Abstract
Introduction:
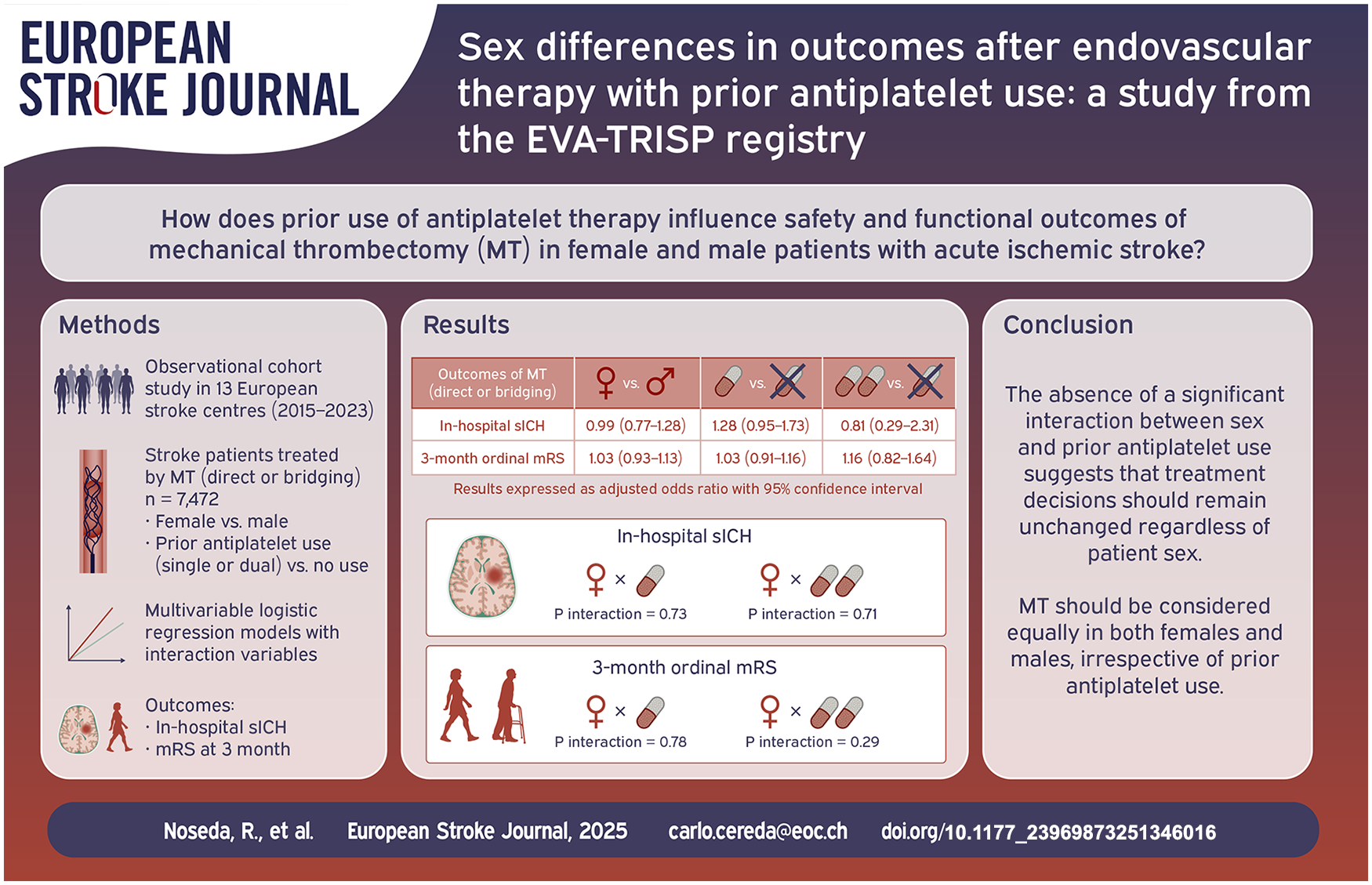
This study assessed sex differences in outcomes after mechanical thrombectomy (MT) with prior antiplatelet use using the EVA-TRISP multinational registry.
Patients and methods:
A cohort of consecutive patients (2015–2023) treated with MT was analysed by sex and prior antiplatelet use in multivariable logistic regression models. Primary outcomes were in-hospital symptomatic intracranial haemorrhage (sICH) and the 3-month modified Rankin Scale (mRS) score.
Results:
Among 7472 patients (47.9% females, older than males: 77 vs 72 years, p < 0.0001), sICH rates were similar (females 4.6%, males 4.4%, p = 0.405). The odds of sICH were not influenced by sex (adjusted odds ratio, aOR, 0.99, 95% confidence interval, CI, 0.77–1.28) or prior antiplatelet use (single therapy: aOR 1.28, 95% CI 0.95–1.73; dual therapy: aOR 0.81, 95% CI 0.29–2.31). No significant interactions were found between sex and antiplatelet use (p = 0.73 for single, p = 0.71 for dual therapy). Females had worse 3-month ordinal mRS scores than males (p < 0.0001), but the odds of a higher mRS score were not associated with sex (aOR 1.03, 95% CI 0.93–1.13) or prior antiplatelet use (single: aOR 1.03, 95% CI 0.91–1.16; dual: aOR 1.16, 95% CI 0.82–1.64). No interactions were found between sex and antiplatelet use for mRS (p = 0.78 for single, p = 0.29 for dual therapy).
Discussion and conclusions:
This study showed that there is no apparent interaction between patient sex and prior use of antiplatelets in relation to safety and functional outcomes of MT. Therefore, there is no reason to support a different decision-making approach by practicing physicians regarding MT in females and males previously taking antiplatelets.
Keywords
Introduction
Endovascular therapy with mechanical thrombectomy (MT) has been established as the standard of care for patients with acute ischaemic stroke (AIS) caused by large artery occlusion in the anterior circulation.1 –5 Sex differences in platelet reactivity and platelet counts have been described.6 –8 Among mechanism(s) responsible for sex differences in platelet reactivity are intrinsic and extrinsic determinants that upregulate the pro-thrombotic and pro-inflammatory functions of platelets in females compared to males. 6
A few studies have systematically assessed the influence of biological sex on outcomes after MT, however, they are mostly post hoc analyses of randomized clinical trial (RCT) data and thus not sufficiently powered to address the question appropriately,9,10 or observational studies showing heterogeneous results limited by single-centre study designs, use of national registries, restricted patient cohorts and/or lack of interaction assessment between patient sex and other variables of interest.11 –18 Regardless of patient sex, several studies have assessed prior use of antiplatelets (before stroke onset) as a potential determinant of MT safety and efficacy outcomes in AIS patients reporting conflicting findings.19 –23 Despite the existing bulk of evidence suggesting distinct effects of patient sex and the prior use of antiplatelets on outcomes after MT, to date, studies specifically evaluating the interaction between patient sex and prior use of antiplatelets in relation to safety and functional outcomes after MT in patients with AIS are lacking.
The aim of this cohort study was to compare safety and functional outcomes after MT (direct, i.e. immediate initiation of endovascular treatment without prior administration of intravenous thrombolysis, IVT, or bridging, i.e. administration of IVT followed by endovascular treatment, hereafter in the manuscript and in the tables referred to as direct MT or bridging) between females and males with prior use of antiplatelets by querying the multicentre, multinational prospective registry, EndoVAscular treatment and ThRombolysis for Ischaemic Stroke Patients (EVA-TRISP).
Methods
Study design, setting and participants
This was an observational cohort study based on the EVA-TRISP registry that collects standardized data from participating European stroke centres of consecutive AIS patients treated with MT (direct or bridging). Currently, the registry comprises more than 20 centres collecting data on more than 10,000 patients. Details of the EVA-TRISP aims, collaborating stroke centres and included patient demographics, pre-stroke history, acute phase management and long-term outcome have been previously comprehensively described. 24
For the present study, consecutive AIS patients admitted between 2015 and 2023, aged 18 years or older, were included. The exclusion criteria outlined in Figure 1 were applied according to the sequence provided. Patients were excluded if information on pre-stroke disability (defined as modified Rankin scale, mRS) was unavailable; if had stroke mimic as possible cause; if used anticoagulants before stroke onset; and if information on biological sex and prior antiplatelet therapy was missing. Based on the biological sex of the patients, the study population was divided into two groups (females and males) and then evaluated for the use of antiplatelets prior to stroke, either as single or dual therapy.
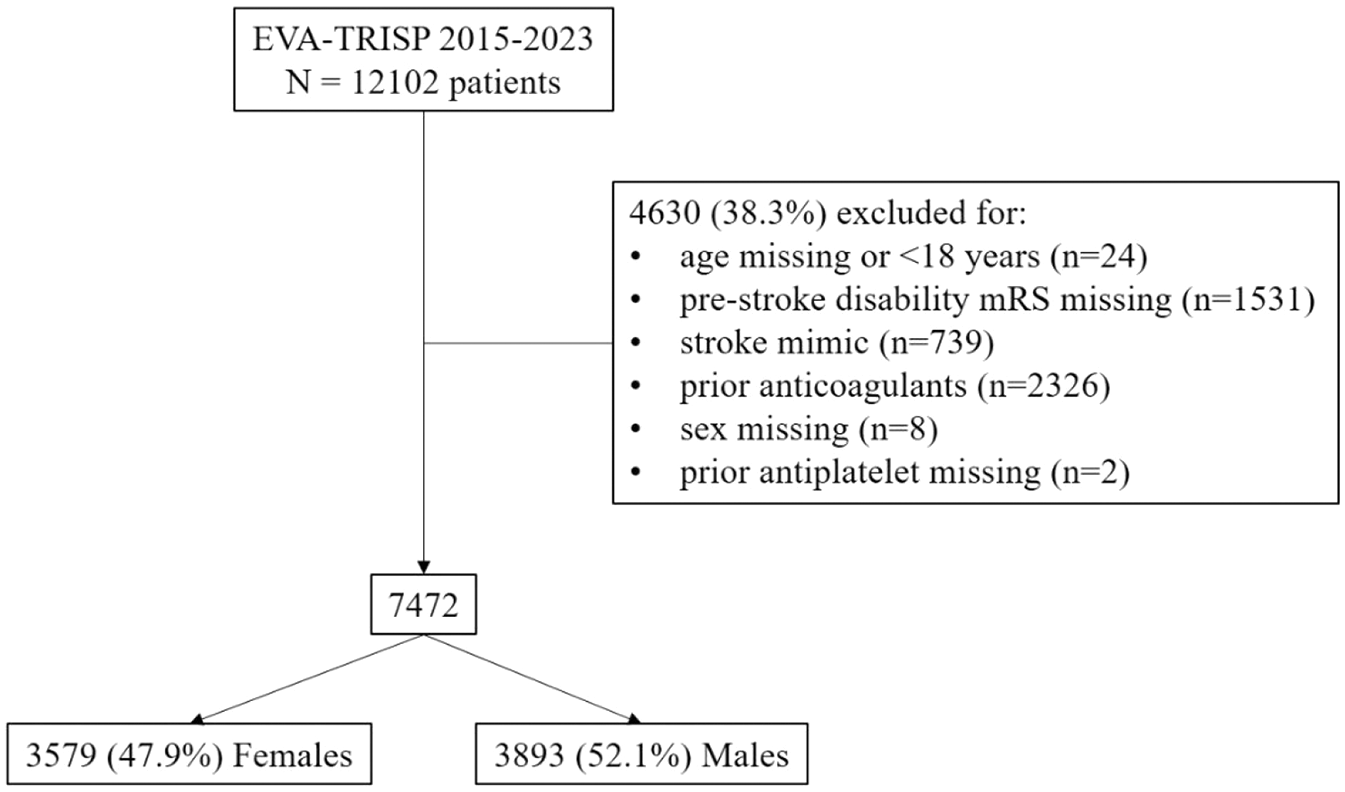
Flowchart of the selection process of patients included in the study.
This study was approved by the local ethics committee (CE Req-2023-01533). The dataset covering the study period was provided by the coordinating centre (at the University of Basel) and was composed of anonymized data, without any coded data. This study followed the STrengthening the Reporting of OBservational Studies in Epidemiology (STROBE) recommendations.
Variables
In addition to information on the biological sex of the patient and antiplatelet use prior to stroke, the following variables were described for the study population: contributing stroke centre; patient age at stroke onset and medical history (including previous stroke, hypertension, diabetes, hypercholesterolaemia, smoking, atrial fibrillation and coronary artery disease); pre-stroke disability as mRS score; whether the time of symptom onset was known (i.e. witnessed) or symptoms were first noted upon awakening from sleep (i.e. wake-up stroke); stroke aetiology according to the TOAST classification; neurologic impairment upon admission measured by the National Institutes of Health Stroke Scale (NIHSS); systolic and diastolic blood pressure at admission; type of intervention (direct MT or bridging).
Outcomes
The primary safety outcome was in-hospital symptomatic intracranial haemorrhage (sICH) defined according to the definition used in the European Cooperative Acute Stroke Study II as an ICH causing an increase in the NIHSS score of more than or equal to four points (and occurring within 7 days of AIS onset).
The primary functional outcome was the mRS score at 3 months after discharge, assessed by an ordinal scale ranging from 0 (no symptoms) to 6 (death). Centres participating in the EVA-TRISP collaboration document the mRS score at 3 months after discharge by telephone calls, postal/electronic questionnaires or outpatient visits. If patients cannot be interviewed, close relatives, nurses or family doctors are asked to report on disability status.
Secondary functional outcomes at 3 months after discharge included functional independence (mRS score 0–2) and mRS score 3–5. The achievement of successful reperfusion after MT, that is, Modified Treatment In Cerebral Ischaemia (mTICI) score 2b–3 on first follow-up imaging was assessed as the imaging outcome.
Statistical analysis
The baseline characteristics of the study population were described and compared between females and males. Continuous variables were described as medians with interquartile range and compared using the Wilcoxon rank sum test. Categorical variables were described as counts and percentages and compared using Pearson’s chi-square test.
Multivariable logistic regression was used to model the association between patient sex and each outcome according to prior use of antiplatelets. With this aim, interaction variables between patient sex and prior use of antiplatelets were introduced. All models were adjusted for meaningful demographic and clinical patient variables potentially influencing outcome measures, including patient age, NIHSS score at admission, pre-stroke disability, medical history of hypertension, hypercholesterolaemia, diabetes, coronary heart disease, smoking, atrial fibrillation, previous stroke and use of IVT. An ordinal logistic regression model was fitted to assess the mRS score at 3 months after discharge and an overall point estimate provided as an odds ratio (OR) representing a shift in scores on the mRS (shift analysis). As sensitivity analyses, multilevel logistic regression models were implemented on the study cohort to assess both primary safety and functional outcomes, whereby participating stroke centres were considered as random effect. Logistic regression analyses by patient subgroups based on the type of intervention (direct MT or bridging) were then performed to assess the association of patient sex, prior use of antiplatelets and their interaction with the odds of primary and secondary outcomes. For these subgroup analyses, the models were adjusted for the same variables as in the overall study population, except for use of IVT (which was the defining variable for the subgroups). Sensitivity subgroup analyses were performed in patients with large-artery atherosclerosis and cardioembolic stroke aetiology to evaluate the impact of patient sex, prior antiplatelet use and their interaction on the safety and primary functional outcomes of direct MT and bridging therapy. Significance was determined for variables with the lower bound of the 95% confidence interval (CI) of the adjusted OR (aOR) higher than 1 and a two-tailed p value less than 0.05. All analyses were performed on complete observations by using the Statistical Analysis System Software (version 9.4; SAS Institute, Cary, NC, USA).
Results
Over the study period (2015–2023), 7472 patients from 13 stroke centres contributing to the EVA-TRISP registry met the selection criteria (Figure 1 and Supplemental Table 1). Of these, 3579 (47.9%) were females and 3893 (52.1%) males. Table 1 shows the baseline characteristics of the study population according to patient sex. Females were older than males (77 (66–84) vs 72 (63–80) years, p < 0.0001) and less frequently used antiplatelets prior to stroke onset (32.3% vs 35.4%, p = 0.0009). Hypertension and atrial fibrillation were more frequent in females (71.0% vs 65.5%, p = 0.0001 and 32.9% vs 26.1%, p < 0.0001, respectively), while smoking and coronary artery disease were more common in males (23.4% vs 17.4%, p < 0.0001, and 21.3% vs 12.8%, p < 0.0001, respectively). In terms of pre-stroke disability, females less frequently showed a mRS of 0 (no symptoms) than males (63.6% vs 72.0% p < 0.0001) and had a worse neurologic impairment at hospital admission (NIHSS, 14 (8–19) vs 13 (7–18), p = 0.001). Compared to males, females more frequently received direct MT (46.6% vs 42.7%, p = 0.001).
Baseline characteristics of the study population according to patient sex.
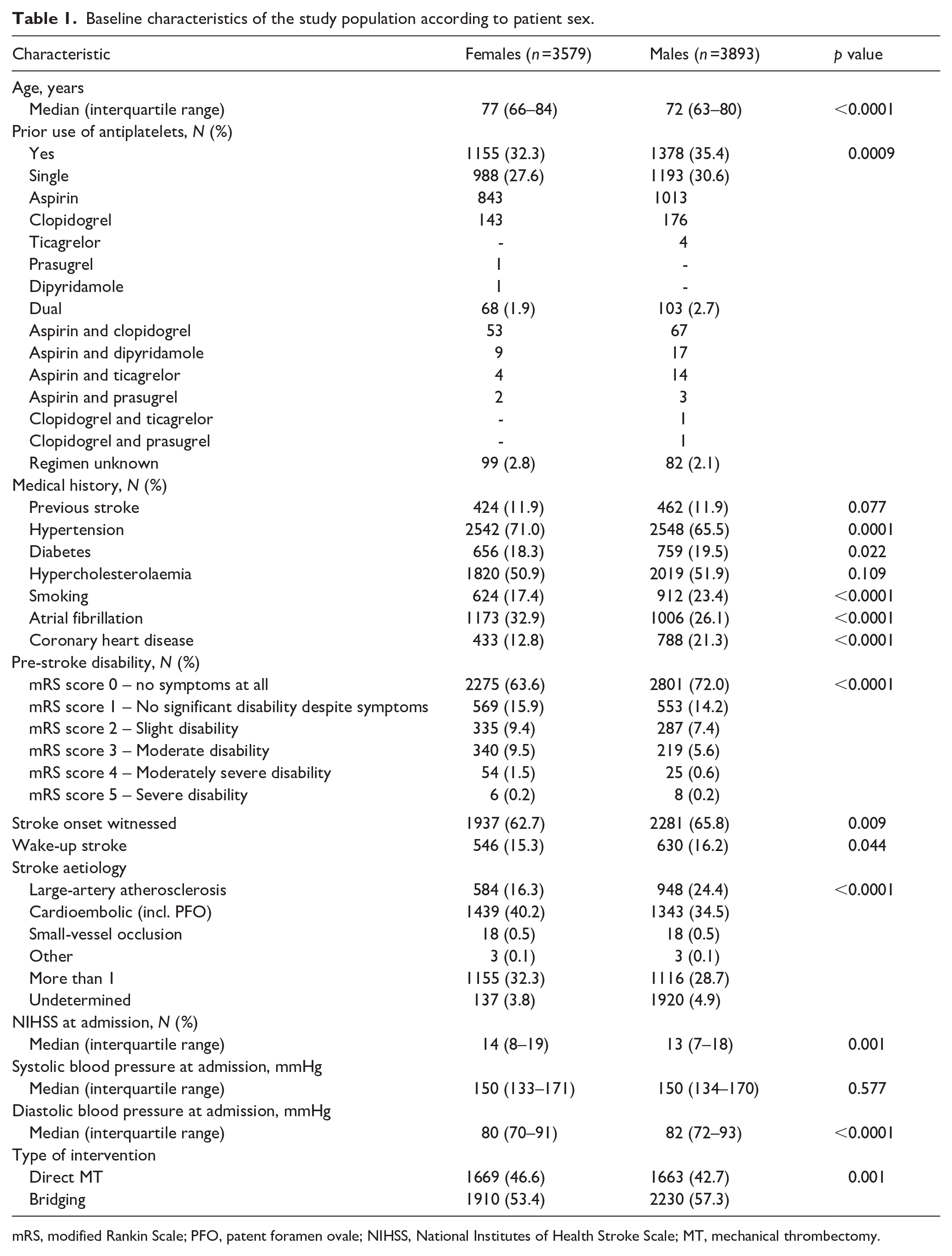
mRS, modified Rankin Scale; PFO, patent foramen ovale; NIHSS, National Institutes of Health Stroke Scale; MT, mechanical thrombectomy.
Primary outcomes
The proportions of females and males with sICH after MT were similar (4.6% and 4.4%, respectively, p = 0.405, Table 2). The multivariable logistic regression analysis showed that there were not sex differences in the odds of sICH (aOR 0.99, 95% CI 0.77–1.28, p = 0.96, Table 3). No association was observed between prior use of antiplatelets and the odds of sICH, either as single (aOR 1.28, 95% CI 0.95–1.73, p = 0.11) or dual therapy (aOR 0.81, 95% CI 0.29–2.31, p = 0.70), compared with no antiplatelet use prior to stroke onset (Table 3). No interaction was found between patient sex and prior use of antiplatelets regarding sICH (Table 3).
Primary and secondary outcomes according to patient sex.
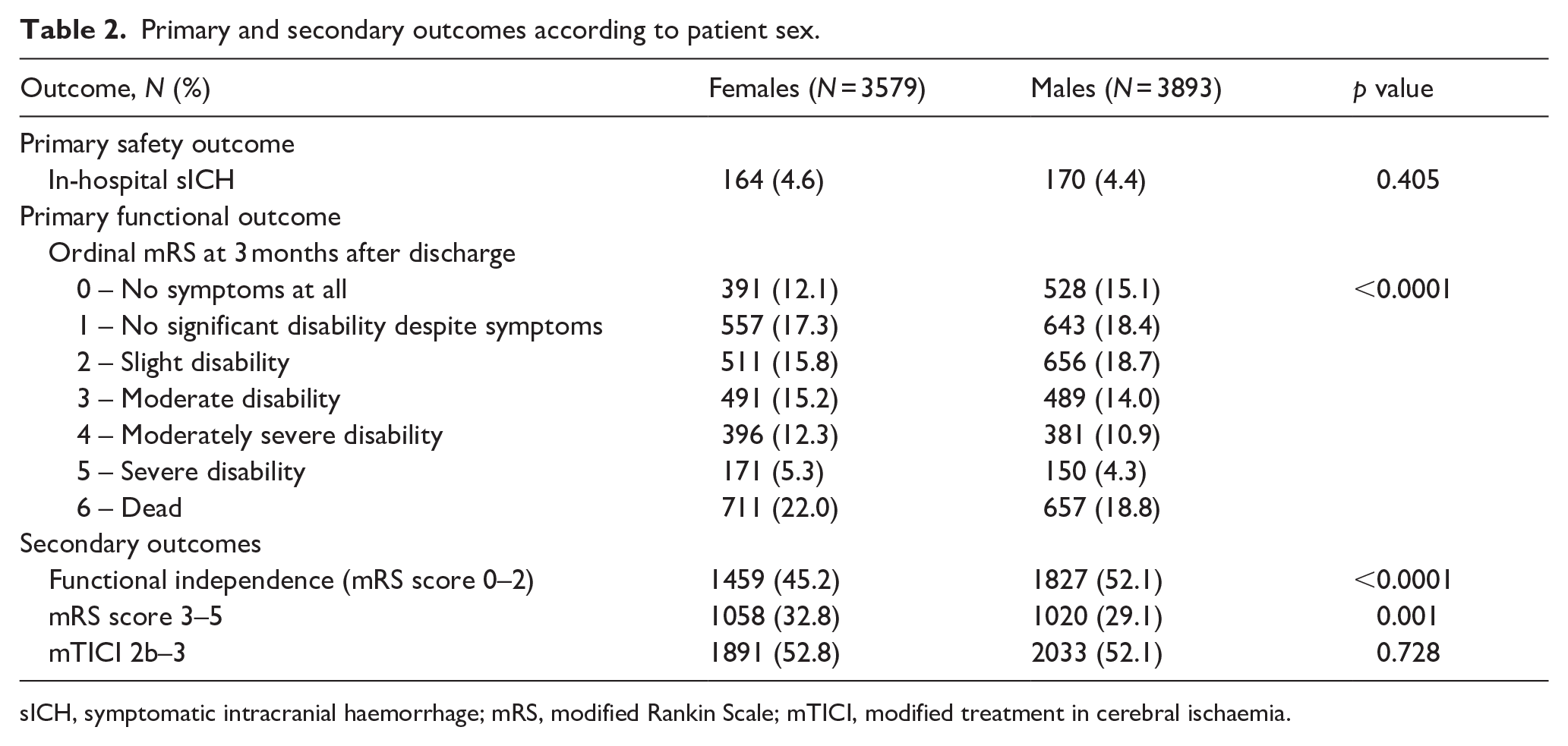
sICH, symptomatic intracranial haemorrhage; mRS, modified Rankin Scale; mTICI, modified treatment in cerebral ischaemia.
Multivariable logistic regression analyses on the association of patient sex, prior use of antiplatelets and their interaction with the odds of primary and secondary outcomes.
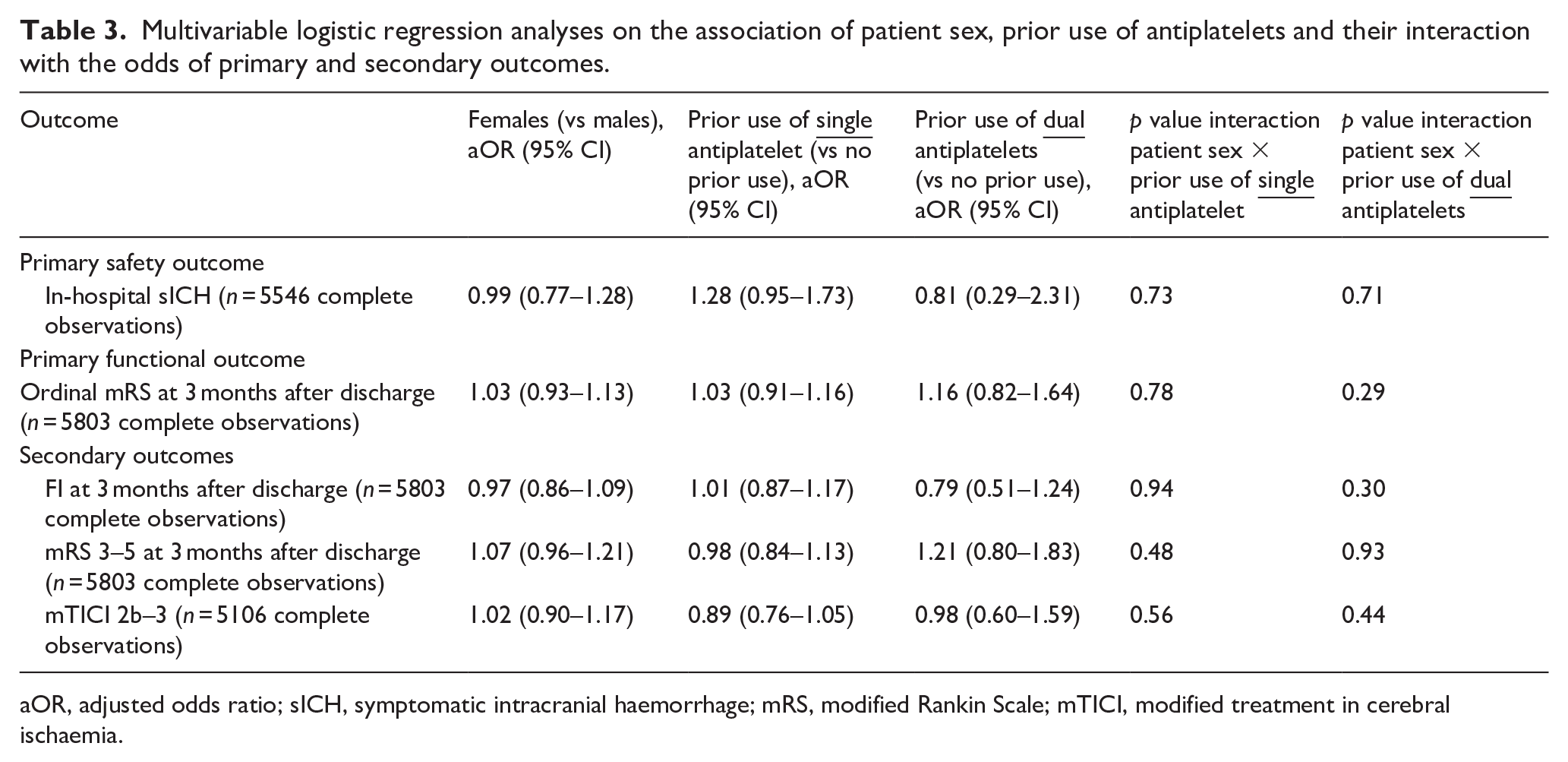
aOR, adjusted odds ratio; sICH, symptomatic intracranial haemorrhage; mRS, modified Rankin Scale; mTICI, modified treatment in cerebral ischaemia.
Compared to males, females showed a worse functional outcome after MT 3 months after discharge across all levels of disability and mortality (ordinal mRS score, p < 0.0001, Table 2). However, the multivariable logistic regression analysis showed that there were not sex differences in the odds of higher mRS scores (aOR 1.03, 95% CI 0.93–1.13, p = 0.61) and no association was observed between prior use of antiplatelets and the odds of a higher mRS score, either as single (aOR 1.03, 95% CI 0.91–1.16, p = 0.68) or dual therapy (aOR 1.16, 95% CI 0.82–1.64, p = 0.40), compared with no antiplatelet use prior to stroke onset (Table 3). No interaction was found between patient sex and prior use of antiplatelets regarding the primary functional outcome (Table 3).
Comparable results were found for sICH and ordinal mRS at 3 months after discharge in multilevel regression logistic models with participating stroke centres as random effect (Supplemental Table 2).
Secondary outcomes
In terms of functional recovery after MT 3 months after discharge, females were less likely to be functionally independent than males (45.2% vs 52.1%, p < 0.0001) and more frequently had mRS scores of 3, 4 or 5 (32.8% vs 29.1%, p = 0.001). Nevertheless, in multivariable logistic regression analyses, patient sex and prior use of antiplatelets (as single or dual therapy) were not associated with the secondary functional outcomes evaluated, either independently or in interaction (Table 3).
The proportion of patients achieving successful reperfusion after MT was similar for females and males (52.8% vs 52.1%, p = 0.728). Patient sex and prior use of antiplatelets were not associated with the odds of mTICI 2b–3, with no interaction between the two variables either (Table 3).
Subgroup analyses by type of intervention: direct MT or bridging
Table 4 summarizes the results of logistic regression analyses in patient subgroups defined according to the type of intervention received (direct MT or bridging). Neither biological sex, nor prior use of antiplatelets (single or dual) nor their interaction had an impact on the odds of sICH after direct MT. In the bridging subgroup, prior use of antiplatelets as single therapy was associated with increased odds of sICH (aOR 1.50, 95% CI 1.01–2.23, p = 0.04), without sex differences (p interaction = 0.45).
Logistic regression analyses by patient subgroups based on the type of intervention (direct mechanical thrombectomy or bridging) assessing the association of patient sex, prior use of antiplatelets and their interaction with the odds of primary and secondary outcomes.
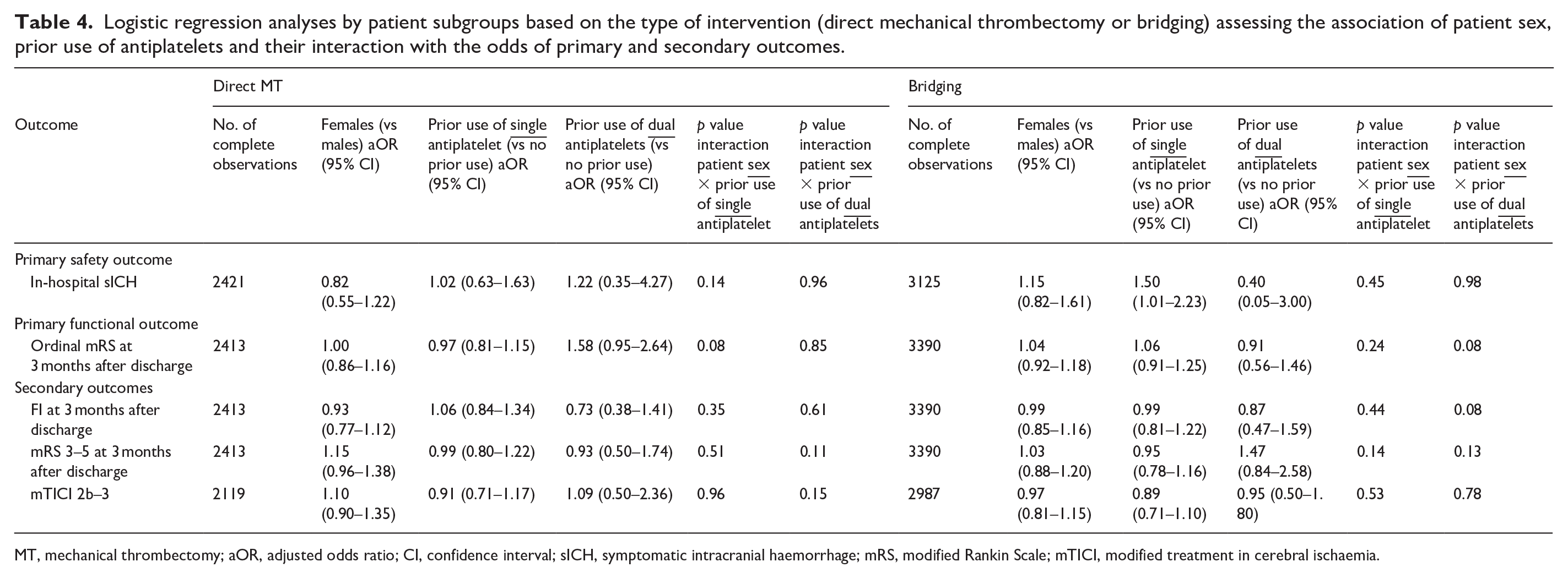
MT, mechanical thrombectomy; aOR, adjusted odds ratio; CI, confidence interval; sICH, symptomatic intracranial haemorrhage; mRS, modified Rankin Scale; mTICI, modified treatment in cerebral ischaemia.
No sex differences were found in the odds of higher mRS scores at 3 months after discharge regardless of the type of intervention received. In both intervention groups, prior use of antiplatelets (single or dual) had no impact on the odds of having a higher mRS score at 3 months after discharge. No associations were found between patient sex, prior use of antiplatelets and their interaction regarding the odds of all secondary outcomes evaluated. Supplemental Table 3 shows the raw data related to subgroup analyses. Sensitivity analyses in cardioembolic stroke patients treated with bridging therapy confirmed that prior use of a single antiplatelet agent was associated with increased odds of in-hospital sICH, regardless of patient sex (aOR 1.98, 95% CI 1.07–3.68, p = 0.03, Table 5). In patients with large-artery atherosclerosis as the stroke aetiology, female patients with prior use of a single antiplatelet agent who were treated with bridging therapy had higher odds of in-hospital sICH (p interaction = 0.04, Table 5).
Sensitivity subgroup analyses in large-artery atherosclerosis and cardioembolic stroke: impact of patient sex, prior use of antiplatelets and their interaction on safety and primary functional outcomes of direct mechanical thrombectomy and bridging therapy. .
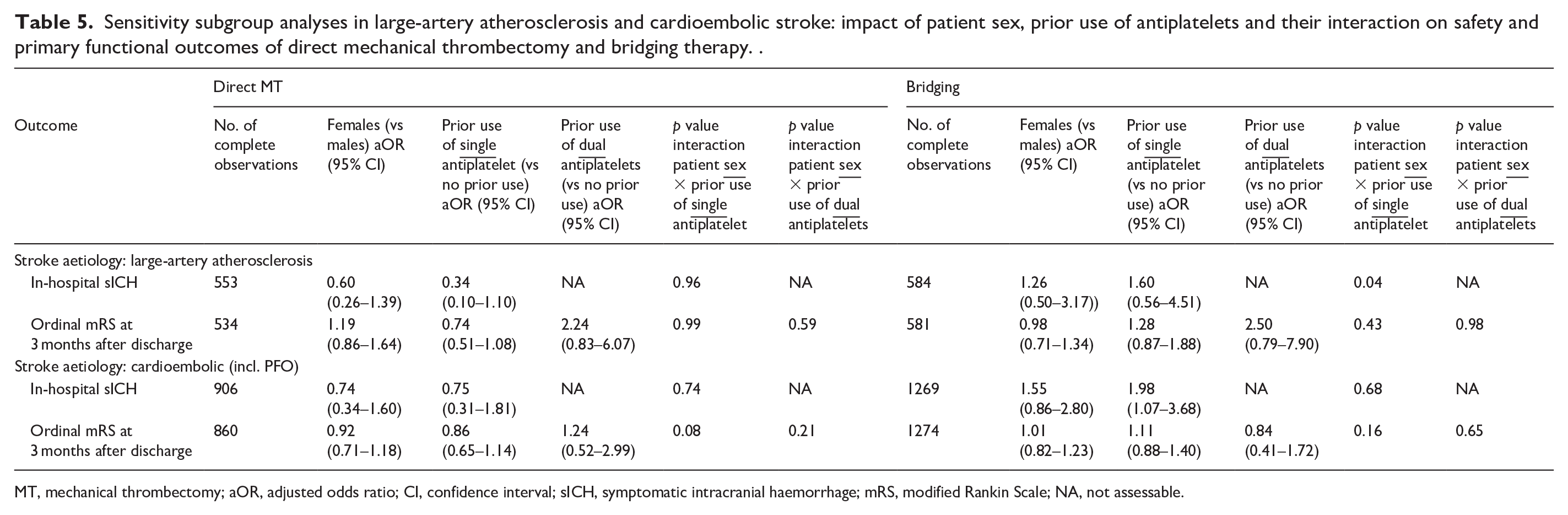
MT, mechanical thrombectomy; aOR, adjusted odds ratio; CI, confidence interval; sICH, symptomatic intracranial haemorrhage; mRS, modified Rankin Scale; NA, not assessable.
Discussion
This study of the large multicentre international EVA-TRISP registry showed no sex differences in safety and functional outcomes after MT (bridging or direct) alongside prior use of antiplatelets. Moreover, in the subgroup of patients receiving bridging therapy, prior use of single antiplatelet therapy did increase the odds of sICH independent of patient sex, however, without effect on functional outcomes at 3 months after discharge.
Concerning sex differences, the baseline characteristics of the study population mirror those described for AIS patients treated with MT from other large registries.12,13 Females were older, more often had hypertension and atrial fibrillation and were more likely to have greater pre-stroke disability. Males more often showed coronary heart disease and were more likely to be taking antiplatelets prior to stroke onset, albeit in both females and males the proportion of patients with pre-stroke antiplatelets did not exceed 40%, consistent with the data reported in the literature. 25 Moreover, we observed that females were more likely to receive direct MT than bridging therapy compared to males. A recent systematic review and meta-analysis that analysed both RCT and non-RCT studies (including hospital-, registry-, and administrative-based data) reported no sex differences in MT utilization following AIS. However, it did not distinguish between direct MT and bridging. 26
sICH following MT: the effects of patient sex, prior use of antiplatelets and their interaction
The proportion of sICH events within the EVA-TRISP population was around 5%, in agreement with previous findings from other registries of AIS patients treated with MT.15 –17 When patient sex was analysed as an independent variable, no effect was observed on the occurrence of sICH following MT in line with previous studies.8,11 –14 Similarly, regarding the association between prior use of antiplatelets and compromised patient safety due to sICH following MT, our analysis in the EVA-TRISP registry is consistent with existing literature, showing no differences between AIS patients with or without pre-stroke antiplatelet use.19 –23 Moreover, our study makes a significant contribution to the existing literature by introducing a novel evaluation of the interaction between patient sex and prior antiplatelet use on the occurrence of sICH following MT. This analysis of the EVA-TRISP registry is the first to show that the safety of MT remains constant between females and males, irrespective of prior antiplatelet use.
Functional outcomes following MT: the effects of patient sex, prior use of antiplatelets and their interaction
Despite the higher proportion of female patients with elevated mRS scores at 3 months after MT, we found no sex differences after multivariable analysis, in which the association between patient sex and 3-month mRS score was adjusted for other clinically relevant variables. Our results are in line with most existing experimental and observational studies reporting that patient sex does not apparently affect functional outcomes after MT,9,10,12,14 –17 in contrast with other studies that observed dissimilar functional results between females and males at 3 months after MT.11,13,18 Noteworthy, in the EVA-TRISP registry, similar functional recovery at 3 months after MT for females and males agreed with comparable reperfusion rates between the sexes. Concerning the association between prior use of antiplatelets and functional outcomes of MT, available evidence is heterogeneous and conflicting. Confirming previous studies,19,21,22 we found that prior use of antiplatelets per se was not associated with changes across all levels of disability and mortality as measured by 3-month mRS shift analysis. On the other hand, differences in functional outcomes after MT depending on whether antiplatelets were used prior to stroke onset have been reported in opposite ways by a national multicentre study conducted in China and a study involving two medical centres in the USA. While the former found that prior use of antiplatelets had a positive effect on functional recovery at 3 months after MT, 20 the latter associated prior use of antiplatelets with a worse functional outcome, reflected by mRS scores of 3–6. 23
As for the safety outcome of sICH, our study provides new insights into the effect of the interaction between prior use of antiplatelets and patient sex on functional outcome after MT, showing for the first time that the efficacy of MT intervention in females and males does not change with prior use or not of antiplatelets.
Bridging therapy versus direct MT: the effects of patient sex, prior use of antiplatelets and their interaction on safety and functional outcomes
Our subgroup analyses based on the type of intervention demonstrated that both bridging and direct MT are equally effective and safe in females and males. Moreover, we observed that among patients treated with bridging, both females and males with prior use of single antiplatelet therapy had higher odds of experiencing sICH events compared to those without prior use of antiplatelets, nevertheless with no impact on 3-month functional recovery. This was not the case for prior use of dual antiplatelet therapy, most likely due to the small sample size of this patient group. Interestingly, sensitivity subgroup analyses in patients with cardioembolic stroke confirmed that prior use of a single antiplatelet agent was associated with increased odds of in-hospital sICH regardless of patient sex. Conversely, among patients with large-artery atherosclerosis, a significant interaction between female sex and prior use of a single antiplatelet agent was observed in relation to higher odds of in-hospital sICH. Since antiplatelet therapy decreases platelet aggregation and inhibits thrombus formation, haemorrhagic complications in AIS patients with prior use of antiplatelets who received IVT before MT are challenging physiological effects that have been documented in previous studies and also without effect on the 3-month functional outcome. 27 Our findings fit into the ongoing debate on whether ‘to bridge or not to bridge’ before MT,28 –36 particularly in that bridging has potential advantages including complete or partial lysis of the thrombus that caused the occlusion of the large vessel, lysis of thrombotic emboli in distal vessels outside the reach of MT and more rapid resolution of cerebral ischaemia. 37 However, there are potential disadvantages to administering IVT first, including delaying the time to start the MT procedure, an increased risk of sICH, and partial lysis of the thrombus in the large vessels allowing it to spread to more distal vessels, outside the reach of MT. 38 The only RCT that evaluated the effects of a loading dose of antiplatelet therapy and/or prophylactic anticoagulation given intravenously in the acute setting before MT (with bridging in about 3/4 of patients), was prematurely stopped because of safety concerns: acute treatment with antiplatelet therapy and/or prophylactic anticoagulation was associated with higher risk of sICH, without apparent influence on functional outcome at 3 months. 39 Although our data do not support a differential approach in terms of IVT for female and male patients with prior antiplatelet therapy, the question is still open and further studies are needed to investigate the impact of prior use of antiplatelets on safety and functional outcomes of direct MT.
Strengths
Our study has several strengths. The broad geographic distribution of participating stroke centres across Europe provides a large and representative sample of AIS patients treated with MT in real-life conditions. This aspect also contributes to a second strength of our study: the heterogeneity of the study population, with baseline characteristics that were not strictly selected, as opposed to the more rigid eligibility criteria used in RCTs (e.g. patient age and comorbidities). Nevertheless, excluding patients on anticoagulant therapy prior to stroke onset from the study population helped to control for a potential bias arising from the association between anticoagulant therapy, bleeding risk, and possible impact on functional outcomes after MT. For the same reason, excluding patients with a high degree of pre-stroke disability allowed us to control for confounding due to the higher prevalence of pre-stroke disability in females compared to males. A third strength lies in the use of multilevel analyses with the stroke centres as a random effect. This method allowed us to account for differences between the various centres in the management of AIS patients and to confirm that the stroke centre itself had no effect on safety and functional outcome assessment in MT according to patient sex and prior use of antiplatelets. Further strengths of the study include the design based on patient stratification by sex, the analytical approach using multivariable regression models adjusted for a range of clinically relevant variables potentially associated with the assessed safety and functional MT outcomes and the addition of an interaction term between patient sex and prior use of antiplatelets. Because of these considerations, to the best of our knowledge, this study provides new, sex-specific evidence on the association between prior use of antiplatelets and MT outcomes.
Limitations
We should also consider the limitations when interpreting the findings of this study. First, we acknowledge the inherent constraints of observational research using patient registries, including the retrospective nature of the analysis and the extent of missing data, resulting in performing all analyses on a subset of complete observations. In particular, the unexpectedly low rates of successful reperfusion observed in the study population may be partly explained by the high proportion of missing data for this variable in the EVA-TRISP registry (up to 30% in both female and male groups). Regarding prior use of antiplatelets, we did not include the duration of treatment and patient adherence, as this information is not collected in the EVA-TRISP registry. Moreover, the relatively small sample size of patients with prior use of antiplatelets, particularly in the dual regimen, limited the study’s power to detect sex differences in safety and functional outcomes following MT. As such, our findings should be interpreted with caution, and further validation in larger cohorts is needed. Another limitation of our study is that it focussed on sex differences without considering gender-related differences encompassing socio-cultural factors. 40 Indeed, the lack of gender-related information in the EVA-TRISP database, such as social support networks, marital status, type of occupation and level of education, precluded the possibility of adopting an intersectional approach that might have contributed to a more comprehensive assessment of the research question.
Conclusion
Based on a large and heterogeneous population from stroke centres across Europe, our results provide evidence that any plausible differences between females and males in the use of antiplatelets prior to stroke onset based on known biological differences in platelet reactivity between the sexes have no impact on safety and functional outcomes of MT. While evaluating sex differences in the efficacy and safety of pre-stroke antiplatelet therapy per se was outside the scope of this study, and such data were not available within the EVA-TRISP database, our findings are consistent with current clinical practice guidelines that do not support sex-specific indications or contraindications for antiplatelet therapy in the context of primary or secondary prevention.41,42 In this study, the absence of a significant interaction between patient sex and prior use of antiplatelets suggests that treatment decisions by practicing physicians should remain unchanged regardless of sex, meaning that MT should be considered equally in both females and males, irrespective of prior antiplatelet use. A broader complementary evaluation including gender differences would be warranted to develop personalized treatment strategies aimed to maximize individual outcomes of MT intervention.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251346016 – Supplemental material for Sex differences in outcomes after endovascular therapy with prior antiplatelet use: A study from the EVA-TRISP registry
Supplemental material, sj-docx-1-eso-10.1177_23969873251346016 for Sex differences in outcomes after endovascular therapy with prior antiplatelet use: A study from the EVA-TRISP registry by Roberta Noseda, Federico Rea, Giovanni Bianco, Elias Auer, Mirjam R Heldner, Eike I Piechowiak, Tomas Dobrocky, Patrik Michel, Susanne Wegener, Hakim Baazaoui, Philipp Baumgartner, Sami Curtze, Nicolas Martinez-Majander, Miranda Nybondas, Andrea Zini, Stefano Forlivesi, Matteo Paolucci, Gian Marco De Marchis, Tolga Dittrich, Paul J Nederkoorn, Nabila Wali, Mira Katan, Stefan Engelter, Henrik Gensicke, Simon Trüssel, Alessandro Pezzini, Mauro Magoni, João Pedro Marto, Maria Brás Monteiro, Ronen R Leker, Tamer Jubeh, Visnja Padjen and Carlo W Cereda in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873251346016 – Supplemental material for Sex differences in outcomes after endovascular therapy with prior antiplatelet use: A study from the EVA-TRISP registry
Supplemental material, sj-docx-2-eso-10.1177_23969873251346016 for Sex differences in outcomes after endovascular therapy with prior antiplatelet use: A study from the EVA-TRISP registry by Roberta Noseda, Federico Rea, Giovanni Bianco, Elias Auer, Mirjam R Heldner, Eike I Piechowiak, Tomas Dobrocky, Patrik Michel, Susanne Wegener, Hakim Baazaoui, Philipp Baumgartner, Sami Curtze, Nicolas Martinez-Majander, Miranda Nybondas, Andrea Zini, Stefano Forlivesi, Matteo Paolucci, Gian Marco De Marchis, Tolga Dittrich, Paul J Nederkoorn, Nabila Wali, Mira Katan, Stefan Engelter, Henrik Gensicke, Simon Trüssel, Alessandro Pezzini, Mauro Magoni, João Pedro Marto, Maria Brás Monteiro, Ronen R Leker, Tamer Jubeh, Visnja Padjen and Carlo W Cereda in European Stroke Journal
Supplemental Material
sj-docx-3-eso-10.1177_23969873251346016 – Supplemental material for Sex differences in outcomes after endovascular therapy with prior antiplatelet use: A study from the EVA-TRISP registry
Supplemental material, sj-docx-3-eso-10.1177_23969873251346016 for Sex differences in outcomes after endovascular therapy with prior antiplatelet use: A study from the EVA-TRISP registry by Roberta Noseda, Federico Rea, Giovanni Bianco, Elias Auer, Mirjam R Heldner, Eike I Piechowiak, Tomas Dobrocky, Patrik Michel, Susanne Wegener, Hakim Baazaoui, Philipp Baumgartner, Sami Curtze, Nicolas Martinez-Majander, Miranda Nybondas, Andrea Zini, Stefano Forlivesi, Matteo Paolucci, Gian Marco De Marchis, Tolga Dittrich, Paul J Nederkoorn, Nabila Wali, Mira Katan, Stefan Engelter, Henrik Gensicke, Simon Trüssel, Alessandro Pezzini, Mauro Magoni, João Pedro Marto, Maria Brás Monteiro, Ronen R Leker, Tamer Jubeh, Visnja Padjen and Carlo W Cereda in European Stroke Journal
Footnotes
Acknowledgements
Data used in this article were obtained from the EVA-TRISP registry. We express our gratitude to the patients, families and clinical and scientific staff for their cooperation throughout the course of this study.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SW received research funds by the Swiss National Science Foundation, the UZH Clinical research priority programme (CRPP) stroke, the Zurich Neuroscience Centre (ZNZ), the Baugarten foundation, the Hartmann Müller Foundation, the Koetser Foundation, the Swiss Heart Foundation; and speaker honoraria from Amgen, Springer, Advisis AG, Teva Pharma, Boehringer Ingelheim, Lundbeck, Astra Zeneca, FoMF and a consultancy fee from Bayer and Novartis via institution for research – all not related to this study.
AZ received consulting and speaker fees from Angels Initiative, Boehringer-Ingelheim, Alexion, Daiichi Sankyo, Pfizer, PIAM, fees for Advisory Board from Daiichi Sankyo, Bayer and Astra Zeneca, not related to this study.
STE received funding for travel or speaker honoraria from Bayer, Boehringer Ingelheim and Daiichi-Sankyo. He has served on scientific advisory boards for Bayer, Boehringer Ingelheim, BMS/Pfizer and MindMaze and on the editorial board of Stroke. His institutions have received an educational grant from Pfizer, compensation from Stago for educational efforts and research support from Daiichi-Sankyo, the Science Funds [Wissenschaftsfonds] of the University Hospital Basel, the University Basel, from the ‘Wissenschaftsfonds Rehabilitation’ of the University Hospital for Geriatric Medicine Felix Platter, the ‘Freiwillige Akademische Gesellschaft Basel’, the Swiss Heart Foundation and the Swiss National Science Foundation.
HG received research support from the Swiss National Science Foundation, AbbVie, Merz and Ipsen, and advisory board honoraria from Daiichi Sankyo and AbbVie and funding for travel from BMS/Pfizer.
RRL granted from EU Horizon 2022, the Pertiz and Chantal Scheinberg Cerebrovascular Fund and received speaker honoraria from IscemaView, Boehringer Ingelheim, Pfizer, Biogen, Medtronic and Abott and advisory board honoraria from Bayer and Filterlex.
VP received travel or speaker honoraria from Boehringer Ingelheim; honoraria from scientific advisory board from Medtronic, outside the submitted work.
CWC is a member of the iSchema View Medical and Scientific Advisory Board.
All other authors declare no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The registry and the present study were both approved by the responsible ethics committee (Comitato etico cantonale, Ticino, Req-2023-01533).
Informed consent
Informed consent was obtained unless it was waived by the relevant ethics committees at the participating stroke centres.
Consent for publication
Not applicable.
Guarantor
Dr. Carlo Walter Cereda.
Contributorship
R.N. and C.W.C. conceived the study. Data collection was performed by G.B., E.A., M.R.H., E.I.P., T.D., P.M., S.W., H.B., P.B., S.C., N.M.-M., M.N., A.Z., S.F., M.P., G.M.D.M., T.D., P.J.N., N.W., M.K., S.E., H.G., S.T., A.P., M.M., J.P.M., M.B.M., R.R.L., T.J., V.P. and C.W.C. R.N. and F.R. performed the data analysis. R.N., F.R. and C.W.C. contributed to data interpretation. R.N. drafted the manuscript. All authors critically revised the manuscript and approved the final version.
ORCID iDs
Data availability
The data supporting this study are available from the Corresponding Author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
