Abstract
Introduction
Antiplatelet therapy is routinely prescribed early after ischaemic stroke. Many patients will already be taking antiplatelet therapy and it is unknown whether these patients should continue the same antiplatelet treatment or switch to a different regimen.
Methods
We selected patients with ischaemic stroke from the Virtual International Stroke Trials Archive database who were prescribed antiplatelets both before and after their stroke and who had detailed records of adverse events after stroke. We compared patients who changed to a new antiplatelet regimen after their stroke to those who continued the same regimen. The primary outcome was recurrent ischaemic stroke within 90 days after their index stroke and the secondary outcome was intracranial haemorrhage (ICH) or extracranial haemorrhage (ECH). We used logistic regression analysis and adjusted for age and baseline NIHSS.
Results
A total of 1129 participants were included. Of these, 538 subjects changed antiplatelet regimen post stroke and 591 continued the same regimen. A recurrent ischaemic event occurred in 4.1% of subjects who changed regimen and 4.3% who continued unchanged (adjusted OR = 0.93; 95% CI 0.54–1.75, p = 0.929). The incidence of ICH and ECH within the first 90 days was similar in both groups (2.4% vs. 2.6% (adjusted OR = 1.02; 95% CI 0.48–2.18, p = 0.955) and 4.7% vs. 2.9% (adjusted OR = 1.82; 95% CI 0.96–3.43, p = 0.065), respectively).
Discussion
The analysis was performed using a non-randomised registry data.
Conclusion
In patients who suffer ischaemic stroke whilst taking antiplatelets, a change in antiplatelet regimen was not associated with an altered risk of early recurrent ischaemic stroke rate or bleeding. However, the results must be interpreted in view of the low event rates.
Keywords
Introduction
Antiplatelet therapy is recommended for patients with ischaemic stroke or transient ischaemic attack (TIA) due to atherosclerotic disease. 1 Antiplatelet drugs are prescribed to prevent further ischaemic stroke, minimise the risk of early death and to improve long-term outcome.2,3
Guidelines recommend that antiplatelet regimen choices are individualised based on numerous factors including patient risk factor profiles, cost, tolerability and relative effectiveness. 4 One potentially important factor is pre-existing antiplatelet therapy. In the recently published CHANCE study, at least 20% of patients had history of previous stroke and 11% of patients had their index stroke whilst taking antiplatelet therapy. 5 Guidelines acknowledge that in patients who experience a stroke whilst taking antiplatelet therapy, there are no clinical trials to show whether switching antiplatelet agents decreases the risk of subsequent events. 6 There are reasons why switching regimen may be advantageous. Failure of antiplatelet treatment could be due to disease severity (no drug will be 100% effective), lack of adherence or resistance to effect of the antiplatelet drug. Resistance to clopidogrel and aspirin exists and studies have shown that aspirin resistance is associated with an increased risk of cardiovascular events. 7 It is possible that resistant patients should be switched to another class of antiplatelet.
In the absence of clinical trial data, we sought to assess whether switching antiplatelet regimens associated with better outcome than continuing the same therapy in patients already taking antiplatelet therapy at the time of acute ischaemic stroke. We hypothesised that a change of antiplatelet regimen would reduce cardiovascular event rate without increasing bleeding.
Methods
We used data contained in the Virtual International Stroke Trials Archive (VISTA) database which have been described previously. 8 VISTA data are stored at the Robertson Centre for Biostatistics, University of Glasgow, Glasgow, UK. It provides access to anonymised data collated from completed and monitored clinical trials. Within VISTA, demographic data such as age, sex and ethnicity; smoking history and comorbidities as well as details on the index stroke and functional outcome measures are contained. Information on adverse events (AE), serious adverse events (SAE), laboratory measurements and prescribed medications are also available from certain trials. All clinical trials within VISTA already have local institutional review board-approved procedures in accordance with the Declaration of Helsinki. Access to data was approved by the VISTA steering committee.
We included patients who had suffered ischaemic stroke whilst taking antiplatelet drugs (for at least seven days prior) and who were recommenced on antiplatelet therapy within 90 days after ischaemic stroke where data on baseline characteristics, medical history, baseline National Institutes of Health Stroke Scale (NIHSS), occurrence of adverse and serious adverse events, and medication use with start dates were recorded in VISTA. We extracted data for these patients.
Data on antiplatelet treatments were based on platelet aggregation inhibitors (which includes derivatives of salicylic acid, thienopyridine and dipyridamole). Participants were grouped according to whether they changed antiplatelet regimen (change group) or continued on the same antiplatelet therapy (continue group).
Our primary outcome was the occurrence of recurrent ischaemic stroke and the secondary outcome was bleeding complications within 90 days post-stroke. The modified Rankin Scale (mRS) at 90 days was used to measure the functional outcomes. We extracted information about recurrent ischaemic stroke and bleeding complications in all patients from AE and SAE reports. We defined a recurrent ischaemic stroke as a clear statement of a new adverse event including the terms such as ischaemic stroke, cerebral infarction or cerebrovascular accident. We did not include events such as worsening of initial stroke symptoms or oedema. For bleeding complication, the event was identified using these key terms: hematoma, haemorrhage, bleeding, blood or melaena. Bleeding complications were categorised into two, i.e. intracranial haemorrhage (ICH) and extracranial haemorrhage (ECH). Intracranial haemorrhage was defined as any ICH but excluding haemorrhagic transformation infarction 1 and 2, whereas ECH was defined as any bleeding episode from another source.
Statistical analysis
We compared baseline variables in the change and continue groups. Categorical variables were summarised using frequencies and proportions and were compared using the Chi-square test or Fisher’s exact test, where appropriate. Continuous variables were summarised using mean [standard deviation (SD)] or median [interquartile range (IQR)] and were compared using Student’s t-test or non-parametric Mann–Whitney test.
We calculated adjusted odds ratios (OR) and corresponding 95% confidence intervals (95% CI) using binary logistic regression for occurrence of outcomes and ordinal shift of the mRS at day 90 using the full scale for functional outcomes. Adjustment was made for age and baseline NIHSS.9,10 We also assessed the effect of changed or continued antiplatelet regimen on outcome measures in subgroups: clinical condition (atrial fibrillation or not, prior stroke or not and treated with intravenous thrombolysis or not). We conducted subgroup analyses in these populations as their stroke risk recurrence11,12 and neurological recovery 13 are different. A p < 0.05 was used to define statistical significance. All analyses were performed using IBM SPSS Version 21.0. 14
Results
A total of 1129 stroke patients met our inclusion criteria (Figure 1). Forty-eight percent were changed to a new antiplatelet drug post-stroke. Patient characteristics are shown in Table 1.
Flow diagram describing the selection of data from the VISTA for the analysis reported. Baseline characteristics. Note: All values are reported as no. (%) unless otherwise noted. AF: atrial fibrillation; AP: antiplatelet; IHD: ischaemic heart disease; NIHSS: National Institutes of Health Stroke Scale; rt-PA: recombinant tissue plasminogen activator; TIA: transient ischaemic attack; SD: standard deviation; IQR: interquartile range. Values are reported as mean (SD). Median (IQR).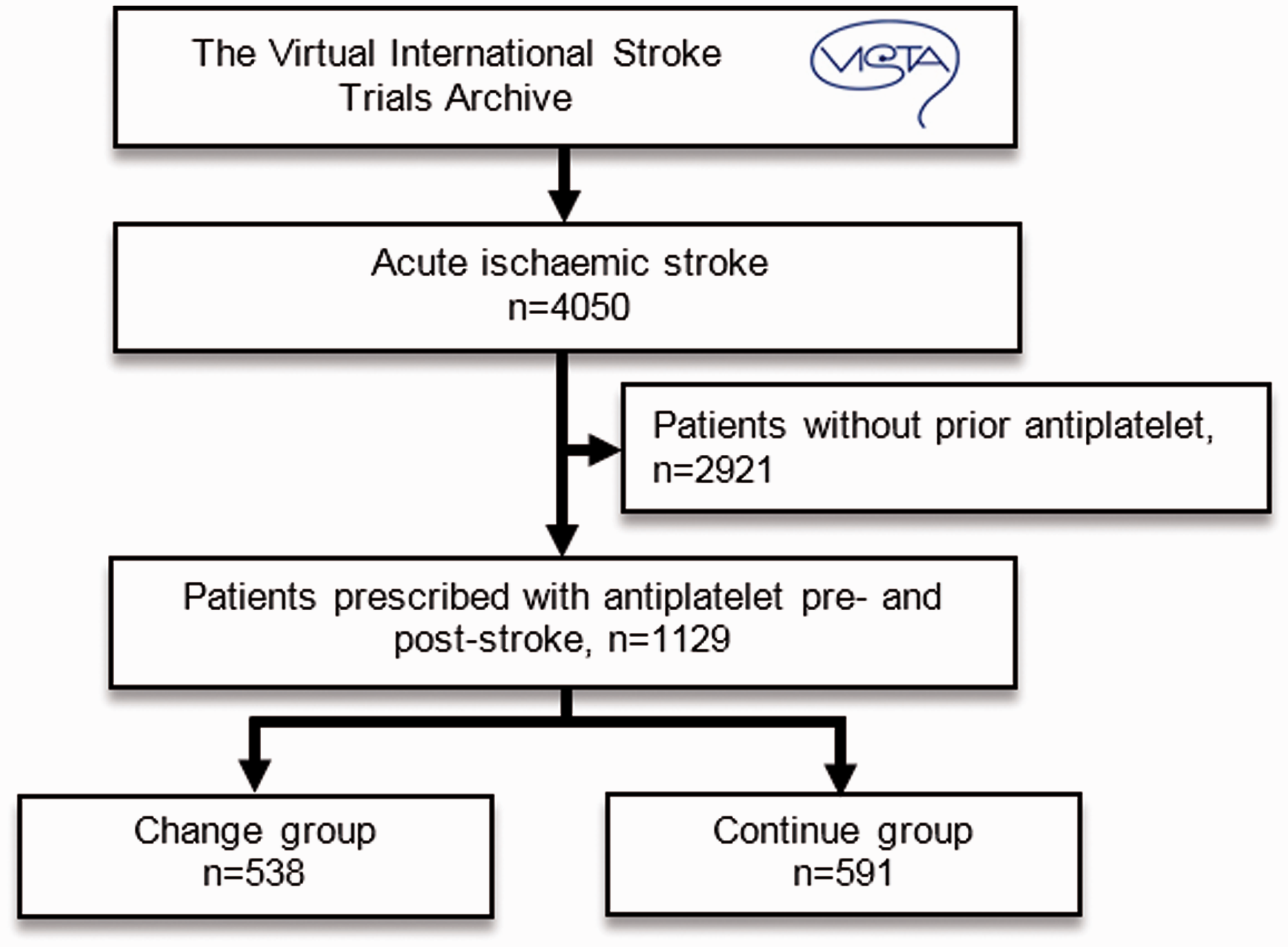
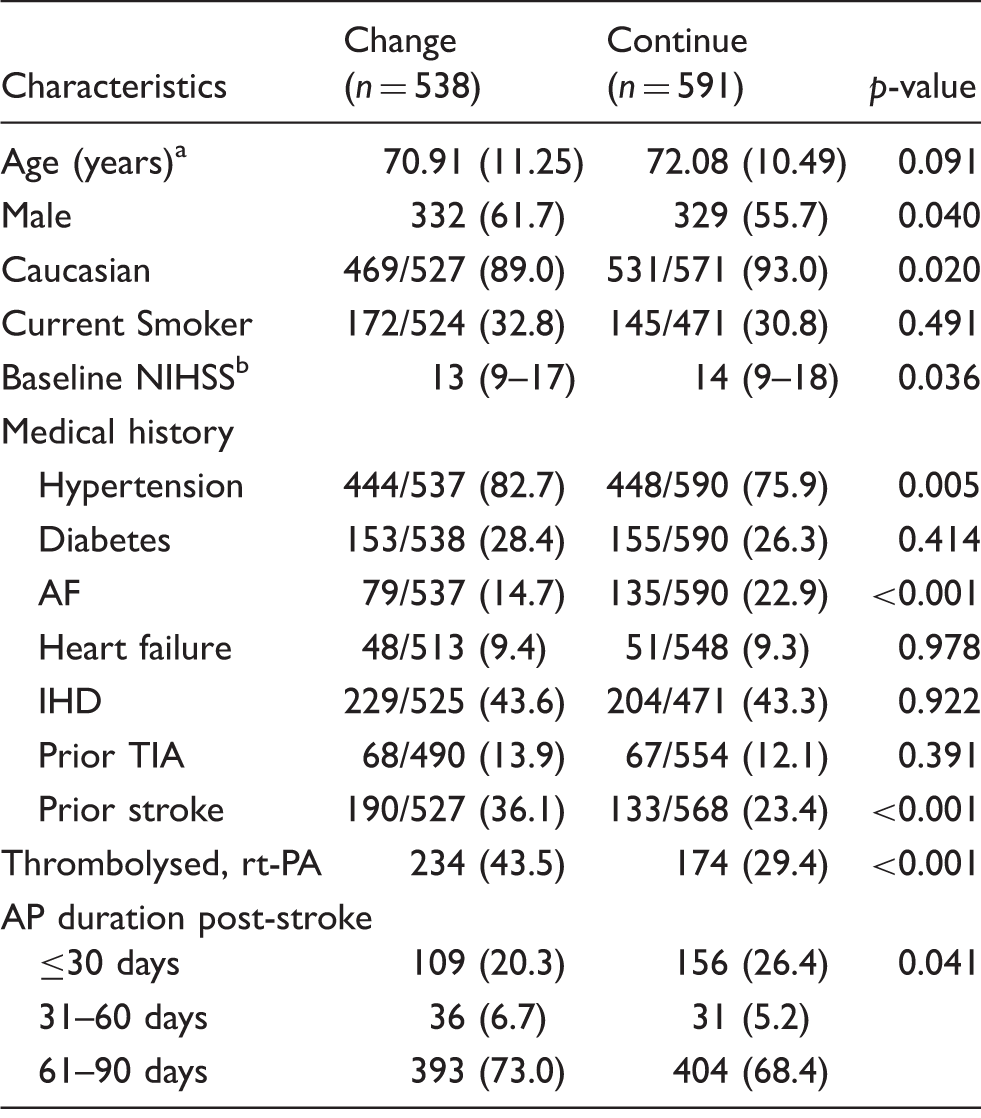
Clinical outcomes at 90 days (adjusted for age and baseline NIHSS).
Note: All values are reported as no. (%) unless otherwise noted. CI: confidence interval; ICH: intracranial haemorrhage; ECH: extracranial haemorrhage; OR: odds ratio.
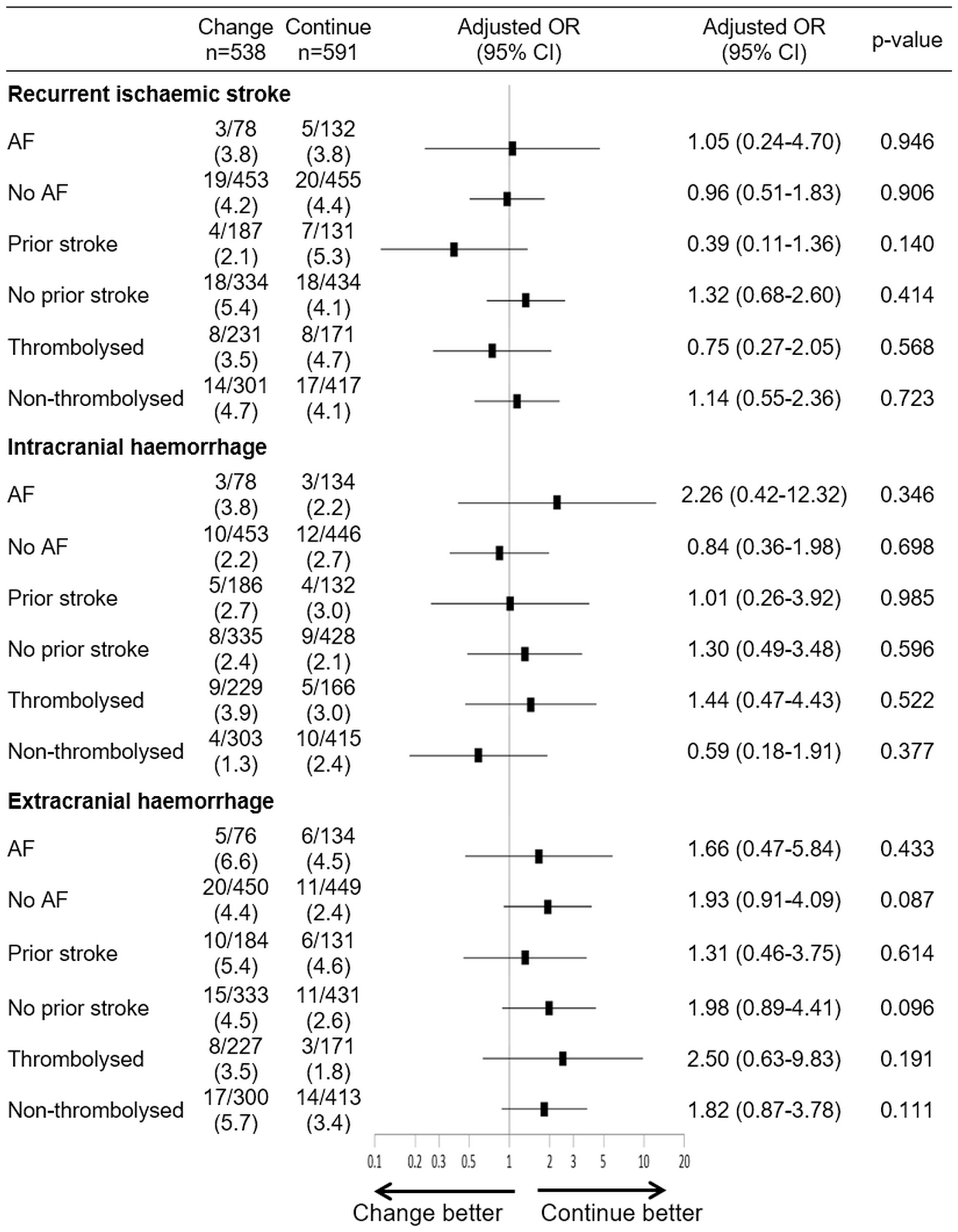
Clinical outcomes at 90 days (adjusted for age and baseline NIHSS) by subgroup. All values are reported as no. (%) unless otherwise noted. AF: atrial fibrillation; OR: odds ratio, CI: confidence interval.
The distribution of mRS at 90 days for change versus continue group is shown in Figure 3. We found that change to a new antiplatelet post-stroke was associated with more favourable functional outcome across a full-scale mRS at 90 days after adjustments for age and baseline NIHSS (adjusted OR = 1.48; 95% CI 1.19–1.86, p = 0.0006), compared with the continue group.
Distribution of mRS outcome at day 90 in patients suffering ischaemic stroke. Diagram showing association of functional outcome at day 90 between change and continue group. Values provided in each box denote the percentage of patients belonging to a specific treatment category (change or continue) and representing the mRS score corresponding to the box.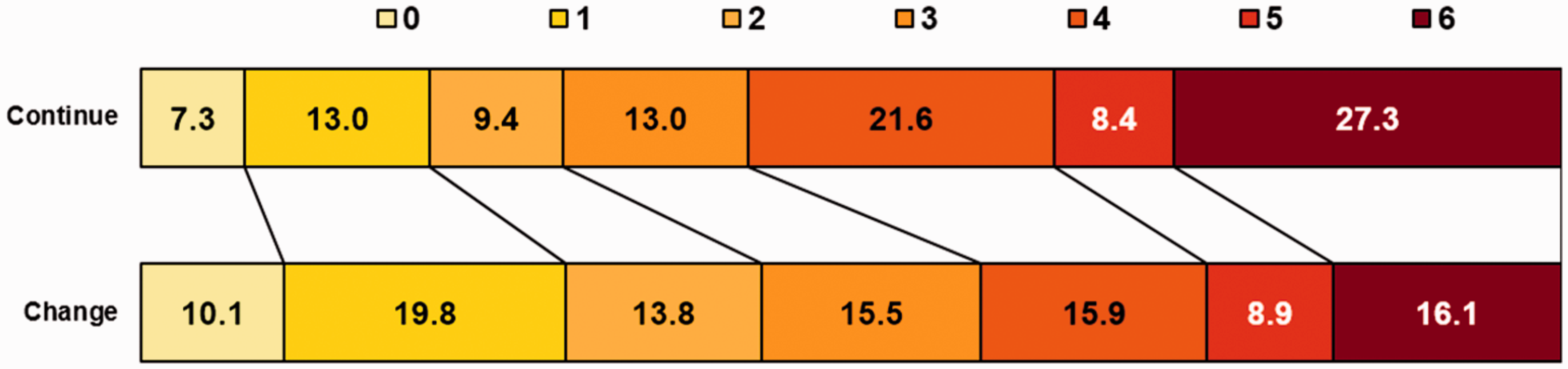
Discussion
In the present study, we found that subjects who experience stroke while on antiplatelet therapy and who switch to a new class of antiplatelet regimen after stroke do not have a lower early recurrence rate than subjects who continue with the same antiplatelet therapy. We also found no difference in bleeding outcomes. Furthermore, the clinical outcomes were not significantly different across pre-defined subgroups. We found that change group was associated with an increase in the odds of a favourable functional outcome.
Many patients suffer ischaemic stroke despite taking antiplatelet therapy. Some studies show that, in ischaemic heart disease, patients who are non-responders to aspirin and clopidogrel are at greater risk of subsequent ischaemic vascular event and death. 15 It is therefore attractive to suggest that changing antiplatelet class will be of benefit, and reduce subsequent recurrence, in patients who are admitted with ischaemic stroke despite antiplatelets. This would be a cheap, immediately available way of reducing risk. Another study found the rate of adverse clinical outcomes was significantly higher in antiplatelet-regimen-modified group than those without modification. 16
There are no trials that show changed antiplatelet therapy after a stroke event reducing the risk of subsequent stroke. 4 A recent retrospective cohort study by Lee et al. 17 compared clopidogrel initiation versus aspirin re-initiation among patients with ischaemic stroke who took aspirin at least 30 days prior to their index of stroke. They found that those who changed to clopidogrel after a stroke were associated with fewer vascular events than aspirin. In contrast, we did not find any significant relation between those who change and continue antiplatelet therapy. The reasons might be because our study follow-up was shorter, i.e. 90 days, compared to the study by Lee et al. 17 and other related trials which are normally more than two years. 18 Furthermore, we divided patients into two different groups, whether they change or continue with the same antiplatelet therapy and not according to specific antiplatelet agent.
We found that those who changed antiplatelet regimen had better functional outcome at 90 days than those in the continue group. Although we tried to adjust for differences in baseline factors, this most likely reflects residual confounding and may reflect regional differences in stroke care. A physician may choose a more aggressive (or change) approach in patients with better prognosis. Such differences may affect functional outcomes but not recurrence risk. In our study, the change group had lower stroke severity and fewer patients with atrial fibrillation at baseline; both factors are known to be associated with an increased risk of poor outcome following an ischaemic stroke.19–23
There are several limitations to this study. The analysis was performed using a non-randomised registry data that derived from various clinical trials. Thus, selection and other biases could have confounded antiplatelet therapy choices. The number of included participants was low due to the selection criteria, the need to be on antiplatelet therapy before and after stroke and the need for the start dates of prior and new medications to be recorded. Although the recurrent ischaemic stroke rate was identical between groups, we lack statistical power to exclude a potentially clinically significant difference between groups. We are unable to investigate stroke recurrence based on stroke subtype as the information is not available in most of the patients. We lack specific measures of medication compliance, particularly before the stroke, which could be a cause of failure of treatment. The effects of switching drugs class in such patients may be different and this will need to be addressed by further study. We also lack information on discharge time, which could be related to compliance and to functional outcomes. We did not have information on all variables to calculate CHA2DS2-VASc score and only 19% of our patients had atrial fibrillation.
There are also strengths to our study. The data are derived from prospective trials with standardised assessments and careful data monitoring, providing high quality data. In addition, we were able to adjust for the most important prognostic variables.
In summary, we found no difference in outcomes in patients who continued the same antiplatelets after stroke or who changed. Despite no clear evidence for changing antiplatelet strategies following a cardiovascular event, over 50% of patients had their medication changed. This may reflect uncertainty or the fact guidelines. As recommended by the guideline, the selection of antiplatelet regimen after ischaemic stroke, however, should be individualised on the basis of patient risk factor profiles, cost, tolerance and other clinical characteristics. 4 However, this was a small retrospective analysis and future prospective trials will be needed.
VISTA-Acute Steering committee members
AV Alexandrov, PMW Bath, E Bluhmki, L Claesson, J Curram, SM Davis, G Donnan, HC Diener, M Fisher, B Gregson, J Grotta, W Hacke, MG Hennerici, M Hommel, M Kaste, KR Lees (Chair), P Lyden, J Marler, K Muir, R Sacco, A Shuaib, P Teal, NG Wahlgren, S Warach, and C Weimar.
Footnotes
Acknowledgements
We thank all of the investigators, coordinators, and patients who contributed data to the VISTA; and the VISTA Steering Committee for providing access to the data.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PMWB is Stroke Association Professor of Stroke Medicine and a member of the VISTA Executive Committee.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: WMK was supported by Ministry of Health scholarship Malaysia. Others – none.
Ethical approval
Not required.
Informed consent
Not applicable.
Guarantor
JD.
Contributorship
The study concept was undertaken by WM-K and JD; data acquisition was done by WM-K, RLM and JD; data interpretation was done by WM-K, RLM and JD; preparation of initial draft was done by WM-K; critical revision of the manuscript was done by WM-K, RLM, MW, PMWB and JD; study supervision was done by JD.
