Abstract
Keywords
Irritable bowel syndrome (IBS) is a common condition impacting over 35 million Americans, 1 leading to health-related quality-of-life losses similar to congestive heart failure or rheumatoid arthritis when severe, 2 and accounting for several billion dollars in health care costs.3,4 IBS is the second-most common cause of missed work-days only to the common cold. 5 In choosing how to treat IBS, costs and health outcomes are equally important factors to IBS patients and their treating providers engaging in shared decisions making, to industry sponsors seeking clinical adoption of approved drugs, and to payers whose essential function is pay for necessary healthcare to manage IBS.
Cost is an increasingly prohibitive barrier to effective patient care yet remains understudied in diseases such as IBS.6–9 To date, most comparative effectiveness studies on IBS involve just that—how efficacy, tolerability, and safety profiles might impact clinical outcomes in ideal scenarios—and do not consider the financial or time burden that add significant barriers to improved health with well-intentioned medical care.10–13 Cost is often less important than clinical outcomes in usual development processes for clinical practice guidelines, and cost is legally prohibited from consideration by the Food and Drug Administration (FDA) in the approval process for new drugs. 14 Despite the lack of published evidence on which to base financial decisions, commercial payers and the Centers for Medicare and Medicaid Services (CMS) remain charged with making necessary coverage decisions which ultimately account for 17.8% of the US gross domestic product. 15 As a result, pricing for IBS treatments and other chronic illnesses remains largely disconnected from clinical outcomes.16,17
Cost-utility analysis maximizes the outcomes achieved per dollar spent from the perspectives of each relevant stakeholder involved in the process of providing or paying for care. 18 These analyses are particularly applicable toward efforts to define the value of competing care options in managed care settings. Recognizing that IBS is most common reason for referral to gastroenterology and accounts for 15% of primary care referrals, providers, and patients urgently need comparative cost and outcomes data in a manner that recognizes limitations of existing evidence. 3 We performed a cost-utility analysis of treatments for constipation-predominant IBS (IBS-C), the most expensive IBS subtype, and to identify important factors which affect treatment preferences to patients and their managed care plans.
Methods
We developed a Markov model to determine the quality-of-life and health care utilization associated with treatment interventions for IBS-C. This study adhered to the CHEERS checklist.
We constructed a panel of IBS experts (study authors) to identify and group treatments for a care-seeking population of patients with moderate-to-severe IBS as well as mild IBS requiring only on-demand symptom-directed therapy. We defined our base-case using disease severity, noting that approaches to IBS-C are similar regardless of age and sex. Toward this end, we included interventions which received 1) a positive recommendation in the American Gastroenterological Association practice guideline for IBS or in recent AGA clinical practice updates, 19 2) a positive recommendation in the American College of Gastroenterology (ACG) monograph, 20 or 3) FDA approval since publication of these documents. The design of each treatment intervention is outlined in Appendix Table 1.
Model Inputs a

CBT, cognitive behavioral therapy; CI, confidence interval; FDA, Food and Drug Administration; FODMAP = fermentable oligosaccharides, disaccharides, monosaccharides, and polyols; QALY, quality-adjusted life year; SSRI, selective serotonin reuptake inhibitor.
Binomial data were handled using a beta distribution in the model.
We stratified analyses into two groups using quality-of-evidence ratings based on GRADE methodology in the ACG monograph: 1) at least moderate quality of evidence (representing patients with moderate-to-severe IBS failing over-the-counter or symptom-directed options; including linaclotide, plecanatide, lubiprostone, and neuromodulators), or 2) low or very low quality of supporting evidence (representing patients with mild IBS seeking only on-demand symptom relief; including peppermint oil, probiotics, anticholinergic antispasmodics, and soluble fiber). Low FODMAP (fermentable oligosaccharides, disaccharides, monosaccharides, and polyols) and cognitive behavioral therapy (CBT) strategies were included in both analyses consistent with usual practice by the expert panel.12,20–22 Our panel deemed these groupings to be consistent with usual care in IBS. Details are provided in Supplementary Table 1 on dosing for each intervention.
Model Design
The model diagram is reported in Figure 1. Assumptions of the model included the following: 1) treatment began immediately and 2) patients continued treatment unless treatment was poorly tolerated or until the patient experienced a serious adverse event requiring discontinuation. The model followed individuals in 4-week cycles to a 1-year time horizon, or until 3 months after the intervention was stopped, such that the model tracked only specific treatment-associated effects based on initial choice of therapy. Health utilities were assigned to individuals based on their IBS symptom severity at the end of each 4-week cycle: treatment response (“adequate relief of global IBS symptoms”) or treatment nonresponse (“not achieving adequate relief of global IBS symptoms”). Patients who are not receiving treatment remain treatment nonresponders. These health states were derived from the PROOF observational cohort study of patients with IBS based on the Rome III definition (79% female gender, mean age of 43.0 [SD = 15.0] years). 2 Changes in costs and health-related quality of life were accumulated at the end of each treatment cycle. A Markov model was used, rather than a decision tree, to account for time-varying effects specifically of treatment discontinuation which generally occur early in therapy. Our time horizon was limited to 1 year to equal the timeframe in which most payer premium and coverage determinations remain stable. This model was evaluated using TreeAge Pro 2019 R1.0 (TreeAge Software Inc., Williamstown, MA).

Model design.
The design of the managed care plan to develop the payer perspective is detailed in the appendix.
Model calibration was performed by comparing the cumulative health care costs of receiving no IBS-C treatment at 1 year of follow-up, 4 and by comparing the cumulative gains in quality-of-life against those extrapolated from 3-month outcomes in the PROOF cohort study. 2
Measuring How Treatment Effects Impacted a Patient’s IBS Symptom Severity
IBS symptom severity was determined on the basis of three distinct treatment factors of efficacy, safety, and tolerability at the end of each 4-week cycle of the model. Efficacy informed the time spent in “treatment responder” health state, while safety and tolerability informed treatment discontinuation rates. Efficacy was defined as the probability of global response to therapy, based on binary endpoints evaluating adequate relief of global IBS symptoms in the active intervention arm of clinical trials. This definition of efficacy allowed the model to account for placebo response, which is 1) an important consideration in managing IBS in clinical practice and 2) varies significantly by intervention. 23 Safety was defined as whether a patient experienced a serious adverse event (SAE), based on Section 5 (“Warnings and Precautions”) of FDA drug labeling, or whether serious adverse events were reported in clinical trials for nondrug treatments (based on the standard FDA definition for an SAE); notably, no SAEs were included in the present study given the safety of IBS-C agents with on-label use. Tolerability was defined as discontinuation of treatment due to an adverse event—noting that this also captures the tradeoffs between the ongoing benefit of taking a drug compared to side effects (or medication ineffectiveness). All model inputs on clinical outcomes were extracted from recent appropriate systematic reviews.10–12,21 However, trials were only included if the methodology matched on-label dosing instructions or usual clinical practice, and if trials adequately reported necessary treatment outcomes.
Translating IBS Symptom Severity into Health-Related Quality-of-Life
Treatment response or nonresponse health states were assigned specific EuroQOL health utility values derived from the PROOF observational cohort study, 2 which were then used to generate quality-adjusted life years (QALYs). In the PROOF cohort, the health utility of global response was 0.78, compared to 0.73 for nonresponse to therapy.
Measuring Treatment Costs
Source for cost inputs are reported in Table 1 and are based on US national data in 2019 US dollars. Payer perspective analysis included all direct costs associated with therapy, as well as outpatient, emergency, and inpatient costs associated with increased health care utilization due to IBS treatment nonresponse. Patient-perspective analyses additionally included work-productivity losses associated with IBS treatment nonresponse, as well as costs of childcare and transportation to medical appointments.
Analyses
Base-case analysis was conducted from payer and patient perspectives. A 1-year time horizon was chosen, consistent with 1) the time horizon for payer budgets, coverage decisions, and premium/deductible determinations; and 2) stability of cost and outcome estimates over this time period. Cost-effectiveness analysis was performed to calculate incremental cost-effectiveness ratios (ICERs), which assess the relative costs needed to achieve the same health gains in choosing one therapy over another. ICERs can be used to compare treatment options, and can also be compared to a payer’s or patient’s willingness-to-pay (WTP) to gain one QALY of health to determine “whether a new treatment is cost-effective” (typically ranging from $50,000 to $150,000/QALY-gained in contemporary economic analyses). ICER values below zero were not reported, due to inherent challenges in interpreting these data. 24 A discount rate of 0% was applied, due to the short time horizon of our study.
Probabilistic sensitivity analyses were performed using a Monte Carlo simulation of 10,000 trials to determine how uncertainty in cost and outcome estimates impacted our findings. Acceptability curves were constructed to understand the likelihood that each treatment would be cost-effective most often across a range of WTP thresholds.
Recognizing limitations in long-term treatment data and heterogeneity in both study design and enrollment numbers in IBS trials, we performed detailed one-way sensitivity analyses for each pair of treatments to account for variation in treatment assumptions within the expected range of values for each model input derived from the literature.
Results
Base-case determinations of costs, health-gains (QALY), and ICER are reported from payer and patient perspectives in Table 2.
Costs and QALYs to Treat IBS-C With Linaclotide, Lubiprostone, Plecanatide, SSRI, Low FODMAP, or CBT a

CBT, cognitive behavioral therapy; FODMAP, fermentable oligosaccharides, disaccharides, monosaccharides, and polyols; ICER, incremental cost effectiveness ratio; QALY, quality-adjusted life year; SSRI, selective serotonin reuptake inhibitor.
The calculated ICERs are based on exact calculations, noting that incremental effectiveness is rounded to the nearest thousandth for the purposes of publication.
Costs and Outcomes of Untreated IBS-C
The baseline cost of untreated IBS-C was $3929.37 from a payer perspective, and $5805.00 from a patient perspective per year. The QALY/year with untreated IBS-C was 0.73.
There were four interventions supported by moderate or strong evidence in clinical practice guidelines for the management of IBS-C, which were included in addition to low FODMAP and CBT: selective serotonin reuptake inhibitors (SSRIs), linaclotide, lubiprostone, and plecanatide. Outcomes for interventions supported by low or very low evidence are reported in the appendix.
Base-Case Analysis From Payer Perspective
From a payer perspective (Figure 2A), SSRI was less expensive and more effective than lubiprostone or plecanatide. Linaclotide was more effective but at significant expense compared to SSRI (ICER = $1,684,547/QALY-gained for linaclotide compared to SSRI). In contrast, low FODMAP and CBT were more effective and only moderately more expensive than SSRI (ICER < $20,000/QALY-gained compared to SSRI). Low FODMAP and CBT were both more effective and less expensive than linaclotide.
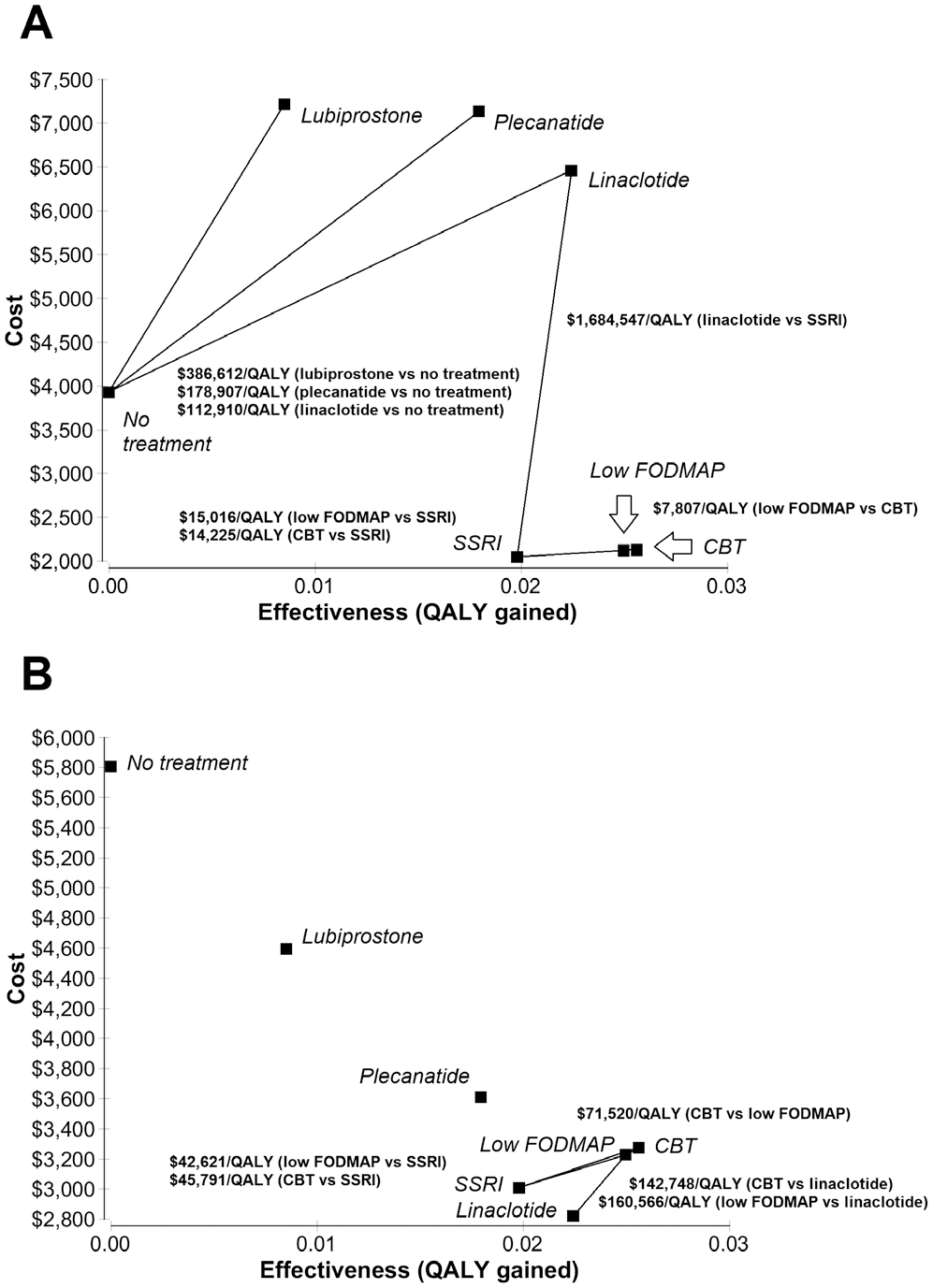
Cost-effectiveness of interventions for IBS-C. Cost-effectiveness is displayed from payer (panel A) and patient (panel B) perspectives for interventions supported by at least moderate level of evidence, as well as low FODMAP and CBT. The horizontal axis shows QALYs (health gains), and the vertical axis shows cost.
SSRI, low FODMAP, and CBT were cost-saving to a payer compared to “no treatment” for IBS-C, with cost savings of approximately $1800 to $1900 per year for patients receiving one of these interventions. In contrast, payers would spend an additional $2531.26 to $3288.63 per-patient annually for patients receiving lubiprostone, plecantide, or linaclotide at their current drug prices compared to “no treatment” for IBS-C.
Base-Case From Patient Perspective
From a patient perspective (Figure 2B), linaclotide was the least expensive intervention for IBS-C and was more effective than SSRI, plecanatide, or lubiprostone. The ICER for low FODMAP ($42,621/QALY-gained) and CBT ($45,791/QALY-gained) were below a willingness-to-pay threshold of $50,000/QALY-gained, compared to SSRI. However, the ICER for low FODMAP ($160,566/QALY-gained) and CBT ($142,748/QALY-gained) were greater than this threshold compared to linaclotide.
All treatments were cost-saving compared to leaving IBS-C untreated. Linaclotide was the most cost-saving IBS-C intervention to a patient at $2982 over 1 year, compared to no treatment. SSRI, low FODMAP, or CBT were less cost saving to patients overall ($2529.21 to $2794.70/year) than linaclotide therapy, but were more cost-saving than plecanatide ($2193.99/year) or lubiprostone ($1208.96/year) referenced against no treatment for IBS-C.
Acceptability Curves
From a payer perspective (Figure 3A), SSRI was more cost-effective than other treatments at low WTP thresholds. Above a WTP threshold of $9000/QALY-gained, low FODMAP or CBT were more cost-effective. From a patient perspective (Figure 3B), linaclotide was the more cost-effective strategy up to a WTP threshold of $110,000/QALY-gained while low FODMAP was the more cost-effective treatment option beyond this threshold. However, it is important to note the significant uncertainty in these findings; no strategy was more cost-effective more than 40% of the time at any WTP threshold.

Acceptability curve of interventions for IBS-C. Acceptability curves are displayed from payer (panel A) and patient (panel B) perspectives.
Specific Factors Influencing Relative Cost-Effectiveness
Appendix Figures 10 to 38 detail the influence of specific model inputs on cost-effectiveness outcomes when comparing two competing IBS-C therapies, from payer perspectives (Appendix Figures 10–23) and patient perspectives (Appendix Figure 24–38). All analyses assessed whether differences in health utility gains with therapy, as well as responder and discontinuation rates would influence treatment preferences based on cost-effectiveness compared to our base-case findings. From a payer perspective, we assessed varying prescription drug costs as well as as direct costs to manage untreated IBS-C or IBS-C with treatment nonresponse. From a patient perspective, we assessed the influence of treatment nonresponse on work productivity and childcare expenses. From a payer perspective, prescription drug costs were the most significant factor influencing treatment preference in these sensitivity analyses. From a patient perspective, loss of work-productivity due to untreated IBS was the most significant factor in sensitivity analyses affecting cost-effectiveness and treatment preference compared to other factors.
Specific to comparison between dietary and psychological treatment options, preference toward a low FODMAP strategy compared to CBT was greater if food costs associated with low FODMAP foods were lower (noting that low FODMAP foods cost an average of 10% more than usual US per-person food costs; Table 1), or if treatment with CBT would require more visits to complete treatment. While preference toward behavioral interventions compared to drug treatments overall was stable in probabilistic sensitivity analysis, the relative treatment preference toward either low FODMAP or CBT varied by the wide range in health outcomes with treatment due to the small size of supporting studies.
Discussion
We performed the first cost-utility analysis for IBS-C that compares cost and health outcomes among competing treatments for patients and payers. To maximize cost-effectiveness and also cost-savings to a payer, SSRI, low FODMAP, and CBT were the least expensive and most cost-effective therapies despite lower levels of supporting evidence, compared to on-label drug treatments. From a patient perspective, on-label drug treatments were the most cost-effective. Among treatments supported by a weak quality-of-evidence, peppermint oil and soluble fiber were the most cost-effective treatment options from a patient perspective.
At a policy level, IBS prescription drug coverage remains just as necessary in the appropriate management of IBS as in other chronic medical conditions. 25 Our findings suggest that dietary and behavioral interventions may appear more cost-effective despite similar clinical effectiveness, a finding largely driven by prescription drug prices. In other words, efforts to address IBS prescription drug prices at a policy level would directly improve treatment satisfaction and adherence,26–28 recognizing that prior authorization restrictions and other insurance barriers directly cause over 1/3 of treatment discontinuations among IBS-C patients who are able to successfully start prescription drug therapy. 29
Factors driving treatment preference based on cost-effectiveness differed between patients and payers in sensitivity analysis. Treatment preferences to payers were largely driven by drug prices and costs of associated IBS care. This contrasted with the patient perspective. In our study, we found that time and financial commitments needed complete treatment (i.e., taking a pill, compared to completing several medical appointments to complete CBT or paying for low FODMAP food) as well as treatment impact on missed work-days due to IBS are important from a patient perspective and should be incorporated into shared decision making.30,31 These findings validate and provide a rationale for common patient questions and discussion topics in managing IBS-C in current practice.32–34
While our model favored SSRI, these drugs are not labeled for an IBS-C indication and have weaker supporting data compared to the FDA-approved IBS-C treatments of linaclotide, plecanatide, and lubiprostone, drugs that were carefully evaluated in rigorous multicenter trials. Payer preference toward SSRI in our model is primarily driven by the comparatively low cost of citalopram ($ 0.03/pill) compared to per-pill costs of linaclotide ($13.57), plecanatide ($13.20), and lubiprostone ($5.92). Even the most expensive SSRI evaluated in an IBS-C trial (paroxetine; $1.90 per pill), resulted in only 14% to 16% the cost of an annual supply of on-label drugs exceeding $4000 annually. Our study does not suggest that SSRI are more effective. Consistent with denials of coverage, prior authorizations, and formulary restrictions which gastroenterologists face in daily practice, 35 our model instead suggests that physicians who use SSRIs in IBS-C may find them more cost-effective than on-label drugs at their current prices as a whole.
Study strengths include rigorous adherence to national guidelines on conducting cost-effectiveness analyses, nationwide data to support generalizability of findings, assessments from patient and payer perspectives, and detailed sensitivity analyses to evaluate the robustness of outcomes for each IBS-C treatment.
We designed our model to evaluate patients covered by a managed care plan with no cost-sharing. This framework remains possible in employer-sponsored plans and Medicaid programs, and exists at the far end of the spectrum from “no coverage at all.” Importantly, the treatment preferences and findings from a payer perspective were not affected by varying reimbursement for the same care (i.e., by varying direct health care costs for untreated IBS-C in sensitivity analysis), suggesting that managed care plans should have similar treatment preferences regardless of funding from private or public sources. However, cost-sharing is increasingly common due to the rise of high-deductible health plans available either in the Healthcare Marketplace (www.healthcare.gov) and employer-sponsored insurance. While we did not explicitly evaluate cost-sharing, cost-sharing would result in the patient perspective aligning more closely with our findings from a managed care perspective.
While our model favored SSRI, these drugs are not labeled for an IBS-C indication and have weaker supporting data compared to the FDA-approved IBS-C treatments of linaclotide, plecanatide, and lubiprostone, drugs that were carefully evaluated in rigorous multicenter trials. Payer preference toward SSRI in our model is primarily driven by the comparatively low cost of citalopram ($ 0.03/pill) compared to per-pill costs of linaclotide ($13.57), plecanatide ($13.20), and lubiprostone ($5.92). Even the most expensive SSRI evaluated in an IBS-C trial (paroxetine; $1.90 per pill), resulted in only 14% to 16% the cost of an annual supply of on-label drugs exceeding $4000 annually. Our study does not suggest that SSRI are more effective. Consistent with denials of coverage, prior authorizations, and formulary restrictions which gastroenterologists face in daily practice, 35 our model instead suggests that providers who use SSRIs in IBS-C may find them more cost-effective than on-label drugs at their current prices as a whole.
Our findings also suggest that dietary and behavioral interventions may be cost-effective; however, this finding has several important limitations. First, dietary and behavioral intervention trials were conducted in pooled IBS or mixed functional bowel disorder populations. Second, the quality-of-evidence to support their use is limited. 21 Third, our findings assume a treatment goal of global symptom improvement, noting that data regarding improvement in constipation in an IBS-C population are lacking with low FODMAP or CBT in the literature.
There are several important limitations toward interpreting our study findings. First, our model inputs are generalized to national data. As with clinical practice guidelines, our model must be adapted to accommodate specific patient and payer characteristics to impact decision making at a local level using our general model construct. Second, adequate relief outcomes on plecanatide are lacking. We mitigated this problem by defining plecanatide efficacy using the FDA abdominal pain responder sub-endpoint and increased plecanatide efficacy in sensitivity analysis to the 95th percentile of response using the FDA bowel symptom responder sub-endpoint (44.6% response rate) as a high estimate, with no change in outcomes. Third, our time horizon was limited to 1 year to equal the timeframe in which most payer premium and coverage determinations remain stable, the lack of availability of clinical trial data beyond 1 year in most cases, the natural history of IBS, and finally the need to monitor IBS patients regularly. Fourth, there are not yet sufficient cost data to include tegaserod or tenapanor in this type of analysis. Fifth, our model does not account for mortality, noting that IBS is a nonlethal illness. Sixth, we did not perform sensitivity analysis on all cost factors, when a lowest fixed cost was available in source data. Instead, we varied only those costs in sensitivity analysis where significant differences exist in practice. Sixth, there is uncertainty in our estimates which is driven as evidenced by the lack of a single dominant treatment in probabilistic sensitivity analysis, which is unsurprising because IBS-C is a complex disease with many possible treatments (many of which are included here). Finally, our findings should only be applied when cost-effective treatments are available and clinically appropriate. Recent data suggest that some IBS individuals have disordered eating behaviors related to long-standing gut symptoms (avoidant restrictive food intake disorder). 36 In these patients low FODMAP may not an appropriate intervention (or at least should be delivered carefully in light of such preexisting behavior patterns). Other individuals with high baseline levels of generalized state-trait anxiety might mitigate the potential benefits of CBT, whereas SSRIs may have multiple benefits for these patients. Thus, it is especially important for payers and providers to understand that treatment selection must depend on individual patient characteristics and not solely on costs alone.
We performed a cost-utility analysis from patient and payer perspectives covering IBS-C treatments recommended in clinical practice guidelines. Our study highlights factors which influence cost-effectiveness from payer and patient perspectives, which are important in patient-centered discussions on appropriate therapy as well as in understanding and guiding payer coverage decisions. Our findings present an opportunity to payers, industry, and policymakers to improve access to effective treatments in broader practice.
Supplemental Material
sj-doc-1-mpp-10.1177_2381468320978417 – Supplemental material for Pharmacologic, Dietary, and Psychological Treatments for Irritable Bowel Syndrome With Constipation: Cost Utility Analysis
Supplemental material, sj-doc-1-mpp-10.1177_2381468320978417 for Pharmacologic, Dietary, and Psychological Treatments for Irritable Bowel Syndrome With Constipation: Cost Utility Analysis by Eric D. Shah, Jessica K. Salwen-Deremer, Peter R. Gibson, Jane G. Muir, Shanti Eswaran and William D. Chey in MDM Policy & Practice
Footnotes
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Shah has no disclosures. Dr. Chey is a consultant for Allergan, Biomerica, IM Health, Ironwood, Outpost, QOL Medical, Ritter, Salix, Urovant and has research grants from Commonwealth Diagnostics, Ironwood, QOL Medical, Salix, Urovant, Vibrant, and Zespri. Dr. Gibson has served as consultant or advisory board member for Atmo Biosciences, Allergan, Celgene, Janssen, MSD, Pfizer, Takeda, and Anatara. His institution has received speaking honoraria from Janssen, BMS, and Pfizer. He has received research grants for investigator-driven studies from MSD and A2 Milk Company.
Monash University financially benefits from the sales of a digital application, online educational course and booklets on the FODMAP diet. The other authors have no disclosures. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Shah is supported by the 2019 American Gastroenterological Association-Shire Research Scholar Award in Functional GI and Motility Disorders, which had no role in the conduct of this study. Financial support for this study was provided in part by a grant from the AGA Research Foundation’s 2019 American Gastroenterological Association-Shire Research Scholar Award in Functional GI and Motility Disorders. The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
