Abstract
Background:
The effects of intravenous alteplase in patients with prior antiplatelet therapy (APT) remain controversial. We aimed to assess the efficacy and safety of imaging-based intravenous alteplase in patients with unknown onset stroke with prior APT.
Methods:
Data from randomized controlled trials comparing alteplase with placebo/standard care in patients with unknown onset acute ischemic stroke from the Evaluation of Unknown Onset Stroke Thrombolysis (EOS) individual patient data meta-analysis collaboration were analyzed. Favorable outcome was defined as a modified Rankin Scale score of 0–1 at 90 days post-stroke. Safety outcomes included symptomatic intracranial hemorrhage (sICH) at 22–36 h and 90-day mortality.
Results:
Overall, 780 patients had available baseline data on prior APT. Compared with the no prior APT group (n = 523), the prior APT group (n = 257) was older (72 vs. 66 years) and had a higher prevalence of vascular risk factors. There was no interaction between prior APT and treatment effects of alteplase (p for interaction = 0.23). In the prior APT patients, 55/125 (45%) patients in the alteplase group and 39/132 (30%) patients in the control group had a favorable outcome (adjusted odds ratio [aOR], 2.07 [95% confidence interval, 1.18–3.64]). The rates of sICH and mortality in the alteplase and control groups were 5.6% and 0.8% (aOR, 7.78 [0.94–63.37]) and 6.5% and 6.1% (aOR, 1.12 [0.38–3.36]), respectively. In the no prior APT patients, 136 patients (50%) in the alteplase group and 112 patients (45%) in the control group had a favorable outcome (aOR, 1.39 [0.94–2.05]). Safety outcomes were not significantly different between the groups (sICH: 3 [1.1%] vs. 1 [0.4%]; mortality: 13 [4.9%] vs. 3 [1.2%]).
Conclusions:
Alteplase has consistent efficacy regardless of prior APT in patients with unknown onset stroke. In addition, prior APT does not significantly increase the risk of sICH or mortality.
Introduction
Intravenous thrombolysis (IVT) with alteplase within 4.5 h of symptom onset has been established as an effective intervention for improving outcomes in acute ischemic stroke. However, the occurrence of symptomatic intracranial hemorrhage (sICH) remains a concern, affecting 2–3% of patients receiving alteplase and potentially contributing to unfavorable outcomes. 1 Approximately 30% of patients receiving time-based intravenous alteplase have been administered antiplatelet therapy (APT) before the onset of stroke. 2 A recent systematic review and meta-analysis revealed a 1.2-fold higher incidence of sICH in patients with prior APT, than in those without prior APT following time-based IVT. Despite this increased risk, no significant association with favorable outcome and death was observed. 3 Notably, most data on time-based IVT for patients with prior APT were derived from observational studies. 3
Evidence supporting the appropriateness of IVT for patients with prior APT has remained inconclusive.3–5 A post hoc analysis of the Thrombolysis for Acute Wake-up and Unclear-onset Strokes with alteplase at 0.6 mg/kg (THAWS) trial showed that prior antithrombotic medication (APT or anticoagulants) was associated with better outcomes without increased mortality or incidence of ICH. 6 The analysis also revealed a significant interaction between prior APT and alteplase, suggesting that prior APT may enhance the therapeutic effects of alteplase. In contrast, a sub-analysis of the efficacy and safety of magnetic resonance imaging (MRI)-based Thrombolysis in Wake-up Stroke (WAKE-UP) trial showed no interaction between antiplatelet use and alteplase use with respect to neurological outcomes. 7 The Enhanced Control of Hypertension and Thrombolysis Stroke Study is the only randomized controlled trial (RCT) to investigate the comparative effect of low-dose (0.6 mg/kg) or standard-dose (0.9 mg/kg) intravenous alteplase in patients with or without prior APT. The results showed that low-dose alteplase tended to achieve better outcomes than standard-dose alteplase in patients on prior APT than in those not on APT. 8 The results of prior RCT subgroup analyses are inconsistent, and further studies are needed to clarify these findings.
Recent meta-analyses of individual patient data by the Evaluation of Unknown Onset Stroke Thrombolysis (EOS) trial investigators revealed the efficacy and safety of alteplase in patients with stroke of unknown onset time by utilizing perfusion imaging or assessing diffusion-weighted imaging (DWI)-fluid-attenuated inversion recovery (FLAIR) mismatch on MRI.9–13 Current guidelines also recommend intravenous alteplase for these patients.14–16 This study aimed to determine the efficacy and safety outcomes of alteplase for patients with stroke of unknown onset time and with prior APT. We hypothesized that patients with prior APT would have favorable outcomes with imaging-based IVT despite the increased risk of bleeding associated with APT.
Methods
Study design and population
This was post hoc secondary analysis of the EOS dataset. Briefly, the dataset consists of a systematic review and meta-analysis of individual patient-level data, including RCTs involving IVT with alteplase for stroke patients with unknown time of onset, derived through advanced imaging diagnostics. The design and primary outcomes of EOS have been previously published. 13 A total of 843 patients were registered in trials based on the DWI-FLAIR or ischemic core/perfusion mismatch criteria.9–12 Patients contraindicated for alteplase treatment were excluded, except for those with unknown stroke onset time. All studies included in this research received approval from the institutional review board at each site, and all participants provided informed consent prior to the initiation of the study.
Outcomes
Following the criteria used in EOS, the primary outcome was a favorable outcome defined as a modified Rankin Scale (mRS) score of 0–1 at 90 days. The secondary outcomes were functional independence (i.e. mRS score of 0–2) and functional improvement across the entire mRS score (mRS shift analysis) at 90 days. The safety outcomes included sICH according to the Safe Implementation of Thrombolysis in Stroke Monitoring Study criteria as a local or remote parenchymal hematoma (PH) type 2 on the imaging scan obtained 22 to 36 h after treatment, accompanied by neurological deterioration indicated by an increase of ⩾4 points in the National Institutes of Health Stroke Scale (NIHSS) score compared to either the baseline value or the lowest value within 24 h, or any hemorrhage leading to death. 17 Additional safety outcomes included radiologically defined parenchymal hemorrhage type 2, 18 death at 7 days after randomization, death at 90 days after randomization, and severe disability or death (i.e. an mRS score of 4–6).
Statistical analysis
Baseline characteristics were expressed as median values (interquartile range [IQR]) or means ± standard deviations for continuous variables and as frequencies or percentages for categorical variables. Significant differences between the prior APT and no prior APT groups were evaluated using Student’s t or the Kruskal–Wallis test for continuous variables and the χ2 or Fisher’s exact test for categorical variables. Meanwhile, the primary and safety outcomes were compared between the assigned treatment groups (alteplase and control groups) in patients with APT and those without APT using a mixed-effect logistic regression model. The treatment effect was reported as odds ratios with 95% confidence intervals (CIs). The categorical shift in the distribution of mRS scores was evaluated by employing a proportional-odds logistic regression model. All models were adjusted for the stratification variables of age, sex, and NIHSS score at randomization. The interaction between alteplase and APT was tested by adding an interaction term to the model for the primary and safety outcomes. For outcome analyses, missing values were not replaced. As a sensitivity analysis to estimate the effect of potential bias caused by missing data, we repeated this regression analysis after imputing missing values using multiple imputations with chained equations (20 imputations). All statistical analyses were conducted using STATA (version 17, Stata Corporation, College Station, TX, USA). A two-sided p-value <0.05 was considered statistically significant.
Results
Patient characteristics
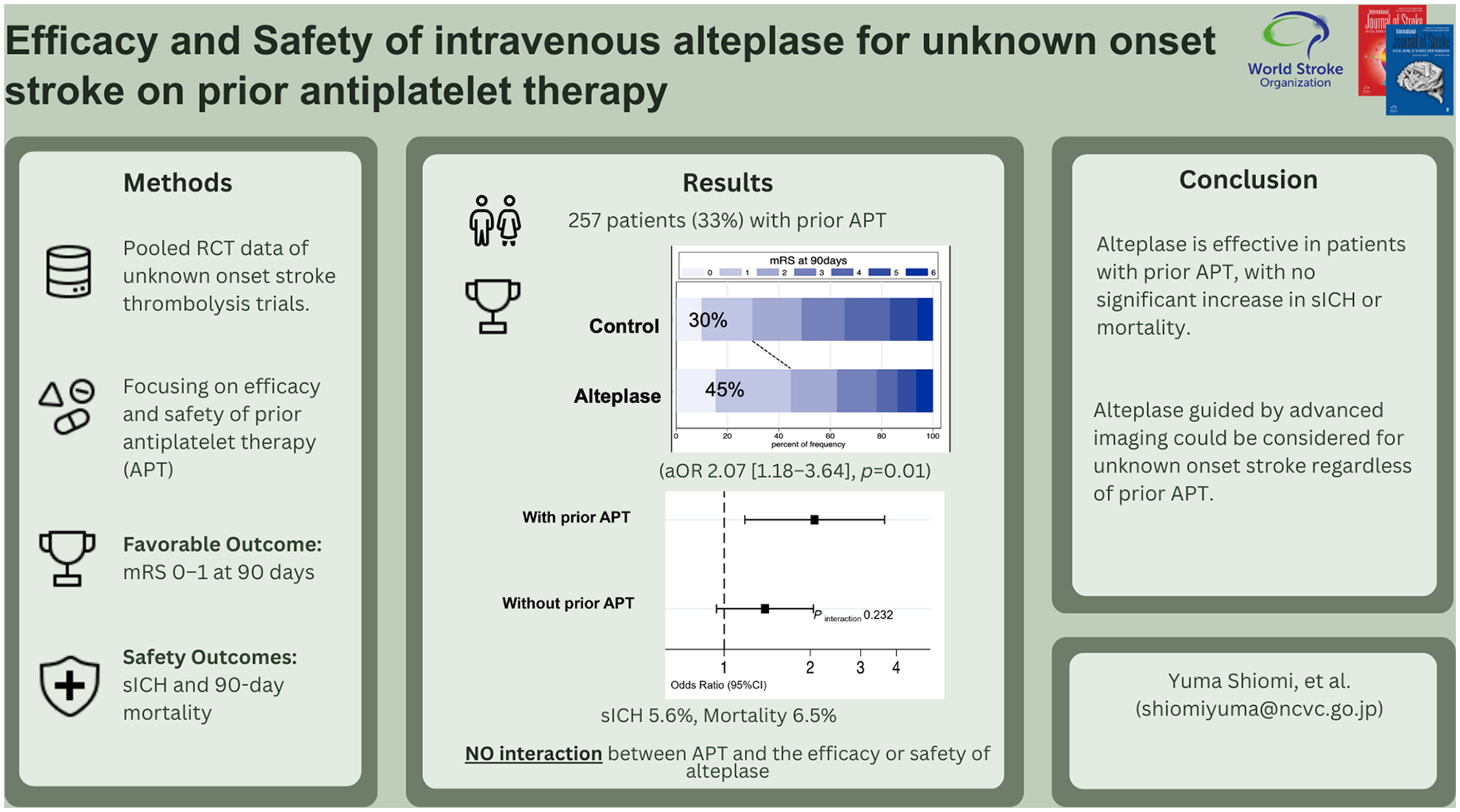
Among the 843 patients included in the EOS dataset, all 63 patients in the ECASS-4 trial were excluded owing to the lack of data on prior APT. A total of 780 patients included in the WAKE-UP, THAWS, and EXTEND trials had available data on prior APT (Figure S1). Of them, 257 (33%) patients were receiving APT prior to stroke. Table 1 shows the baseline clinical characteristics of the prior APT and no prior APT groups. Compared with the no prior APT group, the prior APT group was older (72 vs. 66 years) and had a higher prevalence of hypertension, diabetes mellitus, dyslipidemia, atrial fibrillation, previous ischemic stroke, and transient ischemic attacks (all ps < 0.05). The mRS score at 90 days post-stroke was missing for 14 patients (1.8%), and PH type 2 data were missing for 153 patients (19.6%).
Baseline characteristics between patients with and without prior antiplatelet therapy.
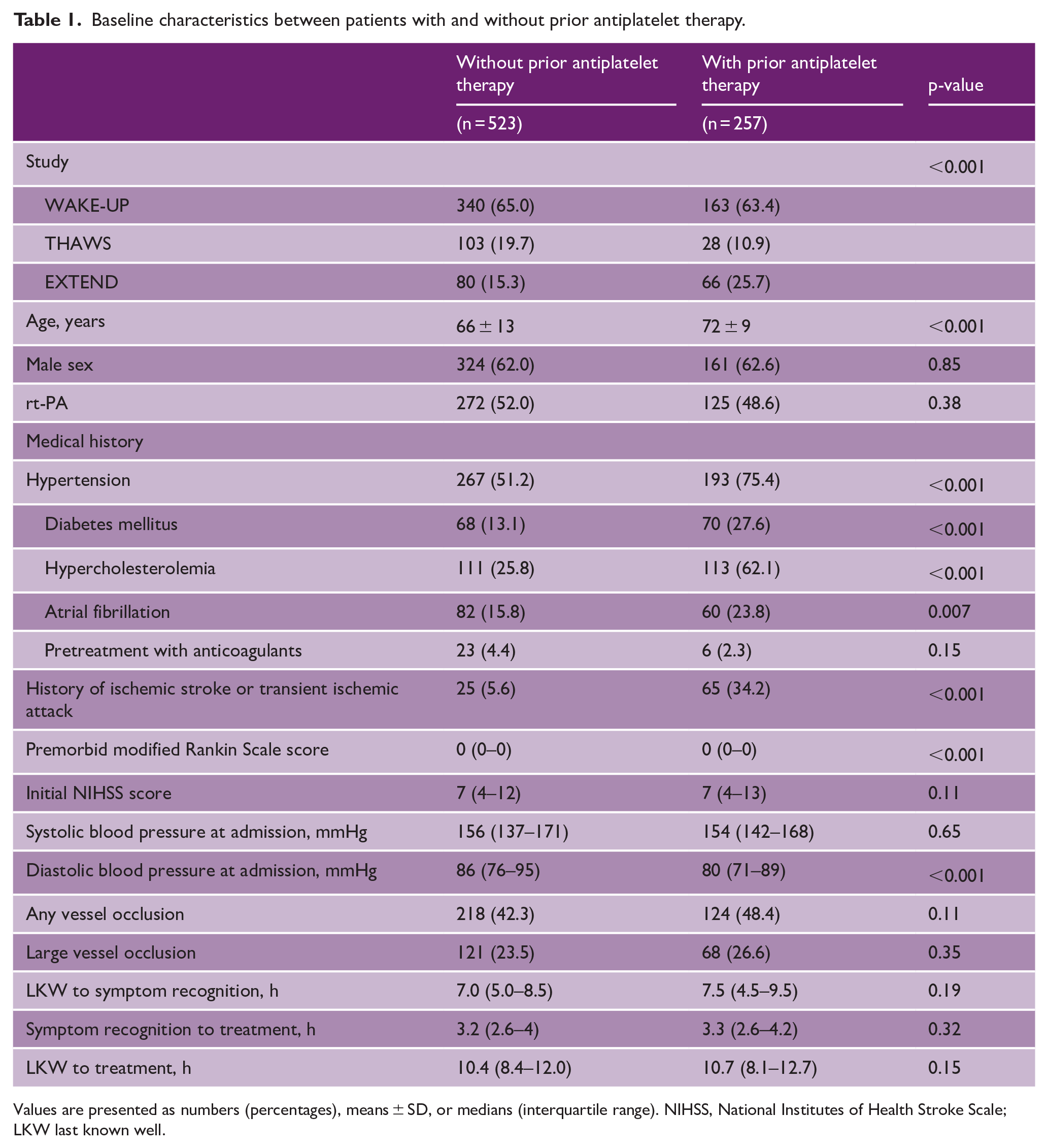
Values are presented as numbers (percentages), means ± SD, or medians (interquartile range). NIHSS, National Institutes of Health Stroke Scale; LKW last known well.
Interaction of efficacy and safety with prior APT in the overall cohort
No interaction between prior APT and treatment effects of alteplase was observed in the overall cohort (p interaction = 0.23; Figure 1). Similarly, no interaction between prior APT and safety outcomes of alteplase was observed (Tables S1A and S1B).
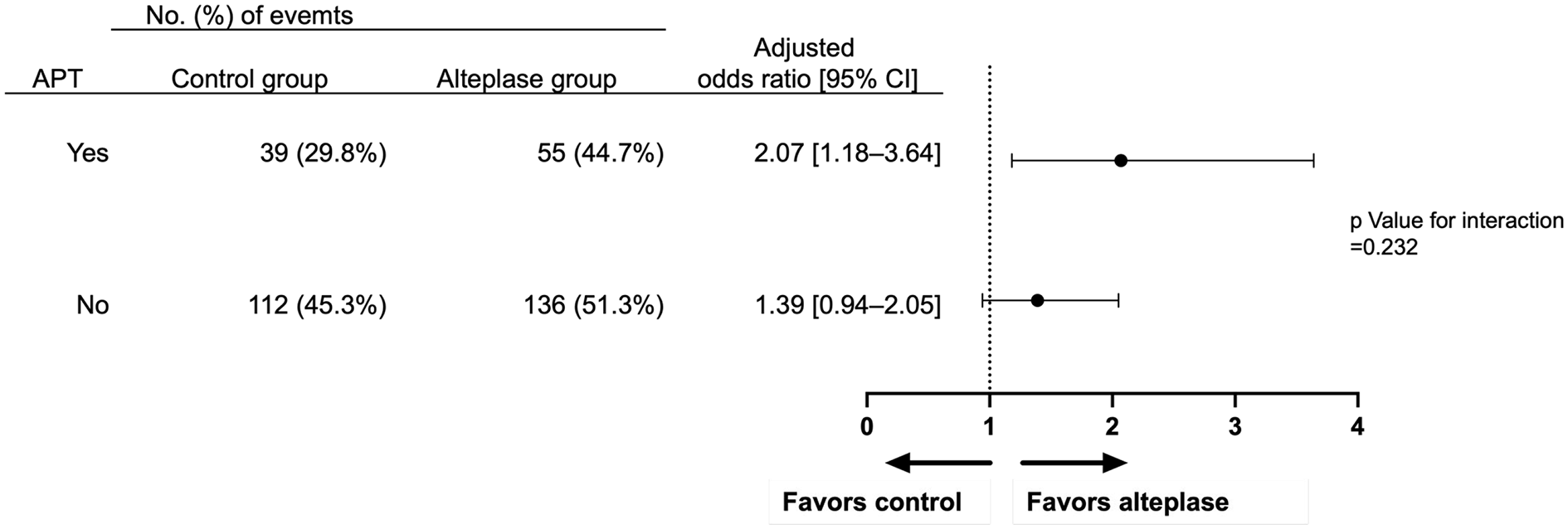
Association of alteplase with favorable outcome. Forest plots for the primary outcome of favorable outcome (modified Rankin Scale score 0–1 at 90 days after stroke) in patients with or without prior antiplatelet therapy (APT).
Efficacy and safety outcomes in patients with APT
Of the 257 patients with prior APT, 125 (49%) and 132 (51%) patients were randomized to receive alteplase and control treatments, respectively. All baseline patient characteristics were comparable between the two groups, except for hypercholesterolemia (Table S2). A favorable outcome was observed in 55 (45%) patients in the alteplase group and 39 (30%) patients in the control group (adjusted odds ratio [aOR], 2.07 [95% CI, 1.18–3.64]; Table 2). Functional independence was observed in 77 (63%) patients in the alteplase group and 64 (49%) patients in the control group (aOR, 2.18 [95% CI, 1.17–4.06]). The mRS score significantly shifted toward better functional outcome at 90 days, favoring the alteplase group (aOR, 1.85 [95% CI, 1.20–2.94]; Figure 2(a)).
Efficacy and safety outcomes of intravenous alteplase in patients with prior antiplatelet therapy (n = 257).
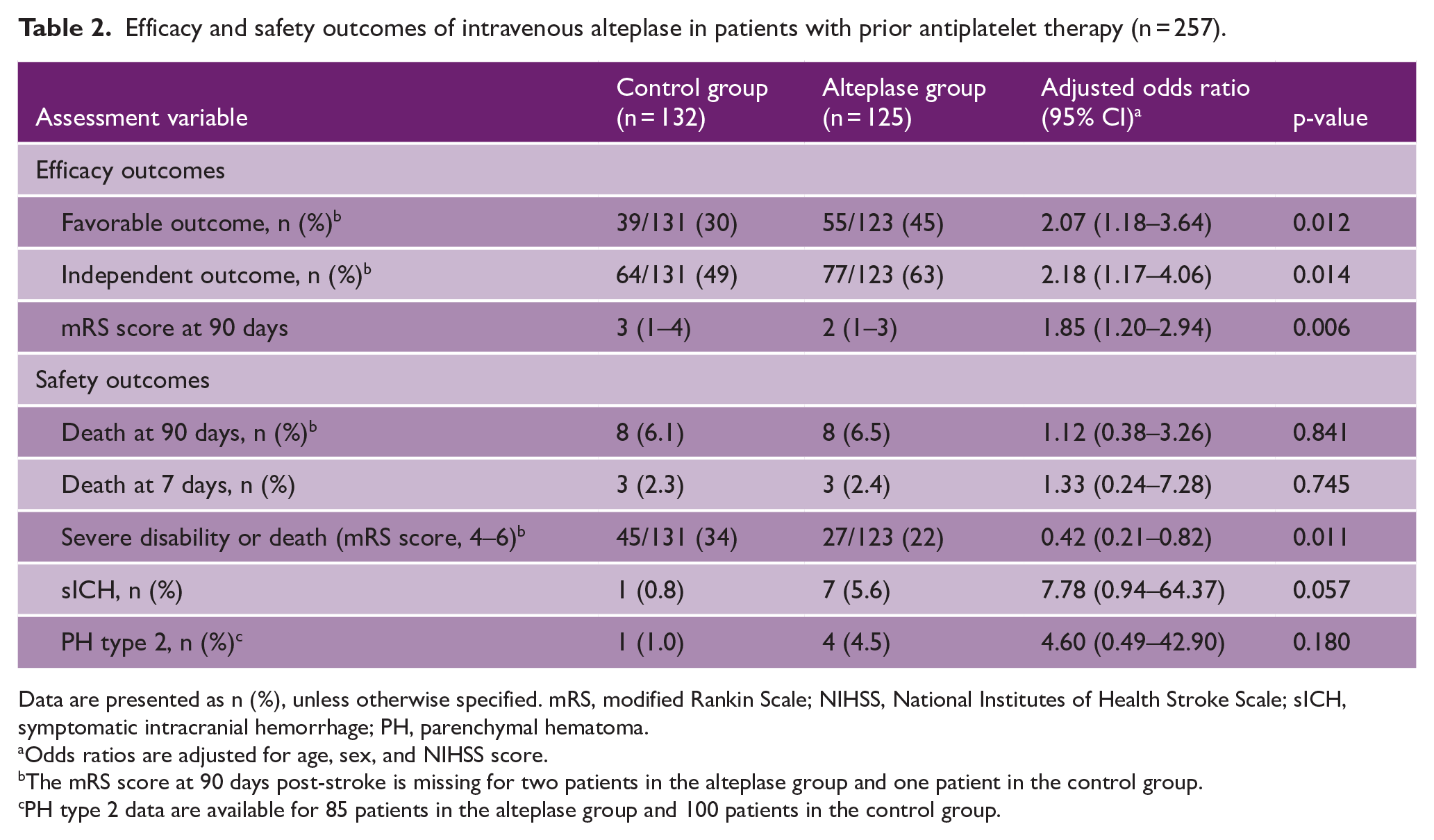
Data are presented as n (%), unless otherwise specified. mRS, modified Rankin Scale; NIHSS, National Institutes of Health Stroke Scale; sICH, symptomatic intracranial hemorrhage; PH, parenchymal hematoma.
Odds ratios are adjusted for age, sex, and NIHSS score.
The mRS score at 90 days post-stroke is missing for two patients in the alteplase group and one patient in the control group.
PH type 2 data are available for 85 patients in the alteplase group and 100 patients in the control group.
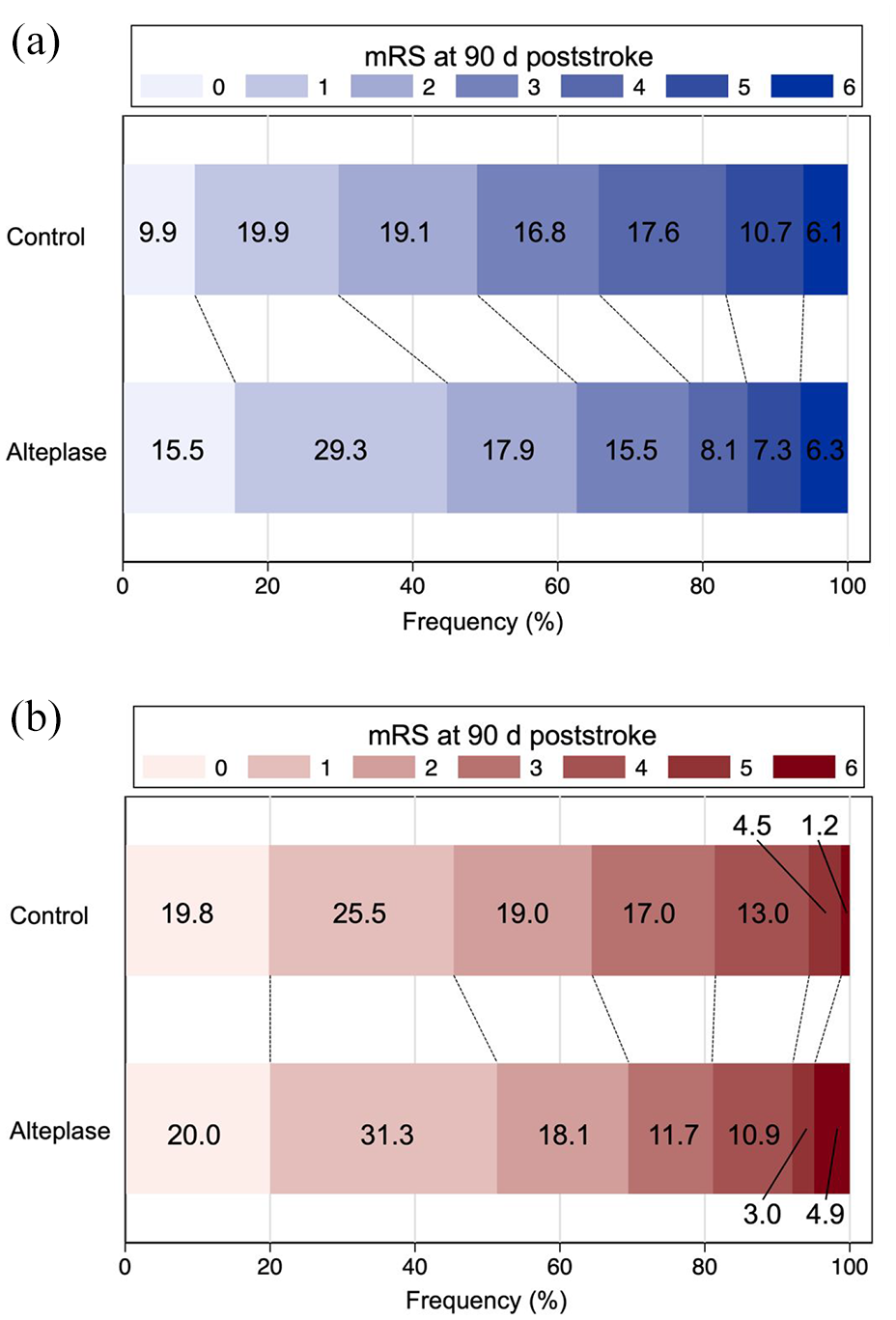
Distribution of modified Rankin scale scores at 90 days post-stroke. Distribution of mRS scores (range, 0–6) in patients with (a) and without (b) prior antiplatelet therapy (APT) treated with alteplase or placebo/control.
For safety outcomes, the 90-day mortality rate was 6.5% (n =8 patients) in the alteplase group and 6.1% (n = 8 patients) in the control group (aOR, 1.12 [95% CI, 0.38–3.26]; Table 2). sICH occurred in seven patients (5.6%) in the alteplase group but in one patient (0.8%) in the control group (aOR, 7.78 [95% CI, 0.94–64.37]; Table 2). There was no significant interaction effect between alteplase and prior APT (p for interaction = 0.485; Table S1A). The sensitivity analysis using an imputed regression model had similar results (Table S3). Among these sICH cases, alteplase was administered at a standard dose (0.9 mg/kg) in six patients and at a low dose (0.6 mg/kg) in one patient. For these sICH cases, the median time from symptom onset or last known well to treatment was 12.2 h (IQR, 9.0–13.8 h), which was comparable to that in the non-sICH group (median 10.9 h [IQR, 7.9–12.5 h]). Functional outcomes among sICH patients in the alteplase group were poor, with all patients demonstrating moderate to severe disability or death (mRS score 4–6) at 90 days, and two patients dying within 7 days. A sensitivity analysis excluding THAWS, the only trial that used a low dose of alteplase, revealed similar findings (Tables S4 and S5).
Efficacy and safety outcomes in patients without APT
Detailed outcomes in patients without prior APT are provided in the Supplemental Material (Figures 1 and 2(b), Tables S6 and S7). No significant differences in sICH or mortality were observed
Discussion
We directly compared IVT-related outcomes in patients with prior APT in pooled RCTs. The results showed no significant interaction for both the efficacy and safety outcomes of alteplase with prior APT. The efficacy and safety of alteplase in patients with ischemic stroke did not differ between those with and without prior APT. This suggests that prior APT does not influence the decision to use imaging-based alteplase in patients with unknown onset stroke. Notably, within the subgroup of patients with prior APT, those who underwent alteplase treatment demonstrated significantly better functional outcomes than did controls. Particularly, the proportion of patients with mRS scores of 0–1 and 0–2 was, respectively, 15% and 14% higher in these patients. In contrast, the rate of sICH following alteplase treatment tended to be numerically higher in patients with prior APT, but there was no significant difference in mortality.
Patients with prior APT were older and had more vascular risk factor profiles than those without APT, although stroke severity was comparable between these patients. Nonetheless, the alteplase group had more favorable outcome than the control group among patients with prior APT. This was not observed in patients without prior APT. These results were confirmed in other efficacy outcomes and indicate the potential antithrombotic effects of prior APT (e.g. inhibition of platelet activation and subsequent thrombus formation) given its long half-life. In addition, the National Institute of Neurological Disorders and Stroke rt-PA stroke trial reported less clinical deterioration following alteplase administration in patients with prior APT. 19 In one observational study, the initiation of antithrombotic therapy within 24 h following alteplase administration was associated with significantly improved vessel patency rates and enhanced functional prognosis. 20 In contrast, the APT in combination with rt-PA Thrombolysis in Ischemic Stroke (ARTIS) trial showed an increased risk of hemorrhage with the concurrent administration of alteplase and aspirin. 21 The ARTIS trial is the sole RCT investigating the combination of APT with alteplase in patients with acute ischemic stroke. It should be noted, however, that aspirin was administered at a dose of 300 mg in the trial, precluding direct comparisons with other trials.
Although there was no significant interaction between alteplase and prior APT with respect to safety outcomes, there were numerically more cases of sICH in the prior APT group than in the no prior APT group. This is expected given the known higher rate of bleeding events with alteplase. 3 However, the 5.6% rate of alteplase-related sICH in the prior APT group was comparable to that in other studies of patients with prior APT who received alteplase. 4 Tenecteplase emerges as a potential alternative to alteplase. Since tenecteplase up to 24 h post-stroke, using imaging-based selection to identify eligible patients, causes sICH in only ≈3% of the patients, this thrombolytic would be safe in terms of assessing a tissue clock.22,23
The background of the patients in the EOS dataset may also have impacted the findings. The 780 patients evaluated in this study had a median initial NIHSS of 7, indicating mostly mild to moderate strokes. For each patient, the decision to perform reperfusion therapy was based on findings of DWI-FLAIR mismatch or ischemic core/perfusion mismatch. In previous observational studies, the rate of sICH following alteplase ranged between 2% and 7%, and the incidence observed in the prior APT group in this study is consistent with these previous findings. 24 The use of advanced imaging techniques might be associated with improved safety of reperfusion therapy. In addition, the sex distribution in our study aligns with trends commonly reported in stroke clinical trials.25,26 The low enrollment rates of women in clinical trials were partly caused by lower ischemic stroke incidence for women than men in middle aged population. 27 This gender inequality was also influenced by barriers specific to their participation, social determinants of health, and differences in healthcare perceptions and experiences.
The strength of the present study was the analysis of a well-organized, pooled individual patient-level database of multiple RCTs. This approach enabled a more accurate evaluation of IVT outcomes in patients with APT. Furthermore, the inclusion of a diverse population from various geographic regions, especially Europe, Australia, and Japan, allowed for broader applicability across different demographics. However, this study also has some limitations that need to be acknowledged. First, we performed a post hoc analysis, and thus, the results should be interpreted cautiously. Second, the study lacked information on prior APT from the ECASS-4 trial. Third, this study did not have detailed information regarding the specific antiplatelet medications used, whether patients were receiving more than one type of these drugs or their dosages. Therefore, we could not evaluate the differential risks associated with specific antiplatelet agents or the potential impact of dual APT. Fourth, the THAWS trial administered an open-label standard of care but not placebo as the control group, and this protocol may have introduced potential modification of treatment effects. However, we conducted sensitivity analysis and observed consistency in the result. Residual confounding from unmeasured variables, such as race, healthcare system differences, or socioeconomic status, may also have influenced the results and limited their generalizability.
In conclusion, imaging-based intravenous alteplase is effective in patients with stroke of unknown onset, with no difference in treatment effects among patients with or without prior APT exposure. Treatment with alteplase appears beneficial in patients with prior APT. Despite the higher rate of treatment-associated sICH, the difference is not significant, suggesting an acceptable risk–benefit ratio. Collectively, our findings suggest that intravenous alteplase guided by advanced neuroimaging could be considered for unknown onset stroke regardless of prior APT.
Supplemental Material
sj-docx-1-wso-10.1177_17474930251322034 – Supplemental material for Efficacy and safety of intravenous alteplase for unknown onset stroke on prior antiplatelet therapy: Post hoc analysis of the EOS individual participant data
Supplemental material, sj-docx-1-wso-10.1177_17474930251322034 for Efficacy and safety of intravenous alteplase for unknown onset stroke on prior antiplatelet therapy: Post hoc analysis of the EOS individual participant data by Yuma Shiomi, Kaori Miwa, Märit Jensen, Manabu Inoue, Sohei Yoshimura, Naruhiko Kamogawa, Mayumi Fukuda-Doi, Henry Ma, Peter Ringleb, Ona Wu, Lee H Schwamm, Stephen M Davis, Geoffrey A Donnan, Christian Gerloff, Jin Nakahara, Kazunori Toyoda, Götz Thomalla and Masatoshi Koga in International Journal of Stroke
Footnotes
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Ma has consulted for Independent Sector. Dr Ringleb has consulted for Boehringer Ingelheim and Daiichi Sankyo Company and reports personal fees from Bayer Healthcare and Bristol-Myers Squibb. Dr Wu reports grants from National Institutes of Health. Dr Schwamm serves as a scientific consultant regarding trial design and conduct to Genentech (late-window thrombolysis) and as a member of steering committee (TIMELESS [A Phase III, Prospective, Double-Blind, Randomized, Placebo-Controlled Trial of Thrombolysis in Imaging-Eligible, Late-Window Patients to Assess the Efficacy and Safety of Tenecteplase] NCT03785678); stroke systems of care consultant to the Massachusetts Department of Public Health; member of a Data Safety Monitoring Boards (DSMB) for Penumbra (MIND NCT03342664) and for Diffusion Pharma PHAST-TSC (Double-Blind, Randomized, Placebo-Controlled, Phase II Study of Efficacy and Safety of Trans Sodium Crocetinate [TSC] Administered Onboard Emergency Vehicles for Treatment of Suspected Stroke; NCT03763929); and delivering continuing medical education lectures for Boehringer Ingelheim and Prime Education: stroke systems of care and improving time to thrombolysis. Dr Warach has consulted for Genentech. Dr Davis reports personal fees from Boehringer Ingelheim. Dr Donnan has consulted for Amgen. Dr Gerloff has consulted for Abbott Canada, Allergan, Amgen, Astra-Zeneca, Bayer Healthcare, and Boehringer Ingelheim and reports grants from Deutsche Forschungsgemeinschaft, Deutsches Zentrum für Luft- und Raumfahrt, European Union, Gemeinnützige Hertie-Stiftung, Merz Farmaceutica Comercial LTDA, and Schilling-Stiftung. Dr Thomalla has consulted for Acandis, Bayer, PORTOLA PHARMACEUTICALS, LLC, and Stryker and reports personal fees from Alexion Pharmaceuticals Inc, Amarin Pharma Inc, Boehringer Ingelheim, Bristol-Myers Squibb, and Daiichi Sankyo Europe GmbH. Dr Kitazono reports grant from Daiichi Sankyo Company LTD. Dr Toyoda has consulted for Otsuka Pharmaceutical and reports personal fees from Abbott Japan, Bayer, Bristol-Myers Squibb, Daiichi Sankyo, and Novartis. Dr Koga has consulted for Janssen Pharmaceuticals, reports personal fees from Daiichi Sankyo Company, Bayer, Bristol-Myers Squibb, Mitsubishi Tanabe Pharma Corporation, and Pfizer, and reports grants from Daiichi Sankyo Company LTD, Nippon Boehringer Ingelheim Co Ltd. The other authors report no conflicts.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The WAKE-UP (Efficacy and Safety of MRI-Based Thrombolysis in Wake-Up Stroke) was supported by a grant (number 278276) from the European Union Seventh Framework program. EXTEND (Extending the Time for Thrombolysis in Emergency Neurological Deficits) was funded by the National Health and Medical Research Council, an Australian Government organization, and Commonwealth Scientific and Industrial Research Organisation. Boehringer Ingelheim provided the study investigational products free of charge. THAWS (Thrombolysis for Acute Wake-up and Unclear-onset Strokes with alteplase at 0.6 mg/kg) was supported by the Japan Agency for Medical Research and Development (AMED; 19ek0210091h0003 and 233fa627011h0002), the Ministry of Health, Labour, and Welfare, and the Mihara Cerebrovascular Disorder Research Promotion Fund. ECASS-4 (European Cooperative Acute Stroke Study-4) was an investigator-initiated trial supported by an unrestricted grant from Boehringer Ingelheim (Germany). MR WITNESS (a Phase IIa Safety Study of Intravenous Thrombolysis with alteplase in MRI-selected patients) was supported by the National Institutes of Health (NIH), National Institute of Neurological Disorders and Stroke (NINDS) SPOTRIAS (Specialized Program of Transitional Research in Acute Stroke; P50-NS051343), and NINDS Division of Intramural Research, was done in part at the Athinoula A Martinos Center for Biomedical Imaging at Massachusetts General Hospital, using resources provided by the Center for Functional Neuroimaging Technologies (P41EB015896), a P41 Biotechnology Resource Grant supported by the NIH National Institute of Biomedical Imaging and Bioengineering. Genentech provided alteplase free of charge to the study for distribution to all sites except to the NINDS intramural branch and starting in year 2 provided supplemental site payments to permit expansion to 14 sites.
Ethical approval and informed consent
The protocol was approved by the ethics committee at each participating site, and all patients gave written informed consent.
ORCID iDs
Data availability statement
The corresponding author will provide this study’s supporting data upon reasonable request with approval from the EOS Steering Committee.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
