Abstract
Introduction:
In recent years, the role of intravenous thrombolysis (IVT) before endovascular stroke treatment (EVT) has been discussed intensively. Whether the discussion was accompanied by changing rates of bridging IVT is unknown.
Methods:
Data were extracted from the prospectively maintained German Stroke Registry, including patients treated with EVT at one of 28 stroke centers in Germany between 2016 and 2021. Primary outcome parameters were the rate of bridging IVT (a) in the entire registry cohort and (b) in patients without formal contraindications to IVT (i.e. recent oral anticoagulants, time window ⩾4.5 h, extensive early ischemic changes) adjusted for demographic and clinical confounders.
Results:
10,162 patients (52.8% women, median age 77 years, median National Institutes of Health Stroke Scale score 14) were analyzed. In the entire cohort, the rate of bridging IVT decreased from 63.8% in 2016 to 43.6% in 2021 (average absolute annual decrease 3.1%, 95% CI 2.4%–3.8%), while the proportion of patients with at least one formal contraindication increased by only 1.2% annually (95% CI 0.6%–1.9%). Among 5460 patients without record of formal contraindications, the rate of bridging IVT decreased from 75.5% in 2016 to 63.2% in 2021 and was significantly associated with admission date in a multivariable model (average absolute annual decrease 1.4%, 95% CI 0.6%–2.2%). Clinical factors associated with lower odds of bridging IVT included diabetes mellitus, carotid-T-occlusion, dual antiplatelet therapy, and direct admission to a thrombectomy center.
Conclusion:
We observed a substantial decline in bridging IVT rates independent of demographic confounders and not explained by an increase in contraindications. This observation deserves further exploration in independent populations.
Keywords
Introduction
Intravenous thrombolysis (IVT) followed by endovascular treatment (EVT) is the recommended treatment for eligible patients with acute ischemic stroke due to anterior circulation large vessel occlusion with no change in guideline recommendations regarding this aspect in recent years.1–3 However, after completion and publication of five pivotal thrombectomy trials that established the superiority of IVT plus EVT over IVT alone in 2015 and 2016, 4 the role of medical treatment with IVT in patients planned to undergo EVT (bridging IVT) has been debated, particularly if bridging IVT was expected to lengthen imaging-to-groin time. The idea to omit IVT was fueled by observations that proximal occlusions with long thrombi have a lower likelihood to recanalize with IVT alone,5,6 higher rates of symptomatic intracranial hemorrhages in patients treated with IVT, 7 and the potential risks of thrombus migration and distal embolization, increasing EVT complexity.8–10 Moreover, an early meta-analysis suggested that EVT alone may be non-inferior to EVT plus IVT. 11 This conclusion, however, was not based on stringent non-inferiority margins and only included data from the first three RCTs. It was not until recently that prospective randomized clinical trials directly comparing EVT alone with EVT plus bridging IVT were completed.12–16 In these studies, direct EVT resulted in lower recanalization rates and was not shown to be non-inferior to EVT with bridging IVT. Combined with the results of 35 observational studies, bridging IVT plus EVT compared to EVT alone improved the odds of functional independence, successful reperfusion, and mortality. 17
In the past two decades, combined efforts of professional organizations, guideline committees, and local institutions have led to a steady increase in IVT rates in eligible stroke patients. 18 It is not known whether the uncertainty about the necessity of bridging IVT may have affected daily clinical practice, that is, IVT rates in large vessel occlusion ischemic stroke patients undergoing EVT. In the current analysis, we aimed to investigate time trends in the rate of bridging IVT before EVT since 2016 in a large German national registry while accounting for changes in clinical and demographic factors and practice patterns.
Patients and methods
Data availability
The data that support the findings of this study can be obtained upon request from the corresponding author after approval of the ethics committee and all participating centers.
Study design and study population
Data were extracted from the multicenter German Stroke Registry – Endovascular Therapy (GSR-ET), a prospectively maintained industry-independent, open-label, academic registry of patients who were treated with EVT for acute large vessel occlusion ischemic stroke as part of routine clinical care in one of 28 both academic and non-academic stroke centers.19,20 Clinical data were assessed locally by trained neuroradiologists and neurologists and underwent centrally coordinated quality control for plausibility and consistency. Patients were included in the current analysis if the following criteria were met: age ⩾18 years, occlusion of the terminal bifurcation of the internal carotid artery (“carotid T”) or the middle cerebral artery, no tandem occlusion (i.e. concurrent occlusion of the extracranial internal carotid artery on admission imaging), admission year between 2016 and 2021 (the latest year available at the date of database export), and available information about whether or not patients received bridging IVT.
Primary outcome parameters were the associations between admission date and bridging IVT (a) in the entire registry population and (b) in patients without formal contraindications to IVT, adjusted for demographic and clinical confounders. Secondary outcome parameters were the associations between admission date and the presence of recorded formal contraindications to IVT. The latter included a symptom-onset/last-seen-well-to-imaging time of more than 4.5 h, prior medication with an oral anticoagulant (apixaban, rivaroxaban, dabigatran, edoxaban, or a vitamin-K-antagonist), or extensive early ischemic changes on brain imaging defined as an Alberta Stroke Program Early CT score (ASPECTS) ⩽5. Clinical factors included as confounders were selected a priori based on clinical experience and/or their status as contraindications to thrombolysis in the past, and included age ⩾80 years, arterial hypertension, diabetes mellitus, carotid-T occlusion, prior dual antiplatelet therapy, and direct admission to a thrombectomy center.
Statistical analysis
Descriptive data are presented as medians with interquartile range (IQR) or absolute numbers with percentages for continuous and categorical variables, respectively. Associations between two or more variables were assessed with a generalized linear model, using a logit link function where appropriate. Analyses involving admission dates were performed with monthly granularity. Multivariable models initially included all admission date × covariate interactive terms to account for varying practice patterns over time; interactive terms were removed from the final model if the p value for the parameter estimate exceeded 0.1. Effect sizes were estimated as odds ratios (OR) with 95% confidence intervals. In addition to ORs, we also calculated absolute average annual changes that were derived from the respective binary logistic regression models by bootstrapping (n = 1000). No imputation of missing values was performed. Using the conservative Bonferroni-Holmes method to adjust for multiple testing, the threshold for statistical significance was set to 0.01. All analyses were carried out using MATLAB version R2022b. 21
Informed consent and ethics approval
Data collection for the GSR-ET was centrally approved by the Ethics Committee of the Ludwig-Maximilians University, Munich (689–15) as the leading ethics committee. Further approval was obtained from local ethics committees or institutional review boards according to local regulations. In accordance with the institutional review board approval, no informed consent was required because no study-specific procedures were performed and data sampling from patients undergoing thrombectomy was already mandated by national law for quality control reasons. 20
Results
A total of 10,162 patients in the registry met inclusion criteria and were analyzed. Information about whether patients received bridging IVT was unavailable for 182 of 13,082 patients (1.4% of the total sample). Baseline variables of patients in whom information on bridging IVT was unavailable were distributed similarly to those in the main analysis. The annual number of patients included in the registry increased steadily from 425 in 2016 to 2362 in 2021. The median number of patients contributed by each center was 185 (IQR 48–290) and there was no association between the date of the first contributed patient per center and overall IVT-rates achieved at the center (β = 0.034 ± 0.228, p = 0.881).
Median age in the study population was 77 years (IQR 65–83) and 5365 patients (52.8%) were women. Median National Institutes of Health Stroke Scale score was 14 (9–18). The prevalences of occlusion of the carotid T, the proximal or distal M1 segment, and the M2 segment of the middle cerebral artery were 18.9%, 38.2%, 25.5%, and 27.3%, respectively. The overall proportion of patients admitted directly to an EVT-center was 59.1% and undulated between 60.4% in 2016, 55.2% in 2019, and 63.9% in 2021, with no evidence of a significant time trend (β = 0.003 ± 0.004, p = 0.485).
Demographic and clinical characteristics according to presence of at least one formal contraindication to IVT and, in patients without formal contraindication(s), according to bridging IVT, are shown in Table 1. Overall, 4702 patients (46.3%) had at least one formal contraindication to IVT. Of the remaining 5460 patients without formal contraindication(s), 3692 patients (67.6%) received IVT before thrombectomy. Amongst all patients (with and without formal contraindications), the rate of bridging thrombolysis decreased from 63.8% in 2016 to 43.6% in 2021 (unadjusted OR 0.88 per year, 95% CI 0.86–0.91, p < 0.001; average absolute annual decrease 3.1%, 95% CI 2.4%–3.8%). In the same period, the proportion of patients receiving imaging more than 4.5 h after symptom onset/last seen well and the proportion of patients taking oral anticoagulants increased from 30.7% to 35.8% (OR 1.05, 95% CI 1.02–1.08, p = 0.003; average absolute annual increase 1.0%, 95% CI 0.3%–1.7%) and from 16.4% to 26.1% (OR 1.09, 95% CI 1.05–1.12, p < 0.001; average absolute annual increase 1.4%, 95% CI 0.9%–2.0%), respectively. Consequently, there was a significant increase in the proportion of patients with at least one formal contraindication to IVT (OR 1.05, 95% CI 1.02–1.08, p < 0.001; average absolute annual increase 1.2%, 95% CI 0.6%–1.9%; Table 2, Figure 1).
Demographic and clinical characteristics according to presence of at least one formal contraindication to IVT and, in patients with no formal contraindication, according to bridging IVT.
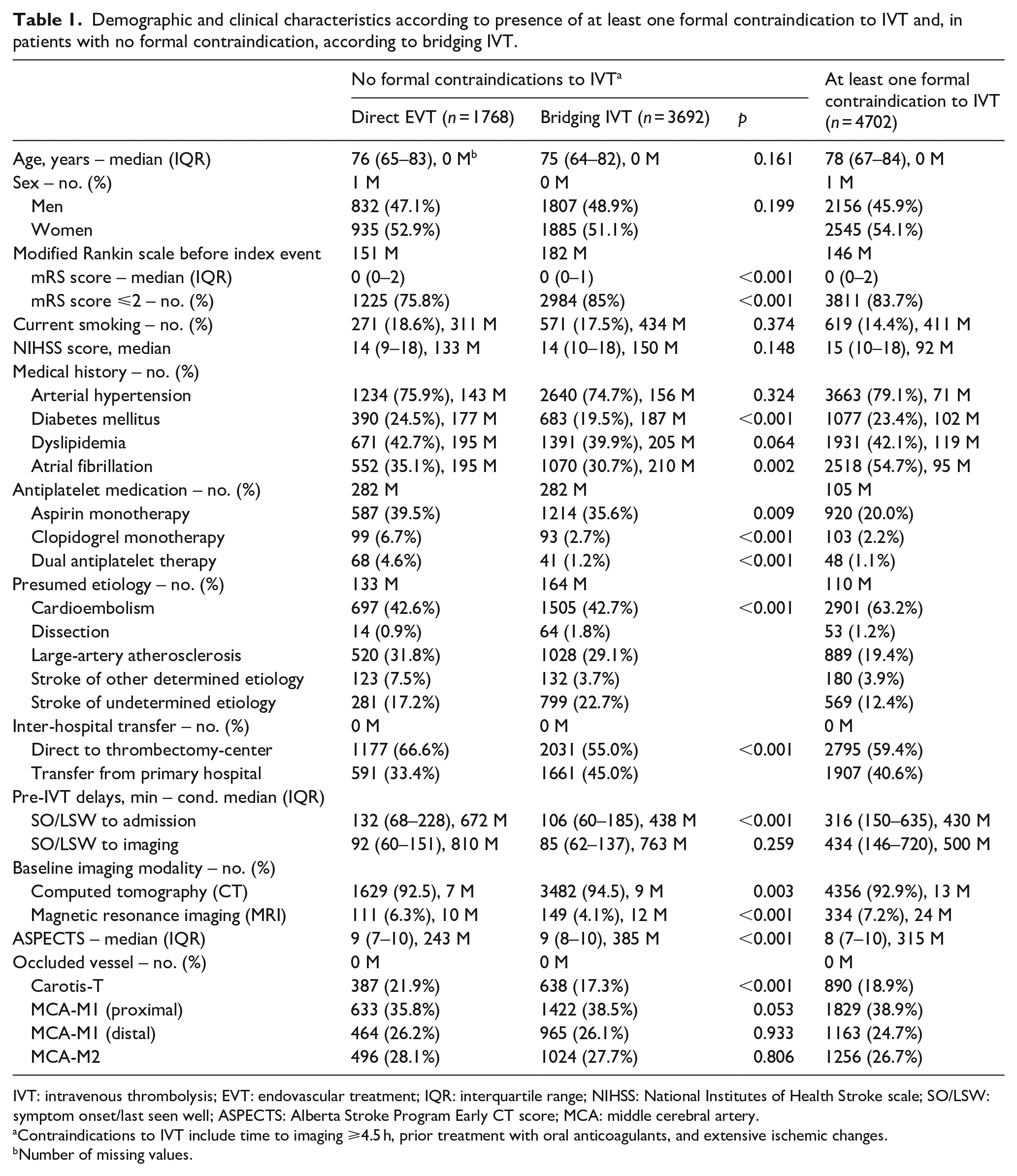
IVT: intravenous thrombolysis; EVT: endovascular treatment; IQR: interquartile range; NIHSS: National Institutes of Health Stroke scale; SO/LSW: symptom onset/last seen well; ASPECTS: Alberta Stroke Program Early CT score; MCA: middle cerebral artery.
Contraindications to IVT include time to imaging ⩾4.5 h, prior treatment with oral anticoagulants, and extensive ischemic changes.
Number of missing values.
Prevalence of formal contraindications to bridging thrombolysis according to year.
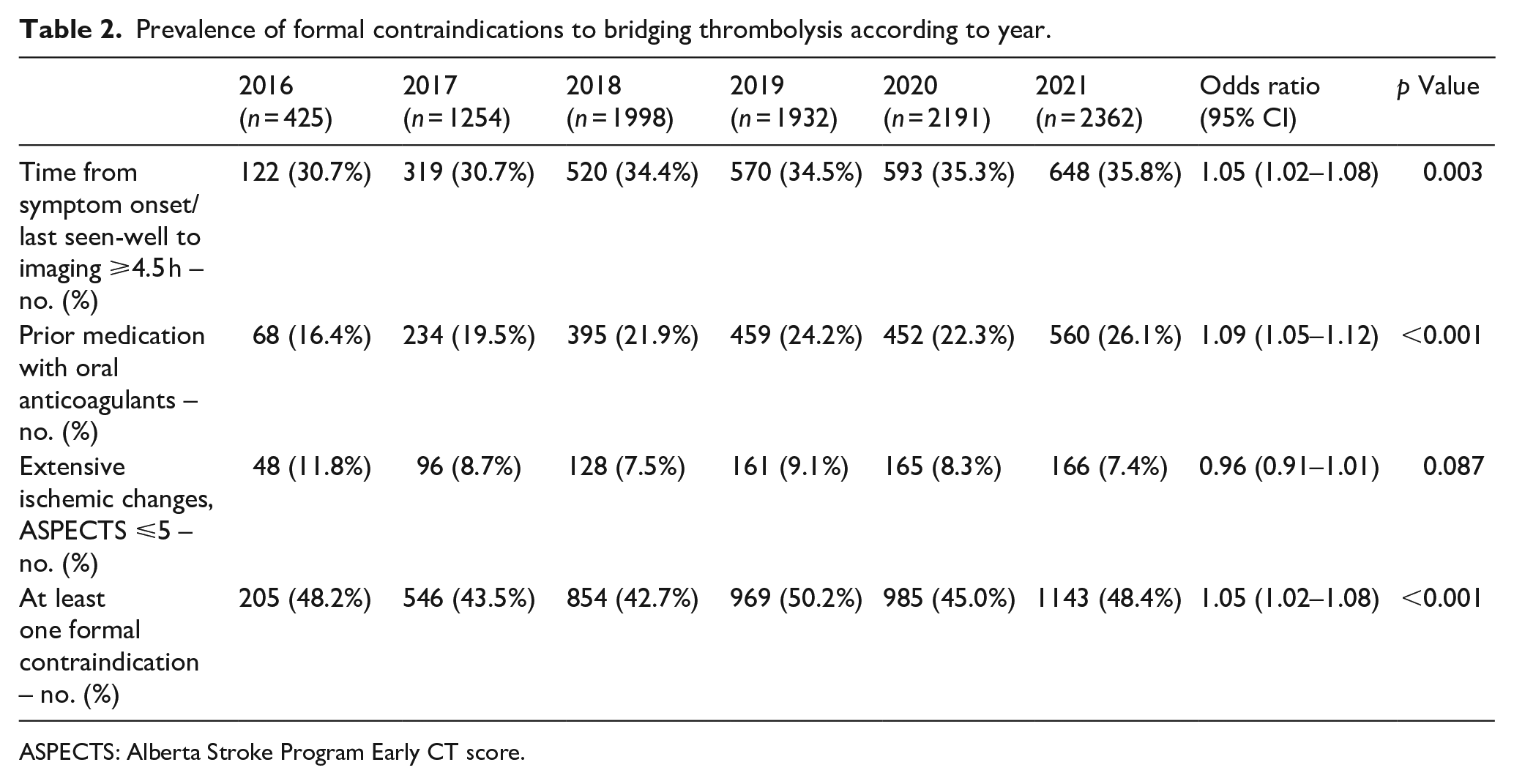
ASPECTS: Alberta Stroke Program Early CT score.
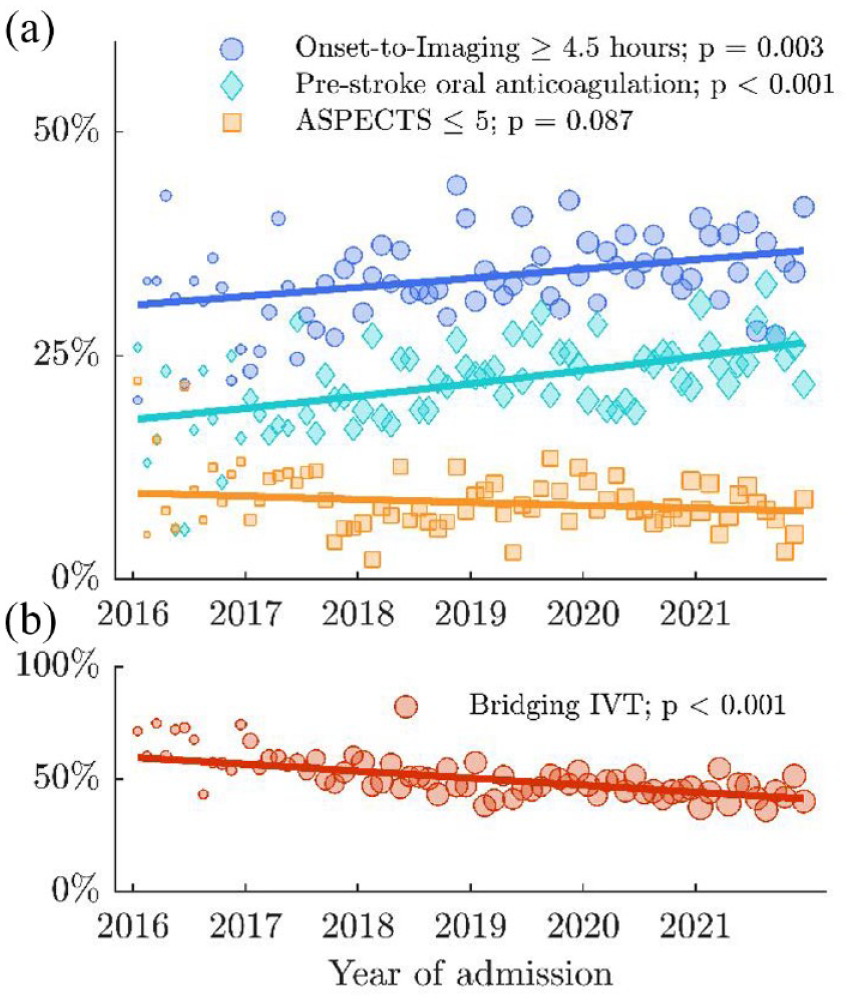
Prevalence of formal contraindications and rate of bridging IVT by admission date.
Among patients without formal contraindication(s) to IVT, the rate of bridging IVT decreased from 75.5% in 2016 to 63.2% in 2021. In a multivariable model, later admission date was significantly associated with lower odds of bridging IVT rate in a multivariable model (aOR 0.93 per year, 95% CI 0.90–0.97; p = 0.001). Clinical factors significantly associated with lower likelihood of bridging IVT included diabetes mellitus, carotid-T occlusion, dual antiplatelet therapy, and direct admission to a thrombectomy center, but not age ⩾80 years and arterial hypertension in the medical record (Table 3). The estimated rate of bridging IVT among patients without formal contraindications derived from the adjusted multivariable model decreased from 74.9% (95% CI 71.8%–77.8%) in 2016 to 66.6% (95% CI 63.9%–69.1%) in 2021, corresponding to an average absolute annual decrease of 1.4% (95% CI 0.6%–2.2%, Figure 2).
Predictors of bridging thrombolysis among patients with no formal contraindications – multivariable regression analysis.
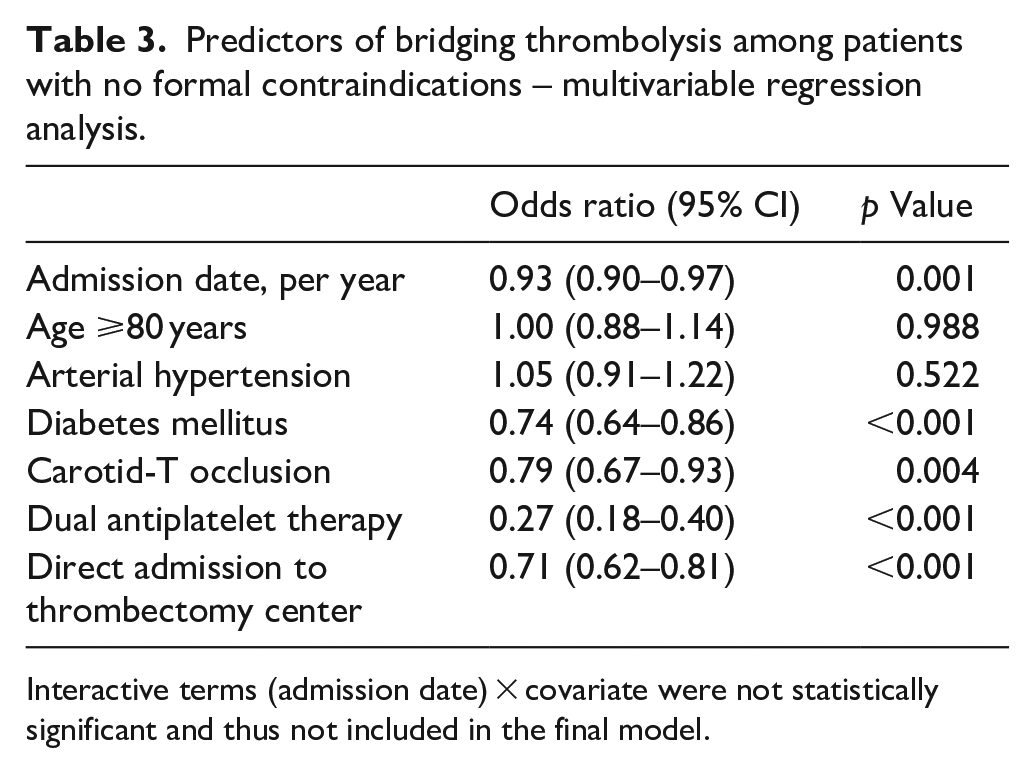
Interactive terms (admission date) × covariate were not statistically significant and thus not included in the final model.
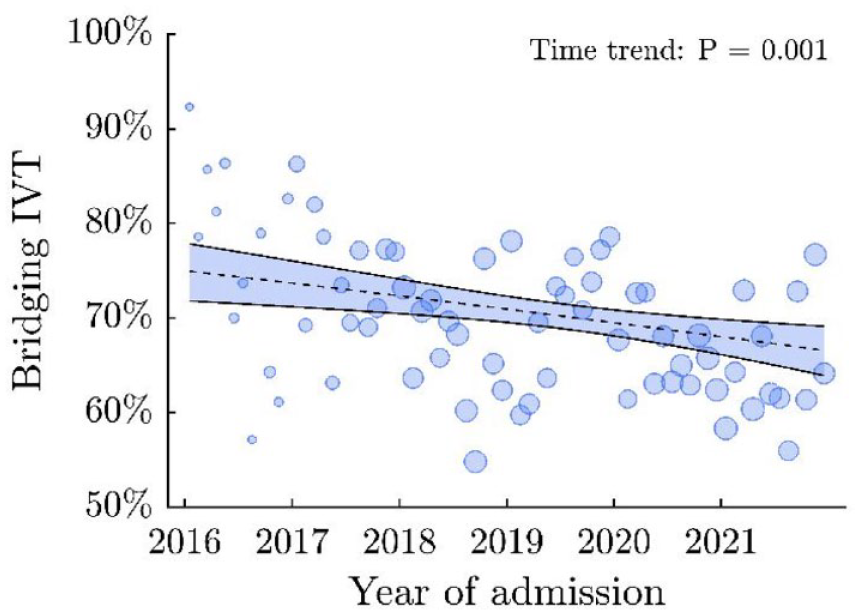
Time trend of bridging IVT in patients without formal contraindications.
Discussion
Main findings
In the current study, we analyzed changes in the rate of bridging IVT in patients with large vessel occlusion stroke in the anterior circulation treated with EVT between 2016 and 2021. In the entire study population over the whole time period, we observed a reduction in the rate of bridging IVT of approximately 20 percentage points, or 3.1% per year. This decline was accompanied by an average annual increase in the prevalence of formal contraindications to IVT of 1.2%, which could not explain the magnitude of the reduction in bridging IVT. Of note, even in the group of patients without formal contraindications to IVT and including adjustment for potential confounders, there was a significant decline in the rate of bridging IVT of 8.3 percentage points, or 1.4% annually.
To our knowledge, this is the first study analyzing the time trend of bridging IVT in patients receiving EVT since the latter became clinical standard in 2016. The observed decline of bridging IVT in patients without formal contraindications was not fully explained by changes in clinical factors alone and is not backed by guidelines which have not changed their recommendations on bridging IVT in the respective period.1,2,11 One hypothesis that could explain the observed trends is a shift in the perceived benefit/risk ratio of bridging IVT after a subset of early non-randomized studies suggested non-inferiority of EVT compared to EVT plus bridging IVT, albeit at generous non-inferiority margins.22,23 We speculate that the psychological aspect that a patient planned for EVT would not be without therapy even if IVT was omitted, and the perceived advantage conveyed by a reduction in complexity of the treatment algorithm without bridging IVT may have been additional contributing factors.
Besides the temporal trend, our study identified several clinical factors that were associated independently with absence of bridging IVT: Diabetes mellitus, prior dual antiplatelet therapy, carotid-T occlusion, and direct admission to a thrombectomy center. Based on our results, we suggest these factors might have influenced clinical decision-making. The negative association of diabetes mellitus and bridging IVT may rest on the exclusion criterion of the ECASS 3 trial that had considered the combined history of diabetes mellitus and previous ischemic stroke as a contraindication. 24 However, careful analysis of published data indicate that these exclusion criteria may not be justified in practice. 1 The younger WAKE UP trial did not consider combined diabetes and previous stroke (or diabetes alone) a contraindication to IVT anymore. 25
Although IVT has been shown to be safe in patients with prior dual antiplatelet therapy,26,27 concerns of an increased risk of symptomatic hemorrhage after IVT may have led clinicians to withhold IVT in a larger proportion of patients. The lower rate of bridging IVT in patients with carotid-T occlusion and direct admission to a thrombectomy center is most likely related to the lower probability of recanalization after IVT alone in these settings. 28
A recent meta-analysis has suggested that bridging IVT before EVT is associated with better clinical outcomes and higher recanalization rates. 17 Current guidelines explicitly state, that patients with anterior circulation LVO stroke should receive IVT in addition to EVT if they have no contraindications. 3 Even though in certain well-defined clinical situations a direct-to-EVT strategy may be appropriate for some patients, the observed decline in the rate of bridging IVT since 2016 is remarkable. It may reflect the willingness to rapidly adopt new treatment concepts even in situations with scarcity of high-level evidence and before publication of updated pertinent guidelines. Future studies should examine if similar trends exist in other health care environments and try to analyze the underlying clinical decision-making processes in greater detail.
Limitations
Our results are based on a large patient cohort that reflect academic and non-academic clinical practice in Germany. Nonetheless, our study has limitations. First, not all potential contraindications to IVT were recorded in the registry and therefore could not be adjusted for, for example, recent major surgery, diffuse hemorrhage-prone metastases, or lack of consent; it is possible that our findings could be explained in part by an increasing prevalence of such patients among the cohort of EVT-treated patients. Second, we have defined a time from symptom onset/last seen well to imaging of more than 4.5 h as formal contraindication to IVT since analysis of the time trend of extended window bridging thrombolysis was not the focus of the current study. In addition, considering potentially more frequent IVT beyond 4.5 h in recent years would have rendered our main result, the adjusted decline in bridging IVT rates, even more pronounced. Last, we did not analyze whether decreasing rates of bridging-IVT affected clinical outcome, as given the non-randomized design of our study spanning a 5-year period, residual confounding with regards to clinical outcome would be likely. Prospective randomized controlled trials, such as those concluded recently,12–16 are required to define more clearly the role of bridging-IVT before endovascular therapy and its relationship with clinical outcome.
Conclusion
In this multicenter center representing a major proportion of academic and non-academic thrombectomy centers in Germany, rates of bridging IVT before EVT in patients with anterior circulation large vessel occlusion stroke have declined between 2016 and 2021. This decline was only partially explained by increases in formal contraindications to IVT and persisted after adjustment for demographic and clinical confounders. Conflicting evidence from non-randomized studies and fear of doing harm may have contributed to uncertainty that resulted in greater reluctance to treat patients with bridging IVT. This observation deserves further exploration in independent populations.
Footnotes
Acknowledgements
We acknowledge the work and support of local GSR-ET investigators.
Abbreviations
GSR-ET German Stroke Registry – Endovascular Treatment
EVT Endovascular treatment
IVT Intravenous thrombolysis
ASPECTS Alberta Stroke Program Early CT score
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Nolte reports grants from the German Ministry of Research and Education, German Center for Neurodegenerative Diseases, and German Center for Cardiovascular Research as well as personal fees from Abbott, Boehringer Ingelheim, Alexion, Bayer Pharma, Bristol Myers Squibb, Daiichi Sankyo, and Pfizer Pharma outside the submitted work. All other authors declare that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Data collection for the GSR-ET was centrally approved by the Ethics Committee of the Ludwig Maximilians University, Munich (689–15) as the leading ethics committee. Further approval was obtained from local ethics committees or institutional review boards according to local regulations.
Informed consent
In accordance with the institutional review board approval, no informed consent was required because no study-specific procedures were performed and data sampling from patients undergoing thrombectomy was already mandated by national law for quality control reasons.
Guarantor
LS and CHN.
Contributorship
LS and CHN researched literature and conceived the study. LS performed the main analyses and wrote the first draft of the manuscript. ES contributed to the data analysis plan. All authors were involved in patient recruitment and data collection, reviewed and edited the manuscript for intellectual content, and approved the final version of the manuscript.
