Abstract
Introduction
Despite a high burden of stroke, access to rapid brain imaging is limited in many middle- and low-income countries. Previous studies have described the astroglial protein GFAP (glial fibrillary acidic protein) as a biomarker of intracerebral hemorrhage. The aim of this study was to test the diagnostic accuracy of GFAP for ruling out intracranial hemorrhage in a prospective cohort of Indian stroke patients.
Patients and methods
This study was conducted in an Indian tertiary hospital (Christian Medical College, Ludhiana). Patients with symptoms suggestive of acute stroke admitted within 12 h of symptom onset were enrolled. Blood samples were collected at hospital admission. Single Molecule Array technology was used for determining serum GFAP concentrations.
Results
A total number of 155 patients were included (70 intracranial hemorrhage, 75 ischemic stroke, 10 stroke mimics). GFAP serum concentrations were elevated in intracranial hemorrhage patients compared to ischemic stroke patients [median (interquartile range) 2.36 µg/L (0.61–7.16) vs. 0.18 µg/L (0.11–0.38), p < 0.001]. Stroke mimics patients had a median GFAP serum level of 0.14 µg/L (0.09–0.26). GFAP values below the cut-off of 0.33 µg/L (area under the curve 0.871) ruled out intracranial hemorrhage with a negative predictive value of 89.7%, (at a sensitivity for detecting intracranial hemorrhage of 90.0%).
Discussion
The high negative predictive value of a GFAP test system allows ruling out patients with intracranial hemorrhage.
Conclusion
In settings where immediate brain imaging is not available, this would enable to implement secondary prevention (e.g., aspirin) in suspected ischemic stroke patients as soon as possible.
Keywords
Introduction
Stroke incidence in low- and middle-income countries has more than doubled in the previous four decades with an early case fatality rate 25% higher than in high income countries. 1 Besides that in the year 2017 only 14% of low-income countries were able to provide at least one CT-scanner per one million people. 2
In consequence, in low- and middle-income countries, many stroke patients do not receive brain imaging for differentiation between intracerebral hemorrhage and ischemic stroke in time. Acute therapy and rapid diagnosis-specific implementation of secondary prevention (in particular, antiplatelet drugs in patients with ischemic stroke) is therefore not possible. A recent analysis suggested to initiate aspirin treatment even in unclassified stroke in resource-limited settings to decrease mortality rates and stroke recurrence. 3 However, this would expose patients with intracerebral hemorrhage to increased risks of re-bleeding. At this point the urgent need for alternative and cost-efficient ways to differentiate between ischemic stroke and intracerebral hemorrhage becomes clear.
The astrocytic protein GFAP (glial fibrillary acidic protein) has been identified as a promising candidate for a point-of-care assay indicating intracerebral hemorrhage in the early phase of stroke.4–6 In intracerebral hemorrhage, GFAP is rapidly released from damaged glial cells as a result of the expanding hematoma. By contrast, in ischemic stroke, a more gradual occurrence of cytolysis and glial necrosis is observed.7,8, Recently, a systematic review confirmed a high diagnostic accuracy for blood GFAP levels as a discriminatory test between intracerebral hemorrhage and ischemic stroke. 9 Whether GFAP is elevated in serum in other forms of intracranial hemorrhage (i.e. subarachnoid hemorrhage and subdural hematoma) is less clear.10,11
Stroke patients in India have an overall limited access to acute brain imaging. Furthermore, incidence rates of intracerebral hemorrhage in Asian countries including India are remarkably higher compared to Western populations. 12 The aim of this study therefore was to evaluate the diagnostic accuracy of GFAP for ruling out intracranial hemorrhage in suspected stroke patients in an Indian tertiary care hospital.
Methods
Standard protocol approvals, registrations, and patient consents
The Biomarker for Rapid Diagnosis of Stroke in India study (BE FAST INDIA) was designed following the guidelines of the Standards for Reporting of Diagnostic Accuracy initiative. 13 The study protocol was approved by the institutional review board of the Christian Medical College (CMC) in Ludhiana, Punjab, India. All patients or legal representatives gave a written informed consent prior to ultimate inclusion in the study. However, as per study protocol, an aliquot of the routine blood sample drawn upon hospital admission before obtaining informed consent was used for study purposes where available.
Study design
BE FAST INDIA was performed at the Department of Neurology of CMC, Punjab, Ludhiana, India. This is a tertiary private hospital serving over 10,000 patients per day. Between November 2018 and May 2019 all patients with clinical symptoms suggestive of acute stroke were prospectively and consecutively included to the study if the following inclusion criteria were fulfilled: (I) age ≥ 18 years, (II) time from symptom onset to hospital admission <12 h (time windows of previous BE FAST studies were extended due to expected delays in the local triage of patients),4,5 and (III) National Institute of Health Stroke Scale (NIHSS) score at hospital admission ≥4 points. The determination of symptom onset time was based on the information provided by patient, relatives, or any other witnesses. Patients were excluded from the study if they had (I) a history of intracranial hemorrhage, ischemic stroke or transient ischemic attack (TIA) in the past 3 months, (II) a history of traumatic brain injury in the past 3 months, (III) a brain tumor at any time in the past medical history,14,15 (IV) no brain imaging available within the first 24 h after hospital admission.
After study inclusion, the following clinical parameters were recorded: age, sex, NIHSS at admission, time interval between symptom onset and blood sampling, time interval between symptom onset and brain imaging, presence of a hemiparesis or cortical signs (i.e. aphasia, neglect, homonymous hemianopia, forced gaze deviation), current anticoagulation or antiplatelet treatment along with INR value at hospital admission, history of vascular risk factors including arterial hypertension, diabetes mellitus, dyslipidemia and smoking (defined according to current guidelines in India),16–18 creatinine level at hospital admission and history of renal dysfunction, malignant neoplasm, and ongoing rheumatic or infectious disease.
Final diagnosis reached at hospital discharge was based on all available clinical data, brain imaging, laboratory results and other examinations, stratified as ischemic stroke (including TIA) or intracranial hemorrhage (including intracerebral hemorrhage, subarachnoid hemorrhage and subdural hematoma). Stroke mimics were also recorded. Stroke mimics were defined as conditions clinically imitating the symptoms of acute stroke such as epileptic seizures, otogenic vertigo, or transient focal neurological deficits secondary to infection, hypoglycemia, or dehydration.
Intracerebral hemorrhage volume quantification
In patients with intracerebral hemorrhage, the localisation of the hematoma was classified into “deep” (striatum, thalamus, brain stem cerebellum) or “lobar” (parietal lobe, frontal lobe, temporal lobe, occipital lobe) on the first available brain scan. Intracerebral hemorrhage volumes were quantified in milliliters by use of the (a × b × c)/2 method. 19 Intraventricular expansion of hematoma if present was also documented. The quantification of infarct sizes in patients suffering from ischemic stroke was not performed. Radiologists were blinded to the clinical data.
Blood sampling
At hospital admission, 2 mL blood was drawn in a serum-gel tube with clotting activator (S-Monovette, 4.7 mL, Sarstedt). The blood tube was centrifuged at 3000 RPM for 12 min at the earliest possibility (max. 6 h after blood sampling). Immediately afterwards the supernatant serum was dispensed into Eppendorf tubes and frozen at –80°C. Although GFAP can stay stable for several days at 4 °C and up to four freezing/thawing cycles, the serum samples were shipped on dry ice as a precaution. 20
GFAP measurements
The serum samples were sent to Quanterix Corporation, Lexington, Massachusetts, USA for GFAP quantification. All laboratory technicians performing the measurements were blinded to the clinical data. The Single Molecule Array (SIMOA) HD-1-Analyzer® was used for determining the serum GFAP concentration. This digital ELISA technology is considered an ultra-sensitive method for GFAP detection. It has so far been used for GFAP measurements in multiple sclerosis, traumatic brain injury and brain tumors.21–24 In a first step, antibody labelled beads were added to 100-μL diluted serum. The high number of beads and the multiple attachment points of each bead added to the solution enable an efficient binding to single protein molecules. Biotinylated monoclonal GFAP antibodies and β-galactosidase–labeled streptavidin were subsequently combined with the solution. Every bead attached to a GFAP molecule was now labelled with an enzyme. Thereafter each bead was loaded into one of the 2,16,000 femtoliter-sized wells of an array. The wells including the beads were then sealed and imaged. The immunocomplex associated enzymes sent fluorescent signals which were measured and converted into the serum protein concentration by SIMOA digitally. The protein concentration was calculated by counting the wells with fluorescent beads relatively to the total number of wells containing beads. 24 All samples were measured twice. The average intra-assay coefficient of variation (CV) was 4.5%. There were blood samples determined with a saturated GFAP signal. We substituted the saturated GFAP signal by the value of 8.291 µg/L, which was 0.001 µg/L above the highest measured GFAP value. In healthy individuals, median serum GFAP concentration was determined as 0.091 µg/L using this test. 25
Statistical analysis
IBM SPSS® Statistics, Version 22 (Statistical Package for the Social Sciences) was applied for statistical analyses. The Mann–Whitney U test was used to compare GFAP serum concentrations between groups. Correlation analyses were made using the Spearman rank test. The calculation of the optimal serum GFAP cut-off concentration to distinguish intracranial hemorrhage from ischemic stroke (and stroke mimicking conditions) was performed by ROC-curve analysis. Sensitivity and specificity measures as well as the positive predictive value (PPV), the negative predictive value (NPV) and the overall diagnostic accuracy were derived from cross tabulations. A significance level of 0.05 was chosen for all tests.
Results
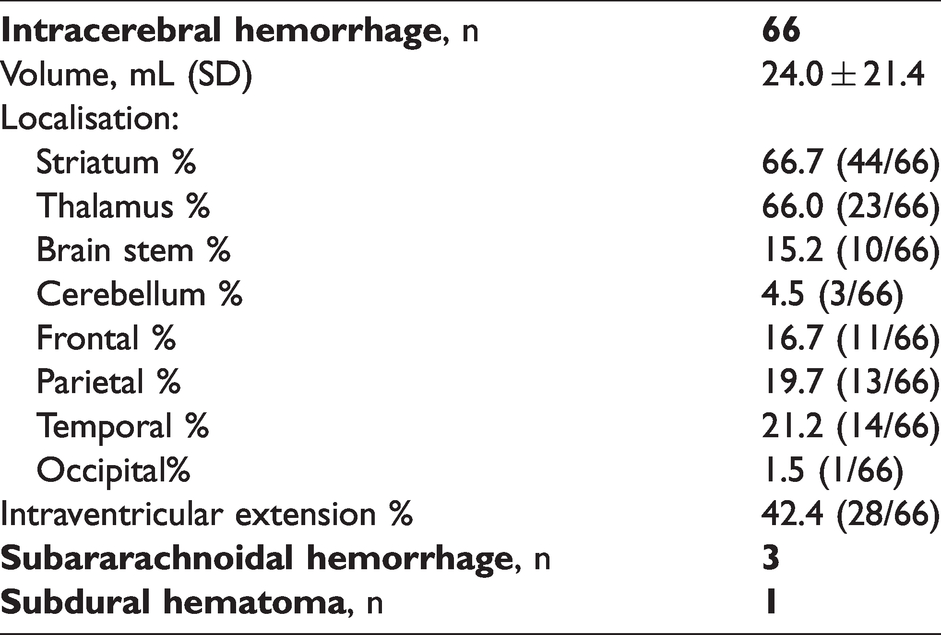
A total number of 155 patients were enrolled after applying all prespecified inclusion and exclusion criteria (Figure 1) in this study. The average age was 61 ± 14 years, and 42% were female. Table 1 depicts baseline variables and clinical characteristics stratified according to the final diagnosis. Seventy patients suffered from an intracranial hemorrhage (66 intracerebral hemorrhage, 3 subarachnoid hemorrhage, 1 subdural hematoma; Table 2), 75 patients were diagnosed with ischemic stroke (Table 3) and 10 patients with stroke mimics. The mean time of symptom onset to blood sampling was 243 ± 212 min.
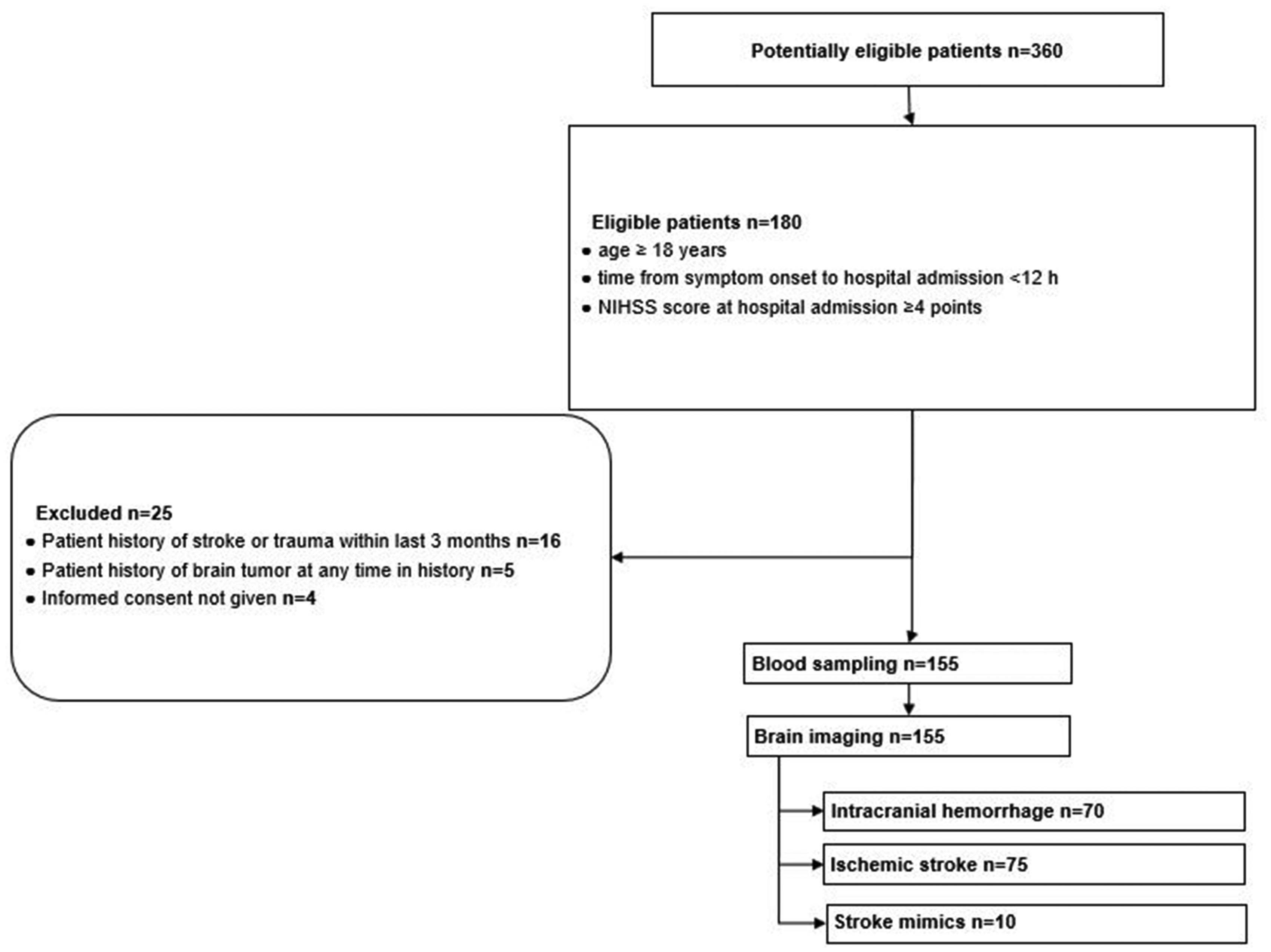
STARD diagram showing flow of patient enrolment.
Baseline characteristics of the study population.
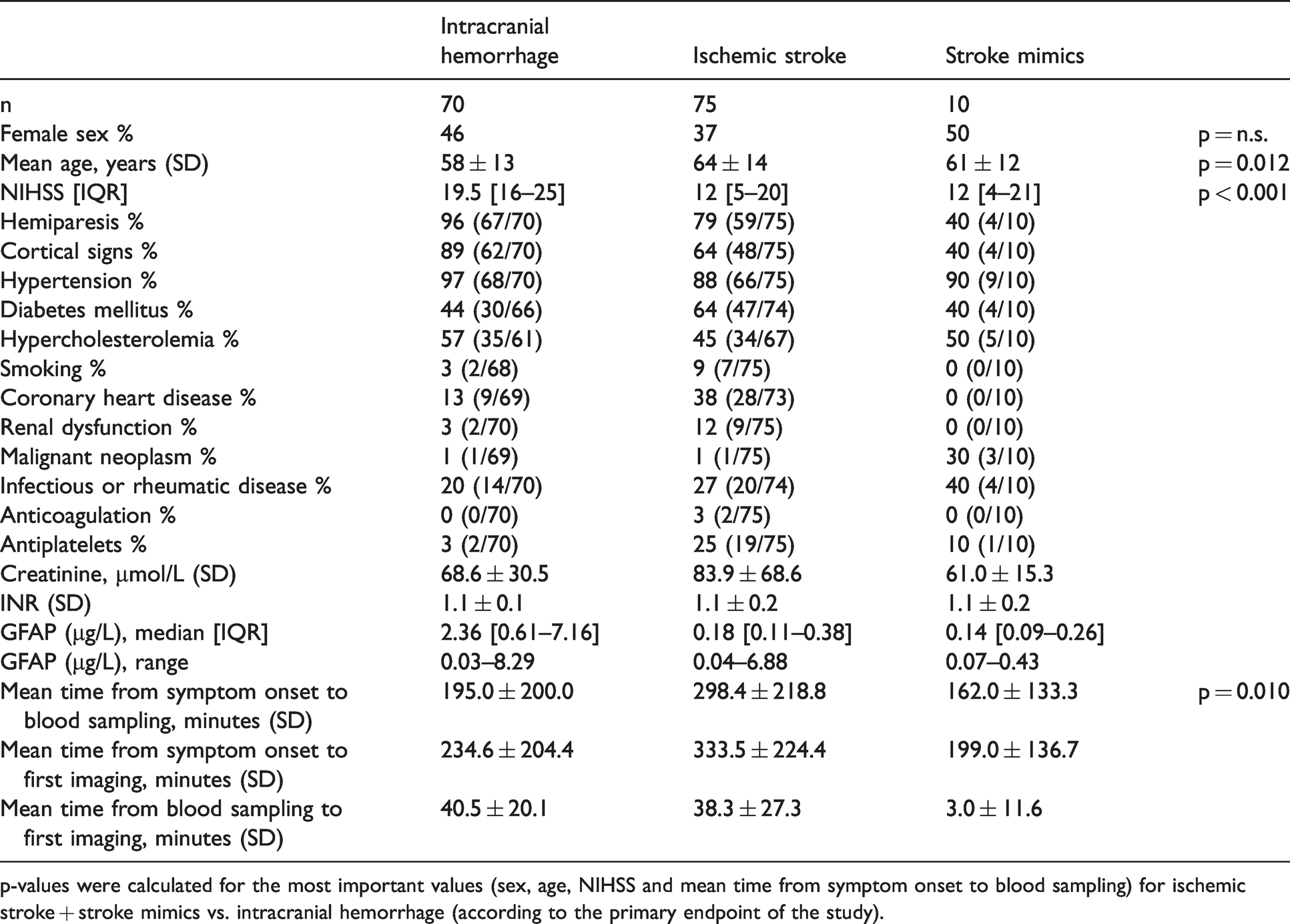
p-values were calculated for the most important values (sex, age, NIHSS and mean time from symptom onset to blood sampling) for ischemic stroke + stroke mimics vs. intracranial hemorrhage (according to the primary endpoint of the study).
Types of intracranial hemorrhage.
Classification and treatment of patients with ischemic stroke.

We found strongly elevated GFAP serum concentrations in intracranial hemorrhage patients compared to patients with ischemic stroke (median [interquartile range, minimum, maximum] 2.36 µg/L [0.61–7.16, 0.03, 8.29] vs. 0.18 µg/L [0.11–0.38, 0.04, 6.88], p < 0.001). Patients classified as stroke mimics had the following final diagnoses (number of patients): basilar migraine (1), functional mimic (1), hyperglycemia induced seizure (1), glioma (2), epilepsy/seizure (4), acute gastritis (1). This patient group had a median GFAP serum level of 0.14 µg/L with the interquartile range of 0.09–0.26 µg/L.
The optimal GFAP cut-off concentration to differentiate intracranial hemorrhage from ischemic stroke and stroke mimicking conditions by ROC analysis (Figure 2(a)) was 0.57 µg/L (area under the curve 0.871 [95% CI 0.810–0.933], p < 0.001). GFAP values below this cut-off ruled out intracranial hemorrhage with a NPV of 82.8% (sensitivity for detecting intracranial hemorrhage 77.1%, specificity 90.6%, PPV 87.1%, overall diagnostic accuracy 84.5%). We then studied cut-off points delivering sensitivities ≥90% and ≥95%, respectively (in order to identify as many intracranial hemorrhage patients as possible). GFAP values below a cut-off of 0.33 µg/L ruled out intracranial hemorrhage with a NPV of 89.7% (sensitivity 90.0%, specificity 71.8%, PPV 72.4%). GFAP values below a cut-off of 0.12 µg/L ruled out intracranial hemorrhage with a NPV of 90.3% (sensitivity 95.7%, specificity 32.9%, PPV 54.0%). The diagnostic accuracy for the discriminatory test improved in patients presenting with more severe stroke (NIHSS > 18; area under the curve 0.926 [95% CI 0.860–0.992], p < 0.001) (Figure 2(b)). In addition, we evaluated diagnostic accuracy in patients who presented within 4.5 h of symptom onset. Here, ROC analysis revealed a cut-off value of 0.57 µg/L. GFAP values below this cut-off ruled out intracranial hemorrhage with a NPV of 79.4% (sensitivity for detecting intracranial hemorrhage 74.4%, specificity 100.0%, PPV 100.0%, overall diagnostic accuracy 87.1%).
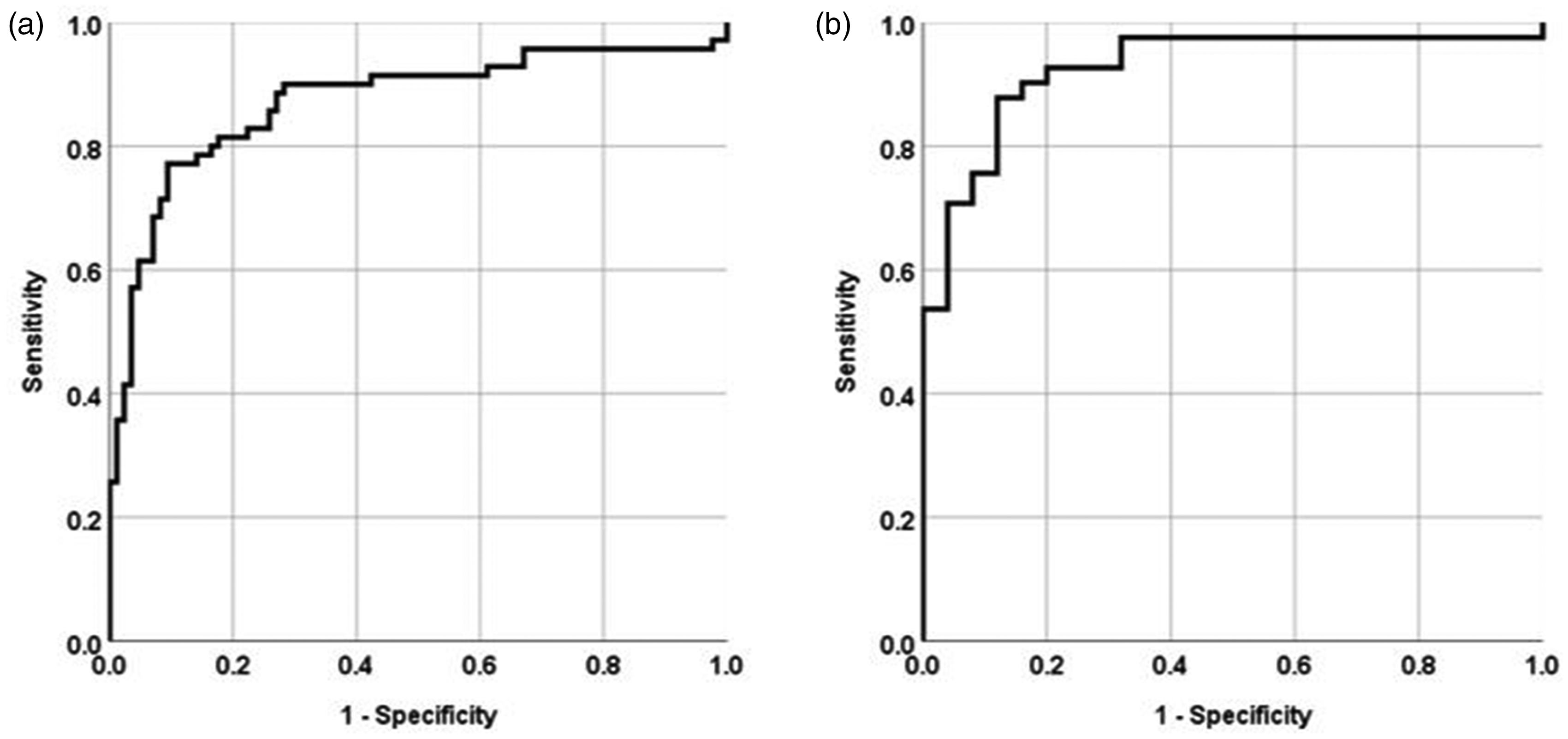
(a) ROC analysis for identifying intracranial hemorrhage. (b) ROC analysis for identifying intracranial hemorrhage in patients with severe neurological deficits (NIHSS > 18).
In patients with intracerebral hemorrhage, the average hematoma volume was 24.0 mL ± 21.4 mL (min. 1.7, max. 102 mL). A significant correlation between intracerebral hemorrhage volume and serum GFAP value was observed (Spearman rho 0.522, p < 0.001; Figure 3). Patients with cortical (lobar) involvement had larger hematoma volumes (p < 0.001) and higher GFAP values (p = 0.012) as compared to patients with deep hemorrhage. Similarly, patients having intracerebral hemorrhage with intraventricular extension had larger (parenchymal) hematoma volumes (p = 0.011) and higher GFAP values (p = 0.041) as compared to patients without intraventricular extension. Three patients with striatal and capsular hemorrhage location with volumes between 4 and 6 mL and two striatal hemorrhages with a volume of 13 mL and 19 mL, respectively, had low GFAP values (below 0.30 µg/L) (supplemental table).
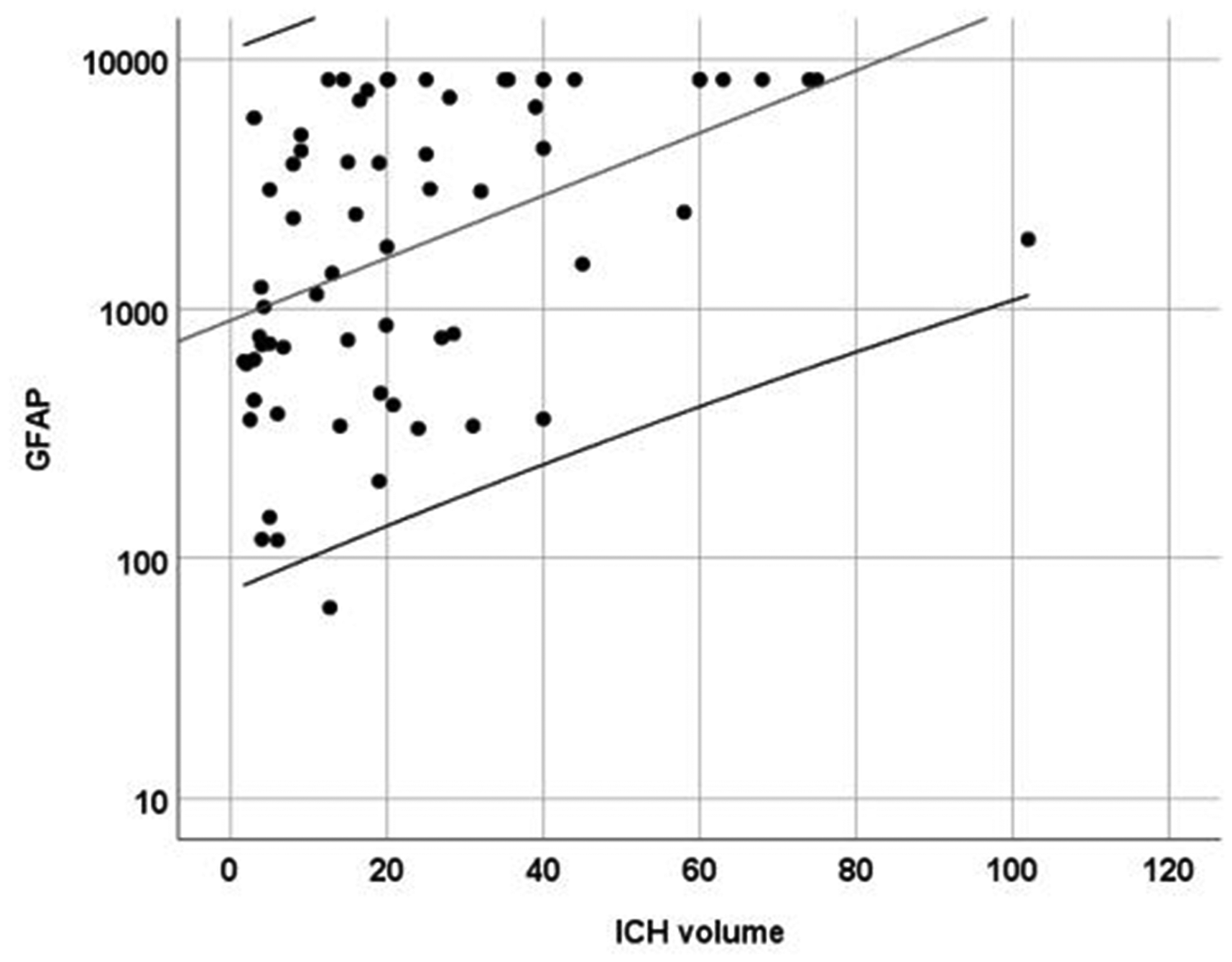
Scatter plot depicting the correlation between intracerebral hemorrhage volume and GFAP serum levels.
The four patients who were diagnosed with other intracranial hemorrhages presented with varying serum GFAP concentration. Subarachnoid hemorrhage in two patients led to elevated GFAP levels (3.46 and 2.53 µg/L), whereas one patient suffering from subarachnoid hemorrhage and another patient suffering from subdural hematoma had a very low GFAP serum concentration (0.03 and 0.04 µg/L).
Discussion
This single-center study performed in an Indian tertiary care hospital revealed a high diagnostic accuracy of a serum GFAP test for ruling out intracranial hemorrhage in patients with symptoms of acute stroke.
In contrast to the previous BE FAST studies conducted in European countries, this study was executed in a region where intracerebral hemorrhage is highly prevalent.4,5 The stroke registry in urban regions of Ludhiana from 2011 to 2013 showed that about 32% of all stroke cases were patients with intracerebral hemorrhage. 26 Likely, this results from genetic factors and the poor control of arterial hypertension in the Indian population. In our study, patients were recruited consecutively, and the proportion of intracerebral hemorrhage patients was even higher than described in the local registry. An explanation could be the seasonal variation of intracerebral hemorrhage incidence which is higher in winter than in summer (recruitment between November and May).27,28
We demonstrated that a GFAP test could be used for excluding patients with intracranial hemorrhage among patients with symptoms of acute stroke. In our dataset, the pre-test probability of having a patient with intracranial hemorrhage was 45% (70/155). This number decreased to 17% (NPV = 83%) in case of a test result below the optimal cut-off point of 0.57 µg/L. By shifting the cut-off point towards lower values (i.e. 0.33 µg/L; thereby identifying more patients with intracranial hemorrhage by positive GFAP results), this probability further decreased to 10% (NPV = 90%). In other words, if the test is below the cut-off point of 0.33 µg/L, one can be 90% sure that this patient does not have intracranial hemorrhage. Still, with a specificity of 72% for this cut-off point the majority of ischemic stroke patients will be captured.
Previous studies used different immunoassays to detect GFAP in patients with acute stroke.4,5,9 For methodological reasons no consensus regarding the optimal GFAP cut-off value (in µg/L) for differentiating between ischemic stroke and intracerebral hemorrhage has been achieved so far. 29 Most studies reported a “sensitivity gap”, leaving some patients with intracerebral hemorrhage undetected. Here, we intended to increase the diagnostic accuracy using the ultra-sensitive SIMOA technology. 24 However, the study results did not reveal an improvement in sensitivity compared to previous studies. False negative results were still recorded in patients with intracerebral hemorrhage who presented with small hematoma volumes (and little astroglial tissue destruction) or short time windows from symptom onset to blood sampling (supplemental table). Hematoma location may also play a role in this context. Similar to previous studies, deep hemorrhages (e.g. basal ganglia) were associated with lower GFAP values than bleedings with lobar involvement. 5 False positive results occurred in a few ischemic stroke patients, and all of them were recruited beyond 6 h of symptom onset. Here, elevated GFAP values might be a consequence of recurrent stroke or of falsely reported time windows (supplemental table).
In our study, serum samples were collected on site, stored and shipped abroad for measurement which is of course impracticable for clinical application. However, GFAP point-of-care tests on hand-hole devices are currently under development which may provide test results in less than 15 min. 36 In countries with limited access to brain imaging a rapid GFAP based discriminatory test could be beneficial in acute stroke management. Many peripheral hospitals in Punjab, India lack brain imaging facilities and refer patients suspected of stroke to tertiary care hospitals. 30 The poor ambulance and traffic infrastructure leads to further delays in acute stroke management. 30 Moreover, many patients from lower- and middle-class families cannot afford acute stroke diagnostics. These obstacles demonstrate the urgent need for an alternative cost-efficient diagnostic method. The sensitivity of a GFAP test system for detecting intracranial hemorrhage is below 100%. Thus, GFAP measurements test cannot replace neuroimaging for ruling out intracranial hemorrhage prior to thrombolytic therapy. However, in settings with absence of neuroimaging the GFAP point-of-care test may help identifying ischemic stroke and initiating antiplatelet therapy. Antiplatelet medication used to prevent ischemic re-events is the more effective the more early it is initiated.31–33 Large randomised trials demonstrated the benefit of early dual antiplatelet therapy in patients with minor stroke and lacunar stroke syndromes.34–36 In resource-limited countries aspirin therapy has led to decrease of stroke related mortality and stroke recurrence even in stroke of unknown etiology. 3
As a precaution, the potential treatment of patients with an intracranial bleeding and low GFAP levels (i.e. false negative findings) with antiplatelets might bear a risk of a greater hemorrhage. Based on available data, these risks might be manageable. 37 Naturally, the GFAP test does not differentiate between ischemic stroke patients and stroke mimics. However, in acute stroke management, CT imaging is primarily used for excluding intracranial hemorrhage, and not for the positive identification of “true” ischemic stroke patients. The treatment of (few) stroke mimics patients with thrombolysis and antiplatelet agents is a broadly accepted condition, and studies did not reveal substantial risks of bleeding events in these patients.
This study has strengths. Firstly, it was conducted in a “true” low- to middle income setting in need of an alternative and cost-efficient testing method for acute stroke patients. This allowed determining the diagnostic accuracy in a stroke collective with a larger proportion of intracerebral hemorrhage cases than in Western populations. Secondly, ultra-sensitive SIMOA technology was used for biomarker detection. As a limitation, GFAP data in intracranial hemorrhage other than intracerebral hemorrhage is scarce, and the heterogeneous GFAP levels determined in this patient cohort (four patients) study does not allow a reliable conclusion. Furthermore, validation of the results is mandatory in larger multi-center cohorts.
To conclude, GFAP is a promising diagnostic marker for management of patients with acute stroke in resource-limited countries. A high diagnostic accuracy was shown in distinguishing intracranial hemorrhage from ischemic stroke and stroke mimicking conditions, especially in patients with high stroke severity. Further studies are needed to evaluate the clinical outcome following treatment initiation with aspirin in the patient group with low GFAP values as well as the cost effectiveness of the measure.
Supplemental Material
sj-pdf-1-eso-10.1177_23969873211010069 - Supplemental material for Serum GFAP for stroke diagnosis in regions with limited access to brain imaging (BE FAST India)
Supplemental material, sj-pdf-1-eso-10.1177_23969873211010069 for Serum GFAP for stroke diagnosis in regions with limited access to brain imaging (BE FAST India) by Love-Preet Kalra, Himani Khatter, Sarvotham Ramanathan, Sameer Sapehia, Kavita Devi, Abirami Kaliyaperumal, Deepti Bal, Ivy Sebastian, Raviteja Kakarla, Anusha Singhania, Shubhra Rathore, Svenja Klinsing, Jeyaraj Durai Pandian and Christian Foerch in European Stroke Journal
Supplemental Material
sj-pdf-2-eso-10.1177_23969873211010069 - Supplemental material for Serum GFAP for stroke diagnosis in regions with limited access to brain imaging (BE FAST India)
Supplemental material, sj-pdf-2-eso-10.1177_23969873211010069 for Serum GFAP for stroke diagnosis in regions with limited access to brain imaging (BE FAST India) by Love-Preet Kalra, Himani Khatter, Sarvotham Ramanathan, Sameer Sapehia, Kavita Devi, Abirami Kaliyaperumal, Deepti Bal, Ivy Sebastian, Raviteja Kakarla, Anusha Singhania, Shubhra Rathore, Svenja Klinsing, Jeyaraj Durai Pandian and Christian Foerch in European Stroke Journal
Footnotes
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Foerch invented the following patent: Use of GFAP for identification of intracerebral hemorrhage (US20150247867). The other authors report no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Department of Neurology, Goethe University, Frankfurt am Main, Germany.
Informed consent
All patients or legal representatives gave a written informed consent prior to ultimate inclusion in the study.
Ethical approval
The ethical approval was obtained from the institutional review board of the Christian Medical College (CMC) in Ludhiana, Punjab, India (20,18,06,270/IECCMCL/Apprvl-BE FAST INDIA/NEUROL).
Guarantor
CF
Contributorship
CF, JDP, LK and AS conceived the study. JDP, AS and LK were involved in gaining ethical approval. JDP, SRathore, RK, HK, SRamanathan, KD, SS, DB, AK, AS, IS and LK acquired the data. CF, HK and LK analyzed the data. CF, JDP, SK and LK drafted the manuscript or revised it critically for important intellectual content.
Acknowledgements
This study was conducted under the patronage of INCERAS e.V. providing non-financial support. The authors are grateful to the healthcare staffs of emergency, radiology, and neurology departments of CMC hospital for their support.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
