Abstract
Background:
Acute blood pressure (BP) reduction is the first-line treatment for acute spontaneous intracerebral hemorrhage (ICH); however, recent research suggests that intensive BP reduction along with cerebral small vessel disease (cSVD) is a risk factor for remote DWI lesions (RDWILs). We aimed to delineate the interplay between cSVD and BP reduction therapy on the risk of RDWILs.
Methods:
We enrolled 303 patients who underwent brain magnetic resonance imaging within 7 days after acute spontaneous ICH. RDWILs were categorized as occurring in borderzone (BZ) or non-BZ areas. We examined the effect of cSVD, acute BP reduction, and their interaction on RDWILs.
Results:
RDWILs were observed in 34 (11%) patients (59.8 ± 10.3-years-old, 24% male). RDWILs were associated with a larger acute weighted average mean arterial pressure (MAP) reduction in the initial 24 h after ICH onset and a higher total cerebral microbleed (CMB) count. Intensive MAP changes (odds ratio (OR) per 10 mmHg 1.76, 95% confidence interval (CI) 1.03–3.20), total CMBs burden (OR per 10 CMBs 1.21, 95% CI 1.08–1.39), and presence of lobar CMBs (OR 7.33, 95% CI 1.59–55.6) were risk factors for RDWILs at BZ, but not at non-BZ. Furthermore, a significant interaction was observed between lobar CMBs and MAP reduction on increased risk of RDWILs at BZ (p = 0.030).
Conclusion:
cSVD modulates the effect of acute BP reduction on the risk of RDWILs. Patients with extensive microangiopathy have a higher risk of developing cerebral ischemic changes in BZ during unstable hemodynamic status.
Keywords
Introduction
Spontaneous intracerebral hemorrhage (ICH) is a common and devastating cerebrovascular disease that is mainly attributed to cerebral small vessel disease (cSVD), including the most common form, hypertensive microangiopathy, and cerebral amyloid angiopathy (CAA). 1 Intensive blood pressure (BP) reduction is considered a safe and gold-standard treatment for acute spontaneous ICH as studies suggest that adequate BP control is associated with better functional outcomes and a lower risk of hematoma expansion.1,2 Nevertheless, the evidence regarding the optimal methods of BP reduction and target BP goal remain conflicting, especially for patients who present with an extremely initial high BP.2,3 In addition, a larger acute BP reduction has been associated with the occurrence of small acute ischemic insults that can be detected by diffusion-weighted imaging (DWI) and that are spatially remote to the hematoma; thus, these lesions have been named remote DWI lesions (RDWILs).4–6
RDWILs are found in around 11%–41% of patients with acute spontaneous ICH.4–6 Although they are usually small and asymptomatic, RDWILs are associated with a higher risk of recurrent cerebrovascular events, higher rate of vascular death, and worse functional outcomes at 3 months.5,7,8 The underlying mechanisms are heterogenous and cSVD pathology plays a major role in the formation of RDWILs.4–6 Emerging research has highlighted that radiological markers of cSVD, including the burden of cerebral microbleeds (CMBs), extension of cortical superficial siderosis (cSS), white matter hyperintensity (WMH) volume, the degree of enlarged perivascular space (EPVS), lacunes count, and total cSVD score are associated with the risk of RDWILs.5,7 Intensive BP reduction has been proposed to lead to cerebral hypoperfusion and consequent ischemic changes at vulnerable areas due to impaired cerebral autoregulation resulting from cSVD. 5 Thus, the cerebral border zones (BZ), which are located at the junction of the distal territories of two main cerebral arteries, may be the regions that are most susceptible to ischemia during acute hemodynamic compromised status due to their low perfusion pressure and poor collaterals. 9
Although intensive BP reduction and cSVD have been found to be independently associated with RDWILs, the influence of the interaction between cSVD and BP reduction is rarely discussed. Therefore, this study aimed to investigate the relationship of RDWILs with cSVD and aggressive BP reduction in the acute stage of spontaneous ICH. We hypothesized that cSVD is one of the main modulating factors associated with RDWILs attributed to intensive BP-lowering therapy. To verify our hypothesis, we categorized RDWILs according to their location into BZ and non-BZ areas to investigate the impact of cSVD, acute BP reduction, and the interaction between cSVD and acute BP reduction on RDWILs in brain regions that are susceptible to hypoperfusion during the acute phase of ICH.
Methods
Study participants
The primary data used in this study were obtained from the National Taiwan University Hospital (NTUH) Stroke Registry, which is a database that prospectively includes all patients with stroke from a tertiary referral center in Taiwan. 10 The Research Ethnics Committee of the NTUH approved the registry. We reviewed the records of 1450 consecutive adult patients (⩾18-years-old) who were admitted to NTUH with acute ICH between February 2012 and December 2019. Patients with known or suspected secondary causes of ICH and patients who did not undergo brain MRI studies within 7 days after the onset of ICH were excluded. Fourty-nine (15.4%) of the remaining 318 patients with acute spontaneous ICH had DWI lesions. Patients who had peri-hematoma DWI lesions, DWI lesions located in the territory of vessels with ⩾50% intracranial/extracranial arterial stenosis or occlusion, or with a history of atrial fibrillation were excluded as these DWI lesions may be related to hematoma, embolism, or arterial stenosis. A final sample of 303 eligible patients with acute spontaneous ICH were included in the final analysis (Figure 1).
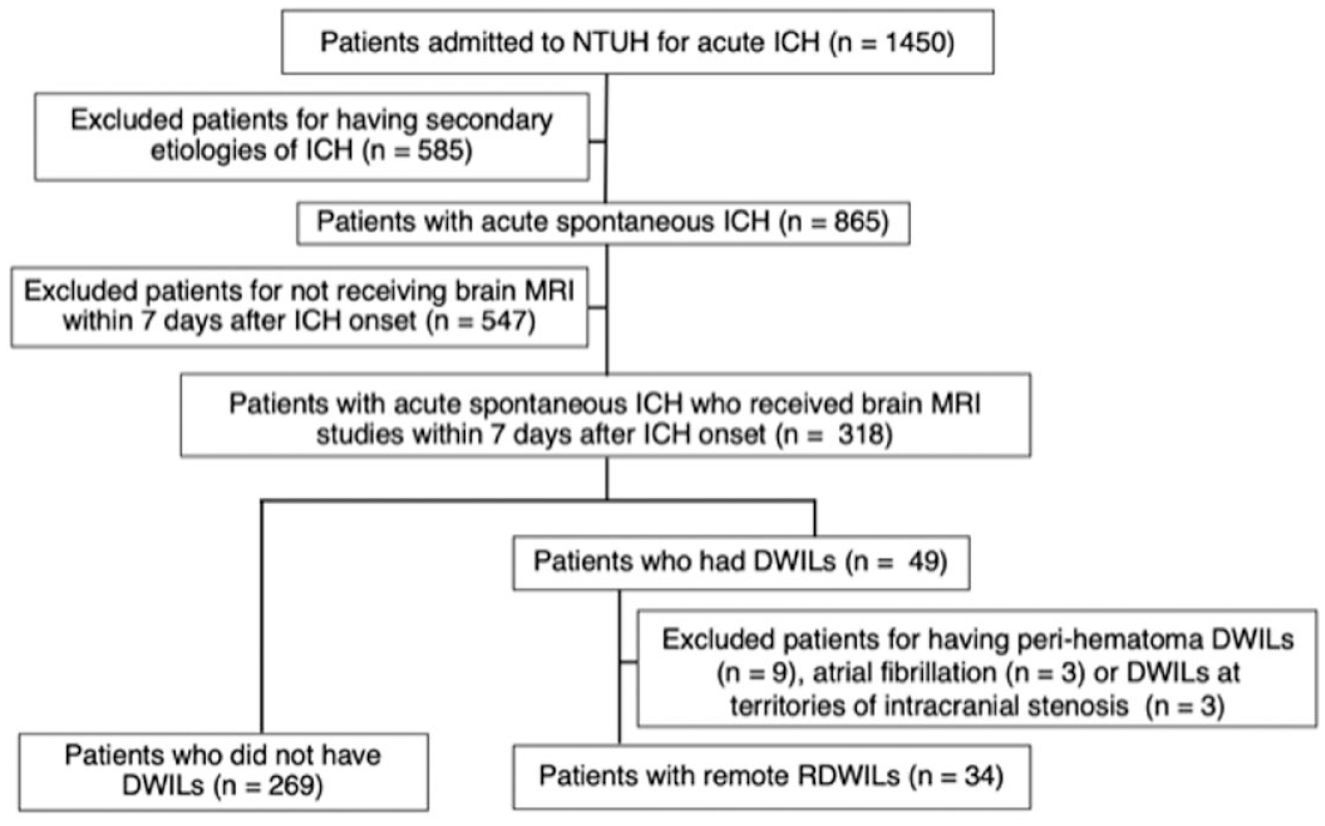
Flow diagram of the study participants.
Demographic information, medical history, the results of physical and laboratory examinations, and BP records were obtained from the patients’ clinical documentation. All patients underwent a BP assessment when they initially presented to the hospital and every hour after admission for at least 24 h. Intensive BP control with target systolic BP of <140 or 160 mmHg depending on each patient’s clinical condition was obtained using intravenous anti-hypertensive medication. 3 The BP values at 0 (i.e. initial BP in the emergency department), 1, 3, 6, 12, and 24 h after the onset of ICH were obtained and manually calculated. Weighted average (wavg) BP change was measured as the area under the curve of the mean arterial pressure (MAP) change in relation to initial MAP over 24 h. 11 Further detailed methods of the patient enrollment and BP management protocols are described in the Supplemental Methods section.
Image analysis
All included patients underwent head computed tomography and 1.5 -T or 3 -T brain MRI. RDWILs were defined as lesions that are hyperintense on a DWI sequence, hypointense on ADC series, and located more than 10 mm away from the acute hematoma. 5 We classified the locations of RDWILs into BZ and non-BZ areas. BZ are regions supplied by the junctions of the distal branches of large cerebral arteries, including the anterior cerebral artery, middle cerebral artery, posterior cerebral artery, Heubner artery, lenticulostriate artery, and anterior choroidal artery (Figure 2). 9 Two trained raters (Chen S. J. and Tsai H. H.) who were blinded to the patients’ clinical information independently reviewed the MRI scans for RDWILs. Excellent interrater agreement was observed for the RDWILs count (kappa value 0.96, 95% confidence interval (CI) 0.92–1.00) and the locations of the lesions (kappa value 0.90, 95% CI 0.78–1.00).
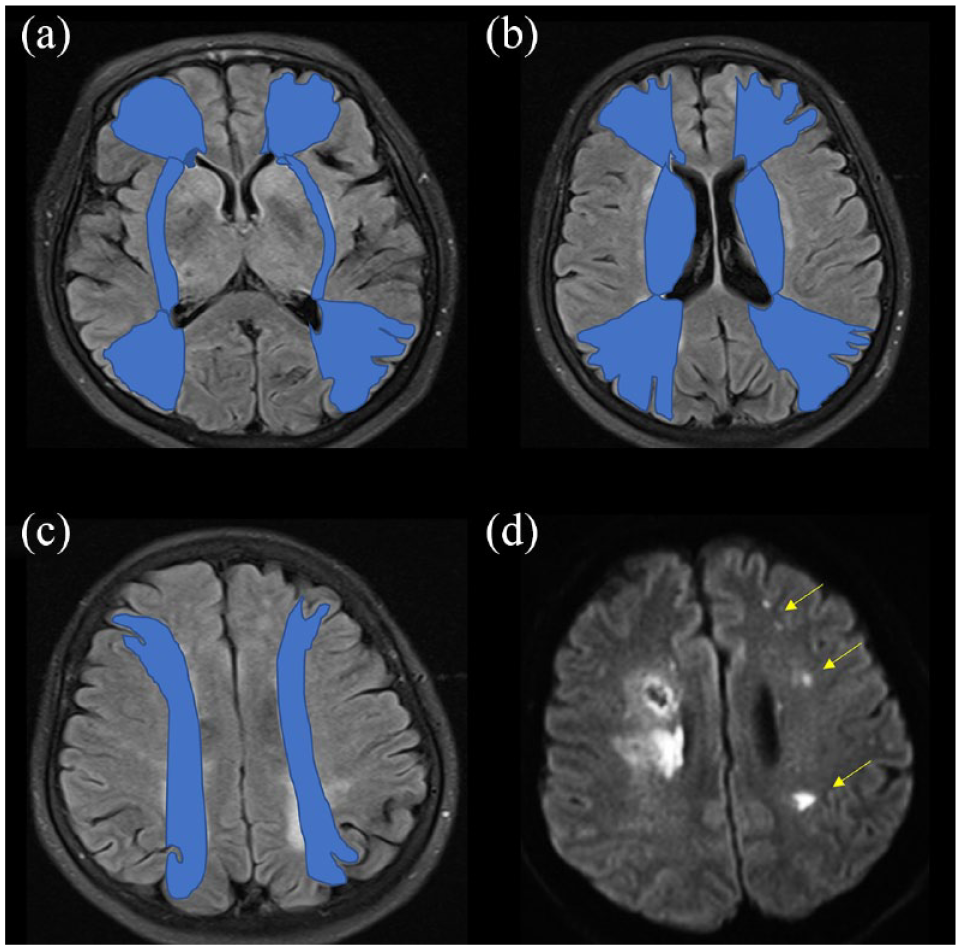
Definition of borderzone RDWILs: (a–c) distribution of borderzone areas on an MRI FLAIR sequence and (d) examples of borderzone RDWILs on a diffusion weighted imaging series (arrows).
MRI markers of cSVD were evaluated as described previously, according to the STRIVE Criteria.12,13 Briefly, cerebral ICH, CMBs, and cSS were identified and counted on the SWI sequences according to the current consensus criteria. 14 CMBs were categorized as lobar or deep CMBs according to their topography, as we previously described. 13 Lacunes were defined as fluid-filled cavities with a diameter of between 3 and 15 mm and were categorized as deep or lobar lacunes based on previous papers. 12 The detailed image analysis method is provided in the Supplemental Methods.
Statistical analysis
Continuous variables are expressed as means ± standard deviations and categorical variables as numbers (percentages). Demographic, clinical, and neuroimaging features were compared between patients with and without RDWILs using the Mann-Whitney test for continuous variables and Fisher’s exact test for categorical factors. Multivariable logistic regression models were applied to assess the impact of the degree of acute BP reduction, radiological markers of cSVD, and their interaction on the risk of RDWILs of interest with adjustment for covariates. Two-tailed p-values ⩽0.05 were considered statistically significant. All analyses were performed using Prism 9 (GraphPad Software, La Jolla, CA, USA).
Results
RDWILs are associated with acute BP drop and cSVD
Among the 303 enrolled patients who suffered acute spontaneous ICH, 34 (11%) had RDWILs. The clinical characteristics, BP variables, and imaging features of the study participants are listed in Table 1 and Supplemental Table 1. The BP trajectories over the initial 24 h for patients with and without RDWILs are shown in Supplemental Figure 1. Patients with and without RDWILs had similar demographics, frequencies of traditional vascular risk factors, and volumes of acute hematoma. Patients with RDWILs had a higher initial SBP and a larger degree of wavg MAP reduction over 24 h (Table 1). Analysis of cSVD markers on neuroimaging studies showed that patients with RDWILs had a higher total CMB burden; however, the severity of cSS, EPVS, WMH volume, and lacunes number did not differ significantly between groups. In univariable logistic regression model (Table 2), a higher wavg MAP change, higher initial SBP, larger total CMB count, and a higher number of deep lacunes were associated with RDWILs. After adjustment for covariates, a higher wavg MAP reduction over 24 h after ICH onset (odds ratio (OR) per 10 mmHg 1.51, 95% CI 1.04–2.25, p = 0.036) and higher total CMB burden (OR per 10 CMBs 1.12, 95% CI 1.02–1.25, p = 0.025) remained significantly associated with the risk of RDWILs (Table 2).
Demographics and characteristics of participants.
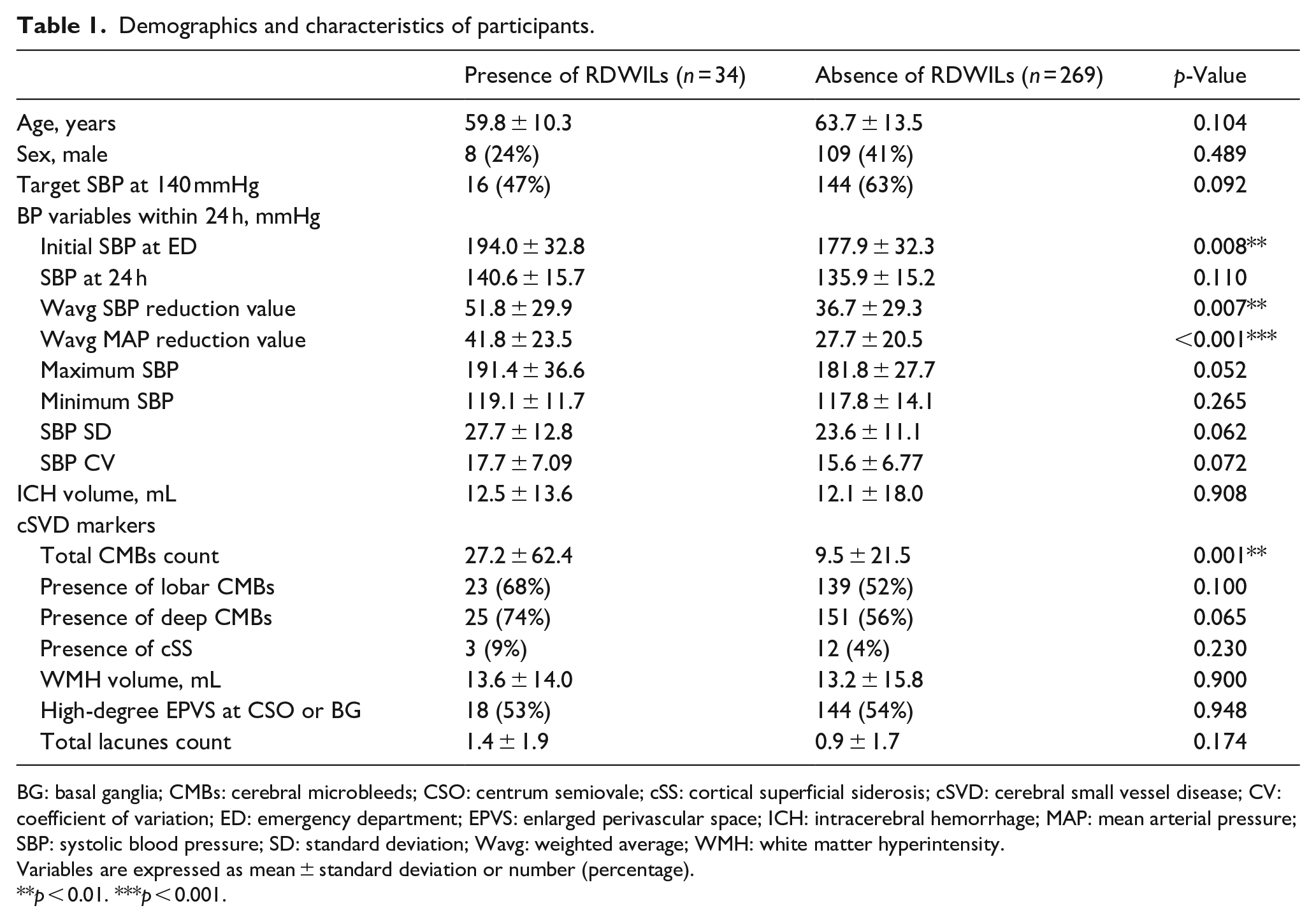
BG: basal ganglia; CMBs: cerebral microbleeds; CSO: centrum semiovale; cSS: cortical superficial siderosis; cSVD: cerebral small vessel disease; CV: coefficient of variation; ED: emergency department; EPVS: enlarged perivascular space; ICH: intracerebral hemorrhage; MAP: mean arterial pressure; SBP: systolic blood pressure; SD: standard deviation; Wavg: weighted average; WMH: white matter hyperintensity.
Variables are expressed as mean ± standard deviation or number (percentage).
p < 0.01. ***p < 0.001.
Factors related to development of RDWILs.
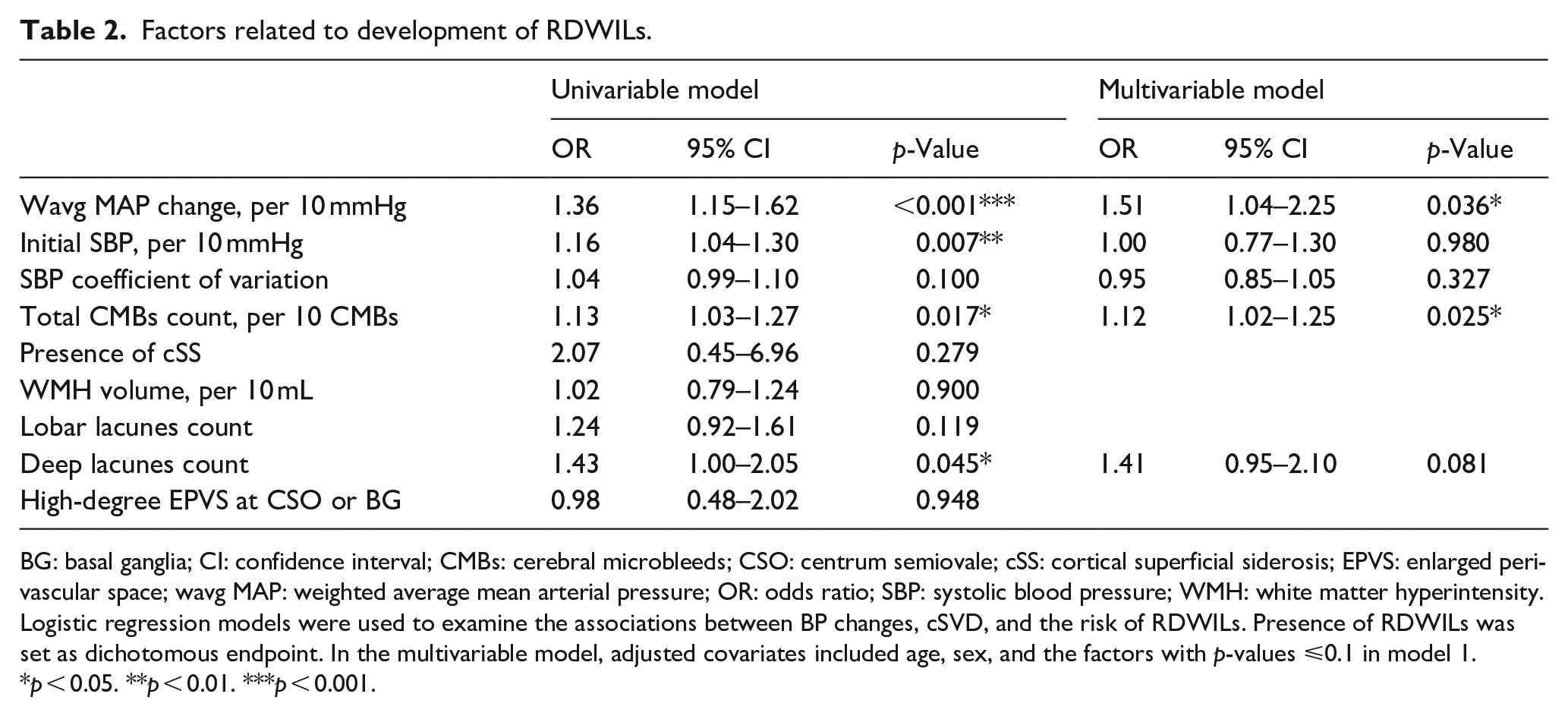
BG: basal ganglia; CI: confidence interval; CMBs: cerebral microbleeds; CSO: centrum semiovale; cSS: cortical superficial siderosis; EPVS: enlarged perivascular space; wavg MAP: weighted average mean arterial pressure; OR: odds ratio; SBP: systolic blood pressure; WMH: white matter hyperintensity.
Logistic regression models were used to examine the associations between BP changes, cSVD, and the risk of RDWILs. Presence of RDWILs was set as dichotomous endpoint. In the multivariable model, adjusted covariates included age, sex, and the factors with p-values ⩽0.1 in model 1.
p < 0.05. **p < 0.01. ***p < 0.001.
Aggressive BP reduction is a risk factor for the occurrence of RDWILs only at borderzone areas
To further delineate the associations between acute intensive BP reduction, cSVD, and RDWILs related to hemodynamic changes, we classified RDWILs according to their topography into RDWILs at BZ (n = 12) and non-BZ (n = 22) areas. Multivariable logistic regression model (Table 3, model 1) showed that an acute change in MAP was a risk factor for RDWILs at BZ (OR per 10 mmHg 1.76, 95% CI 1.03–3.20, p = 0.048) after adjustment for age, sex and initial SBP, but was not related to the development of RDWILs at non-BZ areas (OR per 10 mmHg 1.11, 95% CI 0.74–1.67, p = 0.618). Furthermore, after adjustment for age, sex, BP variables (wavg MAP and initial SBP), and radiological markers of cSVD (Table 3, model 2), a higher total number of CMBs (OR per 10 CMBs 1.21, 95% CI 1.08–1.39, p = 0.003), higher number of lobar lacunes (OR 4.93, 95% CI 1.40–20.2, p = 0.016), and the presence of lobar CMBs (OR 7.33, 95% CI 1.59–55.6, p = 0.022) were associated with the occurrence of RDWILs at BZ. The number of deep lacunes was the only factor associated with the development of RDWILs at non-BZ regions (OR 1.50, 95% CI 1.00–2.22, p = 0.042).
Associations between BP, radiological markers of cSVD, and development of RDWILs at BZ and non-BZ.
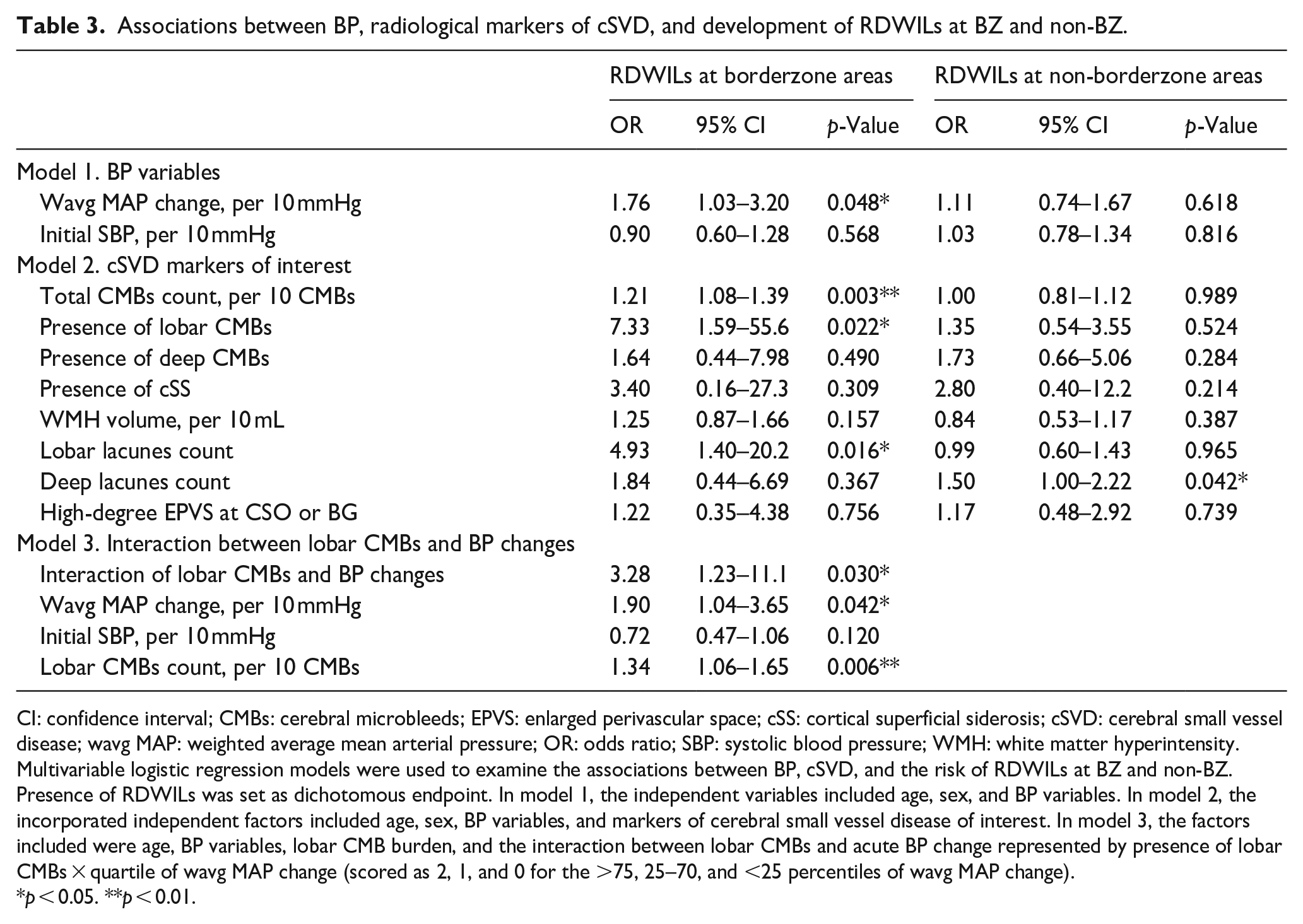
CI: confidence interval; CMBs: cerebral microbleeds; EPVS: enlarged perivascular space; cSS: cortical superficial siderosis; cSVD: cerebral small vessel disease; wavg MAP: weighted average mean arterial pressure; OR: odds ratio; SBP: systolic blood pressure; WMH: white matter hyperintensity.
Multivariable logistic regression models were used to examine the associations between BP, cSVD, and the risk of RDWILs at BZ and non-BZ. Presence of RDWILs was set as dichotomous endpoint. In model 1, the independent variables included age, sex, and BP variables. In model 2, the incorporated independent factors included age, sex, BP variables, and markers of cerebral small vessel disease of interest. In model 3, the factors included were age, BP variables, lobar CMB burden, and the interaction between lobar CMBs and acute BP change represented by presence of lobar CMBs × quartile of wavg MAP change (scored as 2, 1, and 0 for the >75, 25–70, and <25 percentiles of wavg MAP change).
p < 0.05. **p < 0.01.
cSVD aggravates the effect of intensive BP reduction on RDWILs at borderzone areas
We next analyzed the effect of the interaction between cSVD and acute BP changes on RDWILs at BZ. The wavg MAP changes were categorized using the <25, 25–75, and >75 percentiles as 0, 1, or 2 points. The multivariable logistic regression model showed that the presence of lobar CMBs modulates the impact of wavg MAP reduction in the initial 24 h after ICH onset on the risk of developing BZ RDWILs (presence of lobar CMBs × quartile of wavg MAP change: OR 3.28, 95% CI 1.23–11.1, p = 0.030, Table 3, model 3). Subgroup analysis of patients with and without lobar CMBs showed that a higher wavg MAP change was associated with an increased risk of BZ RDWILs in patients with lobar CMBs (OR 2.44, 95% CI 1.28–5.49), but not in patients without lobar CMBs (OR 1.12, 95% CI 0.37–4.93, Figure 3(a)). The respective proportions of RDWILs at BZ and non-BZ classified by the degree of wavg MAP reduction over 24 h after ICH onset in patients with and without lobar CMBs are illustrated in Figure 3(b).
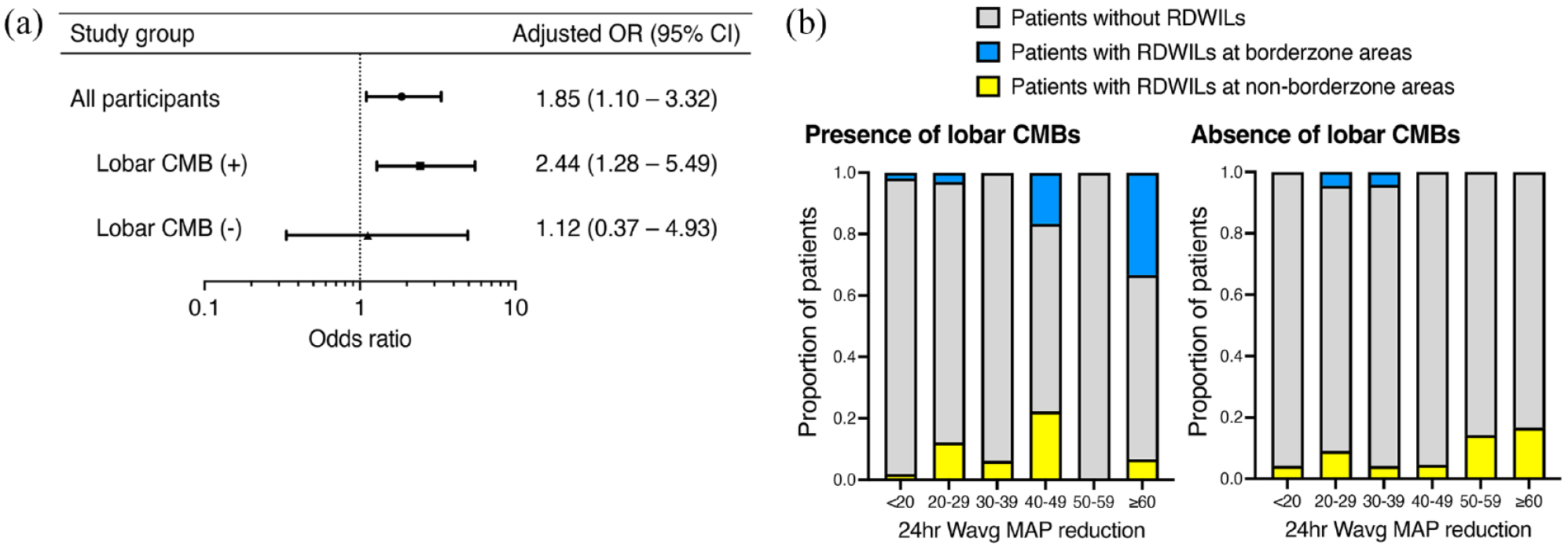
Associations between RDWILs, acute BP reduction, and cSVD: (a) Multiple logistic regression analysis of the influence of the wavg MAP reduction over the initial 24 h after ICH onset on the risk of BZ-RDWILs in the groups of patients with and without lobar CMBs. The model was adjusted for age and initial SBP value. (b) Proportions of patients who developed RDWILs at BZ and non-BZ classified by the degree of wavg MAP reduction over the initial 24 h in patients with and without lobar CMB.
Discussion
This study investigated the relationship between acute BP reduction, cSVD, and the development of RDWILs with consideration of their locations at BZ and non-BZ areas. First, we showed that the factors associated with RDWILs included a larger degree of acute BP reduction and a higher number of CMBs, in agreement with previous literature.4,5 Second, we demonstrated that intensive BP reduction was significantly related to the risk of RDWILs at BZ but not at non-BZ, which supports the hypothesis that RDWILs attributed to BP changes may occur as a consequence of hemodynamic compromise. Lastly, the presence of lobar CMBs modifies the effect of acute BP reduction on RDWILs at BZ, which indicates that microangiopathic processes could increase the susceptibility to ischemic insults at BZ during acute BP-lowering treatment. Thus, patients harboring lobar CMBs are at risk of developing RDWILs during acute BP reduction. Together, our findings provide mechanistic insights to suggest the development of BZ RDWILs can be attributed to the effect of hemodynamic compromise combined with microangiopathy of the cortical vessels. These results also prompt further clinical studies to investigate whether the target BP should be individualized in the acute stage of spontaneous ICH depending on the patient’s lobar CMB status.
DWI can detect ischemic changes with good accuracy and has been increasingly applied to uncover subclinical ischemic changes located at perihematomal and distant areas in patients with acute ICH. 15 Long-term follow-up studies have shown that RDWILs can evolve into permanent cerebral infarcts, lacunes, and WMH, and predispose patients with ICH to poorer functional prognoses. 16 The mechanisms that underlie the development of RDWILs are, to date, unconfirmed. RDWILs were proposed to be surrogate markers of ischemic processes in microangiopathy due to their close associations with the severity of cSVD.6,15 Nevertheless, the causes of RDWILs could be heterogenous, with several other factors also contributing to the insults. These factors include hemodynamic instability, large-artery atherosclerosis, embolic events, and venous drainage disorders. 17 Furthermore, while accumulating evidence suggests a higher initial BP and larger acute reduction in BP are associated with a higher risk of RDWILs in patients with acute spontaneous ICH, other studies reported no associations between BP changes and RDWILs.5,11 Therefore, deeper insight into the characteristics of and mechanisms that lead to RDWILs need to be obtained in order to develop a potential preventive strategy. This study is the first to categorize RDWILs into BZ and non-BZ subtypes, and we demonstrated that a larger acute reduction in MAP increased the risk of RDWILs, but only at BZ. Our finding of a relationship between BP alterations and a specific topographical distribution of RDWILs may partially explain the discrepancies in the previous literature. BZ receive their blood supply from the most marginal branches of the cerebral arteries and have poor collaterals. 9 During compromised hemodynamic status, ischemic changes typically develop at BZ as a result of cerebral hypoperfusion and decreased clearance of emboli due to insufficient blood supply. 9 In our study, both lobar CMBs and a larger BP reduction, as well as their interaction, were significantly associated with the risk of BZ RDWILs; these important findings imply that compromised hemodynamic status may lead to adverse events, especially in patients harboring cSVD involving small cortical vessels. The impaired cerebrovascular reactivity and stenotic arterioles in long cortical perforators may create ischemic penumbra at vulnerable areas, such as BZ, that may cause further ischemic insults during hemodynamic change.18,19
Our study also showed that the subtypes of cSVD markers associated with RDWILs at BZ and non-BZ areas are different, which suggests that BZ and non-BZ RDWILs may be influenced by distinct underlying microangiopathic processes. While lobar CMBs and lobar lacune were associated with RDWILs at BZ, only deep lacune were associated with RDWILs at non-BZ. Moreover, the magnitude of the BP change was not associated with the development of non-BZ RDWILs. These findings suggest that RDWILs at non-BZ are probably related to microangiopathic process per se, especially hypertensive arteriosclerosis, and less closely related to the effects of hemodynamic instability or impaired autoregulation in the deep perforators. The presence of lobar CMBs and lacunes reflects an underlying microangiopathy involving the cortical and subcortical arterioles. 13 Compared to the short, deep, penetrating arteries in the brainstem, thalamus, and BG, the vasculature supplying the subcortical white matter is comprised of long arteries with fewer anastomoses and lower BP gradients, which are thus more vulnerable to ischemia during hemodynamic compromise. 18 Interestingly, only one of the 12 patients with RDWILs at BZ had strictly lobar ICH and CMBs, indicating underlying CAA. The majority of patients with RDWILs at BZs had mixed-location ICH and CMBs in both deep and lobar areas, which are believed to represent severe, chronic hypertensive microangiopathy involving the cortical arterioles in addition to the deep-penetrating vessels. 13 A recent meta-analysis showed that the prevalence of RDWILs is similar in CAA- and hypertensive vasculopathy related-ICH; however, up-to-date information on the topographical distribution of these lesions in different subtypes of cSVD is scarce. 9 We cannot exclude the possibility that CAA contributes to the higher risk of RDWILs at BZ in the current study. Nevertheless, the presence of cSS, a radiological hallmark of CAA, was not related to RDWILs at BZ in our cohort. 14 Additional research in subjects with strictly lobar ICH/CMBs and analysis of other CAA-related biomarkers are required to understand the associations between the severity of CAA and the impact of intensive BP reduction on cerebral ischemic changes.
Our results reinforce the close connection between acute BP reduction and the development of RDWILs. Intensive BP reduction is currently the first-line treatment for patients with acute spontaneous ICH. 1 However, the conclusions of research regarding the clinical benefits of aggressive BP-lowering treatment are heterogenous. While some studies have reported a lower BP target is associated with smaller hematoma expansion and better functional recovery, other studies have demonstrated conflicting results.2,3 One potential explanation for these discrepancies is variation in the patients’ characteristics and severity of cSVD between ethnicities and cohorts. 20 Our results highlight that the severity and extensiveness of cSVD may affect whether patients are susceptible to ischemia during BP-lowering treatment. Therefore, individualized therapeutic targets are necessary in the acute stage of ICH and cSVD status should be considered when setting the optimal BP goal.
Our study has several limitations. First, we enrolled patients who underwent an MRI study within 7 days after ICH onset; thus, we excluded patients with unstable hemodynamic status who could not tolerate MRI or with contraindications for MRI, for example, cardiac pacemakers, which may lead to selection bias. Nevertheless, limiting the MRI study period to 7 days increased the homogeneity of our cohort. Second, because large-artery stenosis and cardiac emboli have been proposed to be major causes of BZ ischemia, we excluded patients with RDWILs in the territory of large artery stenosis or with known cardioembolic sources, which may limit the generalizability of our study. 9 However, this enrollment strategy decreased the risk of possible confounders due to a heterogenous study population and maintained our focus on the impact of BP and cSVD on RDWILs. Third, this was a retrospective observational study. Although all patients were treated using a standardized BP-lowering treatment protocol, variations in the drug responses between individuals may have led to differences in the medication dosages and affected our results. Fourth, as this study had a cross-sectional design, we could not confirm a causal relationship between BP reduction and the formation of RDWILs at BZ. Due to the small sample size, our results only provide exploratory data to indicate that the underlying etiologies of RDWILs differ depending on the location of the lesions. Further larger, prospective studies are needed to verify the impact of the interaction between cSVD and intensive BP reduction on RDWILs and to determine the optimal BP treatment goal in consideration of each individual’s underlying microangiopathy.
In conclusion, this study suggests that cSVD, overly aggressive BP reduction, and their interaction are associated with the development of RDWILs at BZ. While intensive BP reduction is the gold-standard treatment for acute spontaneous ICH, underlying extensive microangiopathy may impair cerebral autoregulation and predispose patients to the formation of ischemic lesions during aggressive BP reduction treatment. The presence of lobar CMBs may be a red-flag sign for a high-risk of RDWILs during acute BP reduction. Future studies that include a risk-benefit assessment are essential to explore appropriate BP-lowering measures for different cSVD burdens in acute spontaneous ICH.
Supplemental Material
sj-docx-2-eso-10.1177_23969873231170989 – Supplemental material for Interaction between cerebral small vessel disease, blood pressure, and remote ischemic lesions in acute spontaneous intracerebral hemorrhage
Supplemental material, sj-docx-2-eso-10.1177_23969873231170989 for Interaction between cerebral small vessel disease, blood pressure, and remote ischemic lesions in acute spontaneous intracerebral hemorrhage by Szu-Ju Chen, Hsin-Hsi Tsai, Yen-Ling Lo, Ya-Fang Chen, Sung-Chun Tang, Jiann-Shing Jeng and Li-Kai Tsai in European Stroke Journal
Supplemental Material
sj-docx-3-eso-10.1177_23969873231170989 – Supplemental material for Interaction between cerebral small vessel disease, blood pressure, and remote ischemic lesions in acute spontaneous intracerebral hemorrhage
Supplemental material, sj-docx-3-eso-10.1177_23969873231170989 for Interaction between cerebral small vessel disease, blood pressure, and remote ischemic lesions in acute spontaneous intracerebral hemorrhage by Szu-Ju Chen, Hsin-Hsi Tsai, Yen-Ling Lo, Ya-Fang Chen, Sung-Chun Tang, Jiann-Shing Jeng and Li-Kai Tsai in European Stroke Journal
Supplemental Material
sj-jpg-1-eso-10.1177_23969873231170989 – Supplemental material for Interaction between cerebral small vessel disease, blood pressure, and remote ischemic lesions in acute spontaneous intracerebral hemorrhage
Supplemental material, sj-jpg-1-eso-10.1177_23969873231170989 for Interaction between cerebral small vessel disease, blood pressure, and remote ischemic lesions in acute spontaneous intracerebral hemorrhage by Szu-Ju Chen, Hsin-Hsi Tsai, Yen-Ling Lo, Ya-Fang Chen, Sung-Chun Tang, Jiann-Shing Jeng and Li-Kai Tsai in European Stroke Journal
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Taiwan National Science and Technology Council (Tsai HH, MOST NSTC 111-2628-B-002-044 and 112-2923-B-002-001-MY3).
Ethical approval
This study was performed with the approval of the institutional review board of NTUH and in accordance with their guidelines.
Informed consent
Written informed consent was waived from the institutional review board of NTUH.
Guarantor
Hsin-Hsi Tsai.
Contributorship
Conception and design: Szu-Ju Chen, Hsin-Hsi Tsai, Li-Kai Tsai. Collection and assembly of data: Szu-Ju Chen, Hsin-His Tsai, Yen-Ling Lo. Data analysis and interpretation: Szu-Ju Chen, Hsin-Hsi Tsai, Li-Kai Tsai. Critical revisions and supervision: Ya-Fang Chen, Sung-Chun Tang, Jiann-Shing Jeng, Li-Kai Tsai. Final approval of manuscript: Szu-Ju Chen, Hsin-His Tsai, Yen-Ling Lo, Ya-Fang Chen, Sung-Chun Tang, Jiann-Shing Jeng, Li-Kai Tsai.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
