Abstract
Rationale:
Early neurological deterioration (END) within 72 h of stroke onset is associated with poor prognosis. Optimizing hydration might reduce the risk of END.
Aims:
This study aimed to determine in acute ischemic stroke patients if enhanced hydration versus standard hydration reduced the incidence of major (primary) and minor (secondary) END, as well as whether it increased the incidence of early neurological improvement (secondary), at 72 h after admission
Sample size estimate:
A total of 244 participants per arm.
Methods and design:
A prospective, double-blinded, multicenter, parallel-group, randomized controlled trial conducted at four hospitals from April 2014 to July 2020, with data analyzed in August 2020. The sample size estimated was 488 participants (244 per arm). Ischemic stroke patients with measurable neurological deficits of onset within 12 h of emergency department presentation and blood urea nitrogen/creatinine (BUN/Cr) ratio ⩾ 15 at point of admission were enrolled and randomized to 0.9% sodium chloride infusions of varying rates—enhanced hydration (20 mL/kg body weight, one-third given via bolus and remainder over 8 h) versus standard hydration (60 mL/h for 8 h), followed by maintenance infusion of 40–80 mL/h for the subsequent 64 h. The primary outcome measure was the incidence of major END at 72 h after admission, defined as an increase in National Institutes of Health Stroke Scale of ⩾ 4 points from baseline.
Results:
Overall, 487 participants were randomized (median age 67 years; 287 females). At 72 h, 7 (2.9%) in the enhanced hydration arm and 5 (2.0%) in the standard hydration developed major END (p = 0.54). The incidence of minor END and early neurological improvement did not differ between treatment arms.
Conclusion and relevance:
Enhanced hydration did not reduce END or improve short-term outcomes in acute ischemic stroke.
Trial Registration:
ClinicalTrials.gov (NCT02099383, https://clinicaltrials.gov/study/NCT02099383)
Keywords
Introduction and rationale
Early neurological deterioration (END) refers to the worsening of neurological symptoms after the initial acute ischemic stroke. Up to one-third of ischemic stroke patients develop END during their acute hospitalization. 1 Its onset within the first 48–72 h of cerebral infarction has a negative impact on prognosis and is associated with long-term neurological and functional decline.2,3
A large proportion of ischemic stroke patients (29–45%) is noted to be hypovolemic at hospital admission.4,5 While hypovolemia can increase the risk of cerebral infarction and lead to acute ischemic stroke, it may also be an independent predictor for the development of END within 72 h of ischemic stroke onset.5,6 Hypovolemia has also been observed to significantly increase the likelihood of death or dependent discharge. 7 Acute stroke guidelines recommend fluid replacement for hypovolemic patients, though they do not specify how rehydration therapy should be executed. 8
We previously established in a preliminary study that hydration therapy guided by blood urea nitrogen/creatinine (BUN/Cr) ratio may reduce the incidence of stroke-in-evolution and improve prognoses of acute ischemic stroke patients.
9
In this current study (hyd
Study aims
Primary objective
The primary objective of this study was to determine in acute ischemic stroke patients if enhanced hydration compared with standard hydration reduces the incidence of major END at 72 h after emergency department (ED) admission.
Secondary objectives
The secondary objectives were to determine in acute ischemic stroke patients if enhanced hydration compared with standard hydration (1) reduces the incidence of minor END at 72 h after ED admission; (2) increases incidence of early neurological improvement (ENI) at 72 h after ED admission; (3) reduces the length of hospital stay; and (4) reduces the incidence of disability in daily activities at 3 months after onset of acute ischemic stroke.
Methods
Design, population, and eligibility criteria
This study was a prospective, double-blinded, multicenter, parallel-group, randomized controlled trial conducted at four branch hospitals of Chang Gung Medical Foundation in line with the Consolidated Standards of reporting Trials (CONSORT) reporting guidelines (Supplemental Figure S1). All adult patients ⩾ 18 years with measurable neurological deficits of onset within 12 h of ED presentation and BUN/Cr ratio ⩾ 15 at point of ED admission were shortlisted for study enrollment.9,10 Participant recruitment strategy and the details of exclusion criteria are described in Supplementary Methods.
This study was reviewed and approved by the Institutional Review Board of Chang Gung Medical Foundation (IRB no. 102-0418A3) and registered in ClinicalTrials.gov (NCT02099383).
Evaluation of stroke
Ischemic stroke subtypes were classified according to the Trial of Org 10172 in Acute Stroke Treatment (TOAST) classification, while severity was evaluated using the National Institutes of Health Stroke Scale (NIHSS).11,12
Management of enrolled patients and study interventions
All enrolled participants underwent computed tomography scans of the brain within 2 h of ED arrival, with follow-up magnetic resonance imaging, if required for diagnostic confirmation, after randomization. Participants randomized to receive standard hydration were intravenously infused with 0.9% sodium chloride (0.9% NaCl) at a rate of 60 mL/h for the first 8 h, whereas those randomized to receive enhanced hydration were intravenously infused with 20 mL/kg body weight of 0.9% NaCl over the same timespan—one-third of this volume was given via bolus with the remainder delivered over 8 h. Thereafter, both groups continued to receive intravenous infusion of 0.9% NaCl at a rate of 40–80 mL/h for the next 64 h. All intravenous infusions were delivered with infusion pumps to ensure precise and accurate infusion rates as intended in accordance with the study protocol.
Randomization process
Enrolled participants were randomized in a 1:1 ratio to the enhanced hydration group and the standard hydration group (control), with stratification by stroke severity (NIHSS 1–5; 6–10; 11–15). Randomization codes were generated using SAS version 9.2 with block sizes of 4.
Blinding
Participants, treating physicians, study sponsor, and other trial staff remained blinded throughout the trial. The infusion pumps, which controlled the intravenous infusion rates, and the 0.9% NaCl bags were covered. After completion, the entire setup, including any leftover saline, was removed and disposed to prevent revealing the treatment allocation. Only the study nurses responsible for preparing and administering the interventions were unblinded.
To maintain consistency and reliability in NIHSS assessments, the evaluations were conducted at both baseline and 72 h by the same expert. This expert was thoroughly trained, certified, and blinded to the participants’ treatment allocations, ensuring methodological, unbiased, and accurate measurement of neurological outcomes.
Study outcomes
Primary outcome
The primary outcome of this study was the incidence of major END at 72 h after ED admission, defined as an increase in NIHSS of ⩾ 4 points from baseline.2,3
Secondary outcomes
The secondary outcomes were assessed at several time points of 72 h, 7 days, and 3 months after the onset of acute ischemic stroke. NIHSS scores were tabulated at 72 h after ED admission to evaluate the incidences of minor END and ENI, while modified Rankin Scale (mRS) and Barthel Index (BI) were scored at 7 days and 3 months after stroke onset to evaluate the incidence of disability in daily activities.13,14 The length of hospital stay was also noted for each patient.
Minor END was defined as an increase in NIHSS of 2 or 3 points from baseline. This conservative definition was used to identify patients with ongoing primary neurological damages earlier, who may benefit from further treatment and intervention. 13 Such a definition is also in keeping with previous studies which generally defined END as an increase in NIHSS of ⩾ 2 points during a 24-h period or hospitalization.12,15 –17 ENI was defined as a decrease in ⩾ 2 points from the baseline NIHSS score, and this correlates with improved 3-month functional outcomes in stroke patients with an initial NIHSS score of 1–15.18,19 In addition, disability in daily activities was defined as having an mRS of ⩾ 2 points.
Comparisons were made between both enhanced hydration and standard hydration groups, and between those with high risk for END (e.g. large vessel disease, high NIHSS at admission) and those without these risk factors.20 –22
Sample size
Sample size calculation was performed using two-sample proportion tests to achieve 80% power at 5% significance level (see Supplementary Methods for additional detail).
Statistical analysis
An intention-to-treat (ITT) analysis was conducted as the primary analysis. All study outcomes of both enhanced hydration and standard hydration groups were compared to evaluate the effectiveness of BUN/Cr-guided hydration therapy in acute ischemic stroke patients. Details of the statistical analysis and regression model are available in the Supplementary Method section.
Results
A total of 17,530 patients were screened against the eligibility criteria between April 2014 and July 2020, with 487 of them proceeding to randomization. Two protocol deviations were reported at this stage due to the use of wrong randomization codes, but the IRB determined that they had a minor impact and the affected participants were allowed to continue in the trial. As such, 242 participants were assigned to the enhanced hydration group and 245 to the standard hydration group. Out of these, 229 enhanced hydration and 239 standard hydration participants successfully completed the study intervention, with 180 enhanced hydration and 183 standard hydration participants included in the final per-protocol (PP) analysis (Supplemental Figure S1).
Baseline characteristics of study participants
Baseline demographics and clinical characteristics were similar in both groups (Table 1). Participants were mostly female (58.9%) with median age of 67 years. NIHSS scores on ED presentation were predominantly in the range of 1–7. Observations in the PP population were similar (Supplemental Table S1).
Summary of participant clinical profiles (ITT).
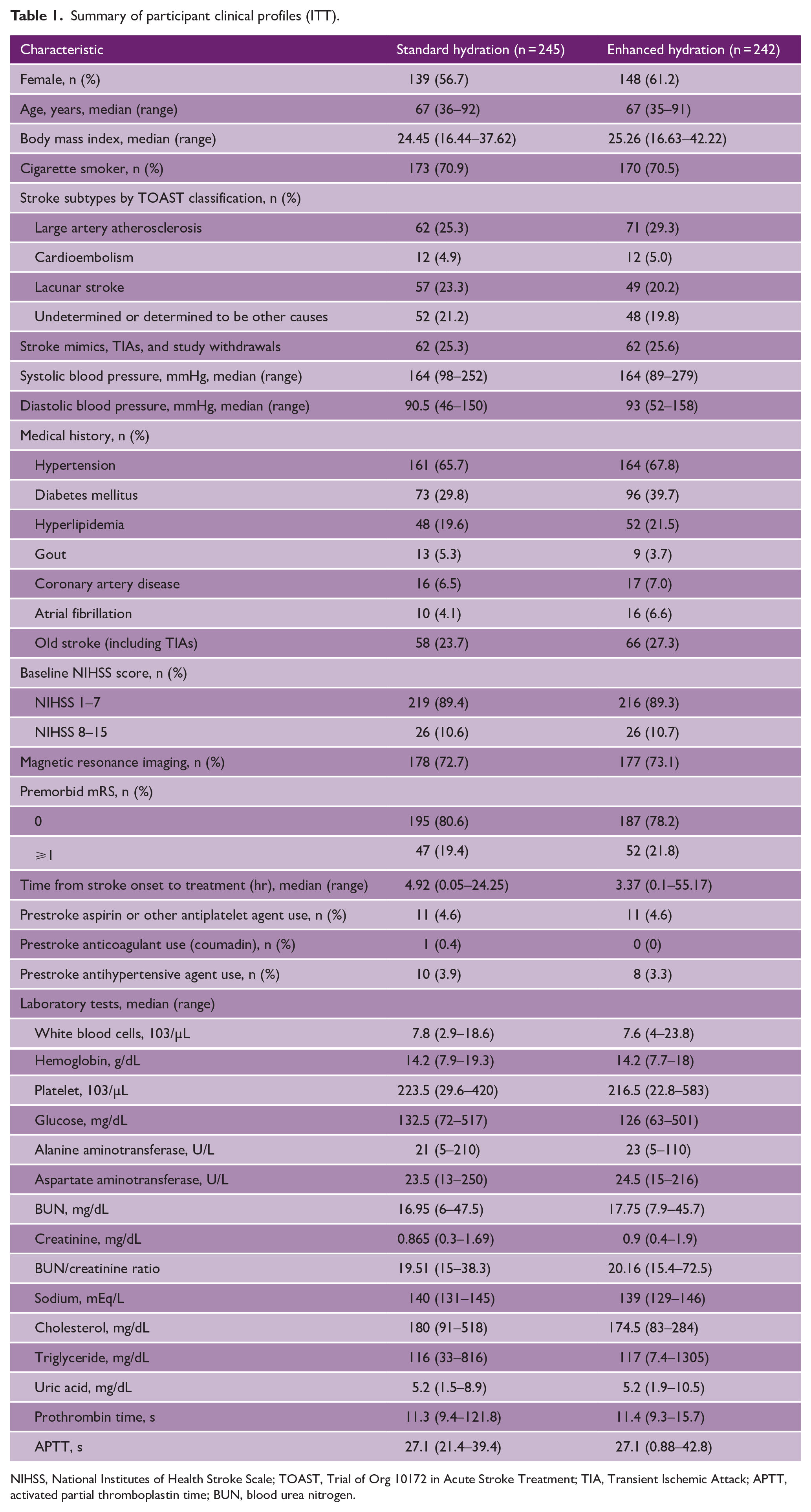
NIHSS, National Institutes of Health Stroke Scale; TOAST, Trial of Org 10172 in Acute Stroke Treatment; TIA, Transient Ischemic Attack; APTT, activated partial thromboplastin time; BUN, blood urea nitrogen.
Primary study outcome
At 72 h after ED presentation, seven participants who received enhanced hydration (2.9%) and five participants who received standard hydration (2.0%) developed major END as defined in this study; this difference of 0.9% points was not statistically significant (p = 0.54) (Table 2). PP analysis yielded similar results (Supplemental Table S2).
Primary and secondary outcomes of both groups (ITT).
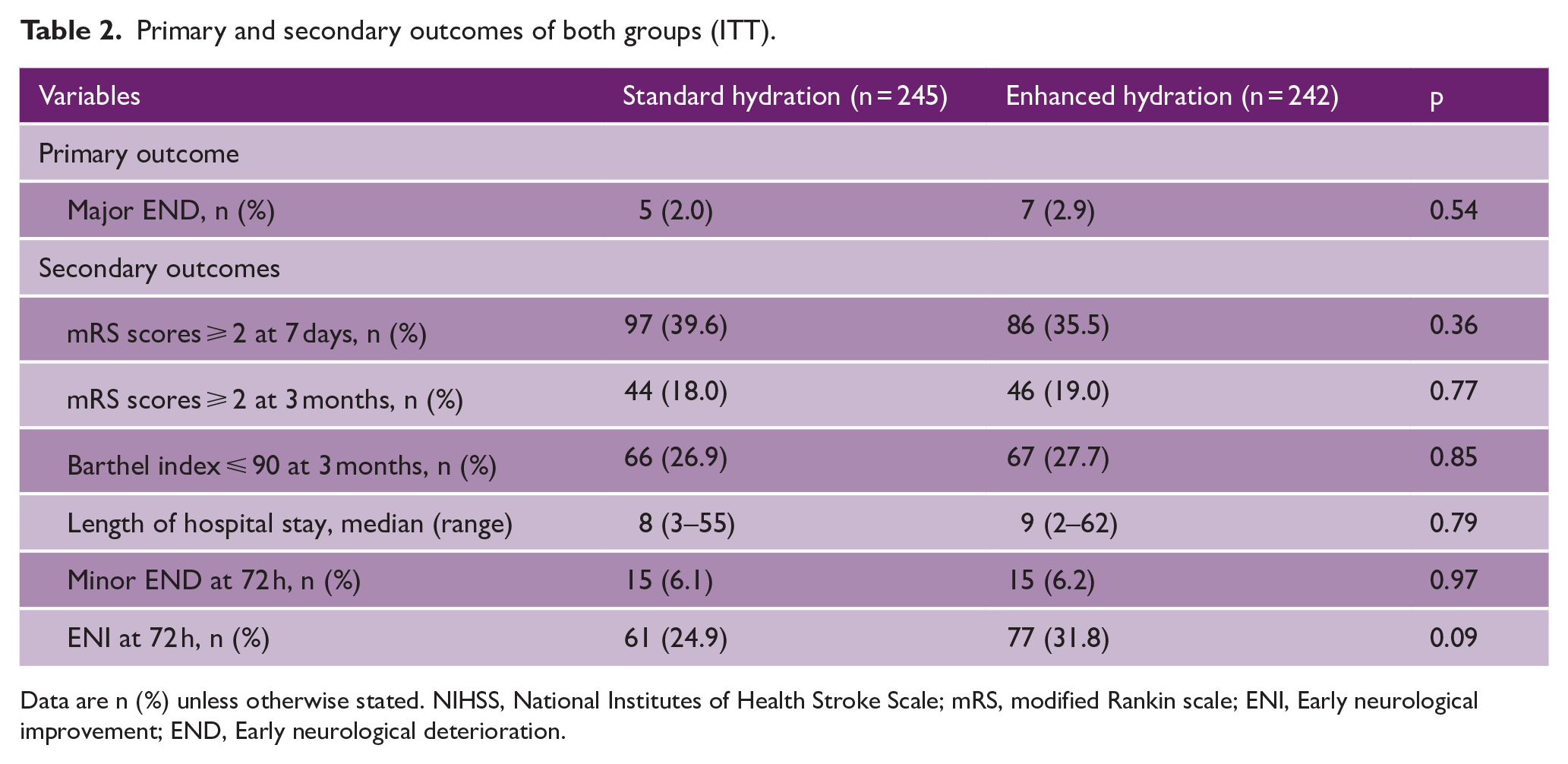
Data are n (%) unless otherwise stated. NIHSS, National Institutes of Health Stroke Scale; mRS, modified Rankin scale; ENI, Early neurological improvement; END, Early neurological deterioration.
Secondary study outcomes
Results for secondary outcomes are presented in Table 2 and Supplemental Table S2. At 72 h, 15 participants in each group developed minor END (p = 0.97), while 61 standard hydration recipients (24.9%) versus 77 enhanced hydration recipients (31.8%) demonstrated ENI (p = 0.09). Incidence of disability in activities of daily living, evaluated via mRS and BI scores at 3 months, did not differ between both groups significantly.
PP analyses of subgroups
After stratified analyses by TOAST classification, patients with lacunar stroke who received enhanced hydration were found to have lower incidences of minor END at 72 h as compared to those who received standard hydration (0.0% vs 10.5% respectively, p = 0.03), and higher incidences of ENI at 72 h (57.1% vs 35.1% respectively, p = 0.03). Patients with cardioembolic stroke who received enhanced hydration were also found to have higher incidences of ENI at 72 h (66.7% vs 16.7% of standard hydration group, p = 0.04). These statistically significant differences persisted even after adjusting for each patient’s baseline NIHSS score on ED presentation via univariable logistic regression analysis (Table 3). Acute ischemic stroke patients with etiology of large artery atherosclerosis in both standard hydration and enhanced hydration groups did not demonstrate any significant differences in study outcomes.
Comparison of outcomes between groups based on different stroke subtypes (PP).
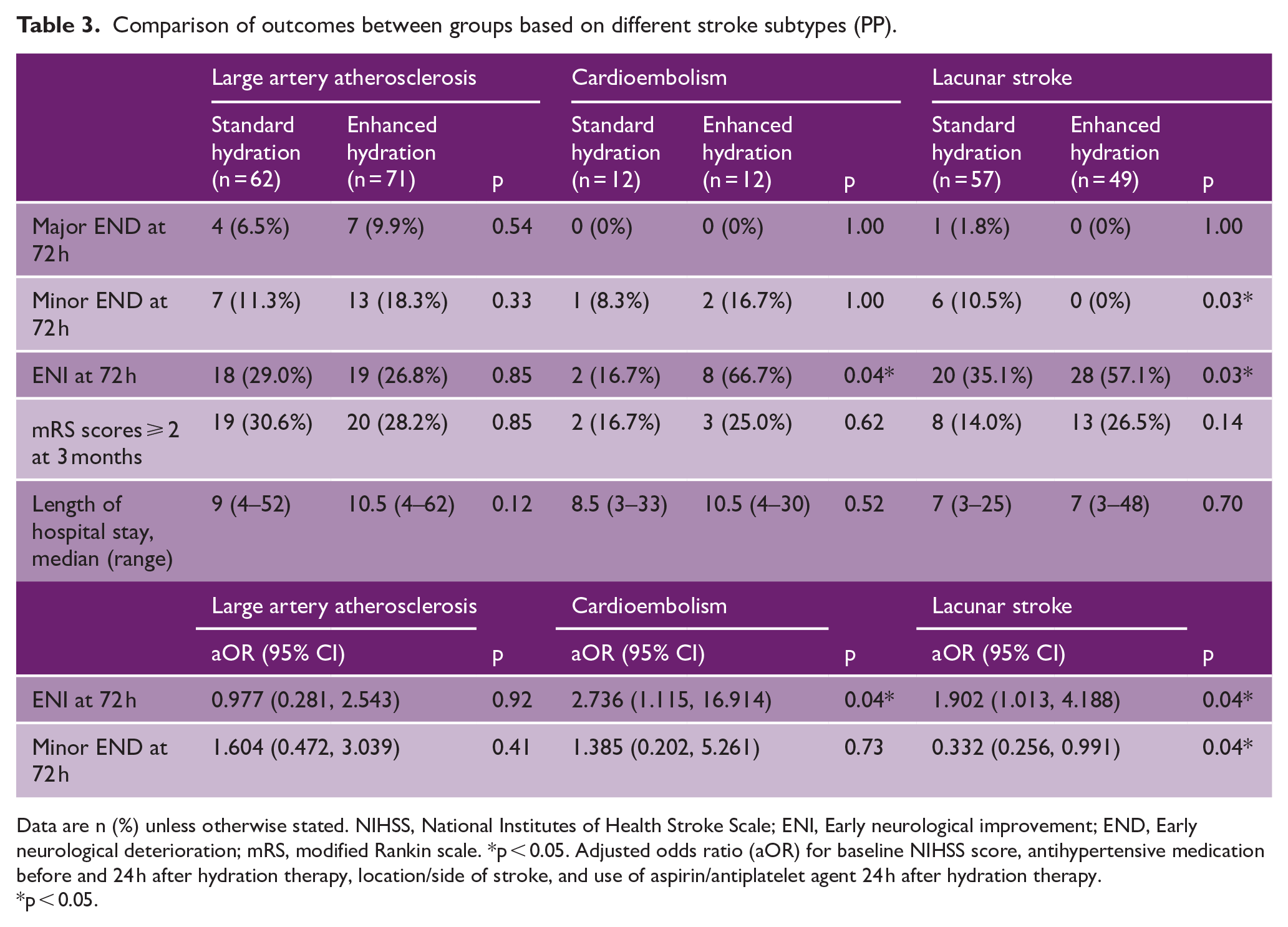
Data are n (%) unless otherwise stated. NIHSS, National Institutes of Health Stroke Scale; ENI, Early neurological improvement; END, Early neurological deterioration; mRS, modified Rankin scale. *p < 0.05. Adjusted odds ratio (aOR) for baseline NIHSS score, antihypertensive medication before and 24 h after hydration therapy, location/side of stroke, and use of aspirin/antiplatelet agent 24 h after hydration therapy.
p < 0.05.
Post hoc PP analyses
On stratification by baseline NIHSS scores, post hoc analysis revealed that patients receiving enhanced hydration demonstrated a higher incidence of ENI at 72 h as compared to those who received standard hydration (45.2% vs 34.6%, p = 0.05; Supplemental Table S3). With regard to time between stroke onset and study interventions, patients in the enhanced hydration group had higher odds of ENI (aOR 1.88; p = 0.03) when treatment was initiated within 7 h of symptom onset, compared to those who started treatment later (Table 4). There were no significant differences in incidences of major END or minor END with respect to timing of intervention.
Comparison of outcomes between groups based on duration from stroke onset to intervention (PP).
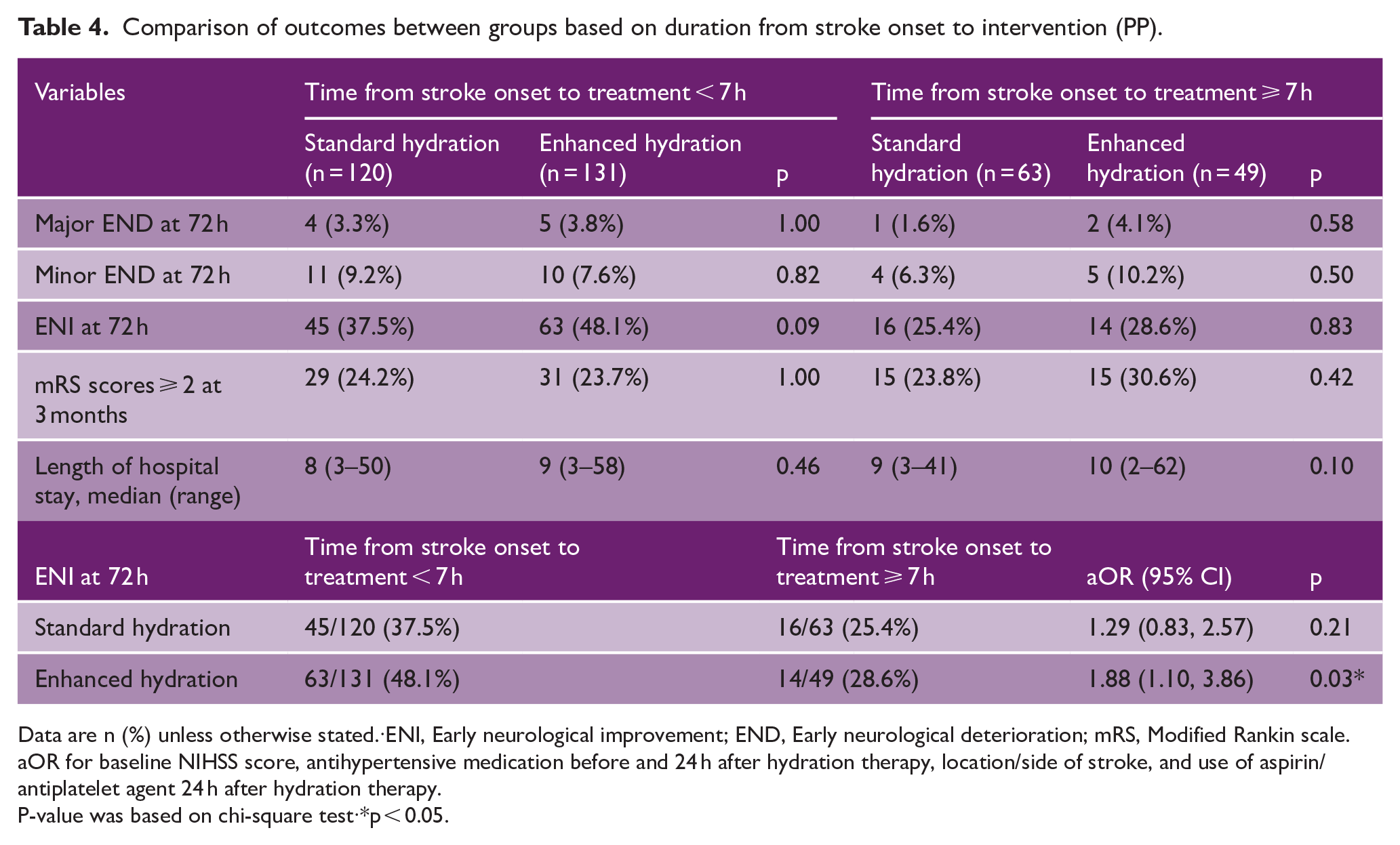
Data are n (%) unless otherwise stated.·ENI, Early neurological improvement; END, Early neurological deterioration; mRS, Modified Rankin scale. aOR for baseline NIHSS score, antihypertensive medication before and 24 h after hydration therapy, location/side of stroke, and use of aspirin/antiplatelet agent 24 h after hydration therapy.
P-value was based on chi-square test·*p < 0.05.
In non-lacunar stroke patients, those without large artery occlusion exhibited a higher rate of ENI at 72 h compared to those with large artery occlusion (aOR 2.49; p = 0.04, Supplemental Tables S4). Age (Supplemental Table S5), baseline systolic blood pressures (Supplemental Table S6), and presence of diabetes mellitus (Supplemental Table S7) did not reveal any statistically significant differences in study outcomes between both enhanced hydration and standard hydration groups, despite these subgroups having a higher prevalence of hypovolemia.23,24
Safety of study interventions
There were nine adverse events reported during the study period, inclusive of one death in the enhanced hydration group. Within the first 12 h, one participant from each hydration group developed breathing difficulties and required unblinding due to suspicion of heart failure, though this working diagnosis was later disproven. All adverse events were deemed by study investigators to be either “not related” or “unlikely related” to study interventions (Supplemental Table S8).
Discussion
This clinical trial investigated whether enhanced hydration therapy guided by BUN/Cr ratio could improve clinical outcomes of acute ischemic stroke patients. No significant differences were observed in the pre-determined primary and secondary study outcomes between enhanced hydration and standard hydration groups.
The possibility of a type II error cannot be ruled out in this study. This is because the initial sample size calculations were based on an estimated END incidence of 10–20%; in contrast, the incidence of either major or minor END observed in our study population ranged from 2.0% to 6.2%. Furthermore, despite attempts to pre-emptively account for 10% losses to our participants, 21.6% of randomized participants ultimately had to be excluded from our analyses (100 with final diagnosis of stroke mimic or transient ischemic attack, and 5 lost to follow-up). These two factors resulted in our study being insufficiently powered to detect differences at 5% level of significance.
Baseline characteristics of our study population likely contributed to the lower-than-expected incidence of END as compared to our pilot study. Our inclusion criteria of “being conscious and able to provide consent,” coupled with our exclusion criteria of “received or plans to receive thrombolytic therapy or surgical intervention,” meant that most patients with major acute ischemic strokes (NIHSS > 7) would have been ineligible for enrollment. This is reflected by our study population predominantly having a minor stroke (NIHSS ⩽ 7; 89.3%), consequently lowering the probability of observing major END in our study.
Nevertheless, subgroup analyses revealed that enhanced hydration therapy resulted in a significantly lower incidence of minor END at 72 h for lacunar stroke patients (p = 0.03) and a significantly higher incidence of ENI at 72 h for patients with either cardioembolic or lacunar strokes (p = 0.04 and p = 0.03, respectively). Statistical significance of these differences persisted even with multiple logistic regression analysis. However, the number of endpoints in each group in these subgroup analyses were very small making the results exploratory. Post hoc analysis with respect to NIHSS scores stratified according to European guidelines 25 also suggested that patients with minor strokes could benefit from enhanced hydration therapy with a higher incidence of ENI at 72 h compared to those in the standard hydration therapy group (45.2% vs 34.6%, p = 0.05).
To avoid microembolism and recurrent ischemic episodes, dual antiplatelet medication has been shown to be effective in stroke patients.26 –28 We hypothesize that enhanced hydration similarly lowers the risk of recurrent ischemic insults by correcting the hypovolemic state of patients with a BUN/Cr ratio of ⩾ 15 and improving microvascular perfusion. This increase in microvascular perfusion is crucial to improving patient outcomes, given that recent MRI perfusion studies have established the presence of END-associated penumbras in lacunar stroke and demonstrated how variations in perfusion due to different microvascular collaterals can affect clinical outcomes.29 –31 Timely correction of hypovolemia may increase collateral circulation and perfusion to the penumbra, hence decreasing minor END incidence and increasing ENI chances in lacunar stroke patients. This hypothesis is supported by our study observations of higher mean arterial pressures 2 h post-treatment in the enhanced hydration group (115.7 mmHg) compared to the standard hydration group (104.3 mmHg) (p = 0.071, Supplemental Figure S2).
Patients with large vessel disease are inherently at risk of END, with stroke progression being a common cause.16,32 While enhanced hydration therapy may initially improve collateral flow in hypoperfused areas caused by large vessel occlusions, this effect may not be adequately sustained to prevent END because of the pre-existing vascular pathology. This is reflected in our findings that non-lacunar stroke participants with an existing arterial occlusion did not benefit from enhanced hydration, unlike those without large artery occlusions who did. Hydration may also contribute to brain edema, and as such counteract and outweigh the initial benefit of restoring perfusion.
The initiation of hydration therapy within 7 h of stroke onset was found to increase the odds of ENI at 72 h in the enhanced hydration group (OR 2.32; p = 0.02). This finding suggests that early prehospital hydration therapy might be beneficial for stroke patients, similar to how dual antiplatelet therapy should be commenced earlier rather than later for maximum benefit. 26
BUN/Cr ratio has been associated with hypovolemia in stroke patients. 10 Current available evidence suggests that hydration therapy prevents END by increasing intravascular volume and cerebral blood flow.33,34 Another hypothesis is that hydration therapy improves blood rheology and limits the pathophysiological development of END.30,35,36 Future studies could be geared toward characterizing and elucidating the exact mechanism by which hydration therapy may reduce the incidence of END in hypovolemic stroke patients. Future studies may also look into incorporating more advanced and accurate measures of hemodynamic status.
Our results suggest that if ENI of stroke patients were to occur, it would be observed by 72 h albeit with no statistically significant long-term outcomes at 3 months. While it may be a testament to our center’s established stroke rehabilitation program, more research can be conducted to examine if this link exists.
Summary and conclusion
No significant differences were observed in our primary and secondary outcomes, though our findings suggest that initiating hydration therapy within 7 h of stroke onset may improve outcomes in patients with lacunar and minor strokes—additional studies are required for confirmation.
Supplemental Material
sj-docx-1-wso-10.1177_17474930241259940 – Supplemental material for Enhanced versus standard hydration in acute ischemic stroke: REVIVE—A randomized clinical trial
Supplemental material, sj-docx-1-wso-10.1177_17474930241259940 for Enhanced versus standard hydration in acute ischemic stroke: REVIVE—A randomized clinical trial by Leng Chieh Lin, Tsong-Hai Lee, Yen Chu Huang, Yuan Hsiung Tsai, Jen Tsung Yang, Lan Yan Yang, Yu-Bin Pan, Meng Lee, Kuan-Fu Chen, Yu-Cheng Hung, Hsien-Hung Cheng, I-Neng Lee, Ming Hsueh Lee, Tefa Chiu, Yeu-Jhy Chang, Zhong Ning Leonard Goh and Chen-June Seak in International Journal of Stroke
Footnotes
Acknowledgements
The authors thank the patients who participated in this study and the Clinical Trial Center at Chang Gung Memorial Hospital for statistical support, funded by the Ministry of Health and Welfare of Taiwan (MOHW110-TDU-B-212-124005).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Chang Gung Healthare Foundation (grant nos. CORPG6D0091–93, CORPG6H0041, CORPG6K0201–02, CORPG6K0141–42, CMRPG6K0091–92, CMRPG6K0041, CMRPG6H0141, CMRPG6H0571, CPRPG6D0011–12, and CMRPG6M0041) to L.C.L., (grant nos. CORPG6D0081–83, CORPG6G0181–83, CORPG6G0231–33) to J.T.Y., (grant no. CPRPG2D0011–12) to K.F.C., (grant no. CPRPG8D0011–12) to H.H.C., and (grant no. CPRPG3D0011–12) to C.J.S. The funder had no role in study design, data collection, analysis, publication decisions, or article preparation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
