Abstract
Introduction:
To improve our understanding of the relatively poor outcome after endovascular treatment (EVT) in women we assessed possible sex differences in baseline neuroimaging characteristics of acute ischemic stroke patients with large anterior vessel occlusion (LVO).
Patients and methods:
We included all consecutive patients from the MR CLEAN Registry who underwent EVT between 2014 and 2017. On baseline non-contrast CT and CT angiography, we assessed clot location and clot burden score (CBS), vessel characteristics (presence of atherosclerosis, tortuosity, size, and collateral status), and tissue characteristics with the Alberta Stroke Program Early Computed Tomography Score (ASPECTS). Radiological outcome was assessed with the extended thrombolysis in cerebral infarction score (eTICI) and functional outcome with the modified Rankin Scale score (mRS) at 90 days. Sex-differences were assessed with multivariable regression analyses with adjustments for possible confounders.
Results:
3180 patients were included (median age 72 years, 48% women). Clots in women were less often located in the intracranial internal carotid artery (ICA) (25%vs 28%, odds ratio (OR) 0.85;95% confidence interval: 0.73–1.00). CBS was similar between sexes (median 6, IQR 4–8). Intracranial (aOR 0.73;95% CI:0.62–0.87) and extracranial (aOR 0.64;95% CI:0.43–0.95) atherosclerosis was less prevalent in women. Vessel tortuosity was more frequent in women in the cervical ICA (aOR 1.89;95% CI:1.39–2.57) and women more often had severe elongation of the aortic arch (aOR 1.38;95% CI:1.00–1.91). ICA radius was smaller in women (2.3vs 2.5 mm, mean difference 0.22;95% CI:0.09–0.35) while M1 radius was essentially equal (1.6vs 1.7 mm, mean difference 0.09;95% CI:−0.02–0.21). Women had better collateral status (⩾50% filling in 62%vs 53% in men, aOR 1.48;95% CI:1.29–1.70). Finally, ASPECT scores were equal between women and men (median 9 in both sexes, IQR 8–10vs 9–10). Reperfusion rates were similar between women and men (acOR 0.94;95% CI:0.83–1.07). However, women less often reached functional independence than men (34%vs 46%, aOR 0.68;95% CI:0.53–0.86).
Discussion and conclusion:
On baseline imaging of this Dutch Registry, men and women with LVO mainly differ in vessel characteristics such as atherosclerotic burden, extracranial vessel tortuosity, and collateral status. These sex differences do not result in different reperfusion rates and are, therefore, not likely to explain the worse functional outcome in women after EVT.

Introduction
The lifetime prevalence of ischemic stroke is higher in women than in men and women more often have an unfavorable outcome.1–6 Endovascular treatment (EVT) is highly effective for anterior large vessel occlusion (LVO) related stroke and doubles the probability of favorable functional outcome. 7 Most clinical trials reported similar treatment effects and functional outcomes after EVT in men and women, whereas several cohort and registry studies found poorer outcomes in women.8–12 Until now, reports on sex differences in large patient series treated in routine clinical practice are lacking.
Little is known about sex differences in neuroimaging characteristics in the acute phase of LVO stroke, in relation to outcome after EVT. Most data are derived from randomized trials or small cohorts of treated patients. Women have a higher incidence of atrial fibrillation, which might lead to larger clots in more proximal locations.13,14 Also, some studies suggest that women more often have intracranial atherosclerosis, which could impair endovascular access and increase the chance of distal emboli during the procedure. 15 Furthermore, there might be sex differences in vessel tortuosity, vessel size, collateral status, or early ischemic tissue changes.16–20
More insight in possible sex differences in neuroimaging features is needed to understand the possible poorer functional outcome after EVT in women and to optimize personalized treatment in patients with LVO. We aimed to investigate sex differences in clot, vessel and tissue characteristics and assessed radiological and functional outcome after EVT in a large real-world cohort of LVO patients.
Methods
Study design
The MR CLEAN (Multicenter Randomized Clinical Trial of Endovascular Treatment for Acute Ischemic Stroke in The Netherlands) Registry is a national, prospective, open, multicenter, observational monitoring study in which all 16 stroke intervention centers in the Netherlands that perform EVT participated. 21 The registration started after the final inclusion in the original MR CLEAN trial and was set up to study safety and effectiveness of EVT in routine clinical practice. Consecutive patients in the Netherlands were included in the MR CLEAN Registry from March 2014 until January 2019. Data of patients who were treated up to 1 November 2017 were processed and used in the current analysis.
Study population
Inclusion criteria were an age of 18 years and older, arterial puncture within 6.5 h of symptom onset, and LVO of the intracranial anterior circulation (intracranial carotid artery, anterior cerebral artery, or middle cerebral artery). EVT was defined as receiving arterial puncture with the aim to perform thrombectomy. The method of EVT was left to the discretion of the treating physician.
Baseline characteristics
For this analysis, the following baseline clinical characteristics were collected: age, cardiovascular risk factors, medication, pre-stroke modified Rankin Scale (mRS) score, admission National Institutes of Health Stroke Scale (NIHSS) score, blood pressure on admission, and treatment with intravenous thrombolysis (IVT) prior to EVT. Furthermore, time from stroke onset to hospital admission, to IVT, to groin puncture, and to reperfusion was recorded.
Neuroimaging characteristics
All patients underwent non-contrast brain CT followed by a CT angiography (CTA) or MRA and Digital Subtraction Angiography (DSA). The neuroimaging characteristics of interest were divided into three main categories: (1) clot, (2) vessel, and (3) tissue characteristics. These categories were further divided into subcategories.
Clot characteristics were subdivided into clot location and clot extent. Clot location was defined as the most proximal occlusion segment (intracranial internal carotid artery (ICA) / internal carotid artery terminus (ICA-T), middle cerebral artery segment 1 (M1), M2, other). Clot extent was measured on CTA by the clot burden score (CBS). This is a grading scale from 0 to 10, that quantifies the extent of the intracranial thrombus by assigning points for the presence of contrast on imaging within the intracranial ICA, M1, M2, and anterior cerebral artery (A1) vessel segments. A score of 10 indicates that no clot is present and a score of 0 indicates complete occlusion of all vessel segments of the proximal intracranial anterior circulation. 22 We calculated the median CBS and the proportion of participants with a CBS < 7, which is related to poor functional outcome. 23
Vessel characteristics were subdivided into atherosclerosis including stenosis, vessel tortuosity, vessel size, and collateral status. Intracranial atherosclerosis and extracranial atherosclerosis, at the carotid bifurcation, was scored as absent or present on CTA. In addition, the proportion of patients with a cervical ICA stenosis of ⩾70% and ⩾99% was calculated using the North American Symptomatic Carotid Endarterectomy Trial (NASCET) criteria. 24 These cut-offs were chosen based on their relevance when performing EVT. Vessel tortuosity was assessed for supra-aortic arteries, cervical internal carotid arteries, and aortic arch in two ways: the proportion of patients with one or more angles larger than 90° was assessed for the supra-aortic and cervical ICA and the proportion of patients with severe elongation of the aortic arch (aortic arch type III, indicating that at least one of the supra aortic arteries arises below the level of the inner curvature of the aorta) was calculated. 25 Vessel size was measured by calculating the radius of the ICA and M1. Finally, collateral status was divided into absent collaterals (0% filling of the vessel downstream of the occlusion as compared to the contralateral hemisphere), poor collaterals (⩽50% filling), moderate collaterals (51%–99% filling), and excellent collaterals (100% filling). 26
Tissue characteristics were measured using the Alberta Stroke Program Early Computed Tomography Score (ASPECTS) on non-contrast CT. The ASPECTS is a grading scale from 0 to 10 for which the vascular territory of the middle cerebral artery is divided in 10 regions. Points are subtracted in the regions with any evidence of early ischemic tissue changes.27,28 We calculated the median ASPECTS, different categories of ASPECTS, and the proportion of patients with an ASPECTS < 8, which is related to an increased probability of death or dependency. 27
Clot and tissue characteristics, presence of intracranial and extracranial atherosclerosis, and collateral status were evaluated by an independent core lab of the MR CLEAN Registry, blinded to clinical findings. 21 ICA stenosis and vessel tortuosity were evaluated by trained students under supervision of an experienced neuroradiologist. 25 The inter-rated agreement of the assessment of several of the characteristics has been previously described.21,25 To calculate the ICA and M1 radius the intracranial vessels were segmented from the CTA data using custom Convolutional Neural Network (CNN) software (StrokeViewer v2.1.22, NICO.LAB, Amsterdam, the Netherlands http://www.strokeviewer.nico-lab.com). The semi-automatic software iCafe (©2016–2020, The University of Washington, used with permission) was here used to extract the center line graph, to anatomically label arterial segments, and to determine the local vessel radius at each point. 29
Radiological and functional outcome
Radiological outcome was the extended thrombolysis in cerebral infarction (eTICI) score post-EVT, rated on the final DSA run. The eTICI is a grading scale from 0 to 3, and measures the extent of reperfusion in the downstream territory of the occluded vessel. Grade 0 indicates no reperfusion, with grade 1 there is a reduction in thrombus but with no reperfusion, grades 2A, B and C indicate respectively 1%–49%, 50%–89%, and 90%–99% reperfusion, and grade 3 indicates complete reperfusion.30,31 Successful reperfusion is defined as a score of 2B or higher, and excellent reperfusion as a score of 2C or higher. To reach these scores, DSA runs including both anteroposterior and lateral views needed to be completed after EVT. If only an anteroposterior view was present, the maximum score was 2A.
Functional outcome was the modified Rankin Scale (mRS) score at 90 days. 32 The mRS is a grading scale ranging from 0 to 6 in which 0 indicates no symptoms and six indicates dead. Functional independence is defined as a score of 2 or lower. Furthermore we assessed early neurological outcome with the NIHSS at 24–48 h, and the occurrence of symptomatic intracranial hemorrhages, defined as a hemorrhage with a related decline of four points or more on the NIHSS, according to the Heidelberg criteria. 33
Missing data
If the baseline NIHSS score was missing, it was retrospectively scored with a standardized chart based on the neurological examination in the electronic patient record. 34 In case successful reperfusion was not achieved, time of the last contrast bolus was used as a proxy for the time of reperfusion. If the mRS was assessed within 30 days of symptom onset instead of at 90 days and the score was 0–5, it was considered invalid. These values were regarded as missing and replaced by mRS scores resulting from multiple imputations. Regression analyses were performed with imputed data except for part of the vessel characteristics (extracranial atherosclerosis including stenosis, vessel tortuosity, and vessel size). Because the measurements of these characteristics were only available in a small selection of patients, regression analyses in this subset were executed without multiple imputations.
Statistical analysis
We performed descriptive statistics for baseline characteristics. Neuroimaging characteristics were compared between men and women using t-tests for continuous variables and chi-square tests for dichotomous variables. Multivariable regression analyses were performed to assess the association between sex and neuroimaging characteristics: binary logistic regression analyses to assess the association between sex and clot location, atherosclerosis and vessel tortuosity; linear regression analyses to assess the association between sex and CBS, vessel radius and ASPECTS; and ordinal logistic regression analyses to assess the association between sex and collateral status. No adjustments were made in the analyses of clot location and vessel radius, since we assumed it to be unlikely that these characteristics would be significantly influenced by confounders. Analyses of CBS were adjusted for age, onset-to-groin time and collateral status; analyses of vessel tortuosity were adjusted for age and hypertension; analyses of atherosclerosis were adjusted for age, smoking, hypertension, hypercholesterolemia and diabetes mellitus; and analyses of collateral status and ASPECTS were adjusted for age and onset-to-groin time. The association between sex and eTICI post-EVT, mRS, and NIHSS at 24–48 h was assessed using ordinal logistic regression (shift) analyses. Binary logistic regression analyses were performed to assess the association between sex and successful and excellent reperfusion, functional independence, mortality, and symptomatic intracranial hemorrhages. Analyses of eTICI post-EVT were adjusted for age, eTICI pre-EVT, CBS, and additional neuroimaging characteristics that appeared to be significantly different between men and women; analyses of mRS and NIHSS were adjusted for age, diabetes mellitus, pre-stroke mRS, NIHSS on admission (and for mRS NIHSS at 24–48 h), onset to groin time, eTICI post-EVT, and additional neuroimaging characteristics that appeared to be significantly different; and analyses of symptomatic intracranial hemorrhages were adjusted for age, hypertension, NIHSS on admission and NIHSS at 24–48 h. Unadjusted and adjusted (pooled) effect estimates were expressed as mean differences or (common) odds ratios (OR) with 95% confidence intervals. For all statistical analyses SPSS for Windows version 25.0.0 was used.
Results
Of the 3637 consecutive EVT patients, 3180 were included in our analysis, 1526 (48%) of whom were women (Figure 1). Women were 5 years older than men (median age 75vs 70 years, Table 1), more frequently had a history of hypertension and less often a history of smoking or myocardial infarction. Women were more often functionally dependent prior to the stroke (pre-stroke mRS ⩽ 2 in 1259/1526 (85%) vs 1487/1654 (92%)). There was no difference in NIHSS score on admission (median 16 in both groups). The median time from stroke onset to hospital admission (57vs 50 min) and to groin puncture (200vs 189 min) was longer in women, and procedural time was somewhat shorter (60vs 62 min). The overall time from stroke onset to reperfusion or last contrast bolus was longer in women (255vs 246 min).
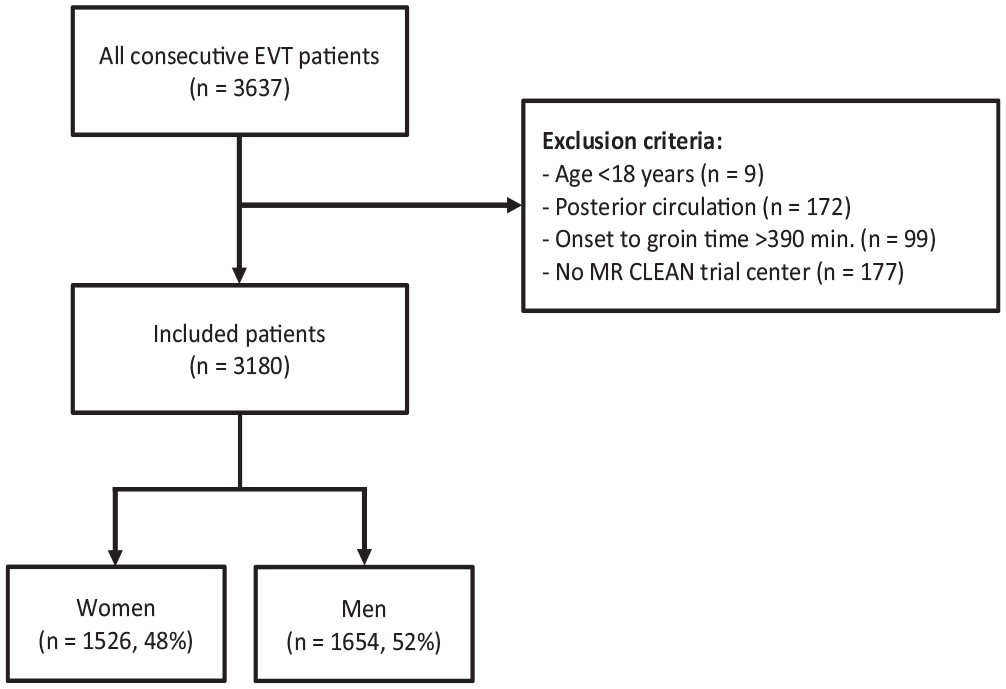
Flow chart of patient selection.
Baseline characteristics of the patients according to sex.
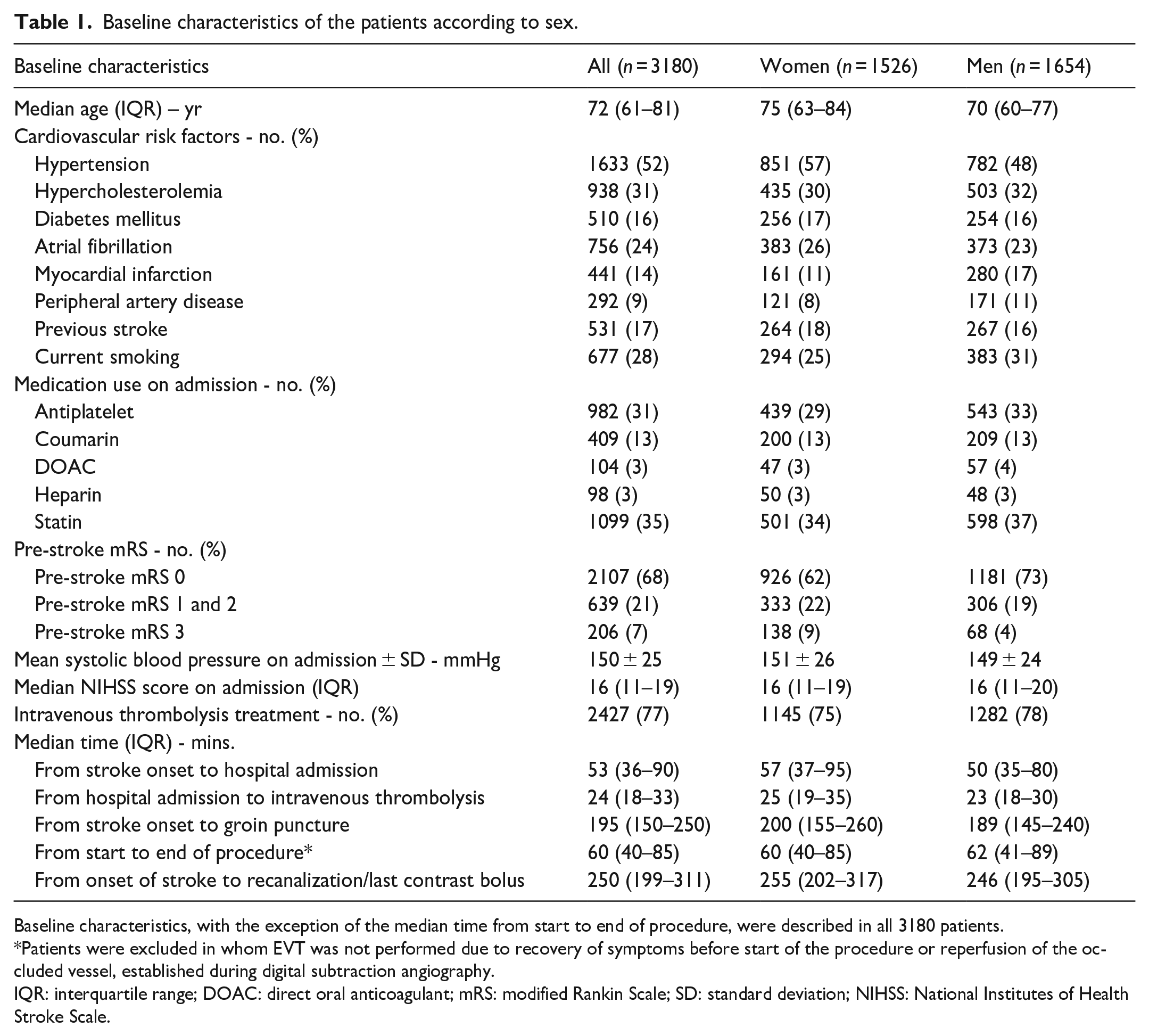
Baseline characteristics, with the exception of the median time from start to end of procedure, were described in all 3180 patients.
Patients were excluded in whom EVT was not performed due to recovery of symptoms before start of the procedure or reperfusion of the occluded vessel, established during digital subtraction angiography.
IQR: interquartile range; DOAC: direct oral anticoagulant; mRS: modified Rankin Scale; SD: standard deviation; NIHSS: National Institutes of Health Stroke Scale.
Neuroimaging characteristics
There were no statistically significant differences in clot location, although in women the clot tended to be less often located in the ICA (360/1458 (25%) vs 435/1566 (28%), OR 0.85; 95% CI 0.73–1.00) and more often in the M1 segment (874/1458 (60%) vs 890/1566 (57%), OR 1.14; 95% CI 0.98–1.31) than in men (Tables 2–4). No difference was seen in CBS (median 6; IQR 4–8 in both groups).
Radiological parameters compared between women and men.
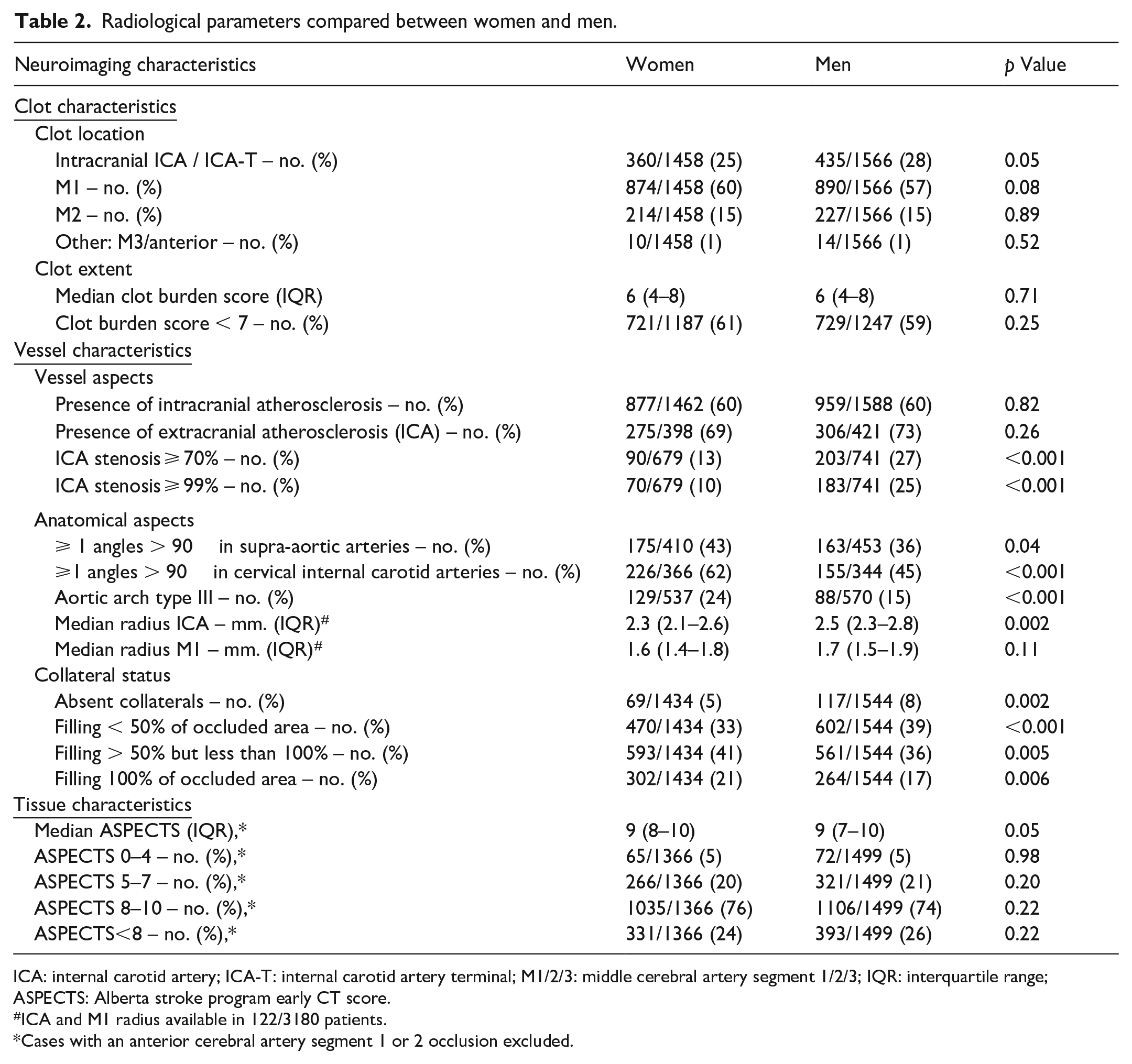
ICA: internal carotid artery; ICA-T: internal carotid artery terminal; M1/2/3: middle cerebral artery segment 1/2/3; IQR: interquartile range; ASPECTS: Alberta stroke program early CT score.
ICA and M1 radius available in 122/3180 patients.
Cases with an anterior cerebral artery segment 1 or 2 occlusion excluded.
Unadjusted and adjusted effect estimates of sex on neuroimaging characteristics (imputed data).
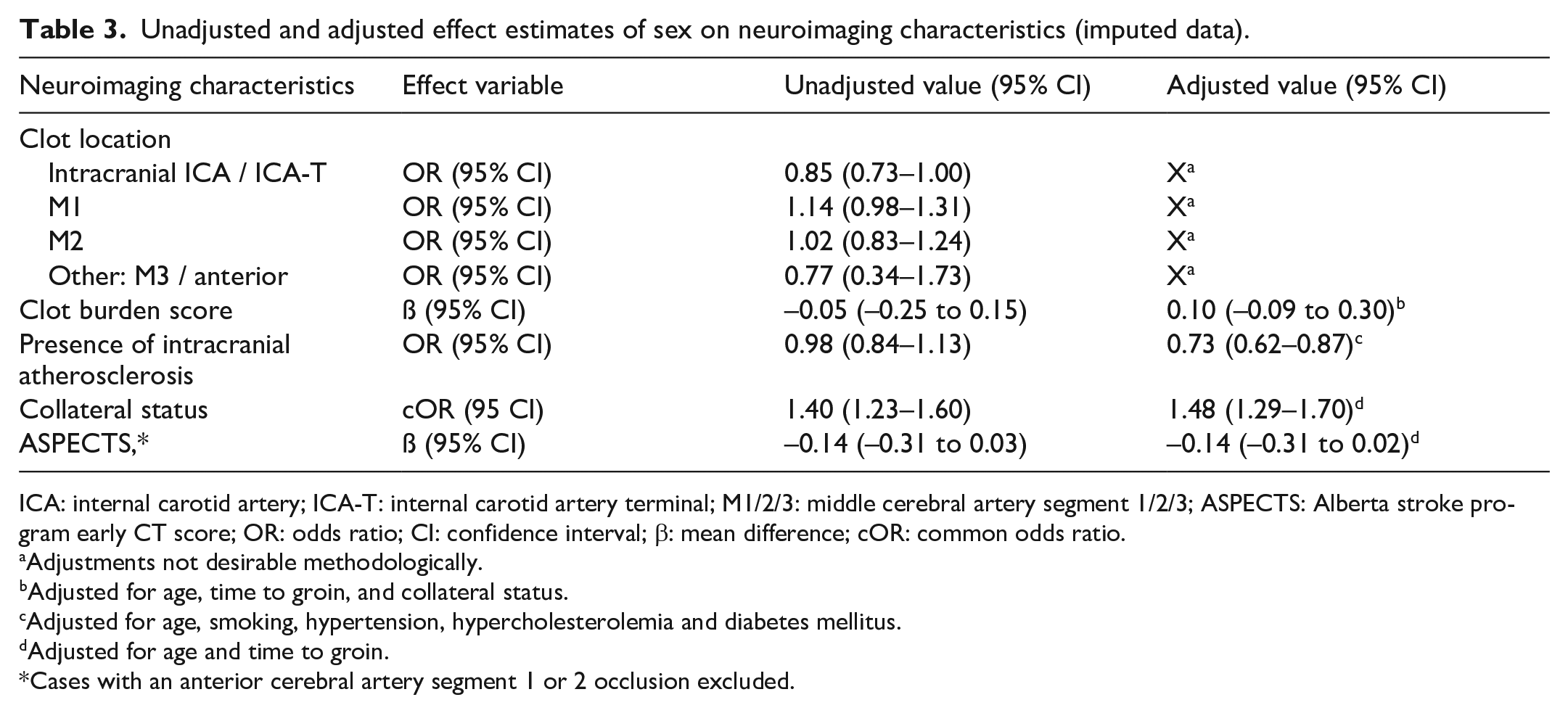
ICA: internal carotid artery; ICA-T: internal carotid artery terminal; M1/2/3: middle cerebral artery segment 1/2/3; ASPECTS: Alberta stroke program early CT score; OR: odds ratio; CI: confidence interval; β: mean difference; cOR: common odds ratio.
Adjustments not desirable methodologically.
Adjusted for age, time to groin, and collateral status.
Adjusted for age, smoking, hypertension, hypercholesterolemia and diabetes mellitus.
Adjusted for age and time to groin.
Cases with an anterior cerebral artery segment 1 or 2 occlusion excluded.
Unadjusted and adjusted effect estimates of sex on neuroimaging characteristics (unimputed data).
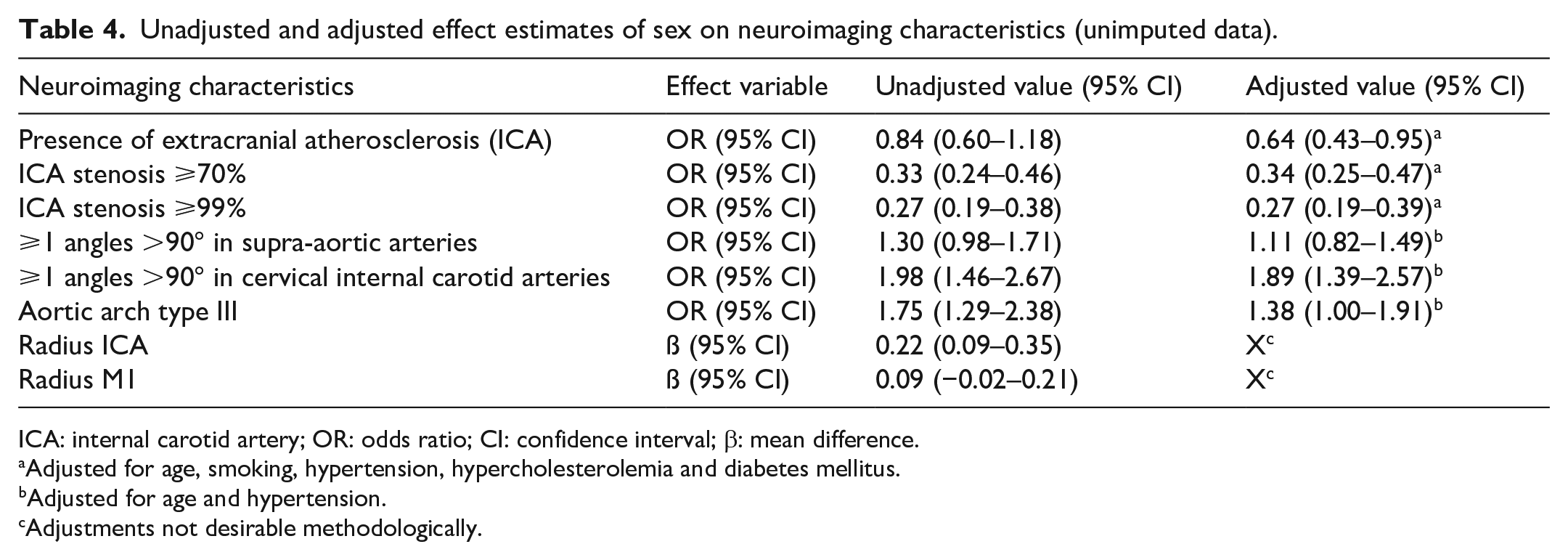
ICA: internal carotid artery; OR: odds ratio; CI: confidence interval; β: mean difference.
Adjusted for age, smoking, hypertension, hypercholesterolemia and diabetes mellitus.
Adjusted for age and hypertension.
Adjustments not desirable methodologically.
The prevalence of intracranial atherosclerosis was equal in women and men (877/1462vs 959/1588, both 60%). However, after adjustment for age, smoking, hypertension, hypercholesterolemia and diabetes mellitus, intracranial atherosclerosis was less frequent in women (adjusted OR (aOR) 0.73; 95% CI 0.62–0.87). Women also less often had extracranial atherosclerosis (275/398 (69%) vs 306/421 (73%), aOR 0.64; 95% CI 0.43–0.95), an ICA stenosis ⩾70% (90/679 (13%) vs 203/471 (27%), aOR 0.34; 95% CI 0.25–0.47), or ⩾99% (70/679 (10%) vs 183/741 (25%), aOR 0.27; 95% CI 0.19–0.39). Women more often had one or more angles ⩾90° in the cervical ICA (226/366 (62%) vs 155/344 (45%), aOR 1.89; 95% CI 1.39–2.57), and more often an aortic arch type III (129/537 (24%) vs 88/570 (15%), aOR 1.38; 95% CI 1.00–1.91). Vessel tortuosity in the supra-aortic arteries was essentially equal (175/410 (43%) vs 163/453 (36%), aOR 1.11; 95% CI 0.82–1.49). The ICA and M1 radius, data available in 122/3180 patients, were smaller in women, but differences were only minimal (median radius ICA: 2.3 mm (IQR 2.1–2.6 mm) vs 2.5 mm (IQR 2.3–2.8 mm), mean difference 0.22; 95% CI 0.09–0.35, median radius M1: 1.6 mm (IQR 1.4–1.8 mm) vs 1.7 mm (IQR 1.5–1.9 mm), mean difference 0.09; 95% CI −0.02 to 0.21). Absence of collaterals or less than 50% filling was less often seen in women (539/1434 (38%) vs 719/1544 (47%) and, overall, women had better collateral status (adjusted common OR (acOR) 1.46; 95% CI 1.27–1.67).
Early tissue changes were essentially similar with a median ASPECTS of nine in both groups and an ASPECTS < 8 in 331/1366 (24%) versus 393/1499 (26%), adjusted mean difference −0.13; 95% CI −0.30 to 0.02.
Radiological and functional outcome
The difference in proportions between the various eTICI and mRS scores are presented in Figure 2(a) and (b). The extent of reperfusion (eTICI) was similar in women and men (acOR 0.94; 95% CI 0.83–1.07). Successful reperfusion (eTICI ⩾ 2B) was reached equally often in both groups (61%vs 63%, aOR 0.94; 95% CI 0.81–1.10) as was excellent reperfusion (eTICI ⩾ 2C) (38%vs 40%, aOR 0.90; 95% CI 0.78–1.05). There was a substantial shift toward a worse functional outcome in women versus men in the distribution of mRS scores at 90 days (acOR 0.81; 95% CI 0.69–0.94) and women less often reached functional independence (34%vs 46%, aOR 0.68; 95% CI 0.53–0.86). Mortality was slightly higher in women compared with men (32%vs 27%, aOR 0.76; 95% CI 0.53–0.99). Median NIHSS after 24–48 h (early neurological outcome) was 11 (IQR 4–18) in women versus 9 (IQR 4–16) in men, acOR 1.08; 95% CI 0.95–1.24. Symptomatic intracranial hemorrhage rates were similar (7%vs 5% (aOR 1.22; 95% CI 1.00–1.48)).
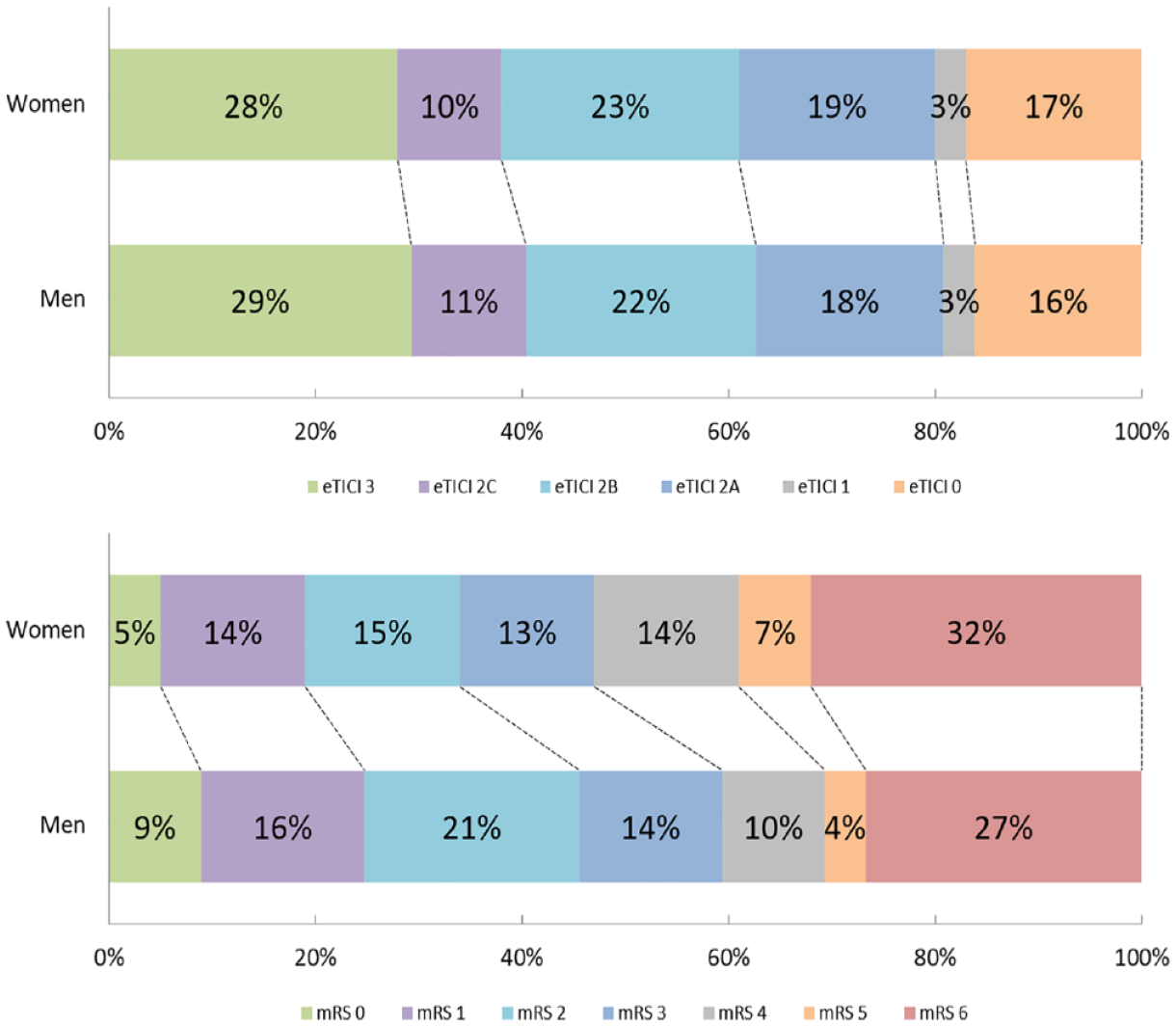
(a) Distribution of eTICI after EVT.
Discussion
In this large, real-world, cohort of patients with acute anterior LVO stroke, performed in the Netherlands, we found sex differences in vessel characteristics. Women less often had intra- and extracranial atherosclerosis and had a better collateral status, whereas vessel tortuosity was less often seen in men. Clot and tissue characteristics were the same or only minimally different in men and women with LVO. Despite sex differences in vessel characteristics, eTICI scores were similar in both sexes. Nevertheless, at 90 days, women had a worse functional outcome.
Women in our study tended to less often have an occlusion in the intracranial ICA and more often an occlusion of the M1 segment than men. This difference was also found in previous smaller studies, but never reached statistical significance (Table 5). Even in our large sample, statistical significance was not reached, implicating that if sex differences in clot location are present they are probably only minimal and will not have a substantial effect on treatment success or outcome. The finding that women had less intracranial atherosclerosis in our study compared to men contradicts with a Chinese study of 2864 patients with an acute ischemic stroke that found a higher frequency of intracranial atherosclerosis in women. 15 A recent analysis of the DUST study group showed comparable intracranial atherosclerosis presence in a general ischemic stroke population of 1397 Dutch men and women. 35 Differences between the current study and the previous studies might be explained by differences in ethnic background or in stroke population. We found that women more often had tortuous vessels than men. Both the presence of atherosclerosis and vessel tortuosity may increase the technical difficulty of EVT and therefore prolong procedural duration, which may negatively affect functional outcome. 36 In our study, procedural times were equal between sexes. However, the number of patients in whom vessel characteristics were complete was relatively small so we refrained from performing a subanalysis investigating a direct relationship between vessel characteristics and procedural time. Finally, in line with a recent meta-analysis and a sub-analysis of the DEFUSE three trial women had a better collateral status.37,38 This better collateral circulation in women could lead to better protection against cerebral ischemia. In addition, early ischemic tissue changes as measured with the ASPECT score were comparable between sexes, but unfortunately no data were available on final infarct volumes. The similar ASPECTS in our study is in accordance with previous studies as presented in Table 5.
Overview of studies comparing sex differences in neuro-imaging characteristics in EVT treated patients.
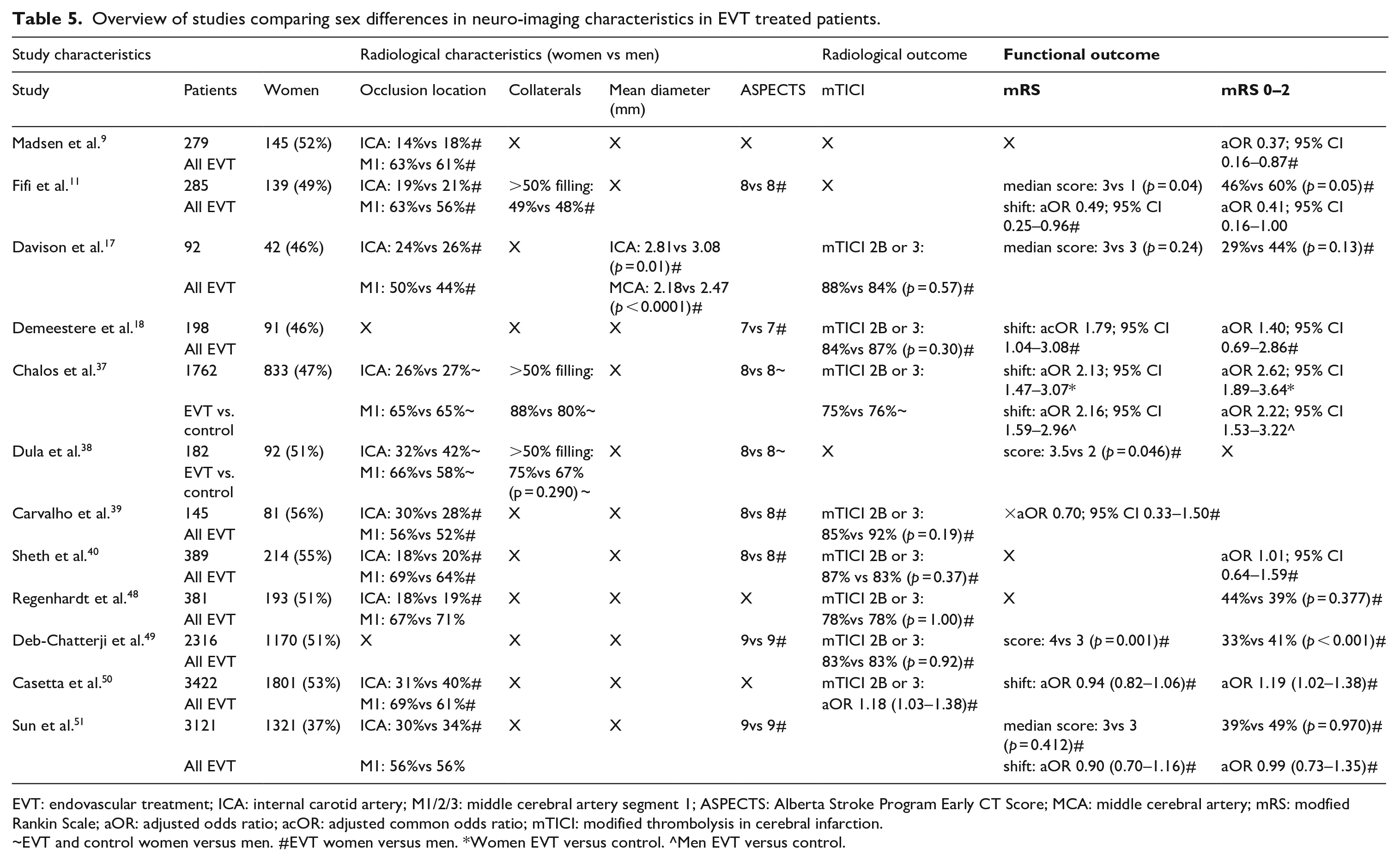
EVT: endovascular treatment; ICA: internal carotid artery; M1/2/3: middle cerebral artery segment 1; ASPECTS: Alberta Stroke Program Early CT Score; MCA: middle cerebral artery; mRS: modfied Rankin Scale; aOR: adjusted odds ratio; acOR: adjusted common odds ratio; mTICI: modified thrombolysis in cerebral infarction.
~EVT and control women versus men. #EVT women versus men. *Women EVT versus control. ^Men EVT versus control.
In our study, women less often reached functional independence after 90 days. In contrast, in most RCTs, functional outcomes were similar in men and women treated with EVT.8,9,39 A subgroup analysis of 1762 patients from seven RCTs found that functional independence was reached in 39% of men and women 90 days after EVT. 37 The difference with our study might be selection of a healthier and younger study population in the RCTs. In our study, both men and women were 5–7 years older than the RCT populations. The worse outcome in women after EVT in our study is also in contrast with two previous small observational studies, that again included a younger study population compared with our study, but in line with another small observational study that found relatively poor functional outcome after 90 days in women (Table 5).9,39,40
Our study has limitations. Because our analyses were exploratory, we did not correct for multiple testing. Results should thus be interpreted with caution due to the potential risk of type I errors. Although we corrected for possible confounders we cannot exclude that residual confounding occurred. Also, because the MR CLEAN Registry was not a randomized trial and did not include a non-treated control group, we were not able to investigate sex differences in treatment effect.8,37 Furthermore, because the MR CLEAN Registry only included patients eligible for EVT, we cannot exclude a selection bias in case there may be sex differences in neuroimaging characteristics leading to abstaining from EVT. Strengths of our study are the prospective study design and the large number of patients studied in a real-world setting.
Why women in our study, a real-world, non-trial setting, do worse than men after EVT remains unclear. In accordance with earlier findings, the time from stroke onset to hospital admission and in-hospital delays were slightly longer in women than in men. This might be because older women more often live alone than men, leading to larger prehospital delays. 14 However, EVT treatment times were slightly shorter in women and the overall difference in onset to recanalization times was less than 10 min. Differences in outcome can also develop as a result of other in-hospital treatments, such as IVT or stroke-unit care. Both better and worse functional outcomes have been reported win women receiving IVT.43,44 In our cohort there were slight differences in the proportion of women receiving IVT compared with men (75%vs 78%) and time from hospital admission to intravenous thrombolysis was on average 2 min longer in women, but it is unlikely that these small differences account for the worse outcome in women. Moreover, a large prospective registry in Austria did not show a difference in quality of stroke unit care between women and men. 43 Another likely explanation is higher frailty in women. Women have a higher life expectancy and more often suffer from stroke at a later age compared with men.13,44,45 Consistent with previous studies, women in our study were more often functionally dependent before their stroke. 46 Although we adjusted for age and pre-stroke mRS, we cannot exclude that women had more additional co-morbidity. This frailty and co-morbidity probably increases the risk of complications or impaired recovery after discharge from the hospital. These social determinants of health might become clearer in a real-world versus a controlled research setting. This hypothesis is in line with two previous studies that found similar outcomes at discharge but impaired functional outcome at 90 days in women compared with men and supported by the similar NIHSS in men and women at 24 h in our study.9,11 Data on outcome at discharge were unfortunately not available. Impaired recovery after discharge from the hospital could be caused by differences between men and women during inpatient rehabilitation. However, a recent Canadian study comparing 9459 women and 10,684 men admitted to inpatient rehabilitation after stroke did not show a difference in length of stay, and women were more likely to be discharged home compared to men. 49 In conclusion, substantial sex differences in vessel characteristics exist in a large population of LVO patients in a real-world setting in the Netherlands. However, these sex differences do not affect radiological outcome and are therefore unlikely to explain the worse functional outcome in women. Our data, therefore, provide no argument that, based on neuroimaging characteristics, decisions on treatment with EVT should be taken differently in men and women with LVO.
Footnotes
Acknowledgements
We would like to thank the MR CLEAN Registry investigators (see Coinvestigators).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IW reports consulting fees from Philips, and is stockholder and inventor of a patent owned by Neurophyxia; PK is a co-founder and shareholder of in Steps B.V.; and is funded by GEMINI (www.dth-gemini.eu): a European Union’s Horizon research and innovation program (Grant Agreement Number: 101136438) and RadPath AI project (2021191). CM reports grants from TWIN Foundation during the conduct of the study (paid to institution), grants from CVON. Dutch Heart Foundation, grants from European Commission, grants from Healthcare Evaluation Netherlands, grants from Stryker outside the submitted work (paid to institution), and is shareholder of Nico-lab; MJHW reports a Vidi grant [project number 91717337] which is a personal grant from the Netherlands Organization of Scientific Research (NWO/ZonMw) and an Aspasia grant.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The MR CLEAN Registry was partly funded by TWIN Foundation, Erasmus MC University Medical Center, Maastricht University Medical Center, and Amsterdam University Medical Center.
Ethical approval
The MR CLEAN Registry was approved by the medical ethics committee of the Erasmus University Medical Center, Rotterdam, the Netherlands (MEC-2014-235).
Informed consent
Informed consent was not sought for the present study. As this is a registry study the medical ethics committee waived the need for obtaining informed consent.
Guarantor
AM.
Contributorship
AM, MW and MJHW researched literature and conceived the study. GH, HO and PK gained data for the study. HO and HL were involved in data analysis. AM wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Coinvestigators
Diederik WJ Dippel1; Aad van der Lugt2; Charles BLM Majoie3; Yvo BWEM Roos4; Robert J van Oostenbrugge5,41; Wim H van Zwam6,41; Jelis Boiten14; Jan Albert Vos8.
Ivo GH Jansen3; Maxim JHL Mulder1,2; Robert Jan B Goldhoorn5,6,41; Kars CJ Compagne2; Manon Kappelhof3; Josje Brouwer4; Sanne J den Hartog1,2,40; Wouter H Hinsenveld 5,6.
Diederik WJ Dippel1; Bob Roozenbeek1; Aad van der Lugt2; Adriaan CGM van Es2; Charles BLM Majoie3; Yvo BWEM Roos4; Bart J Emmer3; Jonathan M Coutinho4; Wouter J Schonewille7; Jan Albert Vos8; Marieke JH Wermer9; Marianne AA van Walderveen10; Julie Staals5,41; Robert J van Oostenbrugge5,41; Wim H van Zwam6,41; Jeannette Hofmeijer11; Jasper M Martens12; Geert J Lycklama à Nijeholt13; Jelis Boiten14; Sebastiaan F de Bruijn15; Lukas C van Dijk16; H Bart van der Worp17; Rob H Lo18; Ewoud J van Dijk19; Hieronymus D Boogaarts20; J de Vries22; Paul LM de Kort21; Julia van Tuijl21; Jo P Peluso26; Puck Fransen22; Jan SP van den Berg22; Boudewijn AAM van Hasselt23; Leo AM Aerden24; René J Dallinga25; Maarten Uyttenboogaart28; Omid Eschgi29; Reinoud PH Bokkers29; Tobien HCML Schreuder30; Roel JJ Heijboer31; Koos Keizer32; Lonneke SF Yo33; Heleen M den Hertog22; Tomas Bulut35; Paul JAM Brouwers34.
Charles BLM Majoie3(chair); Wim H van Zwam6,41; Aad van der Lugt2; Geert J Lycklama à Nijeholt13; Marianne AA van Walderveen10; Marieke ES Sprengers3; Sjoerd FM Jenniskens27; René van den Berg3; Albert J Yoo38; Ludo FM Beenen3; Alida A Postma6,42; Stefan D Roosendaal3; Bas FW van der Kallen13; Ido R van den Wijngaard13; Adriaan CGM van Es2; Bart J Emmer3; Jasper M Martens12; Lonneke SF Yo33; Jan Albert Vos8; Joost Bot36; Pieter-Jan van Doormaal2; Anton Meijer27; Elyas Ghariq13; Reinoud PH Bokkers29; Marc P van Proosdij37; G Menno Krietemeijer33; Jo P Peluso26; Hieronymus D Boogaarts20; Rob Lo18; Wouter Dinkelaar2; Auke PA Appelman29; Bas Hammer16; Sjoert Pegge27; Anouk van der Hoorn29; Saman Vinke20.
Diederik WJ Dippel1(chair); Aad van der Lugt2; Charles BLM Majoie3; Yvo BWEM Roos4; Robert J van Oostenbrugge5,41; Wim H van Zwam6,41; Geert J Lycklama à Nijeholt13; Jelis Boiten14; Jan Albert Vos8; Wouter J Schonewille7; Jeannette Hofmeijer11; Jasper M Martens12; H Bart van der Worp17; Rob H Lo18.
Robert J van Oostenbrugge5,41(chair); Jeannette Hofmeijer11; H Zwenneke Flach23.
Hester F. Lingsma40.
Naziha el Ghannouti1; Martin Sterrenberg1; Wilma Pellikaan7; Rita Sprengers4; Marjan Elfrink11; Michelle Simons11; Marjolein Vossers12; Joke de Meris14; Tamara Vermeulen14; Annet Geerlings19; Gina van Vemde22; Tiny Simons30; Gert Messchendorp28; Nynke Nicolaij28; Hester Bongenaar32; Karin Bodde24; Sandra Kleijn34; Jasmijn Lodico34; Hanneke Droste34; Maureen Wollaert5; Sabrina Verheesen5; D Jeurrissen5; Erna Bos9; Yvonne Drabbe15; Michelle Sandiman15; Nicoline Aaldering11; Berber Zweedijk17; Jocova Vervoort21; Eva Ponjee22; Sharon Romviel19; Karin Kanselaar19; Denn Barning10.
Esmee Venema40; Vicky Chalos1,40; Ralph R Geuskens3; Tim van Straaten19; Saliha Ergezen1; Roger RM Harmsma1; Daan Muijres1; Anouk de Jong1; Olvert A Berkhemer1,3,6; Anna MM Boers3,39; J Huguet3; PFC Groot3; Marieke A Mens3; Katinka R van Kranendonk3; Kilian M Treurniet3; Manon L Tolhuisen3,39; Heitor Alves3; Annick J Weterings3; Eleonora LF Kirkels3; Eva JHF Voogd11; Lieve M Schupp3; Sabine L Collette28,29; Adrien ED Groot4; Natalie E LeCouffe4; Praneeta R Konduri39; Haryadi Prasetya39; Nerea Arrarte-Terreros39; Lucas A Ramos39.
Department of Neurology1, Radiology2, Public Health40, Erasmus MC University Medical Center;
Department of Radiology and Nuclear Medicine3, Neurology4, Biomedical Engineering & Physics39,
Amsterdam UMC, location University of Amsterdam;
Department of Neurology5, Radiology & Nuclear Medicine6, Maastricht University Medical Center+; School for Cardiovascular Diseases Maastricht (CARIM)41; and MHeNs School for Mental Health and Neuroscience, Maastricht, the Netherlands42;
Department of Neurology7, Radiology8, Sint Antonius Hospital, Nieuwegein;
Department of Neurology9, Radiology10, Leiden University Medical Center;
Department of Neurology11, Radiology12, Rijnstate Hospital, Arnhem;
Department of Radiology13, Neurology14, Haaglanden MC, the Hague;
Department of Neurology15, Radiology16, HAGA Hospital, the Hague;
Department of Neurology17, Radiology18, University Medical Center Utrecht;
Department of Neurology19, Neurosurgery20, Radiology27, Radboud University Medical Center, Nijmegen;
Department of Neurology21, Radiology26, Elisabeth-TweeSteden ziekenhuis, Tilburg;
Department of Neurology22, Radiology23, Isala Klinieken, Zwolle;
Department of Neurology24, Radiology25, Reinier de Graaf Gasthuis, Delft;
Department of Neurology28, Radiology29, University Medical Center Groningen;
Department of Neurology30, Radiology31, Atrium Medical Center, Heerlen;
Department of Neurology32, Radiology33, Catharina Hospital, Eindhoven;
Department of Neurology34, Radiology35, Medisch Spectrum Twente, Enschede;
Department of Radiology36, Amsterdam UMC, Vrije Universiteit van Amsterdam, Amsterdam;
Department of Radiology37, Noordwest Ziekenhuisgroep, Alkmaar;
Department of Radiology38, Texas Stroke Institute, Texas, United States of America.
