Abstract
Introduction
Cystatin C has been suggested as a sensitive marker of renal function. A high level of cystatin C is related to cardiovascular disease and stroke in elderly patients. We investigated the relationship between levels of cystatin C and early neurological deterioration with acute ischaemic stroke in elderly patients without chronic kidney disease.
Patients and methods
We evaluated a total of 771 elderly patients (mean age, 72.2; male, 59.0%) without chronic kidney disease who were admitted following acute ischaemic stroke between March 2010 and January 2015. The patients were divided into four groups based on the quartiles of serum cystatin C values. Early neurological deterioration was defined as an increase of ≥2 points from the baseline National Institutes of Health Stroke Scale score during the 7 days following onset. We compared the clinical characteristics and cystatin C concentrations between patients with and without early neurological deterioration.
Results
Eighty-six patients (11.2%) experienced early neurological deterioration. The percentage values of the higher (third and fourth) quartiles were significantly higher in the early neurological deterioration group (30.2% vs. 24.4% and 34.9% vs. 23.8%,
Discussion and conclusions
This study demonstrated that cystatin C concentrations in elderly patients without chronic kidney disease were associated with early neurological deterioration following acute stroke. This suggests that cystatin C level could be a useful predictor for early neurological deterioration following acute stroke.
Introduction
Cystatin C is a cysteine protease inhibitor produced in all nucleated cells at a constant rate. 1 Cystatin C has been suggested as a sensitive marker of renal function than the creatinine-based equation, because its serum concentrations are independent of muscle mass and do not seem to be affected by age.1–4 It might be also associated with declining kidney function, systemic inflammation and metabolic syndrome.3–6 A high level of cystatin C has been reported to be positively associated with the increased risk of stroke, cardiovascular morbidity and mortality, with renal dysfunction appearing to be the most plausible mechanism related to inflammation, atherosclerosis and vascular risk factors.4–6 Moreover, and paradoxically, a few studies reported that higher cystatin C concentration is more strongly associated with cardiovascular disease, stroke and mortality among elderly persons with normal renal function.4,5,7,8
Early neurological deterioration (END) is classified as substantial neurological deterioration that occurs following an acute ischaemic stroke. END occurs in approximately 13–38% of patients with acute ischaemic stroke.9,10 Although previous studies have reported radiological, biological and clinical factors (including age) that can predict END, many of these factors are not useful because of the difficulties inherent in identifying reliable predictors and their underlying mechanisms.9–11
However, the predictive value of cystatin C for END following ischaemic stroke among elderly patients with normal renal function has not been elucidated. We hypothesized that cystatin C could be an important predictor for the risk of END in elderly patients with normal renal function.
Methods
Study population
We initially screened 1537 patients with acute ischaemic stroke and transient ischaemic attack (TIA), who were admitted within the first 7 days following the onset of stroke. They were registered into our prospective stroke registry system within 7 days of the onset of symptoms at Seoul National University Hospital between March 2010 and January 2015. We excluded patients based on the following criteria: chronic kidney disease (CKD; n = 259), lack of cystatin C data (n = 91), lack of clinical data (n = 42), age younger than 60 years (n = 304) and TIA (n = 70). Eventually, 771 patients (age range, 60–97 years) with acute ischaemic stroke were enrolled. The patients were divided into four groups based on the quartiles of serum cystatin C level. The study was approved by the Institutional Review Board of Seoul National University Hospital (IRB NO H-1009-062-332).
Baseline and clinical assessment
Baseline characteristics were recorded, including demographic data (age and sex), and conventional vascular risk factors (hypertension, diabetes mellitus, hyperlipidaemia, a history of smoking (current or past regular smoking), body mass index (BMI), initial body temperature and a history of stroke/TIA). Several laboratory findings, such as white blood cell (WBC) count, initial fasting blood sugar (FBS), haemoglobin (Hb), triglycerides, total cholesterol (TC), high-density lipoprotein (HDL) cholesterol, fibrinogen, C-reactive protein and microalbuminuria, were also evaluated using routine laboratory methods. Blood samples were drawn after overnight fasting. Serum cystatin C was measured from blood samples using a BN II nephelometer (Siemens Healthcare Diagnostics, Inc.) with a particle-enhanced immunonephelometric assay (N Latex cystatin C, Siemens Healthcare Diagnostics, Inc.). The estimated glomerular filtration rate (eGFR; expressed in mL/min per 1.73 m2) was estimated using the four-variable Modification of Diet in Renal Disease (MDRD) formula, as follows: eGFR = 175 × (serum creatinine−1.154) × (age−0.203) × (1.212 if black) × (0.742 if female). 12
Hypertension was defined as a history of antihypertensive treatment, a systolic blood pressure ≥140 mmHg, or a diastolic blood pressure ≥90 mmHg. 13 Hyperlipidaemia was defined as a history of lipid-lowering medication, a serum TC level >240 mg/dL or a serum level of low-density lipoprotein (LDL) cholesterol >160 mg/dL. 14 Diabetes mellitus was defined as HbA1c ≥6.5%, fasting blood glucose ≥7.0 mmol/L, non-fasting blood glucose ≥11.1 mmol/L, or the use of insulin or oral hypoglycaemic drugs. 15
Ischaemic stroke was categorized as large-artery atherosclerosis, small-vessel occlusion, cardioembolism, other determined and undetermined, based on the Trial of Org 10172 in Acute Stroke Treatment criteria.16,17 The ischaemic lesion locations were divided into the anterior circulation (anterior cerebral artery and/or middle cerebral artery territories), posterior circulation (posterior cerebral artery and/or vertebrobasilar artery territories) and multiple artery territory. Moreover, we collected data regarding infections such as pneumonia, urinary tract infection, etc., occurring in the first week after stroke onset. On admission, we investigated the stroke onset time for all patients, and all the patients were initially graded for stroke severity based on the National Institutes of Health Stroke Scale (NIHSS). NIHSS and neurological evaluation were determined every 4 h in the stroke unit and at least 3 times daily in the general ward by specialized nurses or stroke neurologists. Whenever END was suspected by the patients, caregivers, or nursing staff, NIHSS was evaluated in detail by neurologists. The timing of END was identified by a review of the medical records. Considering the registration requirement of our stroke registry system, END was defined as an increase in NIHSS score by ≥2 points from baseline during the first 7 days following symptom onset. 18
Imaging information
The patients underwent brain MRI on a 1.5 T or 3.0 T superconducting magnet systems within 24 h after admission. We evaluated significant stenosis, which was defined as ≥50% stenosis or occlusion of the corresponding artery. 19 In addition, to evaluate the change in the infarct lesion, follow-up brain MRI or brain CT studies were performed after END (n = 71, 82.6%). We evaluated the follow-up images by dividing the results into five categories: no interval change, enlarged lesion, new lesion in new vascular territory, oedematous change (a swelled initial lesion or presenting oedematous change on brain CT or FLAIR image) and haemorrhagic transformation. 20 Two trained neurologists (T.J.K. and S.J.A.), blinded to the clinical information, assessed the degree of stenosis and the change in the infarct lesion. Disagreement was resolved by discussion and reassessment.
Statistical analysis
Continuous variables and proportions of categorical variables were compared using Student’s
Results
Baseline characteristics
Baseline characteristics by quartiles of cystatin C.
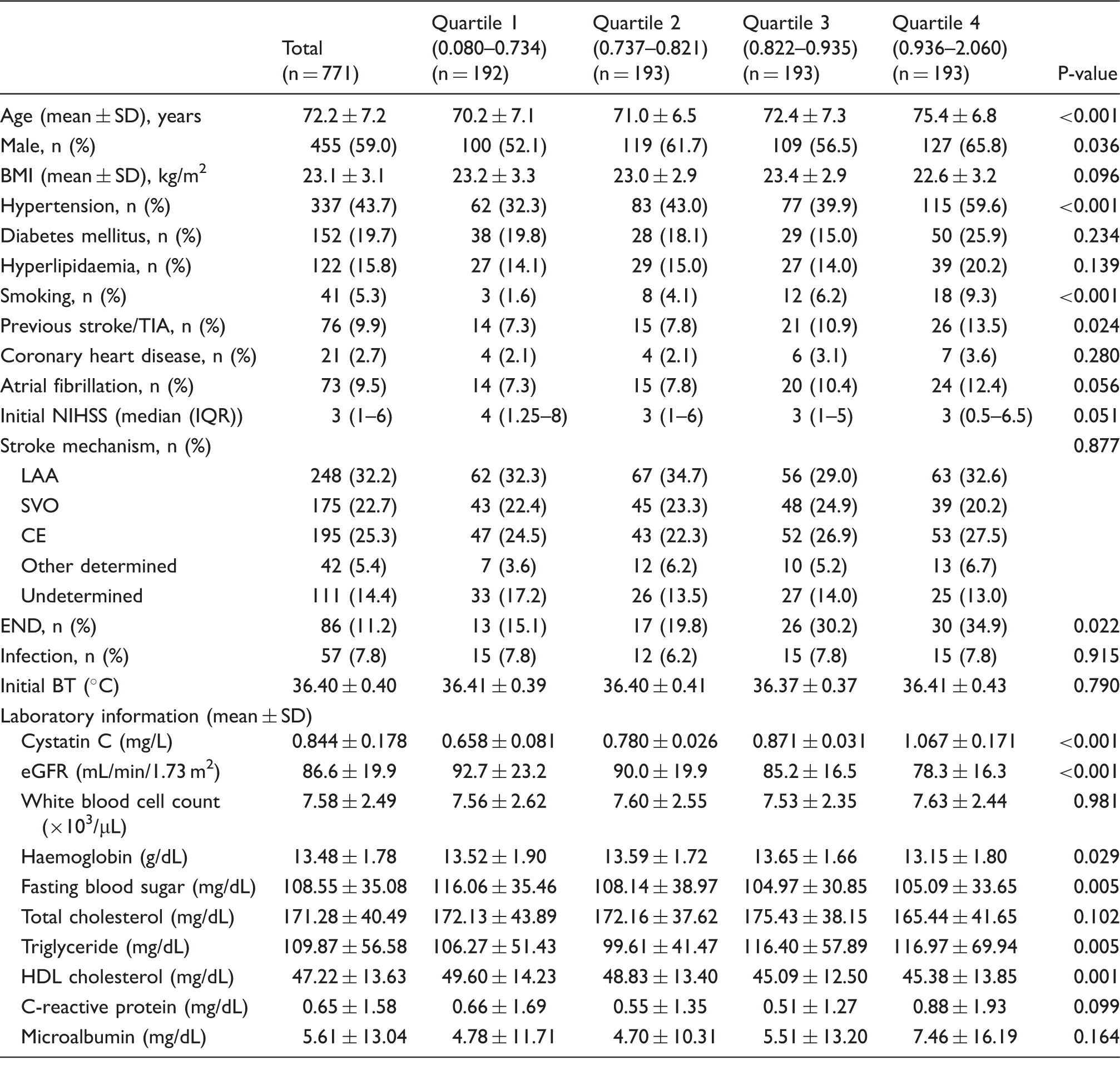
BMI: body mass index; SD: standard deviation; TIA: transient ischaemic stack; LAA: large artery atherosclerosis; SVO: small-vessel occlusion; CE: cardioembolism; END: early neurological deterioration; eGFR: estimated glomerular filtration rate; HDL: high density lipid.
Patients profile according to early neurological deterioration.
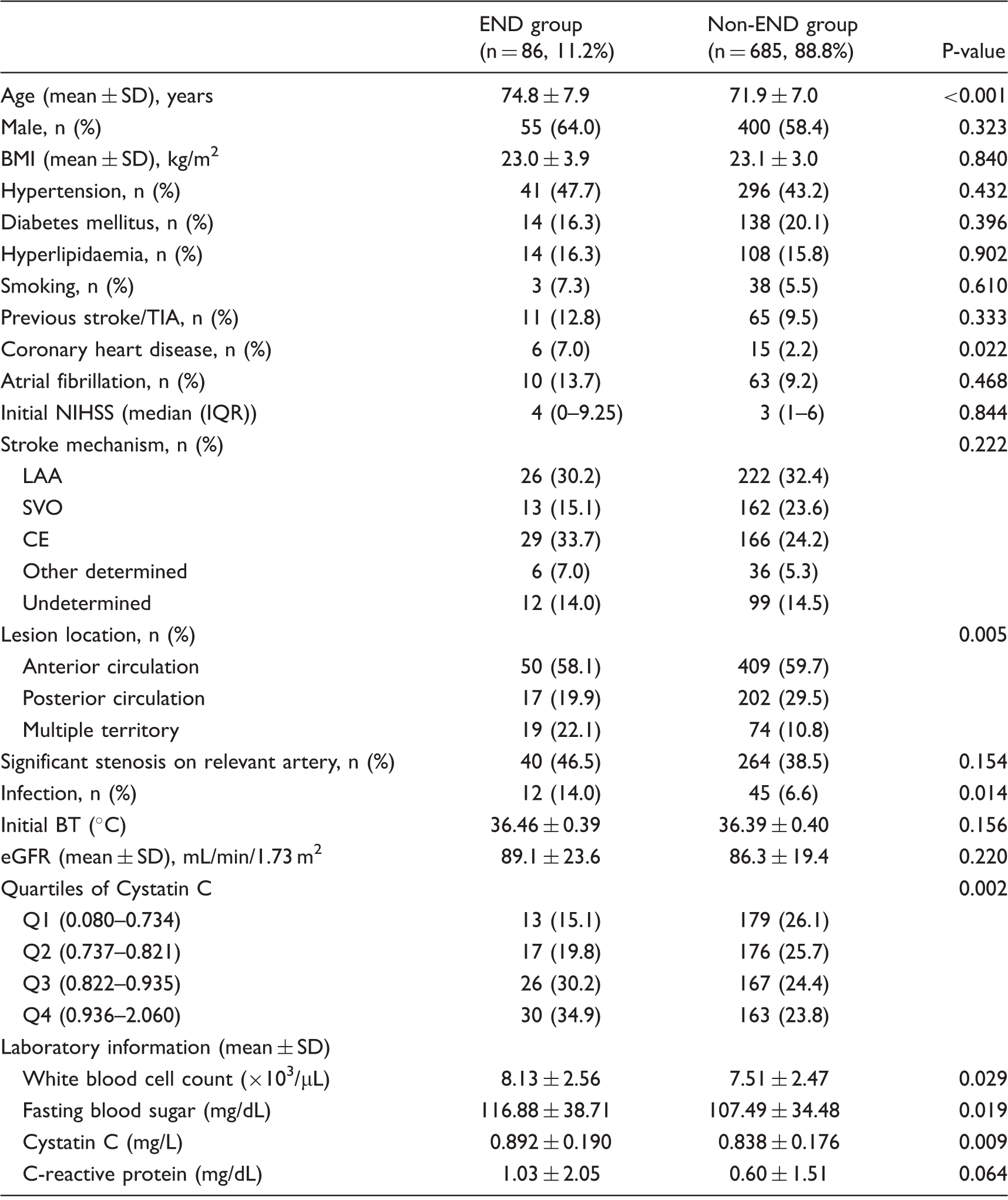
END: early neurological deterioration; BMI: body mass index; TIA: transient ischaemic stack; LAA: large artery atherosclerosis; SVO: small-vessel occlusion; CE: cardioembolism; BT: body temperature; eGFR: estimated glomerular filtration rate.
Among the 86 patients with END, 71 (82.6%) patients underwent follow-up brain CT or brain MRI to identify the possible causes. About one third of patients (35.2%, data not shown) with END had a ‘no interval change’ pattern, while the rate of new lesion occurrence in a new vascular territory was 2.8%. Oedematous change and haemorrhagic transformation were observed in 28.2% of the patients.
The correlation between END and cystatin C
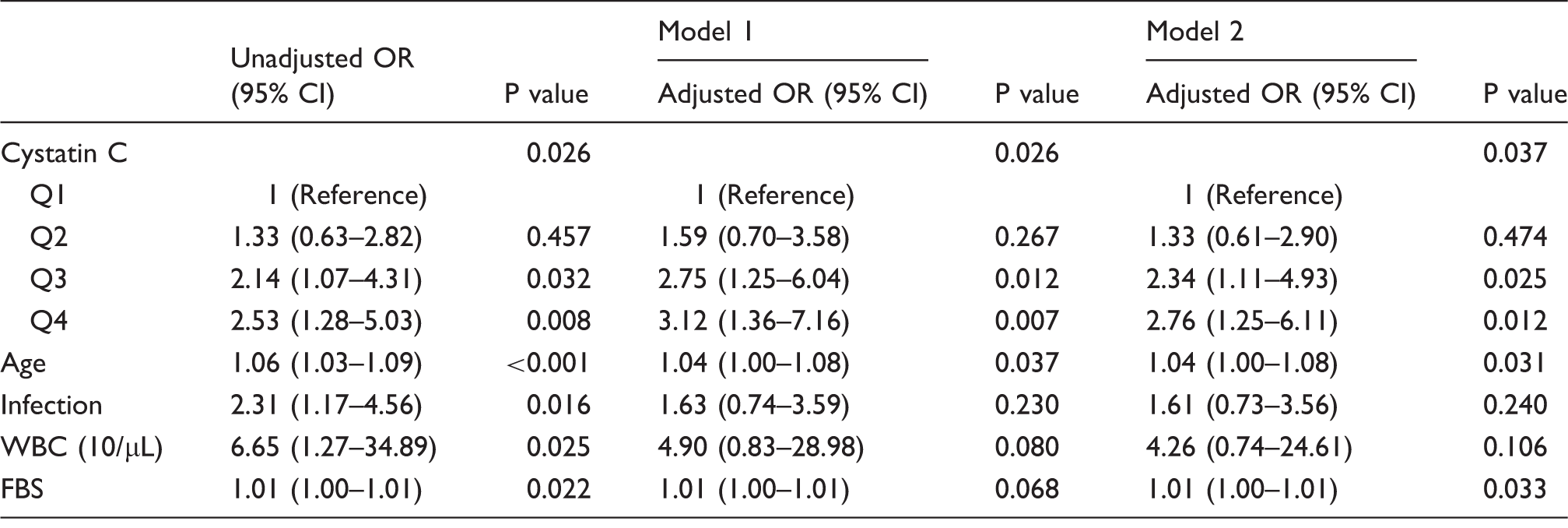
The proportion of subjects with an elevated cystatin C level was significantly higher in the END group than in the non-END group ( Association between levels of cystatin C and the risk of early neurological deterioration (END). The risk of END is increased when the serum level of cystatin C increases. Log odds (solid black line) and 95% confidence intervals (dashed outer bands). Multivariate analysis according to early neurological deterioration. CHD: coronary heart disease; GFR: glomerular filtration rate; WBC: white blood cell; FBS: fasting blood sugar. Model 1: Adjusting for cystatin C, age, sex, hypertension, smoking, CHD, previous stroke/TIA, lesion location, GFR, FBS, WBC, haemoglobin, HDL, TG, infections, significant stenosis. Model 2: Adjusting for cystatin C, age, sex, hypertension, smoking, CHD, previous stroke/TIA, lesion location, FBS, WBC, haemoglobin, HDL, TG, infection, significant stenosis.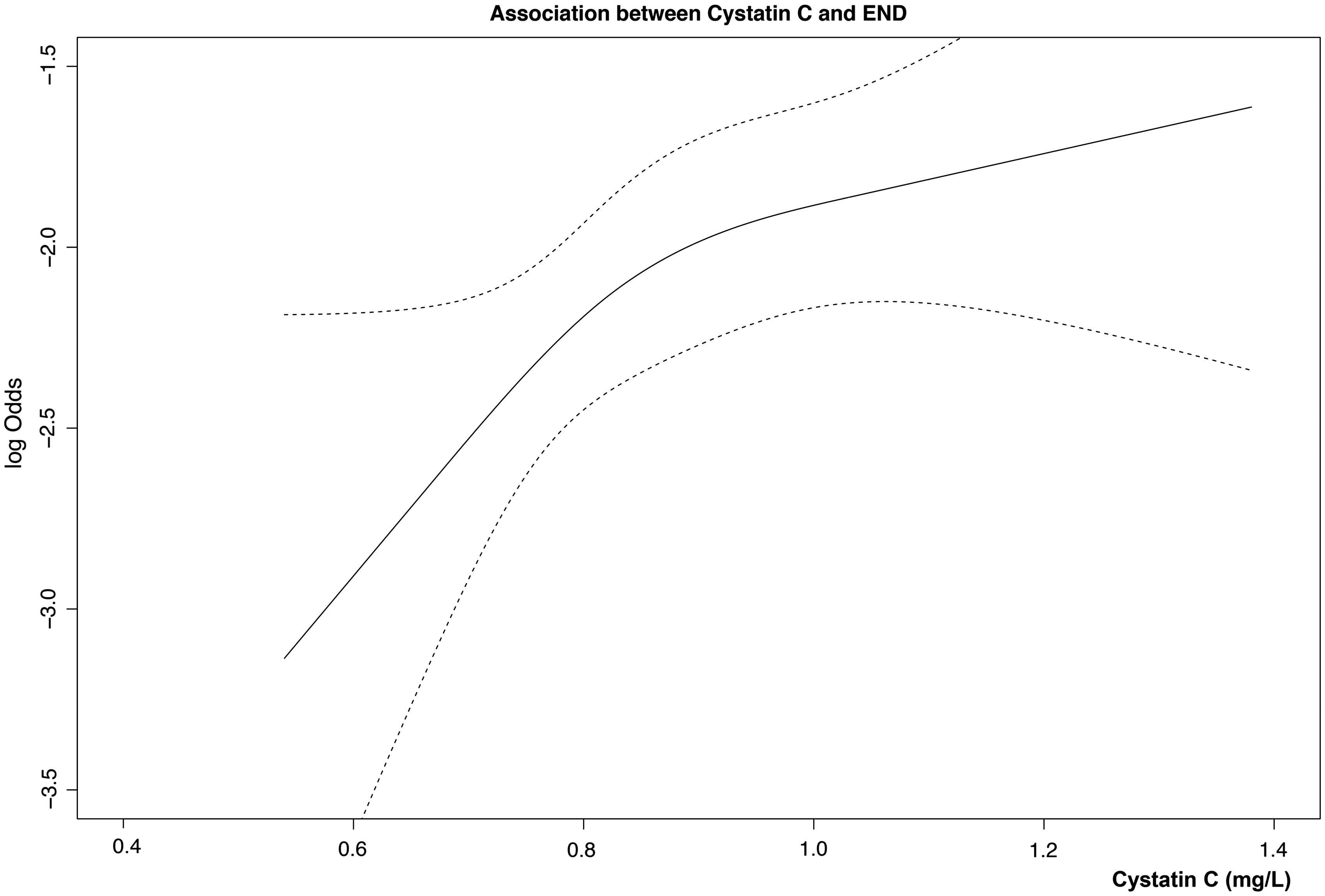
Discussion
In this study, we found that a higher level of cystatin C was an independently significant predictor for END in elderly patients with normal renal function, and that an increased cystatin C level was associated with an approximately 3 times higher risk for END. However, we found that eGFR was not associated with END.
Several factors influence the development of END, such as the severity of the initial stroke, large vessel occlusion, blood glucose, seizure, inflammation, medical complications such as infection, age and low eGFR.9–11,21–23 In line with these observations, our study showed that patients with END tended to be older and higher levels of WBC, and more frequent infection events, and the FBS level was positively correlated with END. However, after adjustment for confounding variables, END had an independent correlation only with age. Moreover, the initial NIHSS score, eGFR and microalbuminuria were similar in the patients with and without END. Higher quartile cystatin C (third and fourth quartiles) accounted for 30.2% and 34.9% of the patients with END compared with 24.4% and 23.8% of the patients with non-END, respectively. These results suggested that a higher level of cystatin C in elderly patients was a possible predictor for the development of END, even if it was a normal level.
Cystatin C is known to be expressed constantly by all nucleated cells. Within the normal range of kidney function, cystatin C has been proposed as a more reliable marker of renal function than serum creatinine and as an indicative marker of preclinical kidney disease.1,4,24,25 A higher level of serum cystatin C, which is a cysteine protease inhibitor, might reflect an imbalance in elastolytic activity. This imbalance between proteases and inhibitors affects the cardiovascular system.7,24,26 Thus, a higher cystatin C concentration is related to inflammation and endothelial dysfunction, and is involved in the pathogenesis of atherosclerosis.27–29 These factors might explain the link between microcirculation disorders and impaired vasodilation, which leads to END. Moreover, a high concentration of cystatin C has also been associated with a hypermetabolic status, and may reflect the duration and severity of other risk factors, such as hypertension and preclinical renal dysfunction.3,6,24,30 Likewise, our analysis showed that patients from groups corresponding to higher quartiles of cystatin C were older and more likely to evidence hypertension, while they had lower eGFR. These mechanisms could affect the process of vascular wall remodelling, directly or indirectly, and could influence the relation between the risk of stroke and other cardiovascular events.4,5,24 In addition, the inflammatory response promotes a local procoagulant state and a direct toxic effect that contribute to clinical neurological deterioration.28,29,31
There are some limitations to our study. First, we did not evaluate medical conditions, such as thyroid dysfunction and the use of glucocorticoid that could have induced an unexpectedly high cystatin C concentration. Second, we did not assess the volume of the infarction. However, the initial NIHSS was similar for the two groups; we therefore assumed that the volume of infarction was not different significantly.
32
Third, we included only elderly patients in our study; therefore, a selection bias was possible and our subject pool might not permit the generalization of our findings. However, cystatin C was not related to a risk of END in the excluded younger patients in our study (
In conclusion, our study found that a higher cystatin C level is associated with END in elderly patients without CKD. In this context, our data may suggest that the cystatin C level could be a useful biomarker for the prediction of END in elderly patients with preserved renal function. Further, large and well-designed prospective studies are required to confirm the true relationship between cystatin C and END using a more accurate method.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Korea Health Technology R&D project (HI10C2020) and by the Ministry of Health and Welfare, Republic of Korea (HI16C1078).
Ethical approval
The study was approved by the Institutional Review Board of Seoul National University Hospital (IRB NO H-1009-062-332).
Informed consent
Written informed consent was obtained from the enrolled patients.
Guarantor
B-WY
Contributorship
TJ Kim had full access to all data, performed statistical analyses, and took responsibility for data integrity and accuracy. S-B Ko and B-W Yoon conceptualized and designed the study. CK Kim, Y Kim, MK Kang, H-G Jeong, K-W Nam, H Mo and SJ An participated in data acquisition. B-W Yoon made intellectual contributions. TJ Kim and B-W Yoon interpreted results and wrote the manuscript.
References
 pro
pro pro
pro arg
arg