorts, making a direct comparison between the two study groups potentially inappropriate. For example, a considerable proportion of participants in the DOAC-non-ICH group underwent head CT for evaluation of stroke-like neurological symptoms (76.3%), possibly introducing a selection bias. Additionally, the timing of DOAC level measurement was random for the DOAC-ICH group and at the lowest point (trough) for the DOAC-non-ICH group. Therefore, we were unable to answer the essential question regarding the contribution of DOAC levels to the occurrence of ICH during DOAC therapy. Second, despite the wide availability of head CT, it was not possible to precisely evaluate the presence of lacunes and calculate the number of cerebral microbleeds. Therefore, the impact of these factors on ICH occurrence during DOAC therapy remains unclear. Third, the DOAC concentration was not disclosed at the time of clinical decision making due to the long turn-around time of UHPLC-MS/MS. Therefore, we could not determine if a concentration-guided management protocol would affect the outcome of DOAC-ICH. Finally, this investigation was conducted at a single center with a limited sample size, potentially limiting the generalizability of the findings and the statistical power. In summary, it is essential to conduct a future large-scale, multicenter study that prospectively follows DOAC users, recording DOAC levels alongside advanced neuroimaging methods under stable DOAC therapy and upon the development of ICH. Such a study would not only facilitate an investigation into the joint contribution of drug levels and SVD to ICH occurrence but also determine a cutoff value for predicting ICH occurrence.
Conclusion
The presence of an SVD burden was associated with DOAC-ICH, with higher SVD severity indicating an increased ICH risk. Additionally, high DOAC levels in ICH cases were associated with unfavorable clinical outcomes. Importantly, to address the potential selection bias related to the different study groups, a prospective study that records drug levels and baseline head CT findings is essential to investigate the co-contribution of drug levels and SVD to DOAC-ICH.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231205673 – Supplemental material for Impact of cerebral small vessel disease burden and drug level at admission on direct oral anticoagulant associated intracerebral hemorrhage
Supplemental material, sj-docx-1-eso-10.1177_23969873231205673 for Impact of cerebral small vessel disease burden and drug level at admission on direct oral anticoagulant associated intracerebral hemorrhage by Shin-Yi Lin, Ya-Fang Chen, Chih-Hao Chen, Ching-Hua Kuo, Yen-Bin Liu, Yuan-Chang Chao, Yu-Fong Peng, Chih-Fen Huang, Sung-Chun Tang and Jiann-Shing Jeng in European Stroke Journal
Footnotes
Acknowledgements
We would like to thank the National Taiwan University School of Pharmacy (NTUSP) Endowment Fund for their generous support, which enabled us to access the facilities for clinical mass spectrometry and NMR structure elucidation. We also thank Chia-Chi Chang from the NTUSP for their assistance in measuring DOAC levels. We would also like to express our gratitude to Professor Chin-Hao Chang in the Department of Medical Research at the NTUH and Dr. Fang-Ju Irene Lin in the Graduate Institute of Clinical Pharmacy at the NTUSP for their valuable statistical counseling assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a research grant from the Ministry of Science and Technology in Taiwan (112-2314-B-002-313), financial assistance from the Ministry of Education, and the NTUH (112-S0123).
Ethical approval
The NTUH International Ethics Committee approved the study protocol (REC number: 201912233RINC and 202003059RINA).
Guarantor
Sung-Chun Tang.
Contributorship
SY Lin, CH Chen, SC Tang, and JS Jeng contributed to the conceptualization and design of the research. SY Lin, YF Chen, CH Chen, YB Liu, YC Chao, and SC Tang performed the research. CH Kuo and YF Peng provided the resources for the concentration analysis. SY Lin, CH Chen, and SC Tang performed the data analysis. SY Lin drafted the original manuscript, and all other authors critically reviewed, edited, and approved the content of the manuscript. SY Lin and CH Kuo contributed to the funding acquisition.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
