Abstract
Purpose:
To review the current evidence on anticoagulation for intracranial haemorrhage (ICrH) survivors with atrial fibrillation (AF).
Method:
Narrative review of the literature.
Findings:
AF and ICrH are age-related conditions whose prevalence and comorbidity is expected to increase with the ageing population. Patients with ICrH were excluded from pivotal randomized trials of anticoagulation in AF and guidelines do not provide strong recommendations on if and when to (re)initiate anticoagulation in patients with AF and ICrH. Pooled analyses of phase II randomized trials have reported reduced risk of ischaemic major adverse cardiovascular events with anticoagulation in this population, but there remains uncertainty regarding the effects of anticoagulation on recurrent ICrH and death, as well as potential heterogeneity of treatment effect in higher risk subgroups, such as patients with cerebral amyloid angiopathy. There are no reported randomized trials investigating the optimal timing of anticoagulation (re)initiation in ICrH survivors with AF and the findings from observational studies have been inconsistent.
Conclusion:
Whether or not OAC should be resumed in ICrH survivors with AF and the optimal timing of OAC (re)initiation are challenging clinical dilemmas that are becoming more frequent with our ageing population. The existing prevalence of AF in patients with ICrH and changing global demographics highlight the importance of ongoing and future randomized trials addressing unresolved questions surrounding optimal stroke prevention strategies in this vulnerable patient population.
Keywords
Introduction
Atrial fibrillation (AF) is the leading cause of cardioembolic ischaemic stroke. AF-associated ischaemic strokes have a 55%–60% likelihood of causing death or disability. 1 Oral anticoagulation (OAC) is highly effective at preventing stroke in the setting of AF, but is consistently underused. 2 Physicians’ and patients’ concerns about bleeding risk are the main impediment to using OAC in this population. 3 This concern is amplified in patients with prior intracranial haemorrhage (ICrH). 4 ICrH, characterized by bleeding within the cranium comprises of various subtypes, including epidural, subdural, subarachnoid and intracerebral (intraparenchymal or intraventricular) haemorrhages. As a history of ICrH was an exclusion criterion for all the pivotal randomized controlled trials (RCT) that established the benefit of OAC in patients with AF, current guidelines do not provide clear recommendations on the use of antithrombotic prophylaxis in these patients.5 –8
Unsurprisingly, physician surveys demonstrate significant uncertainty with broad variations of practice both globally and amongst institutions within the same region. 9 Data from European hospital cohorts showed a 20% antithrombotic therapy resumption rate, with significant variation between institutions (range: 11%–45%). 10 In a 2008 Japanese nationwide survey of ‘neuro-specialists’, 91% of respondents resumed OAC after intracerebral haemorrhage, while 6% avoided antithrombotic therapy altogether. 11 A physician survey of 228 stroke neurologists, neurosurgeons and thrombosis experts demonstrated 30%–98% resumption rate in intracerebral haemorrhage (ICH) survivors with AF depending on the clinical scenario. 4 In this survey, physicians felt most comfortable resuming OAC following traumatic ICH (98% resumption) and were least likely to do so in patients with ICH occurring due to cerebral amyloid angiopathy (CAA), where 30% of respondents indicated they would resume OAC. 4 However, pilot phase RCTs of OAC for AF after ICrH have been completed since these surveys of practice, and main phase RCTs are ongoing.12 –14
In this manuscript, we provide a comprehensive overview of the present literature on OAC in ICrH survivors with AF and outline future directions on the topic, with a focus on patients with spontaneous ICrH resulting from age-related cerebral small vessel diseases (ICH or convexity subarachnoid haemorrhage) and patients with subdural haemorrhages.
Epidemiology
AF and ICrH are age-related conditions that additionally share hypertension as a common risk factor. 15 Indirect comparisons across studies suggest global variation in the prevalence of AF amongst ICrH survivors that range between 10% and 35% in Western cohorts relative to 2%–9% in East-Asian populations.16 –19 This variation is likely a reflection of the younger age of patients with ICrH in East Asia who typically present a decade earlier than their Western counterparts. The prevalence of AF and ICrH, and their ensuing comorbidity, are expected to increase with the ageing global population.20,21 A Spanish population-based study reported a 50% increase in the prevalence of AF in ICH patients between 2003 (10.5%) and 2012 (15.5%). 22 The dilemma of optimal stroke prevention in ICrH survivors with AF is thus expected to become more frequent in the next decades.
Observational studies and RCTs have reported patients with ICrH and AF to be typically in their 7th decade of life and more often male (60% male, 40% female).23 –26 Hypertension (60%–96%) and congestive heart failure (24%–48%) are prevalent comorbidities and roughly a third of the population has had prior history of thromboembolism. The average CHA2DS2-VASc and HAS-BLED scores reported in ICrH survivors with AF range from 4 to 5 and 2–3, respectively, indicating a vulnerable disease population that is at a heightened risk of both thrombotic and haemorrhagic major adverse cardiovascular events (MACE).23 –26 The overlapping risk factors within these scores that are prevalent in patients with ICrH and AF include older age, hypertension and prior history of stroke.
Risk of major adverse cardiovascular events
Recent data have consistently demonstrated elevated risks of ischaemic and haemorrhagic MACE in survivors of ICrH, particularly in the context of comorbid atrial fibrillation.27 –32
In a French prospective observational study (n = 310, 11% with AF), ICH survivors were observed to have cumulative ischaemic stroke and major ischaemic vascular events incidence rates of 9.0% and 15.2% at 5 years, respectively, while the rates of recurrent ICH and major haemorrhagic events were lower (4.9% and 6.2% at 5 years, respectively). 27 This was particularly the case in patients with non-lobar or deep ICH where the rate of ischaemic stroke at 5 years (11.2%) was much greater than ICH recurrence (3.2%). In contrast, amongst patients with lobar ICH the rates of recurrent ICH were slightly greater than ischaemic stroke (7.9% vs 5.3% at 5 years). 27 In a population-based study from China (n = 45,732) the 5-year rates of stroke recurrence (44% vs 41%) and all-cause mortality (28% vs 16%) beyond the first 28 days following the incident stroke were higher in patients with ICH compared to patients with ischaemic stroke. 28 Recurrent strokes in patients with incident ischaemic stroke were infrequently due to ICH (7%), whereas in patients with incident ICH the proportion of recurrent stroke due to ischaemia was common (41%).28,29
The rates of ischaemic vascular events seem to remain heightened indefinitely after ICrH, however there is suggestion of greater rates in the initial few months following the index event. A Dutch study of 19,444 30-day ICH survivors reported a 10-year ischaemic stroke risk of 2.6%–7% (depending on age and sex) that was similar to the risk of recurrent ICH during this time (3.7%–8.1%). 29 In an individual participant data meta-analysis of 47,886 participants from four population-based US cohorts (4% with AF; 318 with incident ICH) the risk of ischaemic stroke and myocardial infarction were 3- (HR 3.1; 95%CI, 2.1–4.5) and 2- (HR 1.9; 95%CI, 1.2–2.9) fold higher, respectively, in participants with ICH compared to participants without. 31 The heightened risk of ischaemic arterial events seemed consistent in the short (<1 year) and long term (>1 year) following ICH. 31 On the other hand, a retrospective cohort study of Medicare beneficiaries in the United States diagnosed with ICH (n = 5924; 28% with AF) observed that the risk of acute ischaemic stroke was greatest in the first 6 months following ICH, and greatest in the first 3 months in patients with ICH and AF. 30
Amongst ICH survivors, the risk of ischaemic vascular events is reported to be greater in patients with AF compared to patients without AF. In a pooled analysis of two UK population based registries, patients with ICH and AF were at an eightfold increased risk of ischaemic stroke compared to patients without AF (6.3 vs 0.7 per 100 patient-years [HR 8·2, 95%CI: 3·3–20·3]), whereas the risk of recurrent ICH seemed similar in both groups (3.3 vs 3.2 per 100 patient-years). 32 In patients with ICH and AF, the rates of ischaemic stroke were greater than recurrent ICH irrespective of haematoma location (non-lobar ICH: 5.6 per 100 patient years rate of ischaemic stroke and 3.6 rate of recurrent ICH; lobar ICH: 7.3 per 100 patient years rate of ischaemic stroke and 4.4 rate of recurrent ICH). 32 Intuitively, the rates of thrombotic events in ICH survivors with AF not taking OAC are higher than that reported in cohorts of patients receiving various degrees of antithrombotic prophylaxis. In a Danish national cohort registry, the rate of ischaemic stroke/systemic embolism in ICrH survivors with AF was 10.4 per 100 patient-years without OAC therapy. 24 Similarly, in the multicentre German RETRACE registry, the rate of ischaemic stroke was 11.2% at 1 year following ICrH in patients with AF not receiving OAC. 25 The rates of ischaemic stroke reported in phase II RCTs comparing OAC versus avoidance of OAC in ICrH survivors with AF have reported annualized ischaemic stroke rates ranging between 6 and 16% in patients assigned to OAC avoidance.12 –14 Interestingly, these rates are higher than would be anticipated based on the median CHADS2 or CHA2DS2-VASc scores of these populations.12 –14,24,25 These findings would suggest that the inherent factors leading to ICrH in patients with AF are also driving an increased risk of arterial thrombotic events through risk factors or pathways that are not currently captured in our traditional AF thrombotic event risk scores.
Patients with ICrH and AF are clearly also at heightened risk of recurrent ICH. As a whole the reported annualized rates of recurrent ICH range between 3% and 7.5%.27 –32 The rate of ICH recurrence differs according to the underlying mechanism and is reported to be twofold to fourfold greater in patients with lobar ICH presumed to result from CAA compared to patients with non-lobar ICH resulting from arteriolosclerosis. 33 In the aforementioned UK population based cohort study, the rate of recurrent ICH was 4.4 per 100 patient-years following lobar ICH and 3.4 per 100 patient-years in patients with non-lobar ICH. 32 Data would indicate that the risk of recurrent ICH is greatest amongst lobar ICH patients with MRI evidence of CAA34,35 who have a 7%/year risk of ICH recurrence and that this is particularly driven by patients with disseminated cortical superficial siderosis (involving four or more non-contiguous sulci) or isolated convexity subarachnoid haemorrhage who are at a 13%/year risk of ICrH recurrence.34,36 The heightened risk of recurrent ICrH following an initial ICrH is lifelong, however data would indicate that patients with CAA and those with subdural haemorrhage are at greater risk of early recurrence.37,38
Risks versus benefits of oral anticoagulation (observational studies)
Data from observational registries comparing OAC with avoidance of OAC in ICrH survivors with AF have been reassuring and suggest persistent benefit with OAC in this vulnerable patient population. In a large Danish national cohort study of 1752 ICrH survivors with AF, the 1 year combined end point of ischaemic stroke/systemic embolism and all-cause mortality was reduced from 27.3% in patients without any antithrombotic treatment to 13.6% in OAC treated patients (adjusted HR 0.55; 95%CI 0.39–0.78), without an increase in recurrent ICH (8.0% vs 8.6%; adjusted HR, 0.91; 95%CI, 0.56–1.49). 24 Similarly data from the German multicentre RETRACE registry demonstrated a 69% relative risk reduction (RRR) in ischaemic stroke with resuming OAC versus avoiding OAC in 261 ICrH survivors with AF, without an accompanying increased risk of recurrent ICH (3.9% per 100 patient-years in both groups). 25 These results are consistent with a study-level meta-analysis of eight studies with total of 5306 patients with ICrH (73% with AF) that reported a 66% RRR in arterial thrombotic events (p for heterogeneity = 0.28), without an increase in recurrent ICrH (RR 1.01; 95%CI 0.58–1.77; p for heterogeneity < 0.001) in these patients, albeit with significant heterogeneity for the effect of OAC on the outcome of recurrent ICrH. 39
An individual participant data meta-analysis of three registries (two from the United States and the German RETRACE registry) with total of 1012 anticoagulant-related ICH survivors with AF further stratified response to treatment according to ICH location. 26 There were 633 non-lobar ICH and 379 lobar ICH in this analysis, of whom 28% and 23% were resumed on OAC, respectively, at a median of 35–44 days. Resuming versus avoiding OAC led to significant reductions in ischaemic stroke in patients with ICH in both locations (HR 0.39, 95%CI 0.21–0.74 in non-lobar ICH; HR 0.48, 95%CI 0.25–0.75 in lobar ICH) and there was no significant excess risk of recurrent ICH with OAC resumption (HR 1.17, 95%CI 0.89–1.54 in non-lobar ICH; HR 1.26, 95%CI 0.88–1.71 in lobar ICH) in either group. 26 Moreover, among 190 patients in this study with adequate MRI data, patients meeting modified Boston Criteria for probable CAA had significant reductions in mortality (HR 0.30, 95% 0.10–0.92) and greater likelihood of achieving a good outcome at 1 year (mRS 0–3; HR 3.11, 95% 1.08–8.97) with resumption of OAC. 26 There were insufficient numbers of ischaemic stroke or recurrent ICH in this smaller subgroup of patients for meaningful analyses of these outcomes according to OAC status in patients with possible or probable CAA. These studies have additionally shown impressive reductions in all-cause mortality associated with resumption of OAC with RRR ranging from 44% to 75%, irrespective of ICH location.24 –26
The totality of the observational literature suggests net benefit from OAC in ICrH survivors with AF, with large reductions in arterial ischaemic events and mortality without an associated increase in the rates of recurrent ICrH. Surprisingly, these reassuring results come predominantly from studies where patients were treated with vitamin K antagonists rather than the safer alternative of direct oral anticoagulants (DOACs).24 –26 Vitamin K antagonists have been reported to increase the risk of recurrent ICH at least twofold compared to placebo in the pivotal AF trials and it would be optimistic to conclude that their safety profile is enhanced in ICrH survivors on the basis of these results. 40 Moreover, randomized trials have only previously indicated a 26% RRR in mortality with warfarin versus control, 40 which is substantially less than the magnitude of mortality benefit being reported in these observational studies. These discrepancies are strong indicators of residual confounding in these observational datasets and in particular confounding by indication and immortal time bias which limit the interpretation and clinical utility of their results, and therefore mandate the conduct of RCTs to resolve this therapeutic dilemma.
Risks versus benefits of oral anticoagulation (randomized trials)
Completed phase II randomized trials
There are three completed phase II open-label RCTs testing OAC versus no OAC in ICrH survivors with AF completed to date.12 –14 These are summarized in Table 1. The first (NASPAF-ICH; NCT02998905) was a small feasibility trial conducted in Canada of 30 participants with ICH and high-risk AF (CHADS65 indicating OAC) who were assigned (2:1) to receive OAC with a DOAC (drug as per discretion of local investigator; standard dosing mandated) or aspirin 81 mg daily. 12 Participants were eligible if at least 2 weeks had passed since their qualifying ICH (median time from index ICH to randomization was 127 days), and all participants were provided with home blood pressure monitors to ensure strict blood pressure control to target of less than 130/80 mm Hg during study participation. The trial only accrued one primary outcome of any stroke (an ischaemic stroke occurring in the aspirin arm; 1/9, 11%) over a median follow-up of 1.5 years, but there were numerical trends suggesting less arterial ischaemic events and net benefit with DOAC therapy, albeit with very few number of events. 12 SoSTART (NCT03153150) was a subsequent non-inferiority trial with internal feasibility phase conducted in the UK. It enrolled 203 patients with high-risk AF (CHA2DS2-VASc score of 2 or more) and ICrH who survived at least 24 h following their index ICrH. 14 Participants were randomized (1:1) to receive OAC (anticoagulant and dose at the discretion of the local investigator) or avoiding OAC (antiplatelet treatment or no antithrombotic therapy). 14 Only 31% of patients assigned to avoiding OAC received antiplatelet therapy and the majority assigned to OAC (96%) received a DOAC at a median of 104 days following the index ICH. Over a median of 1.2 years follow-up, there were eight recurrent symptomatic ICrH (7/8 fatal) in the OAC arm compared with four ICrH (none fatal) in the avoid OAC arm (adjusted HR 2·42, 95% CI 0·72–8·09), which did not provide evidence of non-inferiority for this primary outcome (non-inferiority margin equated to HR of 3.2; estimated as upper limit for ICrH recurrence to offset benefit from ischaemic stroke reduction). 14 Of note, two of the four ICrH occurring in patients assigned to avoiding OAC occurred in patients who had crossed over and were receiving OAC at the time of their recurrent ICrH. This numerical increase in ICrH was offset by a large reduction in ischaemic stroke, which occurred in 19 patients in the avoid OAC arm compared with 3 in the OAC arm, and overall, there was a suggestion of a 47% RRR in the outcome of any stroke and 45% RRR in the combined outcome of any stroke or vascular death with OAC, however these effects did not reach statistical significance. 14 The results of SoSTART were followed by that of the APACHE-AF (NCT02565693) trial. APACHE-AF was a randomized phase II trial conducted in the Netherlands that enrolled 101 patients with high-risk AF (CHA2DS2-VASc score of 2 or more) and OAC-associated ICH occurring within 7–90 days of randomization. Participants were assigned (1:1) to receive apixaban (standard dosing) or avoid OAC (antiplatelet or no antithrombotic therapy). 13 Half (51%) of the 50 participants assigned to avoid OAC received antiplatelet medication and 11 crossed over to receive OAC. The median time from the index ICH to randomization was ~45 days. Over a median follow-up of 1.9 years, the primary outcome of non-fatal stroke or vascular death occurred in 26% (13/50) of patients assigned to apixaban and 24% (12/51) of those assigned to avoiding OAC (adjusted HR 1·05, 95% CI 0·48–2·31). There were four (8%; 1/4 fatal) recurrent ICH in the apixaban arm compared with 1 (2%; none fatal) in the avoid OAC arm (adjusted HR 4.09, 95% CI 0·45–36.91). The recurrent ICH in the avoid OAC arm occurred following cross-over to OAC with rivaroxaban. Perhaps due the small number of events or crossover to no OAC in 9/50 participants initially assigned to apixaban, there was no reduction in ischaemic stroke observed with apixaban versus avoiding OAC (12% in each arm) and there was a numerically excess risk of any stroke with apixaban versus avoiding OAC (20% vs 14%). However, numerical reductions in major occlusive events (12% vs 22%) and vascular death (10% vs 14%) were observed with apixaban in APACHE-AF. 13
Completed phase II randomized trials.
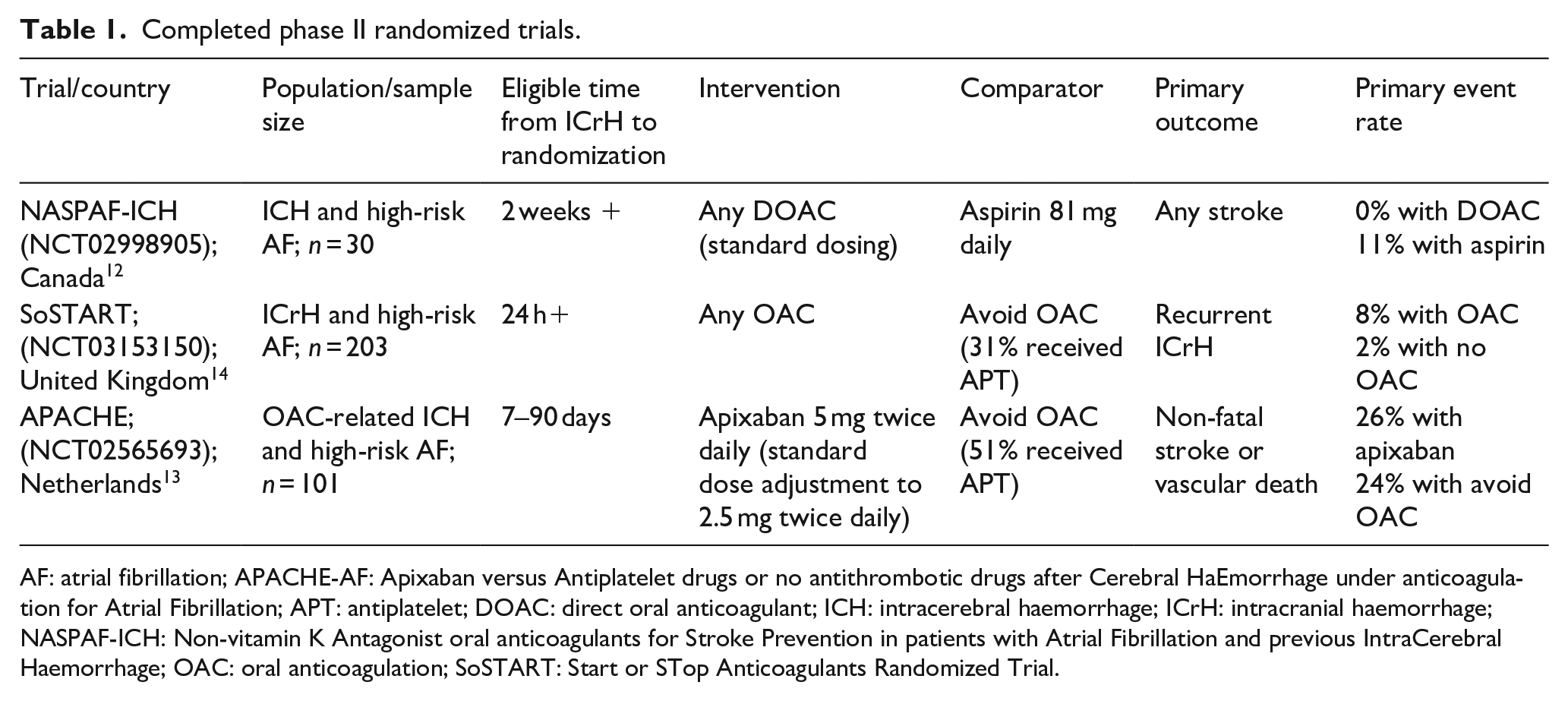
AF: atrial fibrillation; APACHE-AF: Apixaban versus Antiplatelet drugs or no antithrombotic drugs after Cerebral HaEmorrhage under anticoagulation for Atrial Fibrillation; APT: antiplatelet; DOAC: direct oral anticoagulant; ICH: intracerebral haemorrhage; ICrH: intracranial haemorrhage; NASPAF-ICH: Non-vitamin K Antagonist oral anticoagulants for Stroke Prevention in patients with Atrial Fibrillation and previous IntraCerebral Haemorrhage; OAC: oral anticoagulation; SoSTART: Start or STop Anticoagulants Randomized Trial.
Case-fatality rates seemed to be greater with recurrent ICrH compared with incident or recurrent ischaemic stroke in SoSTART participants, where 58% (7/12; all fatal ICH occurred in patients treated with OAC) of recurrent ICH were fatal compared with none of the 22 ischaemic stroke outcomes. 14 However, similar trends were not observed in APACHE-AF. 13
Meta-analyses of existing randomized trials
A Cochrane aggregate meta-analysis of these three phase II RCTs reported a statistically significant 39% RRR in major adverse cardiovascular events (relative risk [RR] 0.61, 95% 0.40–0.94) and 73% RRR in major occlusive vascular events (RR 0.27, 95% 0.14–0.53), a statistically insignificant ~2.5-fold increase in recurrent ICrH (RR 2.43, 95%CI 0.88–6.73), and no meaningful differences in death or functional outcome (mRS 0–2) at 1 year. 41
The COCROACH collaborators (PROSPERO, CRD42021246133) 23 then pooled individual participant data from these three phase II RCTs with that of 80 patients with prior ICrH enrolled into the ELDERCARE-AF RCT (NCT02801669; edoxaban 15 mg daily versus placebo in elderly patients with AF who were deemed unsuitable for standard dosing OAC). 42 In pooled analysis of 412 participants, the median age was 78 years, 40% were female, 32% (n = 106) had qualifying lobar ICH and 1% (n = 5) had isolated convexity subarachnoid haemorrhage. The median time from qualifying ICrH to randomization was 76 days. 23 The primary outcome of any stroke or cardiovascular death occurred in 14% (29/212) of participants assigned to OAC (99% with DOAC) versus 22% (43/200) of participants assigned to avoiding OAC (33% treated with antiplatelet medication) resulting in a HR 0.68 (95%CI 0.42–1.10; I 2 = 0%). OAC reduced the risk of ischaemic major adverse cardiovascular events (HR 0.27, 95%CI 0.13–0.56, I 2 = 0%). There was a numerical increase in haemorrhagic major adverse cardiovascular events (HR 1.80, 95%CI 0.77–4.21; I 2 = 0%), and again, in contrast to results from the aforementioned observational studies, there was no suggestion of reduced mortality (HR 1.29, 95%CI 0.77–4.21, I 2 = 50%) or improved functional outcome (HR 1.12, 95%CI 0.70–1.79, I 2 = 0%) with OAC versus avoidance of OAC. 23 No statistically significant treatment interactions were identified between subgroups for the primary outcome, including between different qualifying ICH locations, but a low number of events led to broad confidence intervals surrounding these estimates. The continued uncertainty regarding the net benefit of OAC in ICrH survivors with AF despite these recent phase II RCTs should encourage recruitment into ongoing trials, which will contribute to a further iteration of the COCROACH individual participant data meta-analysis once the main phase RCTs are complete.
Ongoing randomized trials
There are several ongoing RCTs investigating optimal stroke prevention in ICrH survivors with AF (Table 2). One of these, the ENRICH-AF trial (NCT03950076) trial, is assessing standard dosing edoxaban (60/30 mg daily) compared with non-anticoagulant medical treatment for stroke prevention in ICrH survivors with AF. 43 Following safety review of the first 699 patients (174 [25%] with lobar ICH and 34 [5%] with convexity SAH), the ENRICH-AF Data Safety Monitoring Board (DSMB) recommended that participants with lobar ICH or isolated convexity subarachnoid haemorrhage have study drug terminated as soon as possible and that no further patients with these ICrH subtypes be enrolled. 43 The DSMB indicated that these recommendations were based on observations of unacceptably high risks of recurrent haemorrhagic stroke among patients with these two qualifying ICrH subtypes assigned to the edoxaban arm. The ENRICH-AF trial is continuing to recruit the remainder of the eligible population, and results will not be available until after study completion. Lobar ICH and isolated convexity subarachnoid haemorrhage share CAA as their underlying causative microangiopathy. At present, it is uncertain whether these discrepant conclusions concerning lobar ICH/isolated convexity subarachnoid haemorrhage between the ENRICH-AF data that informed the DSMB’s decision and the COCROACH individual participant data meta-analysis are due to the underpowered nature of the meta-analysis for these subgroups (111 patients with lobar ICH/convexity subarachnoid haemorrhage in COCROACH vs 208 in ENRICH-AF) or enrolment of more aggressive phenotypes of lobar ICH/convexity subarachnoid haemorrhage in ENRICH-AF following the reassurance provided from the initial phase II trial results.23,43 In view of the suggested fourfold excess risk of ICH observed with apixaban in the APACHE-AF trial (albeit with broad uncertainty surrounding the estimate due to a low number of events; n = 5, adjusted HR 4.09, 95%CI 0·45–36.91), it is unlikely that these discrepant results are explained by the use of edoxaban in the OAC arm of ENRICH-AF. Continued enrolment of patients with lobar ICH or isolated convexity subarachnoid haemorrhage into ongoing trials where they have remained eligible (i.e. ASPIRE, A3ICH, PRESTIGE-AF, and STATICH) will ultimately provide greater clarity in this regard.
Ongoing randomized trials of optimal stroke prevention in ICrH survivors with AF.
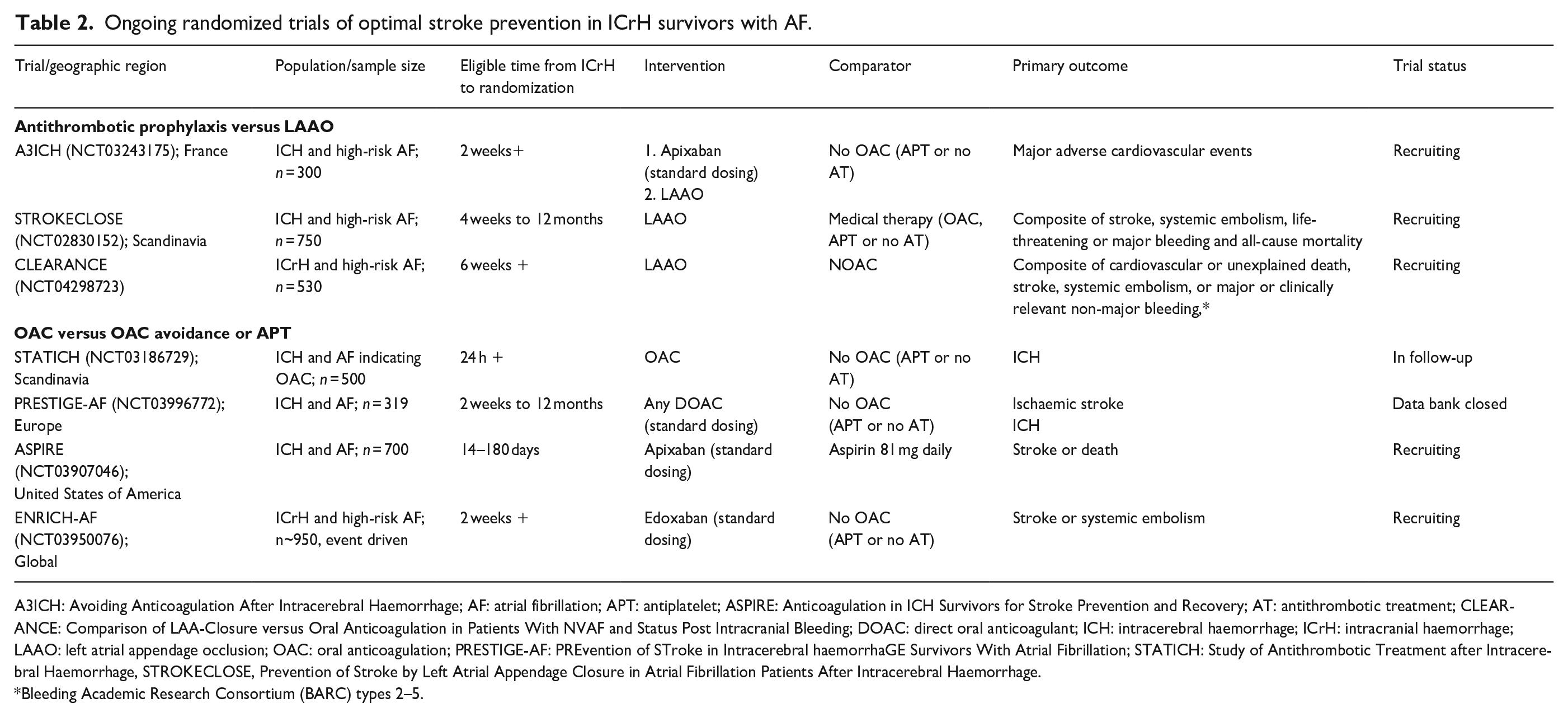
A3ICH: Avoiding Anticoagulation After Intracerebral Haemorrhage; AF: atrial fibrillation; APT: antiplatelet; ASPIRE: Anticoagulation in ICH Survivors for Stroke Prevention and Recovery; AT: antithrombotic treatment; CLEARANCE: Comparison of LAA-Closure versus Oral Anticoagulation in Patients With NVAF and Status Post Intracranial Bleeding; DOAC: direct oral anticoagulant; ICH: intracerebral haemorrhage; ICrH: intracranial haemorrhage; LAAO: left atrial appendage occlusion; OAC: oral anticoagulation; PRESTIGE-AF: PREvention of STroke in Intracerebral haemorrhaGE Survivors With Atrial Fibrillation; STATICH: Study of Antithrombotic Treatment after Intracerebral Haemorrhage, STROKECLOSE, Prevention of Stroke by Left Atrial Appendage Closure in Atrial Fibrillation Patients After Intracerebral Haemorrhage.
Bleeding Academic Research Consortium (BARC) types 2–5.
Timing of OAC following ICH
There exists substantial uncertainty regarding the optimal timing of OAC (re)initiation following ICrH. Although the aforementioned RCTs allowed randomization of patients as early as 24 h to 2 weeks following ICrH, the median timing from ICH to randomization was ~80 days. 23 Literature based estimates on the optimal timing of OAC following ICrH range broadly from 3 days to 30 weeks.38,44 International guidelines have either provided no recommended time window in the absence of definitive evidence or gravitated towards later timing of resumption ranging between 4 and 8 weeks following ICrH (Table 3).5 –7,45 –47 Two studies in particular have been cited to support these later time windows.38,48 The first is a retrospective chart review of 234 patients with warfarin-associated ICrH where the combined risk of recurrent ICrH or ischaemic stroke was observed to reach nadir if warfarin was resumed after approximately 10–30 weeks. 38 However, although subdural haemorrhages accounted for 43% of baseline ICrH, they accounted for 67% of recurrent ICrH. Accordingly, these results may be more relevant to the optimal timing of warfarin in patients with subdural haemorrhages, which are recognized to have a high rate of early recurrence. The second was an analysis of 2619 Swedish patients with AF surviving until hospital discharge following their first-ever ICH identified in the Riksstroke study between 2005 and 2012. 48 Amongst patients deemed at high risk for thrombotic events, the timing of OAC leading to significantly less rates of vascular death or stroke was 7–8 weeks following the index ICH. However, these results should be interpreted carefully as i) all events in the first 4 weeks post ICH (a high risk time for thrombotic events) were censored; ii) there was no significant excess risk of haemorrhagic events observed with OAC at any time point; and iii) there was significantly less thrombotic events with OAC from the very onset of event capture starting at 4 weeks which persisted until 16 weeks. 48 Thus, the net benefit observed with OAC within the 7–8 weeks’ timeframe seemed to have been driven solely by vascular death and superseded the lack of excess haemorrhagic events and significantly lower thrombotic events observed with initiation of OAC at earlier times. This uncoupling of vascular death from thrombotic and haemorrhagic events is difficult to explain and suggests that differences in the outcome of vascular death may have been biased from residual confounding in this study, rendering their results unreliable. 48 Of note, there was no apparent treatment interaction between OAC and time from symptom onset to randomization (dichotomized as within or beyond 8 weeks) for the outcome of stoke or cardiovascular death in the COCROACH individual participant data meta-analysis. 21 In contrast to these two aforementioned observational studies, several others have suggested that the optimal timing of OAC for net benefit is between 2 and 4 weeks following ICrH,17,24,49 and one meta-analysis has even suggested the greatest benefit from OAC occurring with (re)initiation as early as 3 days following ICrH. 44 In the absence of firm evidence or guidelines, survey data indicate that the two most frequently selected timeframes by specialists for OAC resumption following ICH in patients with AF are 14–28 days (21% of respondents) and 4–12 weeks (25%). 4
Guidelines on optimal timing of therapeutic OAC after ICrH (updated in past 10 years).

AF: atrial fibrillation; DOAC: direct oral anticoagulant; ICH: intracerebral haemorrhage; ICrH: intracranial haemorrhage; OAC: oral anticoagulation; RCT: randomized controlled trial.
Minimizing the risk of ICrH recurrence
All efforts should be made to minimize the risk of recurrent ICrH in ICrH survivors with AF. These strategies include ensuring appropriate intensity of OAC (appropriate dosing, education on drug interactions and compliance, and monitoring renal function or INR), falls prevention, and close monitoring and mitigation of modifiable vascular risk factors. One of the most important risk factors for ICrH is hypertension. Hypertension was reported to account for 56% of the population attributable risk (PAR) of ICH in the INTERSTROKE study 50 and was the leading contributor of disability-adjusted life-years (DALY) from ICH and subarachnoid haemorrhage in the Global Burden of Disease Study. 51 High blood pressures increase the risk of both recurrent lobar and non-lobar ICH and are equally important for the prevention of CAA and non-CAA related ICH.52 –54 The optimal blood pressure target for secondary prevention in ICH survivors has yet to be confirmed through randomized trials, but studies have reported heightened risks of incident and recurrent ICH with systolic blood pressures above 120–130 mm Hg.53,55 Current ICH guidelines recommend that ICH survivors have a long-term systolic blood pressure target of less than 130 mm Hg.5,7 Ambulatory blood pressures are reported to increase exponentially in the months leading to an ICH 52 and thus continued close monitoring with home blood pressure monitors and diaries seems essential to ensuring lifelong optimal blood pressure control and ICH prevention. Hypertension additionally contributes to the risk of ischaemic stroke and structural heart disease leading to heart failure (a major cause of mortality) in patients with AF and thus blood pressure control likely provides added holistic benefits in this patient population. 55
In addition to hypertension, six additional modifiable risk factors were reported to contribute to the PAR of ICH in the INTERSTROKE study. 50 These include lack of regular physical activity (35% PAR), unhealthy diet (25%), psychosocial stress (25%), elevated waist-to-hip ratio (13%), current alcohol intake (10%) and current smoking (4%). Together these seven risk factors accounted for 87% of the PAR of ICH. 50 Similarly, apart from hypertension, the top ten contributors to DALYs from ICH and subarachnoid haemorrhage in the Global Burden of Diseases Study included particulate matter pollution, high body-mass index, high fasting blood glucose, kidney dysfunction, smoking, alcohol use, and a diet that is high in sodium, low in fruits and high in red meat. 51 Additional modifiable risk factors specific to OAC-related ICH include the use of a vitamin K antagonist over a DOAC, risk of falling, and the concomitant use of antiplatelet treatment.56 –59 As such, in addition to strict blood pressure monitoring and control, it seems prudent to encourage ICH survivors with AF to abstain from alcohol and tobacco, engage in regular physical activity, maintain a healthy diet and body weight, and seek strategies and tools to reduce psychosocial stress and prevent falling. In those (re)initiated on OAC, DOACs should be prescribed in preference to vitamin K antagonists (unless contraindicated, such as in the presence of mechanical heart valves) and concomitant antiplatelet treatment avoided.
Conclusion
Whether or not OAC should be resumed in ICrH survivors with AF and the optimal timing of OAC (re)initiation are challenging clinical dilemmas that are becoming more frequent with our ageing population. Through the completion of early phase randomized trials to date, we have learned that OAC reduces ischaemic major vascular events in ICrH survivors with AF, but the hazards for death and recurrent ICrH still remain uncertain in these patients. There may also exist heterogeneity in treatment effect of OAC in higher risk subgroups, such as patients with ICrH due to CAA, but this too remains unclear. What is evident, is that we cannot necessarily generalize the findings of randomized trials conducted in safer populations (patients with AF without prior ICrH) to higher risk ones (patients with AF and ICrH). This equally applies to alternative treatments other than OAC, such as left atrial appendage occlusion which has yet to be appropriately tested in RCTs of ICrH survivors with AF. Overall, although there have been significant advances in our understanding of optimal stroke prevention in ICrH survivors with AF in recent years, substantial uncertainty remains. We encourage continued, rapid recruitment to ongoing RCTs to develop high level evidence to resolve these dilemmas and guide treatment decisions in this vulnerable high-risk patient population as soon as possible.
Footnotes
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AS is supported by the Marta and Owen Boris Foundation. He reports consulting and speaker fees from AstraZeneca, Bayer AG, Daiichi Sankyo, and Servier Canada. He additionally reports grant funding from the Canadian Institutes of Health Research, Heart and Stroke Foundation of Canada, National Institutes of Health, Medical Research Future Fund, Daiichi Sankyo, and Bayer AG. He is principal investigator (PI) of the ENRICH-AF trial, co-chair of the ANNEXa-I trial steering committee, and a steering committee member of the ASPIRE trial. CJMK receives funding for research outside the submitted work of the Netherlands Cardiovascular Research Initiative, which is supported by the Dutch Heart Foundation, CVON2015-01: CONTRAST, and the support of the Brain Foundation Netherlands (HA2015.01.06). CONTRAST is additionally financed by the Ministry of Economic Affairs, Top Sector Life Sciences & Health (LSHM17016) and was funded in part through unrestricted funding by Stryker, Medtronic, and Cerenovus. Radboudumc and Erasmus MC received additional unrestricted funding on behalf of CONTRAST, for the execution of the Dutch ICH Surgery Trial pilot study and for the Dutch ICH Surgery Trial from Penumbra Inc. The Radboudumc receives funding through the Promising Care funding scheme of the National Health Care Institute and ZonMw (2021038368). In addition, CJMK reports consulting fees to the Radboudumc from Alnylam for participation in the Therapeutic Steering Committee for the ALN-APP programme, and from Astra Zeneca for participation in an EMCREG International Multidisciplinary Consensus panel. She is PI of the APACHE-AF trial and serves on the DSMB of the ENRICH-AF trial. KNS reports institutional funding from NIH NINDS (U01NS106513, U24NS129500, R01MD016178, R01EB31114, U24NS107215, R01NR018335, R01NS110721), American Heart Association, Hyperfine, Biogen, and Bard, and consulting fees from Astrocyte, Zoll and Sense (DSMB), and CSL Behring. He is PI of the ASPIRE trial and serves on the steering committee of ENRICH-AF. RV reports institutional funding from Bayer, BMS, Pfizer, Daiichi Sankyo, Boehringer, and Medtronic; consulting fees from AstraZeneca; speaker fees from BMS-Pfizer, DSMB participation for Bayer and Portola; stock options in Bayer and Novartis; materials from Medtronic; and chair of the World Stroke Organization research committee. He is PI of PRESTIGE-AF funded by the European Union’s Horizon 2020 research and innovation programme under grant agreement No. 754517. ECS reports honoraria from Boston Scientific, Portola Pharmaceuticals, and Daiichi-Sankyo. She is PI of STATICH and serves on the Steering Committee of ANNEXa-I. CC reports funding from the French Ministry of Health, Novartis (advisory board), and Biogen, Bayer and Bristol Myers Squibb (steering committee). She is PI of A3ICH. RA-SS reports institutional funding from the British Heart Foundation (CS/18/2/33719), the UK National Institute for Health and Care Research and The Stroke Association outside the submitted work, being Data Monitoring Committee chair for ELAN (NCT03148457), and consulting fees paid to the University of Edinburgh from Recursion Pharmaceuticals, Bioxodes, and Population Health Research Institute at McMaster University (Hamilton, ON, Canada). He is the PI of SoSTART and serves on the steering committee of ENRICH-AF.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Not applicable (narrative review of the literature)
Ethical approval
Not applicable (narrative review of the literature
Guarantor
I, Ashkan Shoamanesh, take full responsibility for the article, including for the accuracy and appropriateness of the reference list.
Contributorship
Concept and design: AS; Drafting of the manuscript: AS, Revising the manuscript for content: CJMK, KNS, RV, ECS, CC, RASS
