Abstract
Introduction
Observational studies have suggested increased risk of intracranial haemorrhage (ICrH) in patients receiving selective serotonin reuptake inhibitors (SSRIs). We sought to clarify the impact of SSRIs on ICrH, accounting for study methodology.
Patients and methods
A comprehensive search of Medline, Embase and the Cochrane Library from 1960 to December 2017 identified studies comparing SSRIs with control. The outcomes (first-ever and recurrent ICrH) were meta-analysed using a random effects model.
Results
Twenty-four observational studies and three randomised trials were available for meta-analysis, totalling 4,844,090 patient-years of follow-up. Those receiving SSRIs were more likely to be female (p = 0.01) and have depression (p < 0.001). Compared to controls, SSRI users had a significantly increased risk of ICrH (relative risk (RR) 1.26, 95%CI 1.11–1.42). Although SSRI use was associated with increased ICrH risk in those without previous ICrH (RR 1.31, 95%CI 1.15–1.48), this was not the case in those with previous ICrH (RR 0.95, 95%CI 0.83–1.09). Sensitivity analysis according to the bleeding definition reported demonstrated that although ‘haemorrhagic stroke’ was associated with SSRIs (RR 1.40, 95%CI 1.13–1.72), intracerebral haemorrhage was not (RR 1.11, 95%CI 0.86–1.42). Additional sensitivity analyses demonstrated a stronger association between SSRIs and ICrH in studies with a high (p < 0.001) compared to low risk of bias (p = 0.09) and with retrospective (p < 0.001) compared to prospective (p=0.31) study designs.
Discussion
Although SSRIs are associated with an increased risk of ICrH, the association is partly accounted for by important biases and other methodological limitations in the available observational data.
Conclusion
Our findings suggest there is insufficient high-quality data to advise restriction of SSRIs because of concern regarding ICrH risk.
Keywords
Introduction
Selective serotonin reuptake inhibitors (SSRIs) are frequently used as first-line antidepressants because of their efficacy, tolerability and general safety in overdose. Nevertheless, concerns have been raised regarding whether SSRIs increase the risk of major bleeding. This is plausible pharmacologically: apart from inhibiting presynaptic serotonin reuptake in neurones, SSRIs block serotonin release from platelets, inhibiting both platelet aggregation and vasoconstriction. 1 Observational studies have consistently demonstrated an association between SSRIs and upper gastrointestinal bleeding, 2 but the association with intracranial haemorrhage (ICrH) has not been fully elucidated. Furthermore, given the high prevalence of depression after ICrH, and that SSRIs form the mainstay of therapy in this cohort, a further concern is whether SSRIs increases the risk of ICrH recurrence. 3 To date, there are no systematic reviews assessing SSRI therapy in survivors of ICrH; current European and American stroke guidelines make no recommendations for antidepressant use post-ICrH.4,5
Two previous meta-analyses of observational studies reported a significantly increased risk of ICrH with SSRI use (RR 1.42, 95% CI 1.23–1.65; 6 OR 1.32; 95% CI 1.02–1.71 7 ). Since these meta-analyses were performed, ten relevant observational studies have been published, five of which reported no association with increased risk of ICrH.8–12 Importantly, these meta-analyses did not assess the impact of risk of bias of individual studies on the validity of their conclusions. Studies that do not address differences in confounding variables between groups (through either statistical adjustment techniques or randomisation) may be biased towards overestimating the effect size due to ‘confounding by indication,’ leading to spurious associations between SSRIs and risk of ICrH. Additionally, these meta-analyses did not consider potentially relevant trial data that have assessed SSRIs versus control (placebo or no treatment) irrespective of population studies or the outcome assessed. Such trials may report ICrH in the adverse event table of a trial report, and be missed by search strategies designed to identify studies with ICrH as the primary outcome.
In view of the potential usefulness of SSRIs in psychiatric disorders, and in an attempt to settle this uncertainty over adverse outcomes, we performed an up-to-date comprehensive review and meta-analysis of all available studies investigating the association between SSRIs and ICrH. Our hypothesis was that study quality would influence the observed ICrH risk associated with SSRIs.
Methods
Eligibility criteria and search strategy
All studies comparing SSRI therapy to control (placebo or no treatment), assessing the risk of ICrH as a pre-defined primary or secondary outcome were evaluated, regardless of study design. The definition of ICrH used by each individual study was accepted but careful note was made of the precise outcome definition (ICrH, ‘haemorrhagic stroke’ or intracerebral haemorrhage). We excluded studies (i) not reporting clinical outcomes; (ii) not published as full text articles in English; and (iii) not differentiating between ischaemic and haemorrhagic stroke subtypes in the outcome.
We performed a systematic review of MEDLINE (1960 to December 2017), EMBASE (1980 to December 2017) and the Cochrane Library (until December 2017 Issue). The search strategy included keywords and MeSH terms relating to SSRIs and ICrH (Online Table 1). We manually searched reference lists of relevant studies and included studies after discussion with content experts. To include potentially relevant trial data that have assessed SSRIs versus control (placebo or no treatment) irrespective of population studied or the outcome assessed we also performed a post hoc umbrella review of published meta-analyses. 13
The review was reported according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines. 14 The project was prospectively registered with the PROSPERO database of systematic reviews (CRD42017084513).
Data collection, synthesis and risk of bias
Two investigators (MPJ and OJZ) independently extracted and tabulated data in a standardised data extraction form. Discrepancies and missing data were resolved by group discussion, reference to the original publication and additional independent adjudication (DJW). Where studies reported adjusted and unadjusted effect estimates, the estimate from the model that adjusted for the maximum number of covariates was extracted. 15 Careful note was made of the analysis method (including risk ratio [RR; preferred], odds ratio [OR] or hazard ratio [HR]) and the population studied (first-ever ICrH or recurrent ICrH).
Assessment of risk of bias was performed independently from data extraction, with each study assessed by two authors (OJZ and GB), using the Risk of Bias Assessment Tool for Nonrandomised Studies (RoBANS) for observational studies and the Cochrane Collaboration’s tool for randomised controlled trials. Both tools contain six domains each of which is judged as ‘low risk,’ ‘unclear risk’ or ‘high risk’ of bias. RoBANS domains are selection bias, confounding variables, exposure measurement, blinding, completeness of outcome data and selectivity of reporting. 16 The Cochrane tool domains include random sequence generation, allocation concealment, selective reporting, blinding of participants and personnel, blinding of outcome assessment and incomplete outcome data. 17 The total risk of bias score was calculated by summating the component scores across the six domains (where ‘low risk’ was allocated a score of 0, ‘unclear risk’ a score of 1 and ‘high risk’ a score of 2). Studies with a total risk of bias score of ≥3 were classified as ‘higher risk of bias’ whilst a score of <3 was classified as ‘lower risk of bias.’
Outcomes
The predefined outcome was ICrH, which was stratified according to the population studied: first-ever ICrH and recurrent ICrH. The definitions of these outcomes used by each individual study were accepted. To investigate the possibility that treatment effects vary between the definitions of intracranial bleeding reported, analyses were stratified according to whether they used the less specific term ‘haemorrhagic stroke’ or the more stringent definition ‘intracerebral haemorrhage.’
Statistical analysis
Baseline demographics comparing the SSRI and control groups were meta-analysed from all studies and are summarised as the odds ratio (OR). Random effects meta-analysis was pre-specified to combine estimates from different studies. Pooled binary event data for SSRI and control cohorts were compared using a RR with associated 95% confidence intervals (CI) using the method of DerSimonian and Laird. 18 In cases where the OR was described, these were converted to RR for meta-analysis (RR = OR/([1−pRef] + [pRef*OR]), where pRef is the prevalence of the outcome in the reference group. 19 RR and corresponding confidence intervals were then log-transformed before pooling. Studies reporting HR were included in the systematic review but not meta-analysed due to a scarcity of results presented in this way. Sensitivity analyses were performed according to (i) statistical methods (crude unadjusted or multivariate adjusted), (ii) study risk of bias score, (iii) a post hoc defined assessment of the bleeding definition used, and (iv) study design (prospective or retrospective).
Heterogeneity was quantitatively assessed using the I2 statistic, where an I2 of ≥50% indicates substantial heterogeneity and ≥75% indicates considerable heterogeneity. Meta-regression was performed to quantify the heterogeneity and assess the impact of baseline variables on estimates of ICrH. An exploratory meta-regression was performed according to the risk of bias attributed to each study. Publication bias was assessed by observational analysis of funnel plots and quantitatively assessed using Begg’s test and Egger’s test to identify small-study effects on the outcomes meta-analysed (α-value threshold = 0.05). A two-tailed p-value of 0.05 was considered statistically significant. Analyses were performed using STATA Version 13.1 (StataCorp LP, TX).
Results
The primary search strategy identified 27 observational studies for systematic review.8–12,20–41 We also identified three meta analyses which included 463 unique randomised controlled trials (RCTs).42–44 Of these, three RCTs comparing SSRIs with control reported ICrH as an adverse event and were suitable for quantitative synthesis (Figure 1).45–47 Study descriptors are summarised in Online Table 2. Twenty-seven studies were suitable for inclusion in the quantitative meta-analysis comparing SSRIs with control, including 845,655 patients and including data from 4,844,090 patient-years follow-up. Around 7.7% of patients were exposed to SSRI treatment compared to 92.3% receiving control (placebo or no treatment). The average length of follow-up was 3.23 years (interquartile range 1.82–5.50 years). Differences in key characteristics between SSRI and control groups are summarised in Table 1 (for full baseline demographics, see Online Table 3). Those treated with SSRIs were more likely to be female and have depression. There was no evidence of publication bias in reporting of ICrH (Egger p = 0.77; Begg p = 0.60) (Online Figure 1).
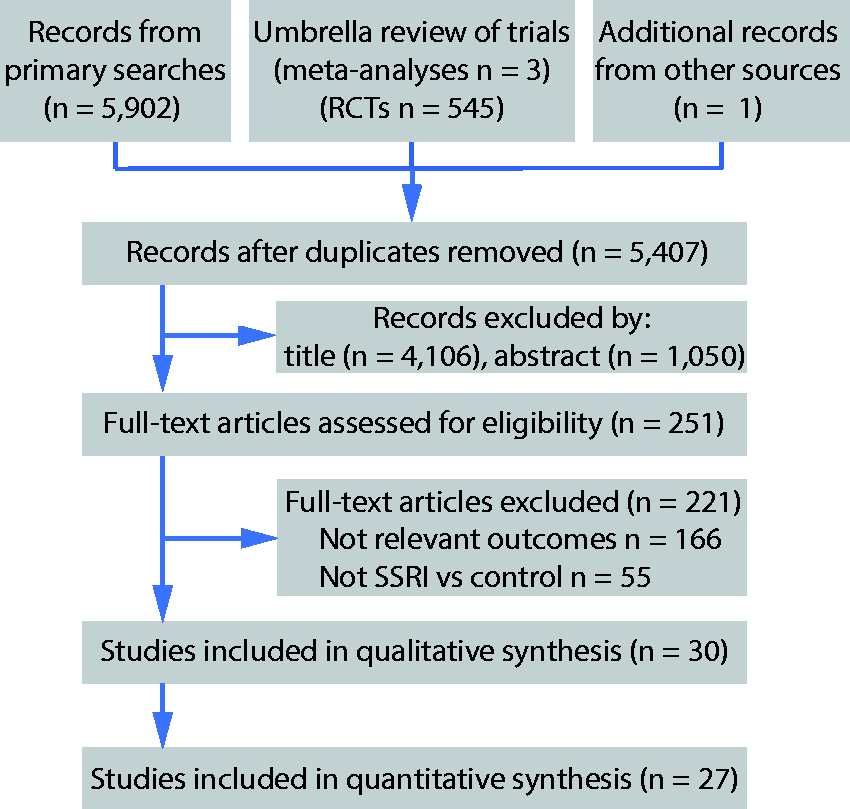
Systematic review flow diagram.
Associations between aggregated patient characteristics and SSRI use.
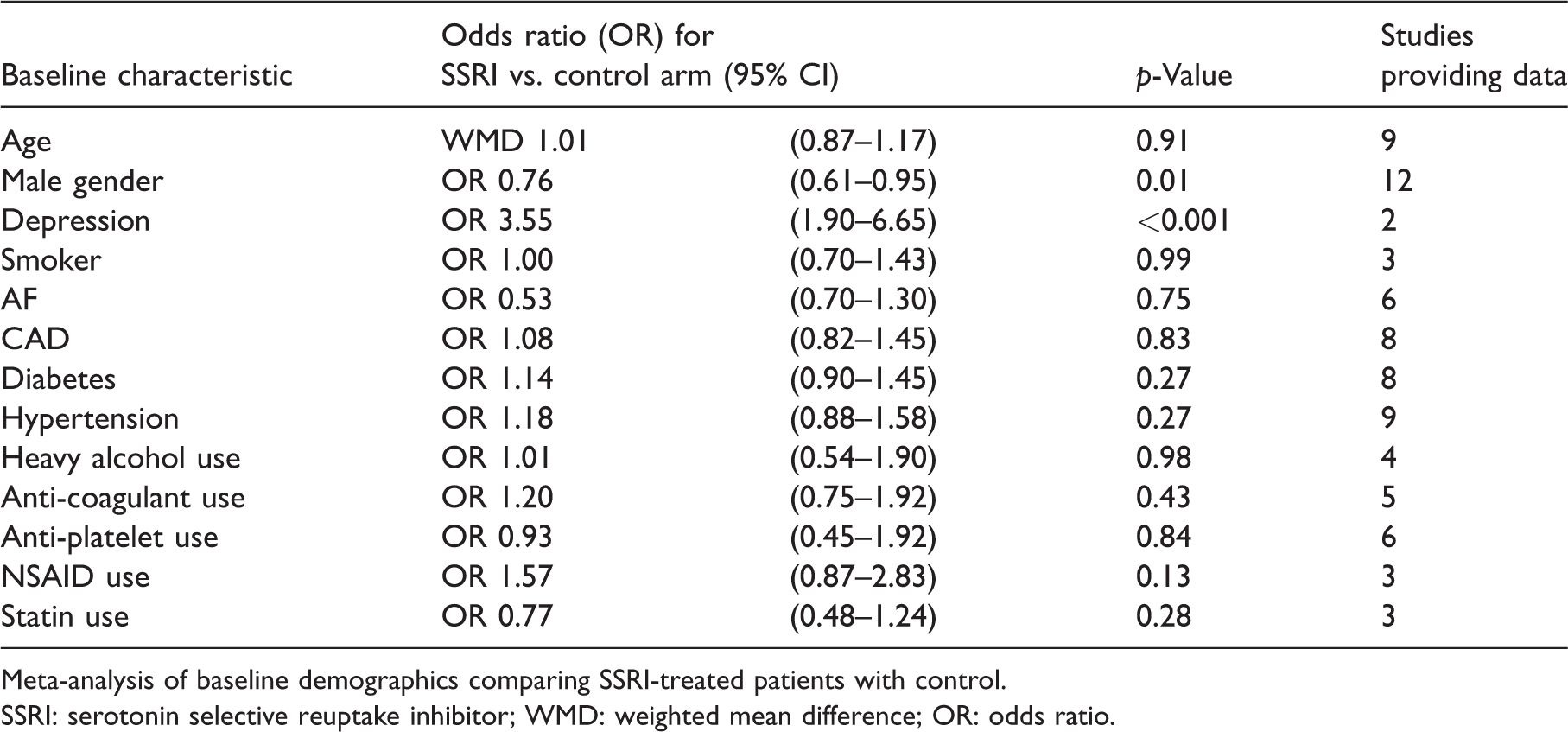
Meta-analysis of baseline demographics comparing SSRI-treated patients with control.
SSRI: serotonin selective reuptake inhibitor; WMD: weighted mean difference; OR: odds ratio.
Outcome: Intracranial haemorrhage
All included studies used CT head imaging or ICD coding to diagnose ICrH. Twenty-seven studies were suitable for meta-analysis for the outcome of ICrH (n = 845,655; Table 2).8,9,11,12,20–26,28–33,36–41,46–49 Overall, there was a significant increase in ICrH risk with SSRI therapy compared to control (RR 1.26, 95% CI 1.11–1.42; p < 0.001) but with substantial heterogeneity across studies (I2=76.1%) (Figure 2). In the subgroup of patients without previous ICrH (24 studies, n = 824,409), there was a significant increase in first-ever ICrH risk with SSRI therapy compared to control (RR 1.31, 95% CI 1.15–1.48; p < 0.001) but with substantial heterogeneity across studies (I2=75.4%).8,9,20–26,28–33,37–41,45–48 In the ICrH survivor subgroup, only three studies were available (n = 21,246), which found no association between SSRI use and recurrent ICrH (RR 0.95, 95% CI 0.83–1.09; p = 0.48) with no heterogeneity across studies (I2=0.0%).11,12,36
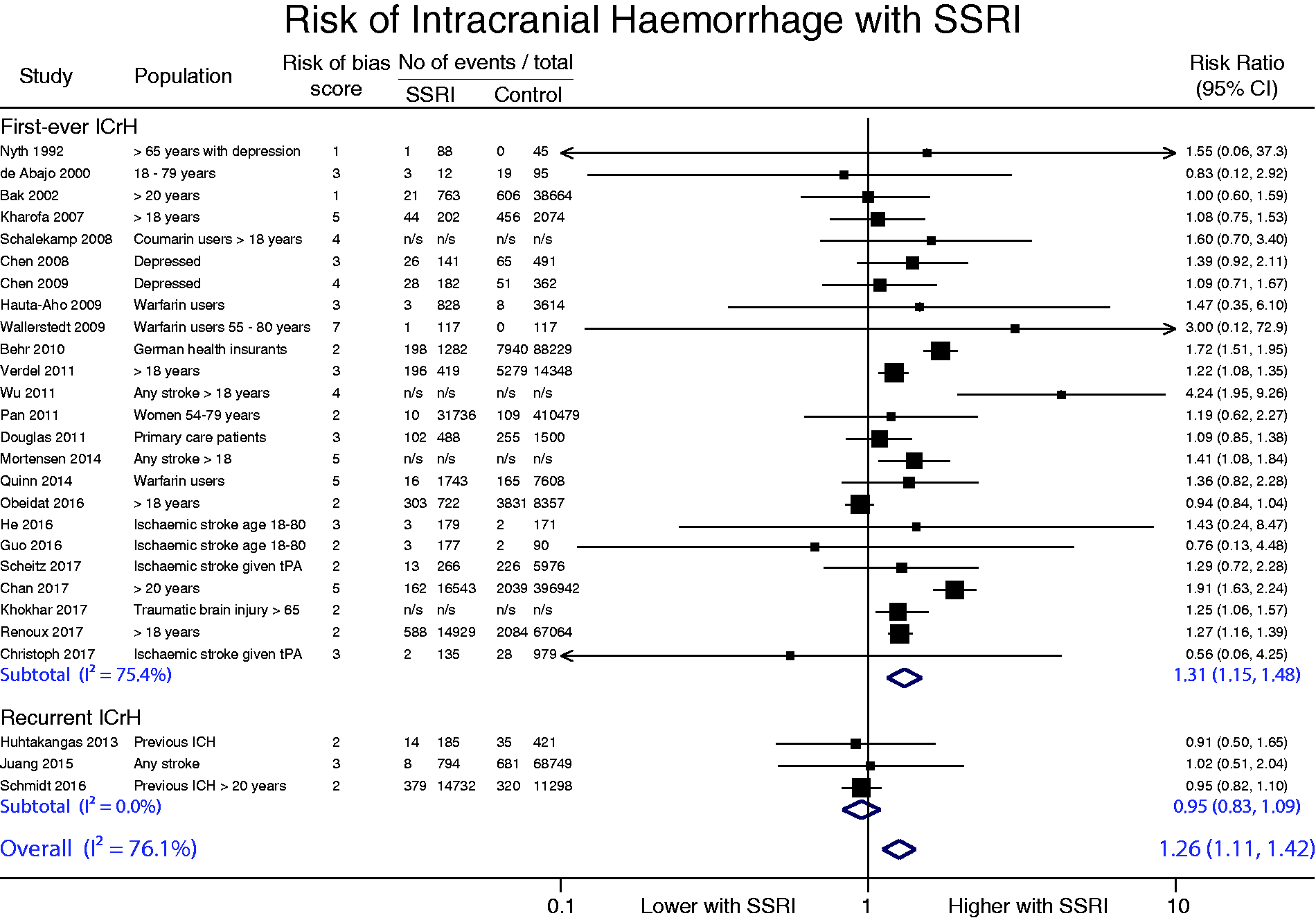
Meta-analysis of serotonin selective reuptake inhibitors on intracranial haemorrhage. Forest plot is subgrouped by subpopulation. First-ever ICrH refers to ICrH events in ICrH naïve individuals and Recurrent ICrH refers to ICrH events in ICrH survivors. The diamond represents the pooled difference using a random-effects model. I2 is the percentage of total variation across studies due to heterogeneity.
Summary of studies.
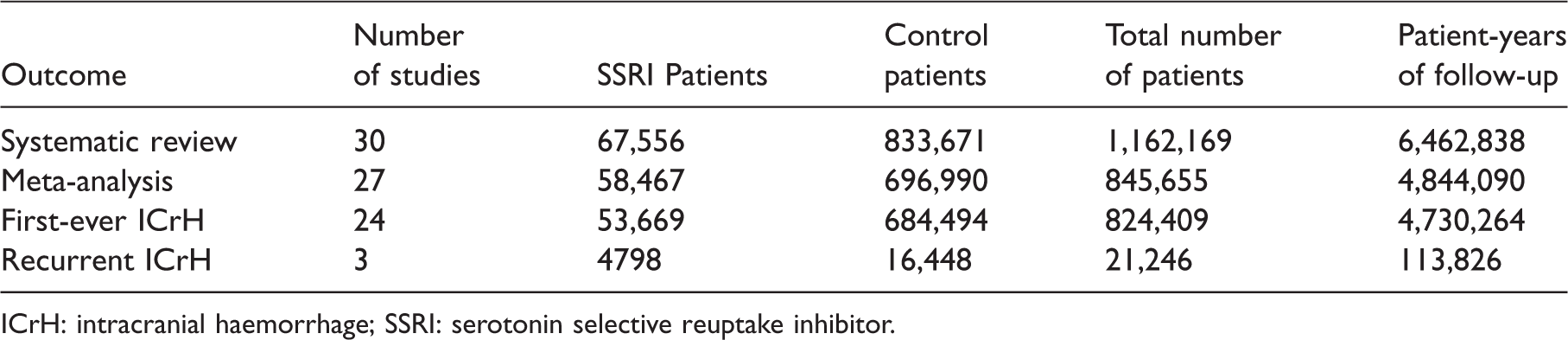
ICrH: intracranial haemorrhage; SSRI: serotonin selective reuptake inhibitor.
We performed a post hoc defined sensitivity analysis to assess the impact of the intracranial bleeding definition reported on event rates. The definitions chosen by component studies were typically based on the International Classification of Diseases (ICD) diagnostic codes used but occasionally were defined by imaging and clinical data. Haemorrhagic stroke mostly included both subarachnoid and intracerebral bleeding although this was often not reported. In studies using the term ‘haemorrhagic stroke,’ SSRIs were significantly associated with increased intracranial bleeding (nine studies,12,23,25,30,31,33,38,39,41 n = 577,473, RR 1.40, 95% CI 1.13–1.72; p = 0.002); however, when ‘intracerebral haemorrhage’ was reported, no association was found (nine studies,9,11,20–22,29,36,46 n = 155,240: RR 1.11, 95% CI 0.86–1.42; p = 0.44) (Online Figure 2). A sensitivity analysis assessing statistical methods revealed that crude unadjusted analyses reported a much stronger association between SSRI and ICrH (RR 1.40, 95% CI 1.18–1.67, p < 0.001) than multivariate adjusted analyses (RR 1.15, 95% CI 1.02–1.31, p = 0.03). Additionally, a sensitivity analysis assessing study design revealed that although retrospective studies reported an increased risk of ICrH with SSRI use (RR 1.34, 95% CI 1.19–1.51, p < 0.001), the was no difference in prospective studies (RR 0.96, 95% CI 0.88–1.04, p = 0.31; Online Figure 3).
The risk of bias in individual studies is presented in Online Tables 4 and 5. The risk of bias category that contributed most to the bias score was due to ‘inadequate blinding of outcome assessments.’ The ‘incomplete outcome data and selective reporting’ category was often unclear. The remaining categories showed generally low risk of bias. We performed an exploratory meta-regression of the effect of study-level bias on ICrH comparing SSRI therapy to control. This revealed that studies with higher risk of bias were more likely to report an association of SSRI and ICrH albeit non-significantly (p = 0.06; Figure 3). This was supported by a sensitivity analyses according to risk of bias, demonstrating that whilst studies with high risk of bias (RoBANS score ≥3) reported a significant association between SSRIs and ICrH (RR 1.36, 95% CI 1.15–1.61, p < 0.001), studies with ROBANS score <3 did not (RR 1.16, 95% CI 0.98–1.39, p = 0.09).
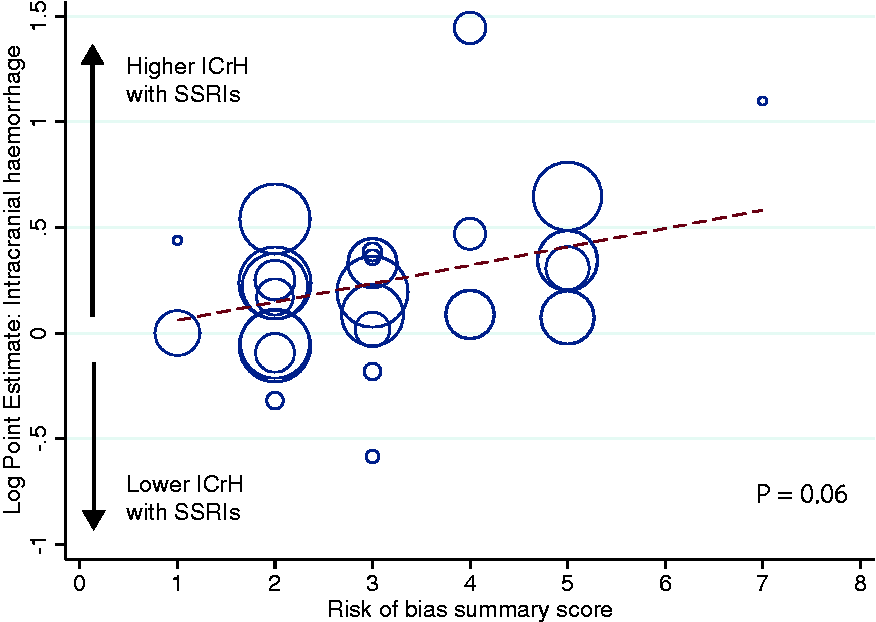
Meta-regression of risk of bias score on the risk of intracranial haemorrhage. All analyses that provided data on rates of intracranial haemorrhage were included, regardless of the study design. See Online Tables 4 and 5 for statistical methods. Each circle represents an individual study, with the circle size dependent on the precision of each estimate in a random-effects model. The risk of bias in each study was summated across all six domains. p = 0.06.
Meta-regression was used to explore the impact of differences in key baseline characteristics between SSRI and control patients with ICrH. There was inadequate reporting of baseline characteristics in individual studies to sufficiently power this meta-regression (Online Table 3).
Discussion
Although SSRI use appears to be associated with an increased risk of ICrH, studies with a higher risk of bias and poorer methodological quality reported a stronger association, raising suspicion that this relationship may be due to extraneous influences rather than direct causation. These findings are based on a comprehensive meta-analysis of 24 observational studies and three randomised trials, including a combined total of 4,844,090 patient-years of follow-up. Based on our analysis, important differences in baseline characteristics between patients in the SSRI and control groups, as well as inadequate statistical adjustment significantly distort effect estimates. However, given the high level of heterogeneity, these results should be interpreted with caution with the need for careful risk–benefit analysis prior to initiating therapy.
To date, no previous meta-analysis assessing ICrH risk with SSRIs has considered the quality of study methodology when synthesising their results. Compared to randomised trials, observational data are less likely to generate unbiased estimates regarding risk and should be viewed as hypothesis-generating, rather than definitive. The prior meta-analyses tended to accept that statistical adjustment for recognised confounders sufficiently accounts for bias associated with observational studies. However, the substantial heterogeneity in treatment effects between studies utilising statistical adjustment suggests that even sophisticated statistical methods should be interpreted with caution and cannot replace randomisation; despite careful adjustment, important confounders can be unmeasured, unidentified or concealed. Even with a reasonable selection of adjustment variables, when treatment and control groups differ vastly in characteristics, reliable effect estimates are not possible without breaching the assumptions of the statistical model. Indeed, there were large variations in the degree to which individual studies adjusted their final analyses. For example, one study performed a Cox regression analysis adjusting for age, sex and medications and reported a 3.5-fold increase in ICrH with SSRIs, 28 whereas another incorporated 14 possible confounders into a bivariable binary logistic regression model and revealed an OR of 0.55, albeit with wide 95% CI (0.06–4.71). 10 This variability can be surpassed by randomisation, which is the optimal method to eliminate selection bias and overcome issues with statistical adjustment. Although randomised trials are less likely to generate unbiased estimates, trial data specifically assessing ICrH is unlikely to become available. Previous trials have not systematically collected data on ICrH, and new trials with sufficient power to assess a potential causal relation between SSRI use and first-ever ICrH are unlikely to be performed. Nonetheless in this meta-analysis, by considering study quality, we identified that well designed observational studies that minimise selection bias and optimise statistical adjustment were less likely to report an association between SSRI and ICrH.
In addition to variations in statistical adjustment techniques, using a sensitivity analysis we found that the substantial heterogeneity is partially explained by differences in how the bleeding event was defined. Studies using the terms ‘haemorrhagic stroke’ and ‘intracranial haemorrhage’ were more likely to find a positive association between SSRIs and bleeding events compared to the definition ‘intracerebral haemorrhage.’ This may reflect risk differences across patient categories (i.e. increased risk of other intracranial haemorrhages other than intracerebral haemorrhage, e.g. subarachnoid, subdural, or extradural haemorrhage) or differences in specificity of definitions. Eleven of the 18 studies using the terms ‘haemorrhagic stroke’ or ‘intracranial haemorrhage’ did not specify how they defined this non-specific category. This further supports the notion that studies with less clearly defined outcome measures and poorer methodology are more likely to report an increase in bleeding events with SSRIs.
Psychiatric conditions, such as depression, are linked to many medical conditions and are itself an independent risk factor for ICrH. 50 As SSRIs are not prescribed until clinicians detect deterioration in patients’ mental health, treatment with SRRIs is likely to be influenced by the probability of these comorbid illnesses, causing ‘confounding by indication.’ In support of this, we found that pooling baseline characteristics amongst the included observational studies identified that, as expected, those on SSRIs had more depression than controls. Furthermore, studies which utilised depression as an inclusion criterion, thus addressing this confound, observed no association between SSRIs and ICrH.23,25
To date, no previous meta-analysis assessing ICrH risk with SSRIs has included data from SSRI randomised trials to determine whether ICrH has been reported as an adverse event, irrespective of the RCT study population or primary outcome assessed. We identified three RCTs reporting ICrH as an adverse event and in all of these studies ICrH rates were not significantly different between the SSRI and control groups, albeit event rates were low. Indeed, in the trial by Nyth et al., 46 the cerebral haemorrhage occurred in one patient in the citalopram group 12 days after initiating therapy, and the authors conclude that a causal relationship to SSRI therapy was improbable.
Although we found a significant increase in first-ever ICrH risk with SSRIs, there was no association with recurrent ICrH. However, since only three small studies assessed recurrent ICrH, this analysis is likely to be underpowered. The absolute risk of recurrent ICrH is low (1.2% per year), 51 emphasising the need to pool results to increase statistical power. Indeed, the association between SSRIs and first-ever ICrH was only recognised after meta-analysis of a large number of individual studies,6,7 each failing to achieve statistical significance alone. 20 Nonetheless, even with adequately powered meta-analyses, randomised trials will still be necessary to clarify any causal association.
Current European and American guidelines make no recommendations on the class of antidepressant to use post-stroke.4,5 From our analyses, there is insufficient data to advise restriction of SSRI post-ICrH. Although results have been conflicting SSRIs appear to have beneficial effects particularly in the treatment of post-stroke depression.52,53 Moreover, recent trial data has shown that SSRIs post-stroke may speed motor recovery post-stroke45,54 and prevent new onset of apathy. 55 These potential benefits need to be balanced against any potential ICrH risks depending on ICrH aetiology and severity, comorbidities and coexisting therapies. 56 Three large multicentre randomised controlled trials of fluoxetine in patients with recent ICrH are in progress (FOCUS, AFFINITY, and EFFECTS ClinicalTrials.gov Identifier: NCT02683213), and should provide important data on how SSRIs modify ICrH risk and affect functional outcome and quality of life.
Strengths and limitations
Our review is based on published data of independent studies, performed in accordance with an explicit, reproducible methodology. We explore, for the first time, the association between SSRIs and both first-ever and recurrent ICrH. Additionally, we utilised a comprehensive risk of bias score to inform the validity of our conclusions, demonstrating that the association between SSRIs and ICrH is at least partly accounted for by poor methodological quality in the available observational data.
We acknowledge drawbacks, primarily reflecting the limitations of component observational studies. There was scarce reporting of baseline characteristics with variation in definitions used. We were unable to meta-analyse SSRI dose, SSRI type or the impact of location of ICH (lobar versus deep) due to insufficient data available. Only aggregate, rather than individual participant data, was available, increasing the risk of falsely inferring individual characteristics from group data. Moreover, selective reporting within studies, in particular outcome non-reporting, which is not fully accounted for by the RoBANS checklist, may have put the overall treatment effect estimate at risk of bias. In addition, due to expected disparities in study design and populations, we pre-specified a random-effects model. Although no significant publication bias was detected, we noted significant heterogeneity for the ICrH outcome, which can be attributed to both effect magnitude and direction.
Conclusion
Although SSRIs appear to be associated with increased risk of ICrH, studies of poorer quality reported a stronger association indicating that this association may be due to unmeasured confounders rather than direct causation. Observational data is subject to inherent confounding, including by indication and due to co-morbidities and co-prescription that cannot be mitigated, even by statistical adjustment. Our data do not support withholding SSRIs to reduce the risk of ICrH and physicians decisions should be based on a carefully considered individualised assessment of indication, comorbidities and the goal of antidepressant therapy.
Supplemental Material
Supplemental material for The impact of selective serotonin reuptake inhibitors on the risk of intracranial haemorrhage: A systematic review and meta-analysis
Supplemental Material for The impact of selective serotonin reuptake inhibitors on the risk of intracranial haemorrhage: A systematic review and meta-analysis by Melanie P Jensen, Oliver J Ziff, Gargi Banerjee, Gareth Ambler and David J Werring in European Stroke Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MPJ, OJZ, GB, and GA have no relevant conflicts. DJW was UK chief investigator for A9951024 (Pfizer) and has received consultancy and lecture fees from Bayer.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was undertaken at University College London Hospitals NHS Foundation Trust/University College London who received a proportion of funding from the Department of Health’s National Institute for Health Research Biomedical Research Centers funding scheme. OJZ is funded by the National Institute of Health Research as an Academic Clinical Fellow. GB received research support from the Rosetrees Trust. DJW receives research funding support from the British Heart Foundation and the Stroke Association (TSA BHF 2009/01; PPA 2015/04).
Ethical approval
Not required.
Guarantor
DJW.
Contributorship
MPJ and OJZ contributed equally by developing the eligibility criteria, performed the primary literature search, contributed to data extraction and drafting of the manuscript. GB contributed to data extraction and critical revision of the manuscript. GA contributed to statistical analysis and critical revision of the manuscript. DJW designed the study concept, led the study group and critically revised the manuscript.
Acknowledgements
We thank Kate Brunskill, Deputy Librarian at the Queens Square Library, UCL Institute of Neurology for her expertise and guidance in finalising the search strategy.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
