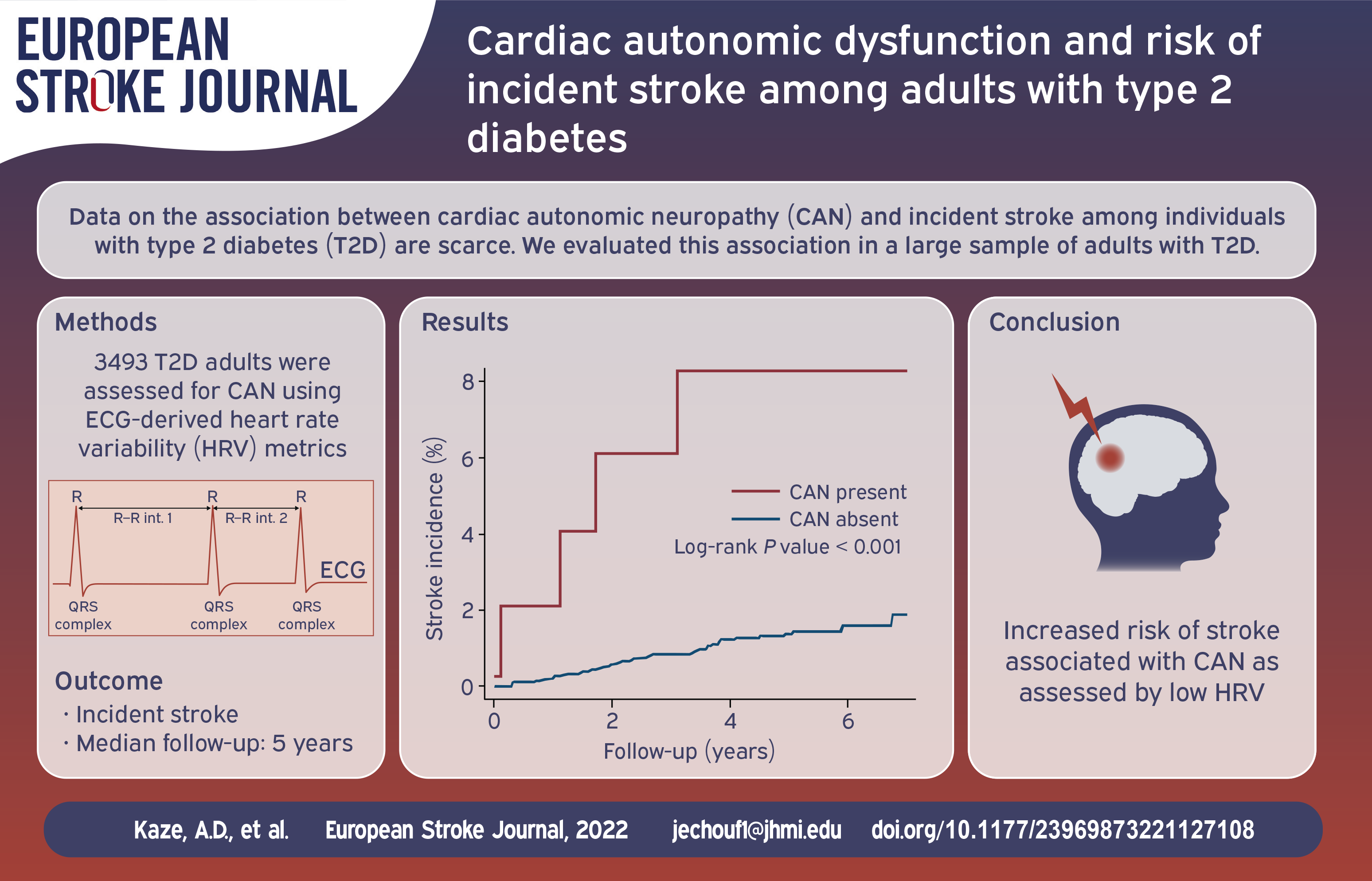
Abstract
Introduction:
There is a dearth of data on the association between cardiac autonomic neuropathy (CAN) with incident stroke among individuals with diabetes mellitus. We evaluated this association in a large sample of adults with type 2 diabetes.
Patients and methods:
Participants with type 2 diabetes from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study without atherosclerotic cardiovascular disease at baseline were included. CAN was assessed at baseline by heart rate variability (HRV) indices and QT index (QTI) calculated from 10-s resting electrocardiograms. HRV was assessed using standard deviation of all normal-to-normal R-Rs intervals (SDNN) and root mean square of successive differences between normal-to-normal R-R intervals (rMSSD). CAN was defined based on several composite measures of SDNN, QTI, resting heart rate and peripheral neuropathy. We used Cox proportional hazards regression to generate hazard ratios (HR) and 95% confidence intervals (CI) for incident stroke in relation to CAN.
Results:
A total of 3493 participants (mean age 62.2 years, 44.5% women, 62.9% White) were included. Over a median follow-up of 5.0 years, 50 stroke cases occurred (incidence rate 3.0/1000 person-years [95% CI 2.2–3.9]). After adjusting for confounders, low HRV was associated with a higher risk of stroke (HR of 2.20 [95% CI 1.23–3.93] and 1.88 [95% CI 1.04–3.41] for low SDNN and rMSSD, respectively). Participants with CAN (defined as lowest quartile of SDNN and highest quartiles of QTI and heart rate) had a 5.7-fold greater risk of stroke (HR 5.70, 95% CI 2.49–13.08).
Discussion and conclusion:
CAN was independently associated with an increased risk of incident stroke in a large cohort of adults with type 2 diabetes.
Introduction
Stroke affects more than 700,000 people every year in the USA and is responsible for huge societal costs and adult disability.1,2 The incidence of stroke is especially higher among people with diabetes mellitus who have a three-fold higher age-adjusted risk of stroke compared to those without diabetes. 2 Additionally, diabetes mellitus is associated with a greater mortality following stroke. 2 While the high burden of traditional cardiovascular disease (CVD) risk factors may partly explain the higher risk of stroke among those with type 2 diabetes, cardiac autonomic dysfunction might play a role.2,3 Indeed, extant evidence suggests that cardiac autonomic neuropathy (CAN) may be associated with increased risks of stroke as well as poststroke morbidity and mortality in the general population.3,4 Moreover, autonomic nervous system dysfunction is highly prevalent in type 2 diabetes compared to the general population. 5
While autonomic dysfunction has been found to predict poor functional outcomes after acute stroke, 6 there is a paucity of epidemiological data on the association of CAN with the incidence of new strokes among adults with type 2 diabetes. We aimed to evaluate the associations of CAN measures with incident stroke in type 2 diabetes, using data from the Action to Control Cardiovascular Risk in Diabetes (ACCORD) study. We hypothesized that CAN would be associated with a greater risk of stroke.
Materials and methods
The data used for the analyses in this article are available at the National Institutes of Health’s Biologic Specimen and Data Repository Information Coordinating Center (BioLINCC).
Study design
The ACCORD study was a randomized double two-by-two factorial trial that enrolled 10,251 people with type 2 diabetes at 77 locations across the US and Canada. The recruitment occurred in two phases with 1174 participants enrolled from January 2001 to June 2001, and 9077 participants from February 2003 to October 2005. They were assigned to either an intensive glucose lowering arm with a goal of a glycated hemoglobin (HbA1C) <6% or a standard treatment arm with an HbA1C goal of 7.0%–7.9%, as well specific blood pressure and lipid intervention arms. The design and methods of the ACCORD study have been reported elswhere. 7
For the purpose of this analysis, participants were excluded if they had a history of atherosclerotic cardiovascular disease (ASCVD) (defined as prior myocardial infarction, angina, stroke, history of coronary revascularization, carotid or peripheral revascularization) at baseline (n = 3609), artificial pacemaker (n = 23), atrioventricular conduction defect (n = 232), atrial fibrillation/flutter (n = 73), premature beats and other arrhythmias (n = 456), missing electrocardiogram (ECG, n = 650), as well as poor quality ECG (n = 236). We further excluded participants (n = 1479) taking medications that can affect heart rate variability (HRV, including calcium-channel blockers, digitalis, beta blockers and other antiarrhythmic medications). After the relevant exclusions, 3493 participants were left of our analyses. The study inclusion process is displayed in Supplemental Figure S1.
Assessment of cardiac autonomic neuropathy
HRV was assessed at baseline using digitalized 12-lead ECG. After an overnight fast, participants were asked to lie supine and ECGs were recorded over 10 consecutive seconds (GE MAC 1200 electrocardiograph system). 8 ECGs were transferred via phone line to the reading center where they were analyzed and reviewed to ensure technical quality. Two HRV time-domain indices were derived from the ECG recordings: standard deviation of all normal-to-normal R-Rs intervals (SDNN) and root mean square of successive differences between normal-to-normal R-R intervals (rMSSD). Additionally, resting heart rate (HR) and the QT index (QTI) were derived from simultaneous recordings. QTI was derived as observed/predicted QT duration with the predicted QT duration computed based on the Bazett’s formula (QTc = QT/R-R1/2).
Low HRV was defined using cutoff values recently derived from healthy US adult populations: low SDNN defined as SDNN < 8.2 ms; low rMSSD as rMSSD < 8.0 ms. 9 Additionally, CAN was defined as a composite measure using four different approaches : 1) CAN1 defined as SDNN and rMSSD both being below the fifth percentile of the general population distribution (SDNN < 8.2 ms and rMSSD < 8.0 ms)9,10; CAN2 as the lowest quartile of SDNN (<7.815 ms) and the highest quartile of QTI (>104.32%); CAN3 as the lowest quartile of SDNN and highest quartiles of QTI and resting HR (>77 bpm); CAN4 as the lowest quartile of SDNN and highest quartiles of QTI and resting HR in the presence of diabetic peripheral neuropathy (defined as a Michigan Neuropathy Screening Instrument Score >2 on the physical exam component).8–10 The various composite definitions of CAN have been shown to have higher predictive value than isolated abnormalities alone for adverse outcomes in individuals with diabetes mellitus.8,9,11,12
Ascertainment of stroke
The participants were prospectively followed from the baseline visit until the occurrence of stroke, death or study end (June 2009). Stroke events were ascertained via 4-monthly clinic visits during which participants were queried about emergency room visits, out-of-hospital procedures and hospitalizations since the prior visit. If participants missed a clinic visit, they were contacted by clinic staff who performed stroke events ascertainment over the phone. A stroke event was defined as a focal neurologic deficit lasting more than 24 h, associated with evidence of brain infarction or hemorrhage by computed tomography, magnetic resonance imaging or autopsy. The events were adjudicated by an expert committee in ACCORD.7,13 Stroke events were not further classified into ischemic versus hemorrhagic.
Covariates
The covariates were selected a priori based on their relation with CAN and/or stroke; they included age, sex, race, treatment arm, use of antihypertensive medications, use of antiarrhythmic medications (calcium-channel blockers, digitalis, beta blockers and other antiarrhythmics), duration of diabetes, cigarette smoking, alcohol consumption, body mass index, blood pressure (BP). 7 Total cholesterol, high-density lipoprotein (HDL) cholesterol, low-density lipoprotein (LDL) cholesterol, hemoglobin A1C (HbA1C) and serum creatinine were measured on blood samples obtained at baseline as previously reported. 7 Estimated glomerular filtration rate was calculated based on the Modification of Diet in Renal Disease equation. 14 Atrial fibrillation (AF) was ascertained (using Minnesota code 8.3) 15 on 12-lead ECG performed at biennial follow-up and exit visits.
Statistical analyses
The baseline characteristics of participants were compared by CAN status and low HRV using the t-test or Kruskal-Wallis test for continuous variables; and the χ2 test for categorical variables. Incidence rates (IR) and 95% confidence interval were calculated by dividing the number of stroke cases by the total at-risk person-years with the person-years estimated from baseline through the earliest of stroke event, date of death, or study termination (June 2009). We evaluated the time-to-event distributions for stroke by CAN status using the Kaplan-Meier curve and compared them using the log-rank test. We used multivariable Cox proportional hazards regression models to generate hazard ratios (HR) and 95% confidence intervals (CI) for stroke. Model 1 adjusted for age, sex, race, and treatment arm; model 2 included variables in model 1 plus duration of diabetes, glycated hemoglobin, cigarette smoking, alcohol intake, body mass index, systolic BP, use of BP-lowering medications, estimated glomerular filtration rate, and total/HDL cholesterol ratio; model 3 included model 2 with further adjustment for interval development of AF during follow-up.
In sensitivity analyses, we repeated analyses after including participants with a history of ASCVD and those on medications that can affect HRV (beta blocker, calcium channel blockers, digitalis and other antiarrhythmic medications); with further adjustment for history of ASCVD and use of HRV-modifying medications.
All analyses were performed using STATA 14.2 (Stata, Inc, College Station, TX). A two-sided P-value of <0.05 was considered statistically significant.
Results
Baseline characteristics by CAN status
The characteristics of participants included (compared to those excluded) in the final sample (reasons for exclusion detailed in Supplemental Figure S1) are displayed in Supplemental Table S1.
The baseline characteristics of study participants by CAN status are displayed in Table 1. A total of 3493 participants were included in our investigation (mean age 62.2 [SD: 5.6] years, 44.5% women, 62.9% white). Of the entire sample, 19.8% of participants (n = 690) had CAN1; 5.7% had CAN2 (n = 199); 3.2% had CAN3 (n = 111); and 1.6% had CAN4 (n = 55). The participants with low HRV had higher BMI, HbA1C, duration of diabetes, lower eGFR, and were more likely to be current smokers or insulin users (Supplemental Table S2).
Baseline characteristics of participants by evidence of cardiac autonomic neuropathy.
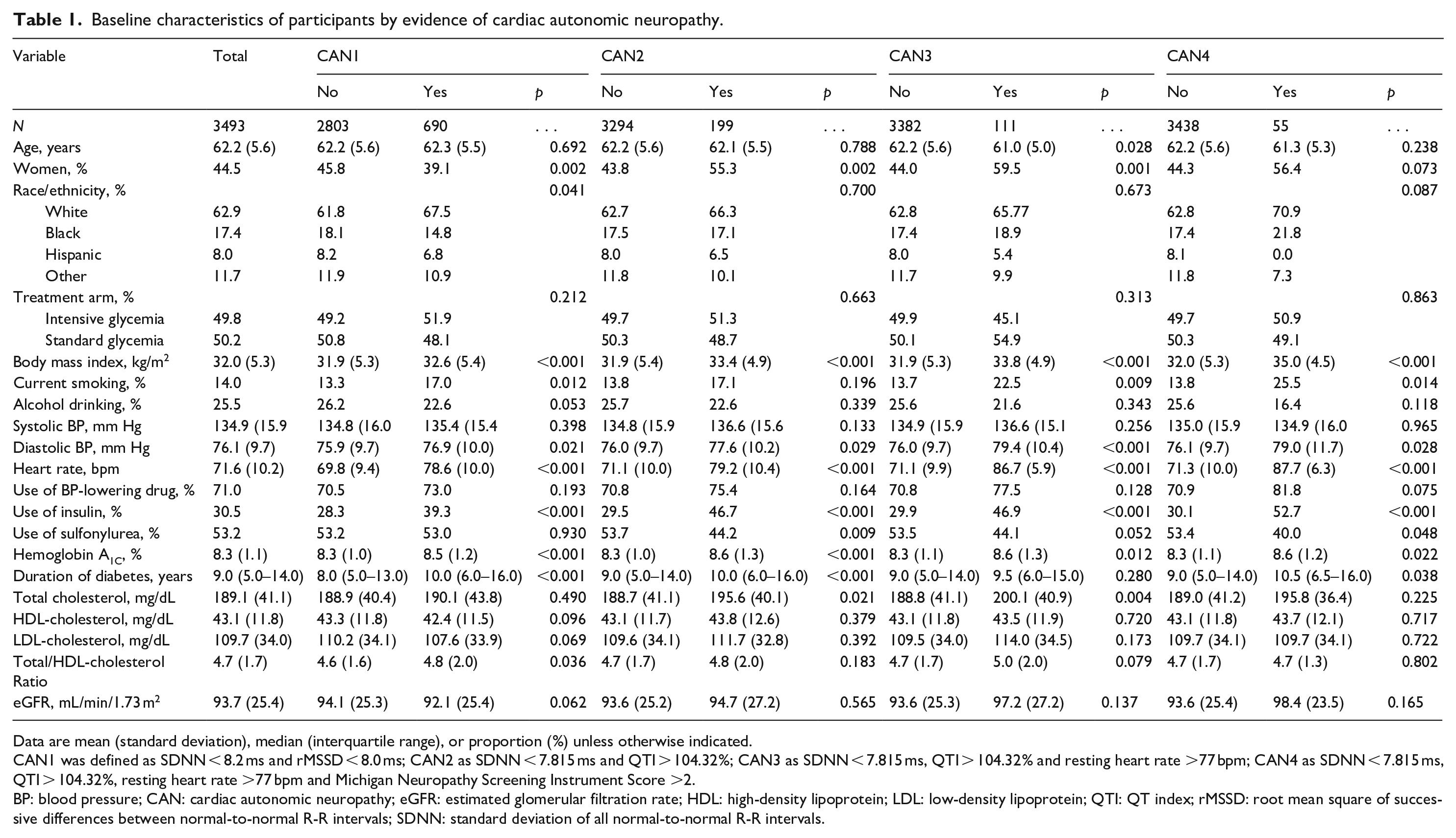
Data are mean (standard deviation), median (interquartile range), or proportion (%) unless otherwise indicated.
CAN1 was defined as SDNN < 8.2 ms and rMSSD < 8.0 ms; CAN2 as SDNN < 7.815 ms and QTI > 104.32%; CAN3 as SDNN < 7.815 ms, QTI > 104.32% and resting heart rate >77 bpm; CAN4 as SDNN < 7.815 ms, QTI > 104.32%, resting heart rate >77 bpm and Michigan Neuropathy Screening Instrument Score >2.
BP: blood pressure; CAN: cardiac autonomic neuropathy; eGFR: estimated glomerular filtration rate; HDL: high-density lipoprotein; LDL: low-density lipoprotein; QTI: QT index; rMSSD: root mean square of successive differences between normal-to-normal R-R intervals; SDNN: standard deviation of all normal-to-normal R-R intervals.
Incident stroke by CAN
Over a median follow-up of 5.0 (interquartile range: 4.0–5.7) years, 50 participants experienced a stroke event (incidence rate 3.0/1000 person-years [95% CI 2.2–3.9]). In unadjusted comparisons, participants with CAN had increased cumulative incidence of stroke as compared to those without CAN (Figure 1, all p-values < 0.05).
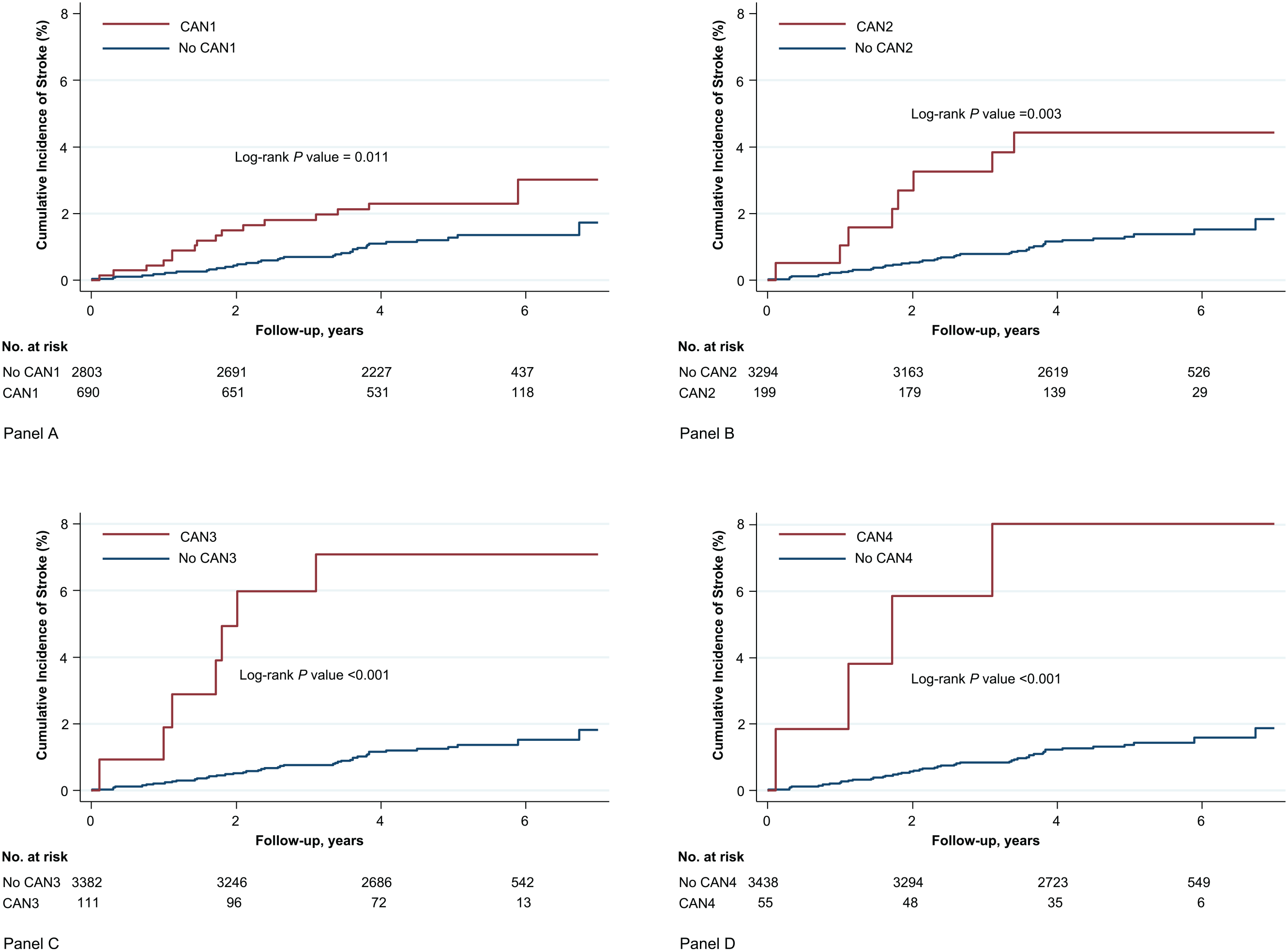
Cumulative Incidence of Stroke by cardiac autonomic dysfunction (CAN) status among individuals with type 2 diabetes. Panel A includes the link between CAN1 and stroke, Panel B includes the link between CAN2 and stroke, Panel C includes the link between CAN3 and stroke, and Panel D includes the link between CAN 4 and stroke.
After multivariable adjustment, low HRV was associated with a higher risk of incident stroke. Indeed, low SDNN was associated with a 2.2-fold greater risk of incident stroke (HR of 2.20 [95% CI 1.23–3.93]). Likewise, participants with low rMSSD had a 1.88-fold higher risk of stroke compared to those with normal rMSSD (HR 1.88, [95% CI 1.04–3.41], Model 2, Table 2).
Regardless of its definition, CAN was associated with greater risks of incident stroke. The HRs of the association of stroke with CAN1, CAN2, CAN3, and CAN4 were 1.83 (95% CI 0.99–3.38), 3.14 (95% CI 1.44–6.82), 5.70 (95% CI 2.49–13.08), and 7.06 95% CI (2.47–20.22), respectively (Model 2, Table 3).
Rates and hazard ratios for incident stroke by evidence of low heart rate variability.
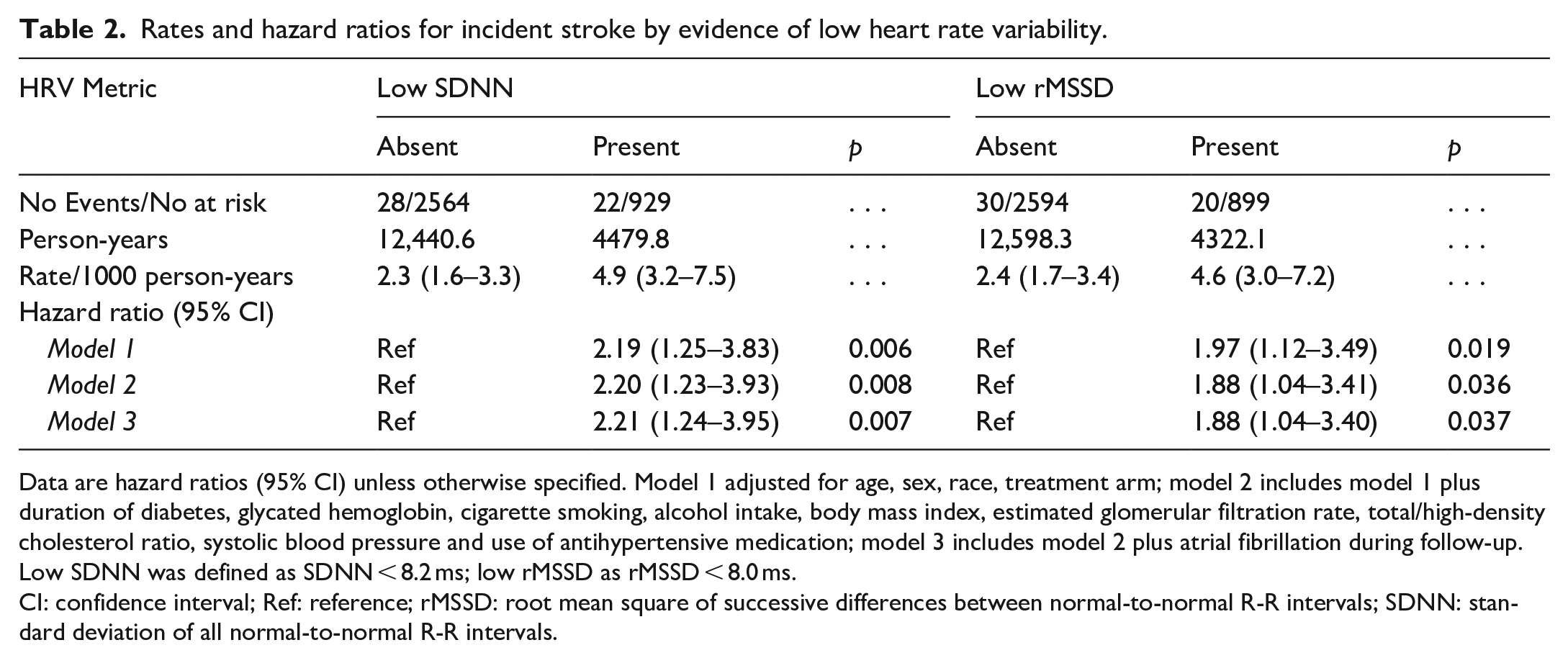
Data are hazard ratios (95% CI) unless otherwise specified. Model 1 adjusted for age, sex, race, treatment arm; model 2 includes model 1 plus duration of diabetes, glycated hemoglobin, cigarette smoking, alcohol intake, body mass index, estimated glomerular filtration rate, total/high-density cholesterol ratio, systolic blood pressure and use of antihypertensive medication; model 3 includes model 2 plus atrial fibrillation during follow-up. Low SDNN was defined as SDNN < 8.2 ms; low rMSSD as rMSSD < 8.0 ms.
CI: confidence interval; Ref: reference; rMSSD: root mean square of successive differences between normal-to-normal R-R intervals; SDNN: standard deviation of all normal-to-normal R-R intervals.
Rates and hazard ratios for incident stroke by evidence of cardiac autonomic neuropathy.
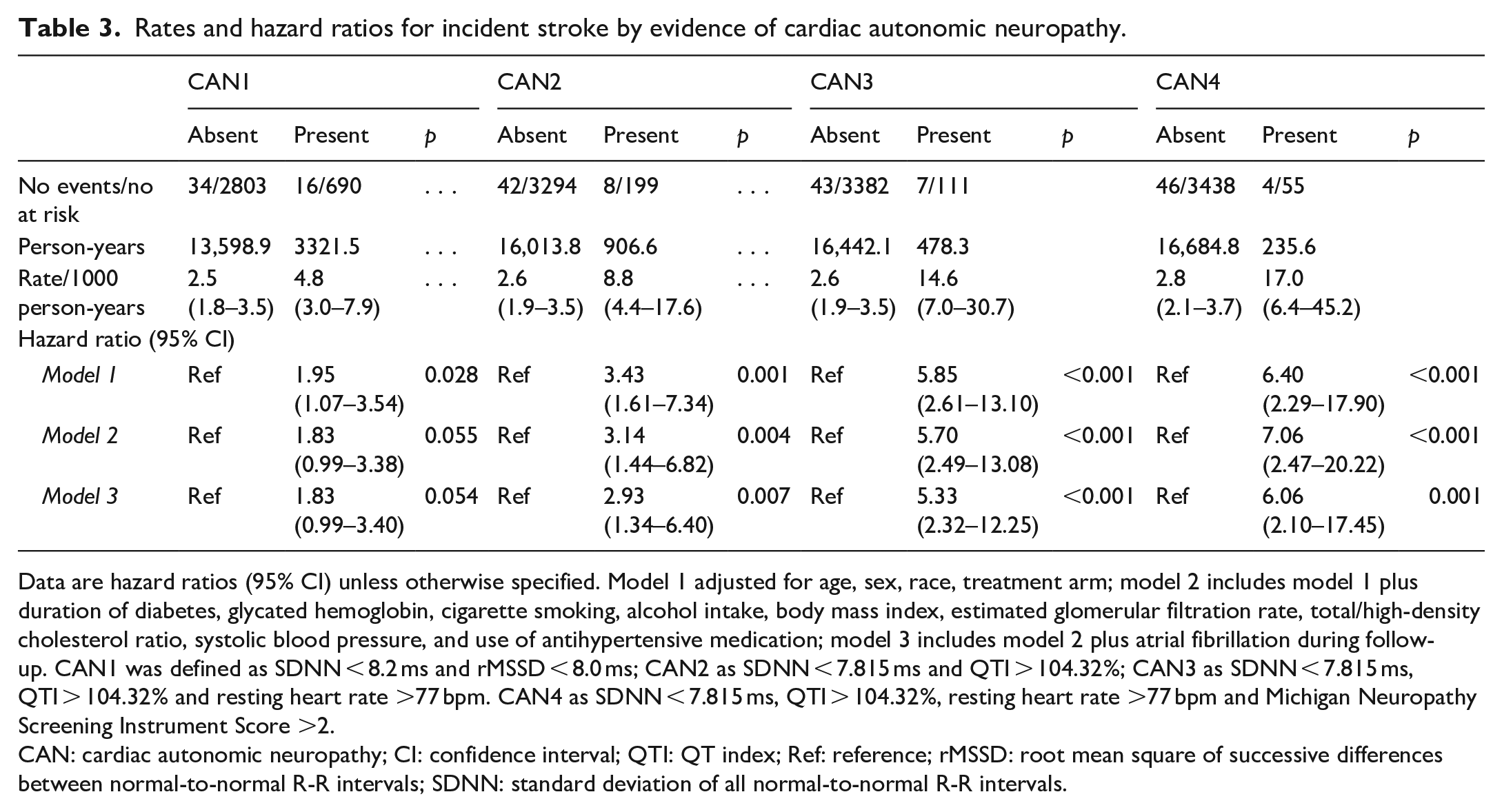
Data are hazard ratios (95% CI) unless otherwise specified. Model 1 adjusted for age, sex, race, treatment arm; model 2 includes model 1 plus duration of diabetes, glycated hemoglobin, cigarette smoking, alcohol intake, body mass index, estimated glomerular filtration rate, total/high-density cholesterol ratio, systolic blood pressure, and use of antihypertensive medication; model 3 includes model 2 plus atrial fibrillation during follow-up. CAN1 was defined as SDNN < 8.2 ms and rMSSD < 8.0 ms; CAN2 as SDNN < 7.815 ms and QTI > 104.32%; CAN3 as SDNN < 7.815 ms, QTI > 104.32% and resting heart rate >77 bpm. CAN4 as SDNN < 7.815 ms, QTI > 104.32%, resting heart rate >77 bpm and Michigan Neuropathy Screening Instrument Score >2.
CAN: cardiac autonomic neuropathy; CI: confidence interval; QTI: QT index; Ref: reference; rMSSD: root mean square of successive differences between normal-to-normal R-R intervals; SDNN: standard deviation of all normal-to-normal R-R intervals.
Further adjustments for interval development of AF during follow-up did not materially affect the magnitude or significance of our findings (Model 3, Tables 2 and 3).
Supplementary analyses
We conducted supplementary analyses on a larger sample that additionally included participants (n = 7421) with ASCVD and those taking medications affecting HRV (beta blocker, calcium channel blockers, digitalis and other antiarrhythmic medications). Cardiac autonomic dysfunction remained associated with an increased risk of incident stroke (Supplemental Tables S3 and S4).
Discussion
We evaluated the association of cardiac autonomic dysfunction with incident stroke in a large cohort of adults with type 2 diabetes. We observed a positive association between CAN with an increased risk of stroke, after accounting for several important stroke risk factors including BP, use of BP-lowering medications, HbA1C and duration of diabetes. Our findings were consistent across several measures of CAN.
Our findings complement the body of evidence on the adverse effects of CAN in people diabetes and corroborate prior reports which also found a positive association between low HRV and stroke, 3 as well as other CVD events in both type 1 and type 2 diabetes; although most of the prior studies did not assess stroke as an individual outcome.8,11,16 –22 CAN has been associated with other microvascular complications. Indeed, we have previously shown that CAN is associated with diabetes retinopathy in the ACCORD study. 23 Our current findings are in line with the results of a previous analysis of the ACCORD data showing a positive association between diabetic retinopathy and an increased risk of stroke. 24 CAN has also been shown to coexist with diabetic peripheral neuropathy. 5
The mechanisms linking CAN to greater risks of stroke are unclear, but may involve the interplay between cerebral blood flow autoregulation and blood pressure, as well as the rate of atherosclerosis progression. In the brain, slow pressure and cerebral blood flow changes are closely regulated by the sympathetic nervous system, whereas rapid changes may not be tightly regulated. 25 It has been suggested that impaired HRV may induce rapid pressure modifications in both the cerebral blood flow and cerebrovascular reactivity,26,27 which may result in a failure of autoregulation, poor cerebral vasculature health, and an increased stroke risk. Another mechanism may be linked to effect of autonomic dysfunction on vascular tone. Indeed, autonomic dysfunction is often associated with arterial stiffness as a result of enhanced autonomic nervous system activity. This may increase the vascular resistance, and increase blood pressure with further increase the risk of stroke. 28 Another mechanism that may explain our findings is the previously reported association between low HRV and the progression of atherosclerosis.29,30 Finally, the positive association between low HRV and stroke could be due to an increased risk of AF among those with low HRV. 31 Notably, in our analysis, adjustment for interval AF did not materially affect the results, suggesting that the observed association may not be explained by AF.
Our findings provide evidence for the role of CAN, a common occurrence in diabetic individuals, in the pathogenesis of stroke in type 2 diabetes and suggest that CAN may prove useful in stroke risk prediction among patients with type 2 diabetes. Our study also suggests that interventions that alter autonomic functions, such as baroreflex modulation for example, may help address stroke among patients with type 2 diabetes; indeed, a blunted baroreflex has been observed repeatedly in acute stroke, and may alter cerebral perfusion. 32 The prevention of CAN in this population could potentially decrease the strain of stroke on the healthcare system. Glycemic control should remain a priority in type 2 diabetes as it may prevent the onset and progression of CAN. 10 The assessment of HRV may especially be relevant among type 2 diabetes individuals who develop cryptogenic stroke. Furthermore, the use of simple tools such as the 12-lead ECG may help in the identification of adults with type 2 diabetes at high risk of stroke. Additional research could help shed light on the pathways relating CAN to stroke. Finally, the incremental predictive value of CAN for stroke discrimination in type 2 diabetes remains to be explored.
The limitations to our study should be acknowledged. First, CAN was assessed using time-domain measures from a 10 second ECG recording only; and we did not evaluate CAN using cardiovascular autonomic reflex tests (CART), 33 the gold standard to diagnose CAN. Therefore, it is possible that we missed certain cases of CAN, leading to the underestimation of the true effect of CAN on incident stroke in people with type 2 diabetes. However, CART may not be feasible in large-scale studies and major professional societies agree with the use of HRV time-domain indices to assess CAN in large clinical studies of adults with diabetes mellitus. 34 Second, in the ACCORD, the subtypes of stroke were not recorded; hence we could not assess the differential effect of CAN on stroke phenotypes (ischemic vs hemorrhagic). Finally, this study was observational, hence there is a possibility of residual unmeasured confounding. Notwithstanding these limitations, the strengths of this study include the use of a large, diverse cohort of type 2 diabetes adults, the rigorous assessment of CAN measures using several definitions, the standardized adjudication of stroke cases, and the robust adjustment of potential confounders.
In conclusion, in a large cohort of adults with type 2 diabetes, cardiac autonomic dysfunction was associated with a higher risk of incident stroke, independently of traditional stroke risk factors. Further research is needed to elucidate the pathways linking CAN to stroke, evaluate its incremental predictive value for stroke risk, and develop strategies targeting both CAN and stroke prevention.
Supplemental Material
sj-docx-1-eso-10.1177_23969873221127108 – Supplemental material for Cardiac autonomic dysfunction and risk of incident stroke among adults with type 2 diabetes
Supplemental material, sj-docx-1-eso-10.1177_23969873221127108 for Cardiac autonomic dysfunction and risk of incident stroke among adults with type 2 diabetes by Arnaud D Kaze, Matthew F Yuyun, Gregg C Fonarow and Justin B Echouffo-Tcheugui in European Stroke Journal
Footnotes
Acknowledgements
The authors wish to thank the staff and participants of the ACCORD study for their valuable contributions.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GCF reports consulting for Abbott, Amgen, AstraZeneca, Bayer, Cytokinetics, Janssen, Medtronic, Merck, and Novartis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Action to Control Cardiovascular Risk in Diabetes (ACCORD) has been funded by Federal funds from the National Heart Lung and Blood Institute (NHLBI). The data from the ACCORD study were supplied to the investigators by the NHLBI through the Central Repository BioLINCC. JBE was supported by NIH/NHLBI grant K23 HL153774.
Informed consent
Informed consent was obtained from each enrolled subject.
Ethical approval
Institutional Review Board approval at each participating center was required before the initiation of study enrollment.
Guarantor
JBE.
Contributorship
JBE researched literature and conceived the study. ADK and JBE were involved in data analysis. ADK researched the literature and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
