Abstract
Highlights
Our comprehensive model can be used to evaluate various health interventions that aim to reduce the disease burden caused by HPV and cervical cancer.
Cytology-based cervical cancer screening tailored to WLWH in Guangxi, China can effectively prevent cervical cancer cases and cancer deaths.
Adopting a 3- or 5-y screening interval may provide a more reasonable balance of benefits and harms to prevent future cancer cases and deaths.
Introduction
Cervical cancer is the fourth most common cancer among women worldwide. 1 Human papillomavirus (HPV) infections cause almost all cases of cervical cancer. 2 Although most women clear HPV infections, certain risk factors can increase the likelihood of progression from cervical HPV infection to cancer such as tobacco smoking and co-infection with human immunodeficiency virus (HIV).3–7 From 2008 to 2011, the age-standardized cervical cancer incidence among Chinese women living with HIV (WLWH) was 47.6 (95% confidence interval [CI] 38.3–57.0) per 100,000, which was 7 times higher than that observed in the general population. 8
Smoking control is a challenge in China. Although the prevalence of smoking is low in women (3%), 52% of men are active smokers, and 70% of Chinese adults are exposed to secondhand smoke (SHS) in a typical week. 9 Published studies have shown a positive association between active smoking exposure and cervical abnormalities among WLWH.10–13 In women without HIV, SHS exposure increases the risk of HPV incidence and low-grade cervical intraepithelial neoplasia (LGCIN) detection by 1.12 and 1.40 times, respectively.14,15 It is reasonable to hypothesize that a similar effect of SHS on HPV and LGCIN might also be occurring among WLWH. Such a high prevalence of SHS exposure in Chinese women implies the need to consider this additional risk factor when evaluating prophylactic health care interventions to reduce the burden of cervical cancer.
Cervical cancer screening with timely treatment is effective in reducing cervical cancer incidence and mortality. 16 Since cervical cancer is an acquired immunodeficiency syndrome (AIDS)–defining condition, the World Health Organization (WHO) recommends regular cervical cancer screening with an earlier screening start age for WLWH than for women in the general population. 17 In some countries, cervical cancer screening guidelines are tailored to WLWH to reflect their higher risk and include recommendations for a shorter screening interval and older age to exit screening.18,19
In 2009, the Chinese government launched the National Cervical Cancer Screening Program in Rural Areas to reduce regional disparities in cervical cancer burden and as a first step toward nationwide provision of population-based cervical cancer screening. However, the current screening guidelines in China do not provide separate screening recommendations for WLWH. 20 The current uptake of cervical cancer screening is estimated to be 20% among the general population of Chinese women. 20 In the absence of tailored screening recommendations, this low level of screening is worrisome for WLWH given their excess risk.
The HIV prevalence in Guangxi is 3 times the national average (1.5% vs. 0.45%), and from 2010 to 2017, more than 85,000 newly diagnosed HIV infections in women were reported to the Guangxi Center for Disease Control and Prevention.21,22 Our previous study has shown that WLWH in Guangxi were willing to be screened and that tailored screening programs carried out at HIV treatment centers might provide an easy way to increase screening. 23 To inform tailored recommendations for WLWH, we developed a comprehensive individual-based model to capture the complex natural history process of cervical cancer in women in Guangxi, China. The model simulates sexual transmission of HPV and also incorporates the additional risk of cervical cancer associated with smoking exposure, which allows us to evaluate screening strategies tailored to WLWH in Guangxi, China. Based on a recent study in Guangxi that showed 91% combination antiretroviral treatment (cART) coverage and 90% cART adherence among WLWH, 24 we assumed that a tailored screening program offered through HIV treatment centers would increase screening uptake from 20% to 80%, since WLWH are already closely followed. In the current modeling study, we aimed to 1) project the benefits of implementing tailored cytology-based cervical cancer screening with 80% uptake compared with the status quo (i.e., cytology-based screening every 3 y from age 21–65 y with 20% uptake) and 2) examine the impact of different combinations of screening end age and interval on the balance between benefits and harms.
Methods
Model Description
An individual-based model of HPV cervical carcinogenesis was implemented in Java. 25 To accurately capture the dynamics of HPV transmission and clearance, each simulation cycle represents 1 mo. The model consists of 4 main modules, which are described below: a demographic module, an HIV dynamics module, a sexual mixing module, and an HPV-cervical carcinogenesis module. The time horizon modeled is 100 y. Note that sources of data and the parameter estimates are presented in the Technical Appendix, under the section “Input Parameters.”
Population Setting
The model can simulate either a cross-sectional population of both men and women or a cohort of women with specific risk exposures. In a cross-sectional population setting, HPV acquisition is through heterosexual activity; in a cohort population setting, HPV acquisition is through age- and sex-specific incidence.
Demographic Module
The demographic module simulates population demographics, including aging, deaths, births, active smoking, exposure to SHS, and benign hysterectomies among women in the population. In the cross-sectional population setting, the initial population starts with the current age distribution of the Chinese population. 26 During each simulation cycle, each individual has a probability of dying based on age-specific all-cause mortality. Each individual is randomly assigned to a smoking exposure based on the active smoking and SHS exposure prevalence in the Chinese population.9,27 Women in the population have a probability of undergoing a hysterectomy for benign reasons. 28
In the cohort population setting, the demographic module simulates a population of women starting at the same age. No births are added to the cohort. The rest of the module remains the same as described above.
HIV Dynamics Module
The HIV module structure is represented in Figure 1A. The HIV module simulates the dynamics of HIV in the population. HIV health states are defined based on the current CD4 cell count of the infected individuals and are categorized as “CD4 > 500,”“CD4 200–500,” or “CD4 < 200.” During a simulation cycle, individuals who are not infected with HIV have a risk of acquiring HIV. The yearly force of HIV infection is parameterized as a Weibull distribution with a shape parameter
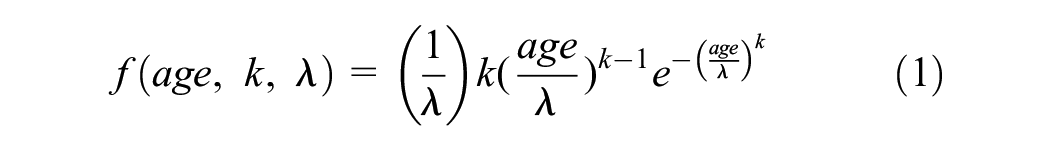
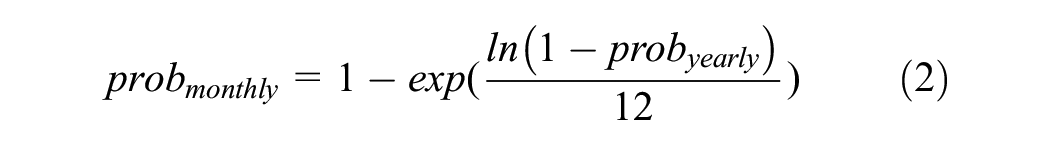

Model structure: (A) HIV dynamics module structure and (B) HPV-cervical cancer natural history module structure. HIV, human immunodeficiency virus; HPV, human papillomavirus.
During a simulation cycle, individuals with undetected HIV infection have a probability of having their HIV detected depending on their current CD4 cell count. Newly detected HIV infections contribute to the observed HIV incidence in the population. Once an HIV infection is detected, the infected individual has a probability of initiating cART. The probability of transitioning between HIV health states depends on the current HIV health state and whether the individual is on cART. At any time, the HIV prevalence of the population is defined as the proportion of individuals with detected HIV infection in the total population.
Risk Strata Definition
Based on the current HIV health state and smoking exposure, each individual is assigned to a specific risk stratum. The risk stratum is updated each month to represent a change in status from the previous simulation cycle. There are 4 levels of HIV infection (i.e., no infection, CD4 > 500, CD4 200–500, and CD4 < 200) and 3 levels of smoking exposure (i.e., no exposure, active smoking, and SHS exposure). As a result, an individual can be in 1 out of 12 possible risk strata during any given cycle. HIV infection and smoking exposure are associated with additional risks of acquiring HPV infection and developing cervical cancer.3,7,30 Individuals who are not infected with HIV and have no smoking exposure have the lowest (i.e., baseline) risk of acquiring HPV infections and/or developing cervical cancer. Individuals in other risk strata have stratum-specific hazard ratios that are applied to the baseline risk. We assumed the risk of HIV+ and smoking are multiplicative. 31
Partnership Formation
In the cross-sectional population setting, at birth, each individual is assigned an age of sexual debut (Technical Appendix Table 1), a fixed number of lifetime sexual partners (Technical Appendix Table 2), and an indicator of whether they will have concurrent partnerships when they are sexually active. The number of lifetime partners is randomly sampled from a sex-specific Poisson distribution. The age of sexual debut is randomly sampled from a sex-specific Weibull distribution. In a given cycle, individuals are eligible to form a partnership if they meet the following conditions:
they have reached the assigned sexual debut age,
they have not reached the assigned number of lifetime partners, and
for individuals who were not indicated as engaging in concurrent partnerships, they are not currently in an active partnership.
Women eligible to look for partners start the process of partnership formation by selecting a man whose age is within a given age interval (Technical Appendix Table 3). Once the partnership is formed, a partnership duration in months is assigned (Technical Appendix Table 4). The partnership dissolves when the duration is reached. During a simulation cycle, any death (i.e., background death, HIV-related death, or cervical cancer death) results in the dissolution of all active partnerships. In this model, we simulated only heterosexual partnership formation. In the cohort population setting, no sexual mixing is considered.
HPV Transmission
The model distinguishes high-risk HPV (hr-HPV) types as the vaccine-preventable type hr-HPV (hereafter referred to as vaccine type) and hr-HPV types that are not prevented by the HPV vaccine (hereafter referred to as nonvaccine type), assuming the use of the nonavalent HPV vaccine (Gardasil-9). In the cross-sectional setting, women who have previously developed natural immunity are assumed to have type-specific immunity against a new HPV infection of that type. Other than the immune individuals, sexual partners who are HPV sero-discordant have a probability of transmitting HPV infections in each simulation cycle. The probability of acquiring HPV infection depends on an individual’s biological sex, age, and the additional risk attributable to HIV infection and smoking exposure. Type- and sex-specific HPV transmission probabilities are modeled assuming a linear relationship with age, as shown in equation (3). In the cohort population setting, individuals can be infected with HPV based on the age-specific HPV incidence.
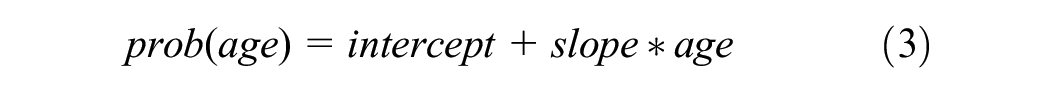
HPV-Cervical Carcinogenesis
The HPV-cervical carcinogenesis module simulates the natural history from HPV infection to cervical cancer development. The model structure is shown in Figure 1B. In each cycle, HPV-infected individuals have a probability of clearing the infection. Type- and sex-specific clearance probabilities are modeled as a linear relationship with age, as shown in equation (4). A certain proportion of women who clear an HPV infection develop type-specific natural immunity. 31 Natural immunity is assumed to be lifelong without waning. Men who clear infections do not develop natural immunity. 31 Persistent HPV infections can progress to LGCIN, high-grade cervical intraepithelial neoplasia (HGCIN), and cervical cancer.
Women in the LGCIN and HGCIN health states can have their lesions regress. However, once a lesion progresses to cancer, regression will not occur. Progression and regression probabilities depend on an individual’s age and the current risk stratum that they are in, which is defined by the current HIV infection status and smoking exposure. The additional risks associated with HIV infection and smoking exposure are summarized in Technical Appendix Table 5. During each cycle, an individual can transition between health states based on progression/regression probabilities. Except for the HPV clearance probabilities, all other transitions are modeled as Weibull rates as a function of age, as shown in equation (5). The rates are transformed to probabilities using equation (6).

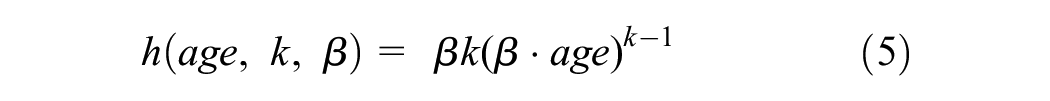
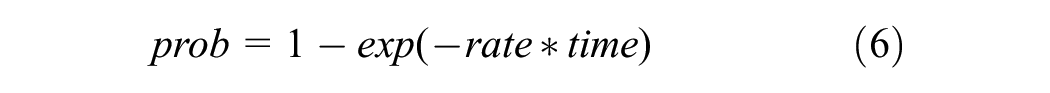
Cancer Stages
Cancer stages are defined as early (i.e., stage I) and late (i.e., stage II through IV). Women with HGCIN can transition to the early preclinical cancer stage. Undetected early-stage cancers can progress to a late preclinical cancer stage. The probability of having cancer symptoms is higher in the late preclinical cancer stage than in the early preclinical cancer stage. Detected cancer cases contribute to the observed cervical cancer incidence in the population. Women who have their cancer detected transition to the clinical cancer stage and can die of cancer based on 10-y survival estimates. Early-stage cancer has a higher survival compared with late-stage cancer. Women who survive 10 y after their cancer is detected transition to a “survivor” health state where they are assumed to be at risk only of death from other causes.
Model Calibration
The HIV module and the HPV-cervical carcinogenesis module are calibrated separately. The HIV incidence (Technical Appendix Table 6), prevalence, and cART coverage in Guangxi are the calibration targets for the HIV dynamics module. Sex-specific HIV incidence is captured as newly detected HIV infections divided by the total population of women or men who are alive. HIV prevalence is calculated as all live detected HIV cases divided by the total population. The calibrated parameters include the following:
the Weibull shape and scale for HIV infection,
the transition probabilities between HIV health states,
the probabilities of being detected at each HIV health state, and
the probability of initiating cART treatment in each month.
The HIV module calibration was accomplished using the Bayesian incremental mixture importance sampling (IMIS) method. 32 The technical details of the IMIS method can be found in the Technical Appendix. The calibrated HIV parameters are considered fixed for calibration of the HPV-cervical carcinogenesis module.
The type- and sex-specific HPV prevalence and cervical cancer incidence are the calibration targets for the HPV-cervical carcinogenesis module. The calibration parameters include
the sex-specific probabilities of HPV transmission and HPV clearance,
CIN progression and regression probabilities, and
the percentage of women who develop natural immunity.
Both manual calibration and the IMIS method are used. The calibrated parameters’ values are summarized in Technical Appendix Table 5.
Model Validation
Internal validation is achieved by comparing the model-predicted outcomes with the calibration targets. External validation is achieved by comparing the model-predicted outcomes to the external validation targets, including cervical cancer mortality in the general population, the relative risk (RR) of HPV prevalence among WLWH compared with the women in the general population, and the RR of cervical cancer incidence among WLWH compared with the women in the general population. The details of the calibration and validation targets can be found in the Technical Appendix, under the section “Calibration and Validation Targets.”
Screening Strategies
We first simulated a cross-sectional population with the size of 5 million to determine the age-specific HPV incidence as a function of heterosexual HPV transmission. This age-specific HPV incidence was then used as an input for the cohort model, which simulates a population of WLWH, age 15 to 100 y, and who are assumed to be newly detected HIV cases with CD4 > 500 (Figure 2).

Model population settings.
We evaluated the status quo scenario and 9 other cytology-based cervical cancer screening strategies for WLWH. In all scenarios, screening is assumed to start at age 21 y, with the primary screening test being cytology with HPV triage for atypical squamous cells of undetermined significance. The screening algorithm is shown in Technical Appendix Figure 1. The status quo scenario (i.e., cytology-based screening every 3 y from age 21–65 y) assumes that without tailored recommendations, WLWH have the same screening uptake (i.e., 20%) as the general population does. We assumed that a tailored screening program offered through HIV-treatment centers would increase screening uptake from 20% to 80%. To examine the impact of screening uptake, we examined this assumption by increasing the uptake from 20% to 50% in a sensitivity analysis. The 9 tailored screening strategies vary by screening interval (every year, every 3 y, or every 5 y) and age to end screening (age 65, 85, or 100 y, equivalent as lifetime screening). Detailed descriptions of each strategy are presented in Technical Appendix Table 7.
Data Analysis
We estimated the following outcomes in WLWH associated with the different scenarios: the number of cytology and HPV tests, the number of false-positive test results, colposcopies, treatments, total cancers, and cancer deaths. The number of cancers and cancer deaths prevented reflect the benefits of cervical cancer screening. The total number of screening tests, false-positive results, colposcopies, and treatments reflect potential harms of screening. For aim 1, the benefit of tailored screening for WLWH compared with the status quo is evaluated as the percentage reduction in cancers and cancer deaths. For aim 2, the impact on the benefit-to-harm balance associated with varied screening interval and end age is examined using the following ratios: the total number of screening tests (i.e., cytology and HPV tests) per cancer and per cancer death prevented and the number of colposcopies per cancer and per cancer death prevented. Higher ratios indicate more tests and potential harms associated with preventing cervical cancers and deaths. Lower ratios indicate more efficient screening. Frontier plots are also presented to reflect a strategy’s “value” in terms of the harm-benefit tradeoff. Four distinct measures of efficiency were selected to evaluate the harm-benefit tradeoffs associated with the screening strategies: 1) the number of colposcopies per total cancer, 2) the number of colposcopies per cancer death, 3) the number of total tests per total cancer, and (4) the total number of tests per cancer death.
Patient and Public Involvement
Patients or the public were not involved in the design, conduct, reporting, or dissemination plans of our research.
Results
Model Validation
We validated the model-predicted outcomes to internal and external validation targets. The model-predicted values compared with the HPV prevalence, cervical cancer incidence, and cancer mortality targets for the general population are shown in Technical Appendix Figure 2. The RR of vaccine-type HPV prevalence among HIV-infected individuals was approximately 1.37 times higher than the general population, falling within the range of the validation targets that were observed among Chinese women (e.g., the RR for HPV 16 was 1.4 with a 95% CI of 1.0–2.1; the RR for HPV 18 was 2.1 with 95% CI of 1.2–3.9). 33 Similarly, the model predicted that the cancer incidence among WLWH was 2.4 times higher than that of the general population, which is in the range of the pooled RR from a meta-analysis that compared cervical cancer incidence between WLWH to women without HIV infection (RR = 4.1, 95% CI 2.3–6.6). 30
Status Quo
Under the status quo assumption of cytology-based screening every 3 y from age 21 to 65 y with 20% uptake among WLWH in Guangxi, the number of HGCIN treatments, the number of cervical cancers detected, and cancer deaths among WLWH was estimated to be 12, 4.8, and 2.6 per 1,000 WLWH, respectively. Approximately 95% of cancer cases in this scenario were detected based on cancer symptoms. With the current 20% screening coverage, approximately 10% of cervical cancers and cervical cancer deaths could be prevented compared to without screening in this population.
Effect of Tailored Screening
All screening strategies evaluated were effective in reducing the total number of cervical cancers and cervical cancer deaths among WLWH. Table 1 presents the outcomes per 1,000 WLWH associated with no screening, the status quo, and the 9 cytology-based screening strategies tailored to WLWH. Under the 9 tailored cytology-based screening scenarios, for every 1,000 WLWH in Guangxi, HGCIN treatments ranged from 33 to 75, total cancers ranged from 1.95 to 3.96, and cancer deaths ranged from 1.00 to 2.10.
Outcomes of Cervical Cancer Screening Strategies in Women Living with HIV, per 1,000 WLWH over the Time Horizon of 100 y

FP, false-positive; HIV, human immunodeficiency virus; HPV, human papillomavirus; WLWH, women living with HIV.
Screening strategies are abbreviated as interval_end age_uptake. For example, q1_65_80% represents screening every 1 y, end age of 65 y, and 80% uptake.
q3_65_20% refers to the status quo scenario.
Strategies with lifetime screening and a screening interval of 1 y resulted in more screening tests. For example, screening strategy 3, which had a screening interval of 1 y and no screening exit, resulted in the most number of screening tests (14,346 per 1,000 WLWH). It also resulted in the highest number of treatments (75 per 1,000 WLWH).
By implementing tailored cytology-based screening in WLWH that increases screening uptake to 80%, the model predicted a reduced number of cancers and cancer deaths. Compared with the status quo, the percentage reduction in total cancers ranged from 18% to 60%, and the percentage reduction in cancer deaths ranged from 20% to 62%. Strategy 3, with lifetime screening and a screening interval of 1 y, was the most effective screening strategy in terms of preventing cancers and cancer deaths.
The balance of benefits and harms, reflecting the efficiency of tailored screening for WLWH, is summarized in Figure 3. Figure 3A and Figure 3B represents the number of total tests and total colposcopies per cancer and cancer death prevented in every 1,000 WLWH, respectively. Strategy 8 (i.e., end age of 85 y and a 5-y screening interval) had the lowest number of tests and colposcopies per cancer prevented, indicating it was the most efficient strategy of the 9 to prevent total cancers. Strategy 9 (i.e., end age of 100 y and a 5-y screening interval) had the lowest number of tests and colposcopies per cancer death prevented, indicating this was the most efficient strategy to prevent cervical cancer deaths.

Benefits and harms of tailored screening strategiesa for women living with HIV compared with the status quob (per 1,000 WLWH over the time horizon of 100 y). (A) Number of tests needed to prevent cancers and cancer deaths (per 1,000 WLWH). (B) Number of colposcopies needed to prevent cancers and cancer deaths (per 10,00 WLWH). HIV, human immunodeficiency virus; WLWH, women living with HIV.
Figure 4 summarizes the frontier strategies for the 4 measures of efficiency in terms of benefits and harms tradeoffs. The model predicted similar frontier strategies for the 4 efficiency measures. Screening with a 1-y interval clustered at the top left of the frontier plots indicates less efficiency in preventing cervical cancers and cancer deaths. Ending screening at age 85 or 100 y is deemed to be on the frontier.
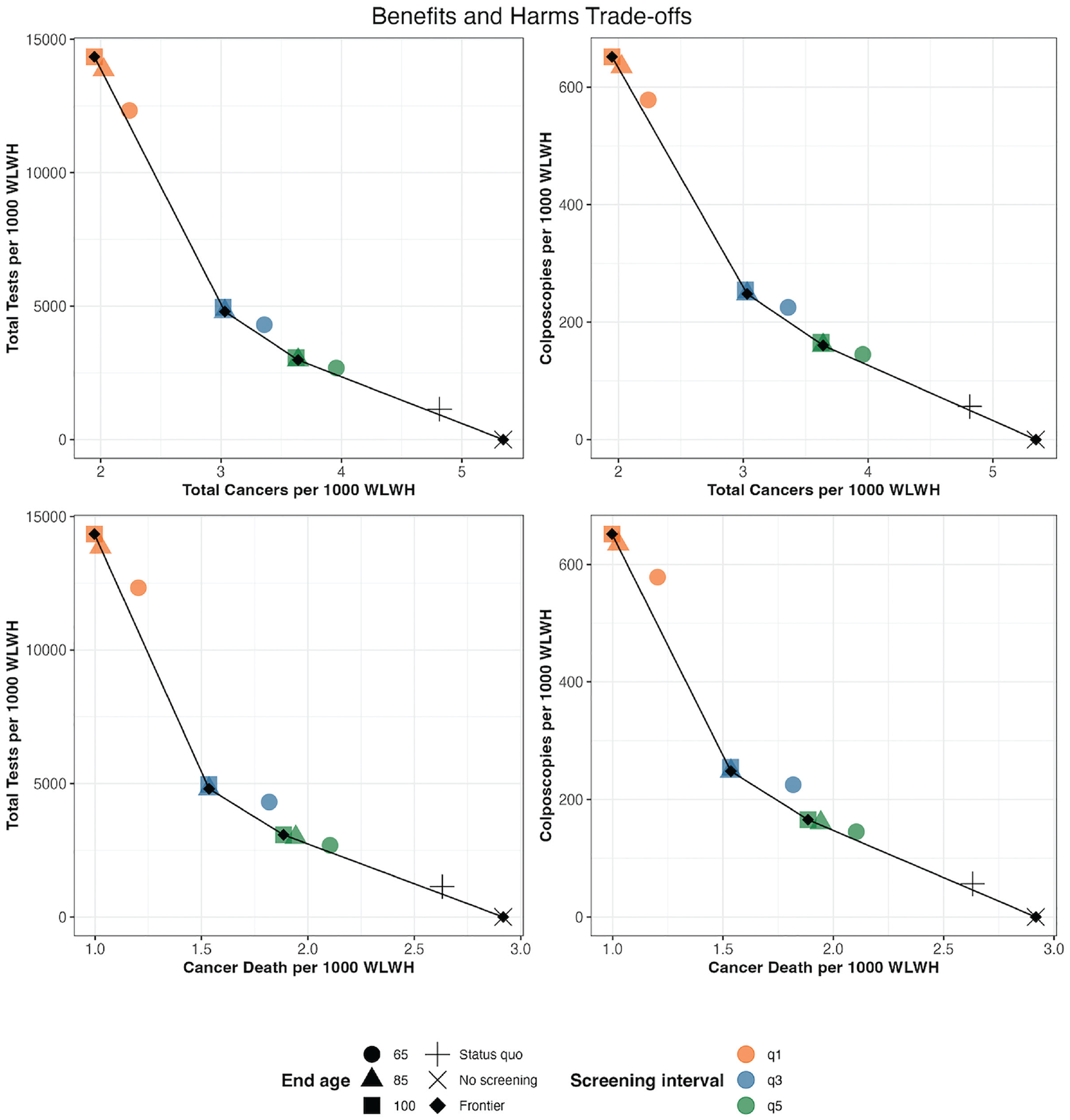
Benefits and harms tradeoffs of tailored screeninga strategies for women living with HIV (per 1,000 WLWH over the time horizon of 100 y). WLWH, women living with HIV.
Discussion
We developed a comprehensive individual-based model to simulate the natural history of cervical cancer in a population from Guangxi, China. Using this model, we were able to replicate sexual HPV transmission between heterosexual partners. We also incorporated smoking exposure as an additional risk factor of HPV acquisition and cervical cancer development, given the high prevalence of SHS reported in China. 9
In this study, the model was used to evaluate the benefits and the harms associated with potential screening strategies targeting WLWH in Guangxi, China. By stratifying on HIV infection status, we were able to evaluate the benefits and the harms of 9 cytology-based screening strategies tailored to WLWH in Guangxi, China, with different end ages and screening intervals. Our model predicted that tailored screening strategies could reduce the total number of cervical cancers and cancer deaths among WLWH. The model predicted that tailored screening with 80% uptake could reduce cancer cases and deaths by 18% to 60% and 20% to 62%, respectively. Lifetime screening with a 1-y screening interval may prevent the most cancers and cancer deaths among WLWH in Guangxi, China. However, screening every year also resulted in more screening tests and false-positive test results, highlighting the need to consider the balance of benefits and harms of different screening strategies in this high-risk population. Our results suggest that a longer screening interval may be associated with a reasonable balance of benefits to harms. In particular, a 3- or 5-y interval was associated with a lower number of screening tests and colposcopies per cancer case and death prevented. We conducted the same set of analyses for women without HIV. Due to the lower burden of disease, the model predicted an even higher number of colposcopies and screening tests needed to prevent per cancer and cancer deaths (Technical Appendix Figure 3) and that a 5-y screening interval would be more reasonable to recommend. Our results are consistent with previous studies that examined the impact of cervical cancer screening among Chinese women without HIV. In a cost-effectiveness analysis of cervical cancer screening strategies in urban China, the authors recommended screening with a 3- or 5-y interval instead of yearly screening. 34 Similarly, in another cost-effectiveness modeling study that evaluated cervical cancer screening effectiveness in rural China, cytology-based screening with a 1-y interval had the highest cost-effective ratio of 44,149 Yuan/quality-adjusted life-year (QALY) compared with screening every 3 y (14,821 Yuan/QALY) and screening every 5 y (9,061 Yuan/QALY). 35
Although the national cervical cancer screening program was launched more than a decade ago, current screening uptake among screening-eligible women remains low.36,37 Studies have identified both individual- and geographic-level factors contributing to the low screening coverage, including gaps in knowledge and health awareness, fear of screening outcomes, cultural barriers to vaginal exams, income, education, and urbanization level.37,39 In a previous study, lack of awareness about available cervical cancer screening services was one of the barriers for WLWH in Guangxi to get screened; women stated that they would be willing to be screened if they knew screening was offered. 23 In the current study, we assumed screening would take place at the HIV treatment centers, where WLWH are already closely followed. This approach to offering cervical cancer screening could be a feasible option to increase cervical cancer screening participation among WLWH. Under the assumption of an increased uptake given this convenient setting, we illustrated the potential benefits of implementing tailored screening for WLWH.
Our model has several strengths. First, by simulating a population with a proportion of HIV-infected individuals, the model can be used to evaluate health interventions targeting the general population as well as individuals living with HIV. The model is calibrated to fit the targets representing the general population but was also validated against HIV-specific targets. It can be used to evaluate potential health interventions that aim to prevent HPV infection in both men and women and cervical cancer among women. For example, the model can be used to evaluate the impact of introducing the HPV vaccine into the Chinese population.
Without a universal HPV vaccination program and a lack of HPV vaccine supply in China, current HPV vaccination levels are low. 40 The coverage is expected to increase as the global supply of HPV vaccines increases in future years. 41 Published studies have illustrated the effectiveness of introducing a universal HPV vaccination program in China, with and without a combination of alternative screening strategies.40,42 With various assumptions, such as the vaccine type, cost, coverage, and vaccination age, these studies reached a consensus that universal HPV vaccination could be cost-effective. By distinguishing the HPV types as the vaccine type and the nonvaccine type, the current model structure is sufficient to evaluate vaccination policies targeting the Chinese population and, importantly, can account for herd immunity effects. Evaluating the epidemiologic and economic impact of a universal HPV vaccination program in China on WLWH requires detailed information that is unavailable at the current stage, such as the types of HPV vaccine that would be made available, realistic coverage, estimated cost, and alternative recommendation for WLWH. Therefore, we conducted a sensitivity analysis that assumes the future HPV vaccination program can reduce the vaccine-type HPV incidence to 50% and 20% of the current incidence. As the overall cervical cancer burden decreases, the total tests and colposcopies needed to prevent per cancer and cancer death increase. However, a 3- or 5-y screening interval is still predicted to provide better benefits to harms balance than screening with a 1-y interval (Technical Appendix Figure 4 and Figure 5).
A second strength of this model is that it allows sexual mixing in sero-discordant partners and so is more realistic than simply simulating a population that includes only HIV-infected individuals. As a result, our model can be used to evaluate health interventions that aim to reduce sexually transmitted HPV, such as condoms. 43 Third, the model can be used to evaluate the impact of population-level smoking control policies on cervical cancer in China. Since 2015, a new comprehensive smoke-free law came into effect in Beijing, leading to the draft of a national smoking control policy. 44 By varying the prevalence of active smoking and SHS exposure in the model, we can examine the effect of such smoking control policies on reducing cervical cancer and/or precancer among Chinese women and among WLWH.
Our model has limitations. The assumption of achieving 80% uptake is based on patient adherence to cART treatment, assuming that a tailored screening program is offered through HIV-treatment centers. However, the actual screening uptake or whether implementing screening at existing HIV-treatment centers is successful is not guaranteed. Therefore, we conducted a sensitivity analysis assuming the screening uptake will be increased from the status quo of 20% to 50%. Although the benefits of cervical cancer and cancer death reduction are lower, the results are similar in terms of the predicted benefits and harms balance (Technical Appendix Figure 6).
Cytology-based screening has been adopted by many national population-based screening programs and is also recommended and available in China, and the “screen-and-treat” strategy with visual inspection with acetic acid or Lugol iodine (VIA/VILI) triage is currently recommended as an alternative to cytology-based screening. 45 The 2021 updated WHO guidelines recommend switching from VIA or cytology to HPV-based screening. 17 We did not evaluate the effect of primary screening with HPV or co-test screening strategies on reducing cervical cancer burden among WLWH. HPV primary tests and co-tests are more sensitive than cytology but are also less specific. 46 As a result, although screening with HPV tests is more effective at detecting cervical disease, to reduce the number of unnecessary follow-up tests and colposcopies, detailed triage strategies are required. 16 In future analyses, we will evaluate the impact of primary HPV and co-test screening strategies targeting WLWH based on the updated WHO guidelines.
In addition, we did not explicitly model HIV transmission in the model. Although modeling HIV transmission explicitly would improve the accuracy of the model, modeling HIV and HPV transmission would complicate the model structure. Therefore, we simplified the HIV module and modeled the acquisition of HIV by applying a fixed incidence rate.
Conclusion
We developed a comprehensive individual-based model that simulates the natural history of cervical cancer in WLWH. To highlight the usefulness of our model for policy, we evaluated the benefits and the harms associated with 9 cytology-based cervical cancer screening strategies tailored to WLWH in Guangxi, China. Implementing cytology-based cervical cancer screening in this high-risk population can effectively prevent cervical cancer cases and cancer deaths. Our results suggest that adopting an older end age and a shorter screening interval might be more effective in preventing cancer cases and deaths but may also be associated with more tests and costs. Adopting a 3- or 5-y screening interval may provide a more reasonable balance of benefits and harms among WLWH in China.
Supplemental Material
sj-docx-1-mpp-10.1177_23814683241308905 – Supplemental material for Cervical Cancer and Cervical Cancer Screening in Women with HIV in Guangxi, China: A Model Simulation Study
Supplemental material, sj-docx-1-mpp-10.1177_23814683241308905 for Cervical Cancer and Cervical Cancer Screening in Women with HIV in Guangxi, China: A Model Simulation Study by Ran Zhao and Shalini Kulasingam in MDM Policy & Practice
Footnotes
Acknowledgements
We would like to express our sincere gratitude to the Cancer Intervention and Surveillance Modeling Network (CISNET) Cervical Cancer HIV working group for providing their valuable feedback in developing the cervical cancer natural history model structure. We would like to thank the CISNET University of Michigan Lung Cancer group for giving feedback in developing the smoking module. We would like to thank Ms. Madalyn Nones for her help in proofreading the manuscript. The work has been presented at the 2022 CISNET Annual Meeting.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided in part by a grant from the National Institutes of Health’s National Cancer Institute (project No. 3U01CA253912). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
Authors’ Note
This study was conducted as part of the PhD thesis of Dr. Ran Zhao at the University of Minnesota–Twin Cities, School of Public Health, Division of Epidemiology and Community Health.
Author Contributions
Ran Zhao, conceptualization, methodology, formal analysis, investigation, resources, data curation, writing–original daft, writing–review and editing, visualization; Shalini Kulasingam, conceptualization, methodology, validation, writing–review and editing.
Ethical Considerations
There was no collection of human subjects data in this study, and ethical approval was not required.
Data Availability
The current article is a computational study, so no data have been generated for this article. This study involved modeling rather than direct analysis of primary datasets. The input data for the current study are available from the corresponding author upon request, with justification as needed. Due to the complexity of the current model code, any request to access and use the model will be based on a review of the request and assumes that there is a sufficient level of resources available for appropriate training and/or University of Minnesota staff time.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
