Abstract
Introduction
Increasing attention is being paid to interventions for cognitive impairment (CI) post-stroke, including for CI that does not meet dementia criteria. The aim of this paper was to conduct a systematic review and meta-analysis of the prevalence of cognitive impairment no dementia (CIND) within one year post-stroke.
Patients and methods
Pubmed, EMBASE and PsychInfo were searched for papers published in English in 1995–2017. Included studies were population or hospital-based cohort studies for first-ever/recurrent stroke, assessing CIND using standardised criteria at 1–12 months post-stroke. Abstracts were screened, followed by full text review of potentially relevant articles. Data were extracted using a standard form, and study quality was appraised using the Crowe Critical Appraisal Tool. A pooled prevalence of CIND with 95% confidence intervals (CI) was estimated using random-effects meta-analysis. Heterogeneity was measured using the I2 statistic.
Results
A total of 7000 abstracts were screened, followed by 1028 full text articles. Twenty-three articles were included in the systematic review, and 21 in the meta-analysis. The pooled CIND prevalence was 38% [95% CI = 32–43%] (I2=92.5%, p < 0.01). Study quality emerged as one source of heterogeneity. The five studies with the highest quality scores had no heterogeneity (I2=0%, p = 0.99), with a similar pooled prevalence (39%, 95%CI = 35–42%). Other sources of heterogeneity were stroke type, inclusion of pre-stroke CI, and age at assessment time.
Introduction
Post-stroke cognitive impairment (CI) is a common consequence of stroke, leading to reduced quality of life. 1 Increasing attention is being paid to identify and develop interventions that can improve cognition post-stroke, and quantifying the potential benefits of such interventions for improving cognition. However, there is an absence of robust evidence on the prevalence of post-stroke CI, based on rigorous systematic review and meta-analysis.
A previous systematic review and meta-analysis estimated the prevalence of dementia in the first year after stroke. 2 In hospital-based studies that included first-ever and recurrent stroke, the pooled prevalence of dementia was 26.5%. 2 However, there has been no meta-analysis of the prevalence of CI that does not meet the criteria for dementia (cognitive impairment no dementia, CIND).
Information on the prevalence of post-stroke CIND is critical in order to alert clinicians to its frequency among stroke patients, informing screening, diagnosis and rehabilitation practices. It can also inform policy and planning for services and interventions for CI, by identifying the number of patients who could potentially benefit from such services. Prevalence estimates are also required for robust epidemiological modelling of post-stroke cognitive disease progression, which is necessary to evaluate the cost-effectiveness of cognitive interventions.
A significant challenge in estimating the prevalence of post-stroke CIND is the lack of uniformity in its definition. Studies vary as to the number of domains that must be impaired, the number of tests per domain, how domains are defined, and specific cut-offs used to define impairment.3,4 Early definitions focussed on the concept of mild cognitive impairment (MCI), primarily framed as a pre-cursor to Alzheimer’s disease. 5 This definition often requires intact basic activities of daily living (ADLs), which can be problematic for stroke patients who may have ADL difficulties due to sensorimotor problems, independent of cognitive function. 6 It has been recommended that requirements for intact ADLs should be specific to instrumental ADLs (IADLs) that are associated with cognition (e.g. managing money). 3 This is consistent with the recent DSM-5 criteria which emphasise impairment in IADLs as a distinguishing factor between major and minor neurocognitive disorder. These new criteria also do not require memory impairment, which is not always present in stroke-related dementia. 7
Population or sampling characteristics, such as study setting (hospital or community), inclusion of recurrent stroke, or of pre-stroke dementia, can also lead to variation in estimates of post-stroke CI prevalence.2,8,9 Exclusion of patients who have difficulty undergoing cognitive testing (e.g. language difficulties) may be unavoidable, but can also effect prevalence estimates. 10
We aimed to systematically review and meta-analyse current evidence on the prevalence of post-stroke cognitive impairment no dementia (CIND) up to one year post-stroke. We included any study that examined a level of CI that did not meet the criteria for dementia. Differences in definition were then explored in stratified analysis, along with other factors including case-mix, follow-up time, definition of CIND, and factors affecting selection.
Method
This study was carried out according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Statement. 11 The protocol was registered in the International Prospective Register of Systematic Reviews in September 2016 (registration number: CRD42016047840). 12
Search strategy
Three databases were searched from January 1995 to 30 September 2017: Pubmed (http://www.ncbi.nlm.nih.gov/pubmed/), EMBASE (https://www.embase.com) and PsycINFO (http://www.apa.org/pubs/databases/psycinfo/index.aspx). Older studies may be less generalisable to the present-day, due to changes in stroke outcomes, casemix and treatment, and 1995 was chosen as an arbitrary cut-point. The search strategy was developed in consultation with a librarian and a stroke specialist. Title and abstract terms for stroke, ischaemic stroke and CI were used, in addition to relevant topic terms (e.g. Medical Subject Headings). A sample search strategy (for PubMed) is displayed in Supplementary Table S1. Following removal of duplicates, each abstract was assessed by one reviewer (ES), with a random 45% of the independently screened by three further reviewers (ND, NAM and DR). Any disagreements were discussed to reach consensus.
Following the abstract screening process, full texts of potentially relevant articles were examined. If there was doubt over inclusion of an article, it was discussed with a further independent reviewer (KB) to reach a consensus decision.
The reference lists of included articles were reviewed to identify any further relevant articles. A citation database (Scopus: https://www.elsevier.com/solutions/scopus) was also searched to identify any relevant papers citing the included articles. Two articles that reviewed available cohort studies on stroke and cognitive impairment were also reviewed to ensure that no relevant study had been overlooked.13,14
Inclusion and exclusion criteria
Studies that included a population of adult stroke patients, either mixed ischaemic and haemorrhagic, or ischaemic stroke alone, were included. Studies that included patients with transient ischemic attack (TIA) were eligible if stroke patients comprised ≥75% of the sample. Hospital-based studies with inclusion of consecutive eligible patients, or community-based studies that included all eligible strokes within a defined geographical area, were included. Included studies assessed cognitive function using a standardised test, at a defined time post-stroke, and reported the percentage of patients classified as having CIND. Studies that used alternative terminology – for example, mild cognitive impairment instead of CIND – were included.
Studies of patient populations that were unlikely to be representative of the total population with stroke were excluded – for example, studies that only focussed on specific stroke sub-types, only included patients with specific deficits or co-morbidities, studies that excluded patients with any disability, or populations recruited via rehabilitation or outpatient settings. Studies that only included haemorrhagic stroke were excluded. Studies where the follow-up time was not known (e.g. stroke was ascertained on imaging or autopsy), or with a broad range of follow-up times (e.g. studies of stroke patients living in the community at varying times post-stroke) were excluded. Studies that assessed cognition less than one month after the index episode were excluded, to account for the influence of post-stroke delirium. Studies that excluded post-stroke dementia, without clearly reporting the number who were excluded for that reason, were excluded from the review, to ensure consistency in the denominator.
Studies that were published in a peer-review journal in English were included. Commentary or review articles, case reports and conference abstracts were excluded. When more than one article based on the same study sample was identified, the most complete and/or relevant paper was identified as the reference article, with supplementary information obtained from the other articles. If it was not possible to establish whether a study should be included on the basis of the published article, the authors were contacted for further information.
Data extraction
Data were extracted using a standard form by ES, and checked by AMc. This included the total study N, the number with CIND and the number with dementia, setting, population (stroke type, first-ever/recurrent stroke, inclusion of pre-stroke dementia/CI, mean sample age), outcome measurement (definition of CIND, definition of dementia) and follow-up time. Details about the definition of CIND and dementia were extracted – e.g. whether intact ADLs were required for a diagnosis of CIND, and whether memory was included as a criterion for dementia. Where sufficient information was available, rates of eligibility (the percentage of patients assessed for eligibility who were included) and response rates (the percentage of eligible patients who participated in the study) were calculated. The CIND prevalence rate for each study was calculated using the total patients with no cognitive impairment (NCI), CIND and dementia as the denominator.
Quality assessment
The Crowe Critical Appraisal Tool was applied independently by two individuals (ES, AMc). 15 Quality criteria are based on reporting quality and suitability of study characteristics for the research questions. This tool includes eight sections, covering areas such as “Design” and “Data Collection”, each scored out of five. We defined suitability criteria in advance of applying the tool, based on the specific purpose of estimating the prevalence of CIND. The full criteria used are available in Supplementary Table S2. If there was a difference between the two appraisers of >2 points in a score for a specific section, or the total score for the study, this was discussed and any adjustments made to the score accordingly.
Consistency in ratings was evaluated with an intra-class correlation co-efficient (ICC), based on the following criteria: <05 = poor; 0.5–0.749 = moderate; 0.75–0.9 = good; >0.9 = excellent. 16 An overall quality score was calculated for each study by averaging the two appraiser scores. For the stratified meta-analysis, the quality score was divided into quartiles, based on the 25th, 50th and 75th percentiles.
Analysis
A pooled prevalence rate was estimated based on all eligible studies, using random effects meta-analysis based on the binomial distribution. 95% confidence intervals were calculated with the exact method, using the metaprop command in Stata. 17 Heterogeneity was quantified using the I2 statistic. The statistical significance and magnitude of the I2 was considered, with values over 40% considered moderate, and values over 60% considered substantial. 18 Reasons for heterogeneity were explored using stratified meta-analysis. Per protocol, 12 stratified meta-analysis was carried out based on the following groups: casemix (stroke type, inclusion of recurrent stroke and pre-stroke dementia), CIND definition; and setting (hospital V community). In addition, we also examined heterogeneity on the basis of quality score quartile and year of data collection.
Results
Included articles
A total of 7000 unique abstracts were identified, of which 5971 were excluded in the initial title/abstract review (see flow chart in Figure 1). The full texts of 1028 articles were reviewed, with 184 of these examined in detail and of these 161 of these were excluded. Following review of the reference lists, articles citing included studies, and two relevant review papers,13,14 three further full text articles were reviewed, none of which were eligible for inclusion. The most frequent reason for exclusion was that the study population was not a general stroke population, e.g. non-stroke participants were included, or only a specific type of stroke patient was included. The next most frequent reason was that CIND was not assessed – for example, CI was measured as a continuous variable, or CI was reported as a single category that included CIND and dementia.
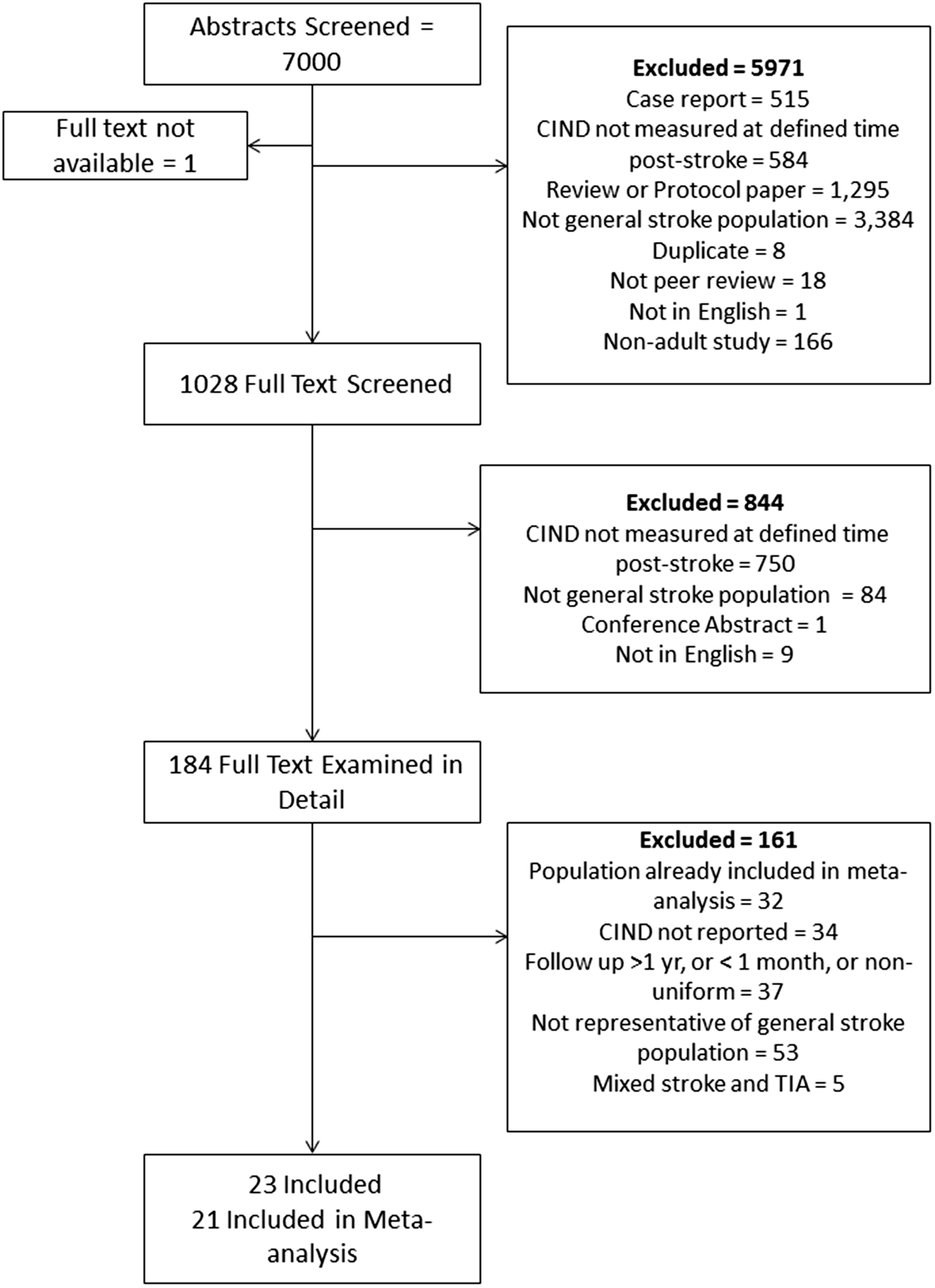
Flow chart of included studies.
There were 23 articles included in the review. Almost half included a mixed ischaemic and haemorrhagic stroke population (k = 11) (see Table 1). Follow-up time ranged from 1 to 12 months, with three months the modal follow-up time (k = 12). The average age ranged from 39 to 81. Most studies (k = 15) had an average age between 65 and 75. One study was population-based, 19 and the remainder were hospital-based. There was some consistency in definitions of dementia, with 15/23 studies using a DSM definition of dementia. Definitions of CIND were much more diverse, with varying tests, domains and cut-offs used.
Characteristics of included studies (n = 23).
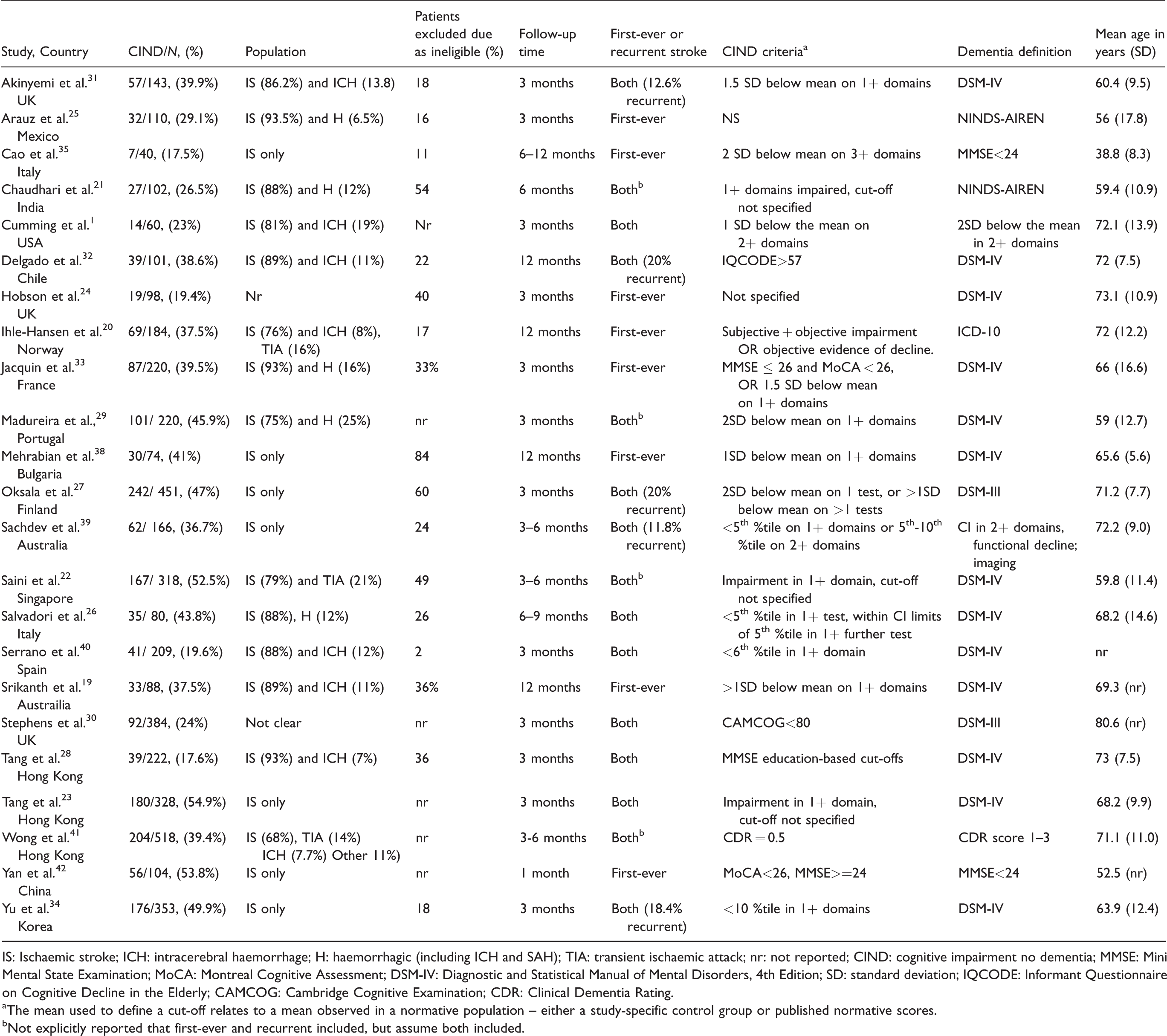
IS: Ischaemic stroke; ICH: intracerebral haemorrhage; H: haemorrhagic (including ICH and SAH); TIA: transient ischaemic attack; nr: not reported; CIND: cognitive impairment no dementia; MMSE: Mini Mental State Examination; MoCA: Montreal Cognitive Assessment; DSM-IV: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition; SD: standard deviation; IQCODE: Informant Questionnaire on Cognitive Decline in the Elderly; CAMCOG: Cambridge Cognitive Examination; CDR: Clinical Dementia Rating.
aThe mean used to define a cut-off relates to a mean observed in a normative population – either a study-specific control group or published normative scores.
bNot explicitly reported that first-ever and recurrent included, but assume both included.
Exclusion criteria related to the capacity to participate in the research were common across studies, including psychiatric or neurological disorders, language or sensory problems, lack of consciousness, refusal to consent. Some studies had criteria beyond these that excluded the most severely affected patients, for example those with mRS > 4 or NIHSS > 4 (see Table S4 for more details).
Study quality
The mean quality score was 30.3/40 (range: 24.8–33.5), or 3.75/5 per section. The intra-class correlation coefficient was 0.82, which indicates good reliability. 16 Quality scores were influenced by multiple factors, with studies performing well in some areas and poorly in others. Almost all studies had at least one problem with their definition of CIND or dementia – for example, a lack of detail on specific cut-offs used20–24 or whether intact ADLs were required for CIND.19,22–30 Many studies reported limited information on how patients were selected into the study, with only 10/23 studies providing a clear flow chart.20–22,25,26,28,31–34 Poor information on patient selection was a key distinguishing characteristic of studies classified as low versus high quality (see Tables S3 and S4 for more details).
Prevalence of CIND
The estimated prevalence of CIND in the first year after stroke ranged from 17.5% 35 to 54.9%. 23 Two studies had restrictive age criteria that meant the mean age was substantially lower (age < 49, mean age = 39 years) 35 or higher (age > 75 years, mean age = 80), 30 and were only included in the age-stratified meta-analysis (see Supplementary Figure S1), leaving 21 studies included in the meta-analysis.
Figure 2 displays the meta-analysis of 21 studies. The pooled prevalence was 38% (95% CI: 32–43%). The total number of patients across studies was 4152, and 1680 were classified as having CIND (n = 1680/4152). However, the heterogeneity was very high (I2 = 92.5%, p < 0.01). This heterogeneity was not explained by factors related to casemix, follow-up time, age, patient selection, or definition of CIND and dementia (see Supplementary Figures S1 to S12). Differences across sub-groups were hard to interpret due to the high heterogeneity, but no substantial differences in prevalence between sub-groups were observed.
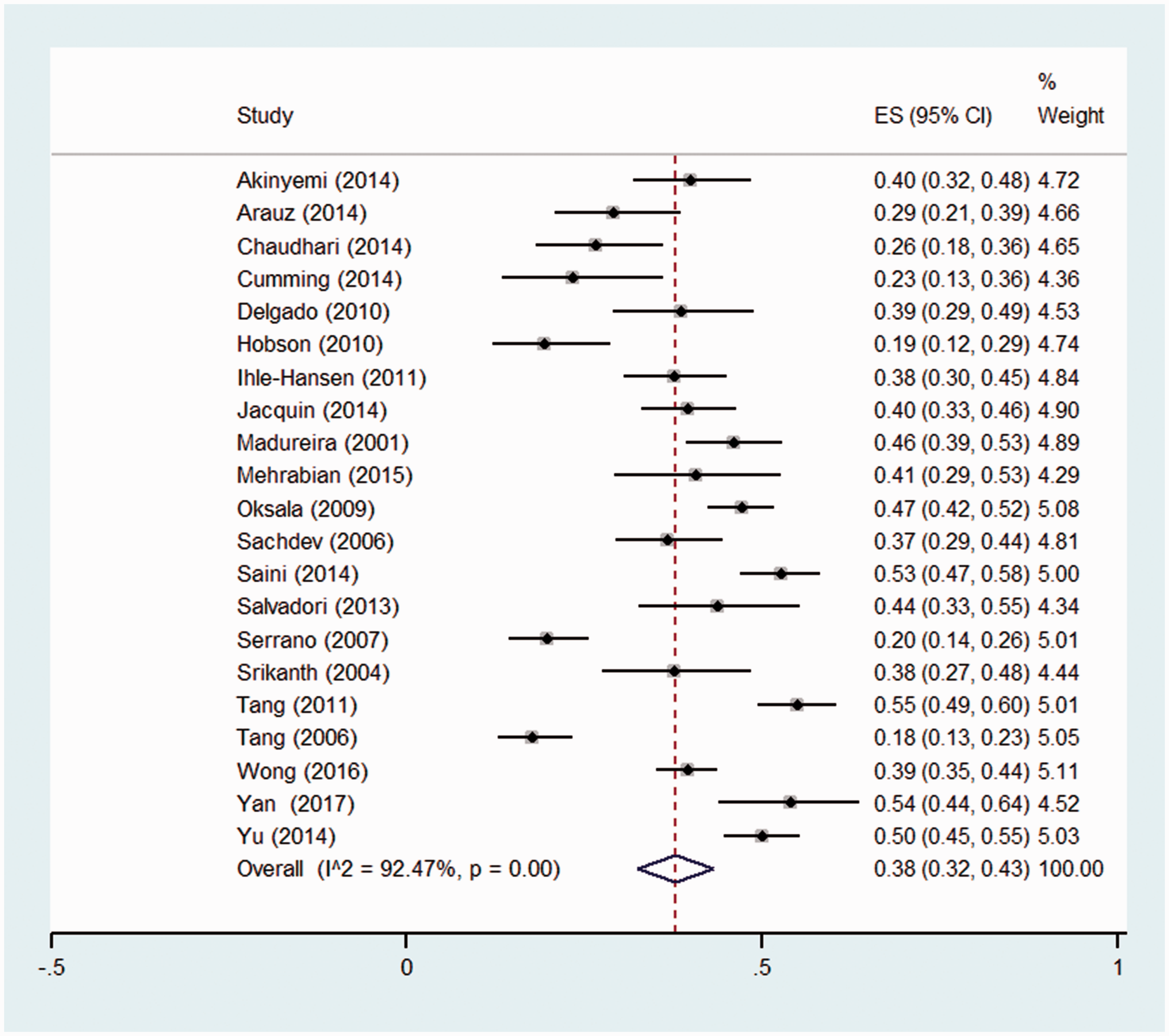
Pooled prevalence of CIND in the first year post-stroke.
However, the heterogeneity did reduce when the studies were stratified by quality score, with no heterogeneity observed in the highest quartile of quality (see Figure 3). In these highest quality studies, the pooled prevalence was 39% (95% CI 35–42%, n = 285/736). This prevalence estimate was similar to the overall estimate, only more precise.
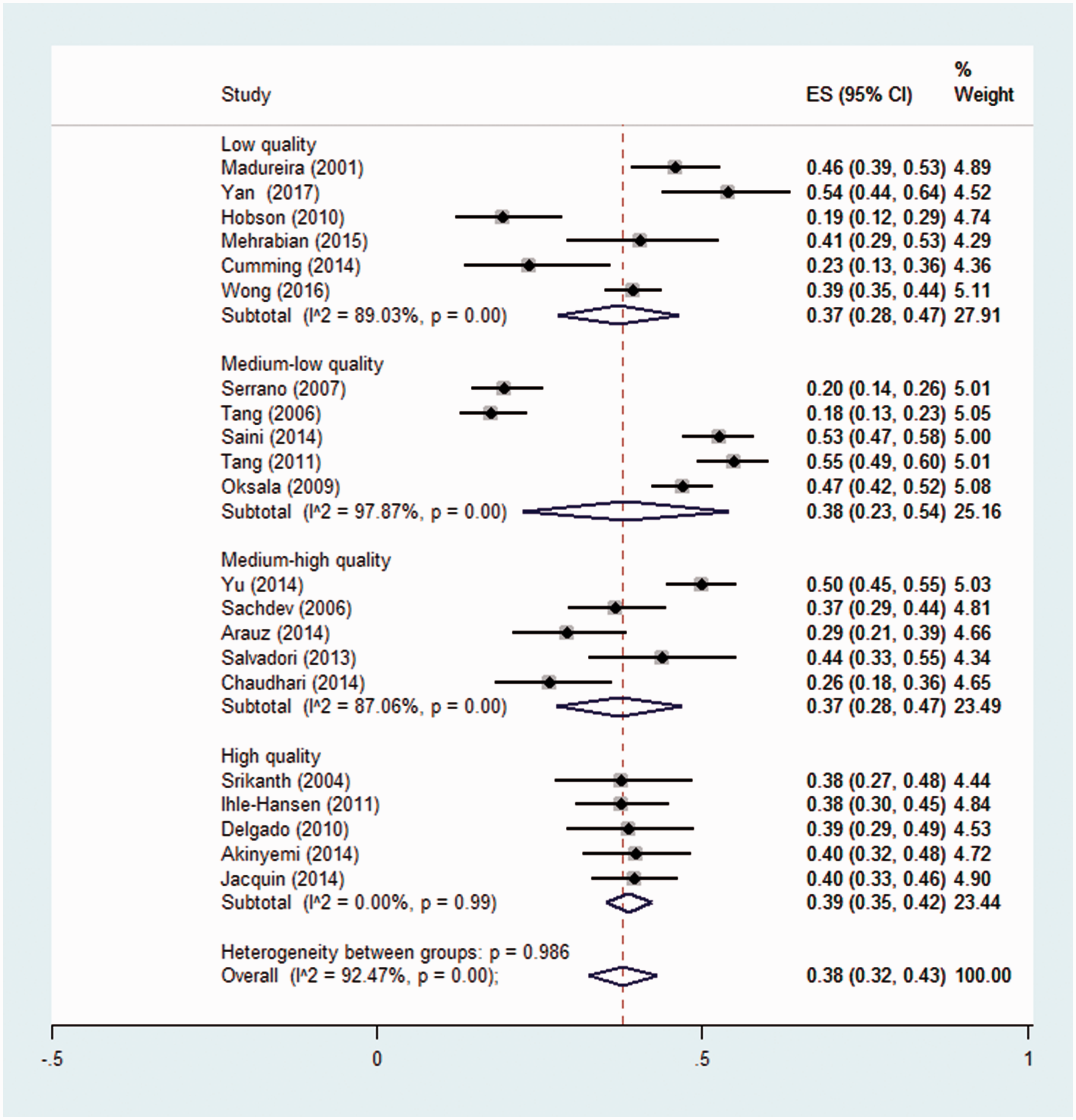
Pooled prevalence of CIND in the first year post-stroke, stratified by quality quartile.
The studies in the two highest quality quartiles (the top 50% of quality scores) were stratified by casemix, follow-up time, age, patient selection, and definition of CIND and dementia (see Figures S13–S22). The analysis by stroke type (see Figure 4) indicated that in higher quality studies with a study population of mixed ischaemic and haemorrhagic stroke, the prevalence estimate was 36% (95% CI 33–43%, n = 310/844), with moderate heterogeneity (I2 = 48.5%, p = 0.07). Stratification by age of the sample suggested a higher pooled prevalence in studies with a mean age greater than 65 years (41%, 95% CI: 36–45%), compared with younger than 65 years (32%, 95% CI: 24–40%), although this difference was not statistically significant (p = 0.06) (see Figure 5). Heterogeneity was also reduced when stratified by inclusion of pre-stroke CI and dementia (Figure S14), and when we only included high-quality studies that used a domain-based definition of CIND (e.g. impairment in one or more cognitive domains) and DSM-IV criteria for dementia (Figure S20).
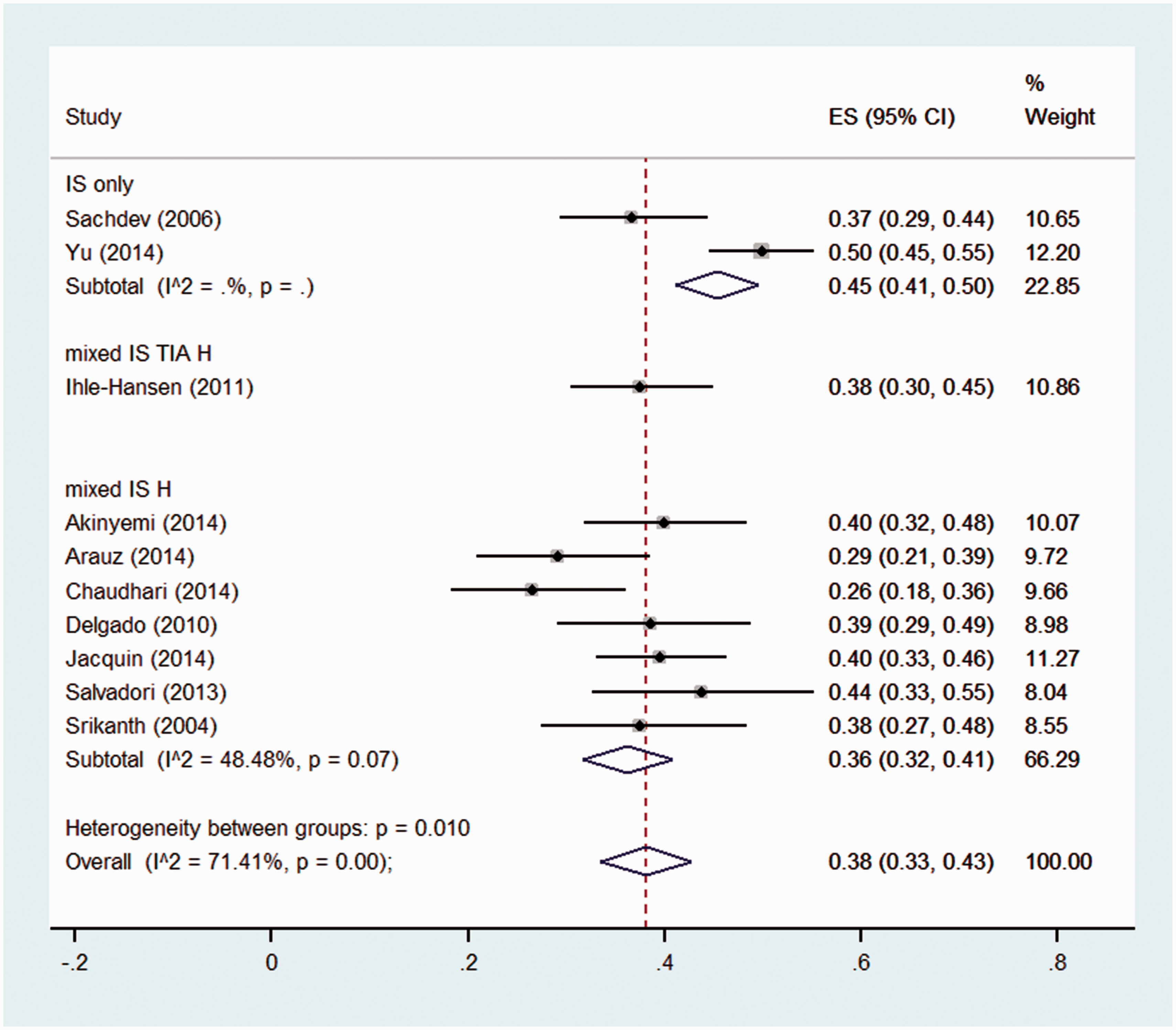
Pooled prevalence of CIND in first year post-stroke, in high and medium-high quality studies, stratified by stroke type. IS: Ischaemic stroke, TIA: Transient Ischaemic Attack; H: Haemorrhagic stroke.
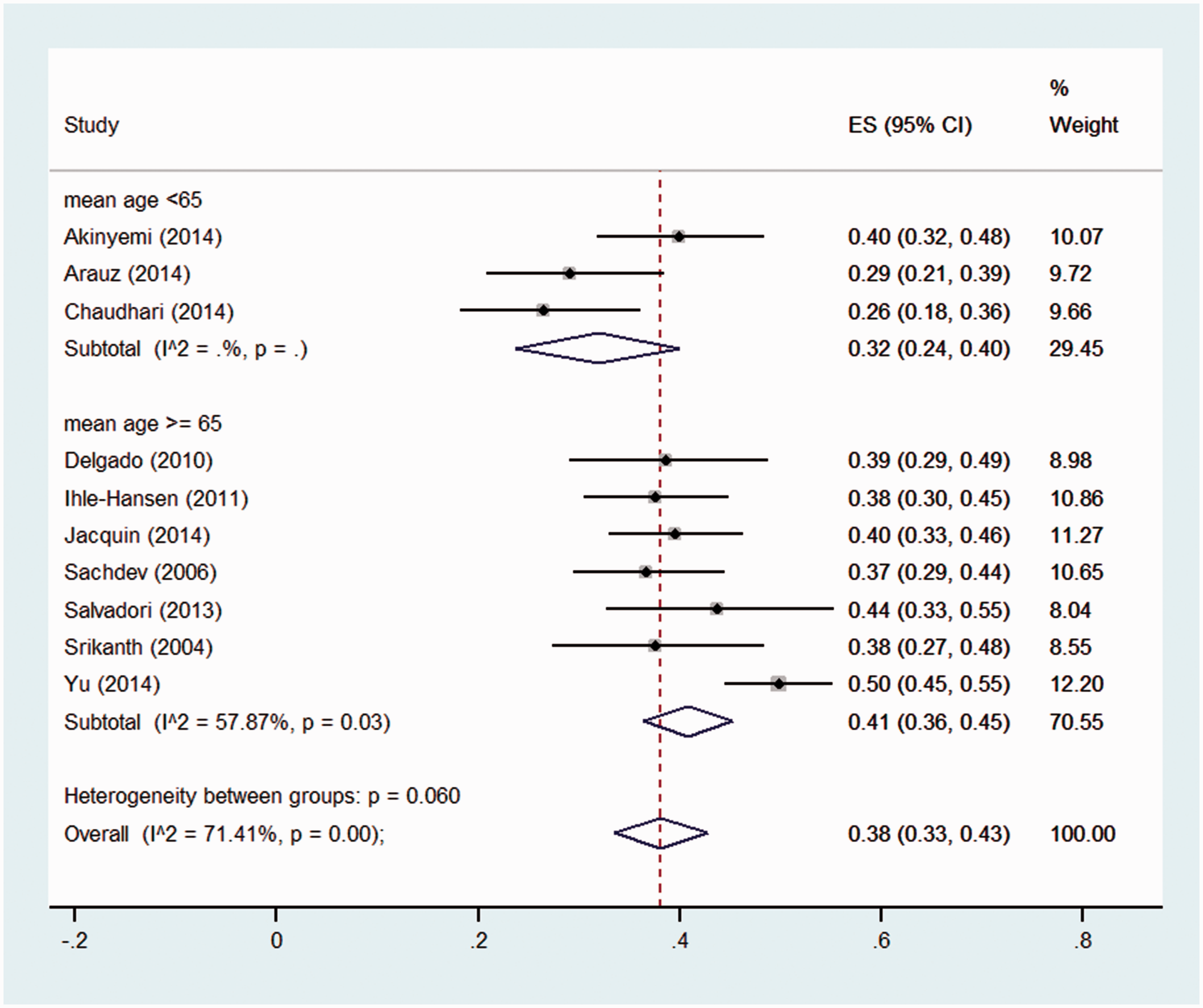
Pooled prevalence of CIND in first year post-stroke, in high and medium-high quality studies, stratified by mean age of the study sample.
The meta-analysis was re-run excluding patients diagnosed with dementia post-stroke from the denominator, to estimate the proportion of stroke survivors without dementia who have CIND. The pooled prevalence was 45% (95% CI: 40–51%), and again heterogeneity was high (I2 = 89.76%, p < 0.01) (see Figure S23). The stratified analysis was similar to the main meta-analysis, with no heterogeneity observed in the highest quartile of quality (see Figure S24).
Discussion
This meta-analysis found a pooled prevalence of CIND in the first year after stroke of 38% (95% CI: 32–43%). However, heterogeneity was very high, indicating that this estimate is not consistent across studies. Sources of heterogeneity related to casemix and study design were explored, with no one factor explaining differences in the estimates obtained. However, overall study quality emerged as a significant source of heterogeneity, with no heterogeneity in the five studies in the highest quartile of quality score. The pooled prevalence for these five studies was 39%, very close to the overall pooled prevalence. As quality scores reflected multiple factors, it may have been a combination of factors that led to variation in estimates, rather than a single factor. High-quality studies had clearer information on patient selection, and may have been more representative of the overall stroke population. Casemix appeared to play a role, as stratified analysis by stroke type and sample age of studies in the top two quality quartiles led to a reduction in heterogeneity. Some key casemix factors, such as inclusion of prior stroke or TIA, were not associated with differences in estimates. Patients with TIA or prior stroke tended to be in a minority in these studies (<20%, see Table 1) and may not have had a substantial influence on the overall estimate.
Study quality
Many studies relied on narrative descriptions of patient selection into the study and follow-up, which were often incomplete or inconsistent. This highlights the need for clear and comprehensive charts describing the flow of participants. Very few studies included detailed information on data collection procedures, for example use of home visits or telephone calls, which can be an important influence on selection bias. 9 There was some consistency in definitions of CI, in that most used the DSM-IV criteria for dementia, and a definition of CIND based on impairment in at least one cognitive domain. However, there was diversity in how these definitions were operationalised, including specific cut-offs for impairment, the use of functional criteria and the requirement for a memory impairment.
Strengths and limitations of the review
Strengths of this meta-analysis include a rigorous and comprehensive database search, covering over 20 years of studies; a detailed quality appraisal process; and a thorough exploration of heterogeneity. A limitation was the inclusion of some lower quality or less relevant studies, such as studies did not provide sufficient detail on study recruitment, or that included TIA patients. However, we were concerned that strict inclusion criteria would exclude some potentially relevant studies. We decided to take a more inclusive approach, but stratify the meta-analysis by these factors, to identify any effect on the results. A further limitation is that studies were predominantly hospital-based (with only one population-based), and higher risk patients were generally excluded due to lack of capacity to participate.
We excluded studies that focussed only on specific stroke sub-types, such as lacunar stroke. This ensured that the study populations were as representative as possible of the overall general stroke population. Lacunar stroke is a very common stroke type, and this approach thus potentially excluded a large body of research. However, we only identified one study that would have been included if we had not excluded lacunar stroke. 36 This study estimated a CIND prevalence of 35%, which is within the confidence intervals of our prevalence estimate. Similarly, a recent systematic review of MCI in lacunar stroke found a pooled prevalence rate of 37% across four studies. 37 .
Implications
The quality of studies in this area could be improved by more consistent definition of CIND across studies, and clearer reporting of patient selection and data collection procedures. However, given the consistency between the overall pooled estimate and the estimate derived from the highest quality studies, further high-quality prevalence studies may not alter the findings of this review. None of the studies identified used the DSM-5 criteria for mild neurocognitive disorders, which is not surprising given the relative recency of their publication. 7 However, it will be interesting to compare the results of this review with any future studies using the DSM-5 criteria.
In addition to being a useful guide for clinicians, this prevalence estimate will be used in the StrokeCog epidemiological modelling platform. This model will apply this estimate and those from other sources to real-world populations to estimate the number of people with post-stroke CIND, and make projections into the future. This will inform policy and planning for future service provision, and facilitate cost-effectiveness analysis.
Conclusions
Approximately 4 in 10 stroke patients who are hospitalised and capable of undergoing cognitive assessment have CIND in the first year post-stroke. This finding is consistent across high-quality studies that include a mixed ischaemic and haemorrhagic stroke population. The prevalence may be lower (3 in 10) in younger age groups. Clinicians need to be mindful of the need to screen for post-stroke CIND, using tools such as the MoCA, and refer for more detailed neuropsychological assessment where necessary. However, the benefits of screening and assessment are limited in the absence of cost-effective interventions for this condition. The prevalence estimate identified in this study is a critical parameter for the StrokeCog epidemiological model that will contribute to identifying such cost-effective interventions.
Supplemental Material
Supplemental material for Systematic review and meta-analysis of the prevalence of cognitive impairment no dementia in the first year post-stroke
Supplemental Material for Systematic review and meta-analysis of the prevalence of cognitive impairment no dementia in the first year post-stroke by Eithne Sexton, Affraic McLoughlin, David J Williams, Niamh A Merriman, Nora Donnelly, Daniela Rohde, Anne Hickey, Maev-Ann Wren and Kathleen Bennett in European Stroke Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Health Research Board (HRB) in Ireland under Grant No. ICE-2015–1048 and award RL-15–1579.
Ethical approval
Ethical approval was not sought for this article because this was a systematic review of existing publications and no new patient data was collected.
Informed consent
Informed consent was not sought for this article because this was a systematic review of existing publications and no new patient data was collected.
Guarantor
ES.
Contributorship
AH, KB and MAW developed the original idea. ES developed the review protocol, and was primarily responsible for the data analysis and drafting the manuscript, under the supervision of KB. The search strategy and inclusion criteria were developed by ES with substantial input from DW and AH. Abstract screening and article review was carried out by ES, NAM, ND, DR and KB. ES and AMc conducted the quality appraisal and data extraction. All authors commented on and gave final approval to the final version of the manuscript.
Acknowledgements
We acknowledge the valuable input or Prof Frances Horgan, Prof Niall Pender and Grainne McCabe. We also acknowledge the participants and authors of the studies included in this meta-analysis.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
