Abstract
Keywords
Atrial fibrillation (AF) is the most common cardiac arrhythmia. 1 Prevalence of AF in the United Kingdom, including undiagnosed cases, has been estimated at 2.4% and prevalence roughly doubles with each decade of age.2,3 AF substantially increases the risk of thromboembolic stroke due to blood pooling in the left atrium and systemic embolization to the brain. More than 20% of the 130,000 annual strokes in England and Wales are attributed to AF. Approximately 2 out of 10 stroke patients do not survive hospital admission, one third recover in 1 month, and the remainder have disabilities needing rehabilitation, making stroke the leading cause of adult disability. 4
Warfarin is an effective oral anticoagulant for the prevention of stroke in patients with AF. 5 Recent estimates suggest approximately 300,000 AF patients in the United Kingdom were prescribed warfarin in 2016.6,7 Warfarin-related bleeding is one of the top five reasons for hospitalization for adverse drug effects in England 8 because of the narrow therapeutic index and numerous drug and dietary interactions. Although the approximate acquisition cost of warfarin is only £10 per patient per year, the requirement for therapeutic monitoring means that the estimated annual cost of managing a patient on warfarin in the National Health Service (NHS) in England and Wales is approximately £283, giving a national annual spend of over £86 million on AF patients. 7 A 2014 NICE (National Institute for Health and Clinical Excellence) report estimated that 29% of AF patients received no treatment, that 22.5% of patients were receiving only aspirin, and that many receiving anticoagulation are not in the optimal therapeutic range. 7
The class of directly acting (or non–vitamin K antagonist) oral anticoagulants (DOACs) include dabigatran (a direct inhibitor of clotting factor II), rivaroxaban, apixaban, edoxaban, otamixaban, and betrixaban (which are factor X inhibitors). These agents have a more rapid onset and offset of action than warfarin and are considered to have more predictable dosing requirements, increasing convenience, and reducing the need for drug monitoring. 9 The estimated annual acquisition cost per patient of new anticoagulants is substantially higher than that of warfarin and will remain so until patent expiry (e.g., 2020 for rivaroxaban). However, the higher acquisition cost could be offset by the reduced need for therapeutic monitoring through anticoagulation services, by increased effectiveness, or by improved safety.
DOACs have been evaluated in clinical trials as an alternative to lifelong warfarin for the prevention of stroke in patients with AF. Some UK studies have assessed cost-effectiveness of individual DOACs10,11 and, more recently, compared multiple DOACs.12–14 However, none of these include edoxaban and all have based their efficacy estimates on individual trials rather than a network meta-analysis (NMA) of all available trials. There is a need to simultaneously compare cost-effectiveness of all DOACs and warfarin in a single model that pools all the available evidence and to estimate the value of further clinical research.
Defining optimal as the strategy with the greatest expected net benefit, our objective is to determine the optimal first-line anticoagulant in the prevention of ischemic stroke for patients with non-valvular AF and not contraindicated to warfarin in the NHS and to estimate the value of further clinical research on the effectiveness of DOACs. Patients include those with paroxysmal, persistent, or permanent AF who are considered eligible for anticoagulation by their physician. The article is organized as follows. The methods section first describes the model structure, then describes treatment strategies and switching rules, then describes sources and assumed or estimated values for the model inputs, and closes by describing sensitivity analyses. We then give the results of our economic evaluation and finish with a discussion of our findings in the context of previous research and implications for policy.
This work was part of a larger project on the effectiveness and cost-effectiveness of DOACs for AF and for the primary prevention, treatment, and secondary prevention of venous thromboembolic disease. 15 The clinical results and headline cost-effectiveness results have been published elsewhere, 16 but the present article gives greater detail on the cost-effectiveness analysis. We additionally present results of a full value of information analysis.
Methods
We performed a search of the literature to identify previous model-based cost-effectiveness analyses addressing our decision question (for full details see Sterne et al 15 and Lopez-Lopez et al 16 ). We developed the structure of our models from a critical appraisal of these previous models together with discussions with clinical experts and patient representatives. We chose to develop a new model structure, as no previous model had compared all relevant DOACs or included all details identified as relevant by our clinical experts; however, our model shared many features with previous models. Our economic evaluation was conducted in parallel to a systematic review and NMA of efficacy and safety data.15,16
Model Structure
We used a Markov model with a cycle length of 3 months and time horizon 30 years to compare patients assigned to warfarin or DOAC as first-line treatment. The model structure is illustrated in Figure 1. Patients enter the model in the AF well state, where they are assumed to have no history of stroke, myocardial infarction (MI), bleeding or intracranial hemorrhage (ICH). At any state in this model, patients can experience an ischemic stroke, extracranial bleed, ICH, MI, transient ischemic attack (TIA), systemic embolism (SE), or all-cause death. No distinction is made between the severities of ischemic stroke. An extracranial bleed includes major or non-major clinically relevant bleed, without distinguishing bleed location (e.g., gastrointestinal bleeding). Non–clinically relevant bleeds are not explicitly modelled as their impact on costs and utilities is assumed to be low. All-cause death includes fatal strokes, bleeds, ICH, and MI as many randomized controlled trials (RCTs) do not report cause specific mortality. States are used to record a history of stroke, MI, bleed, or ICH and states representing a history of one, two, three, or all four of these events differ in their costs, utilities, and risks of future events. SE and TIA have a transient cost and disutility but are assumed not to have permanent effect on event risks. As patients age in the model event rates, utilities, and costs change as described below. Apixaban and dabigatran have different dose recommendations for older and younger patients but our model compared only the higher doses (apixaban 5 mg and dabigatran 150 mg) that are recommended for younger patients, and makes the simplifying assumption that dose does not reduce with age. 17 It should be noted that the price of the different doses are the same (Table 5 in Appendix 1). Lower doses, with different event rates, are explored in sensitivity analysis.
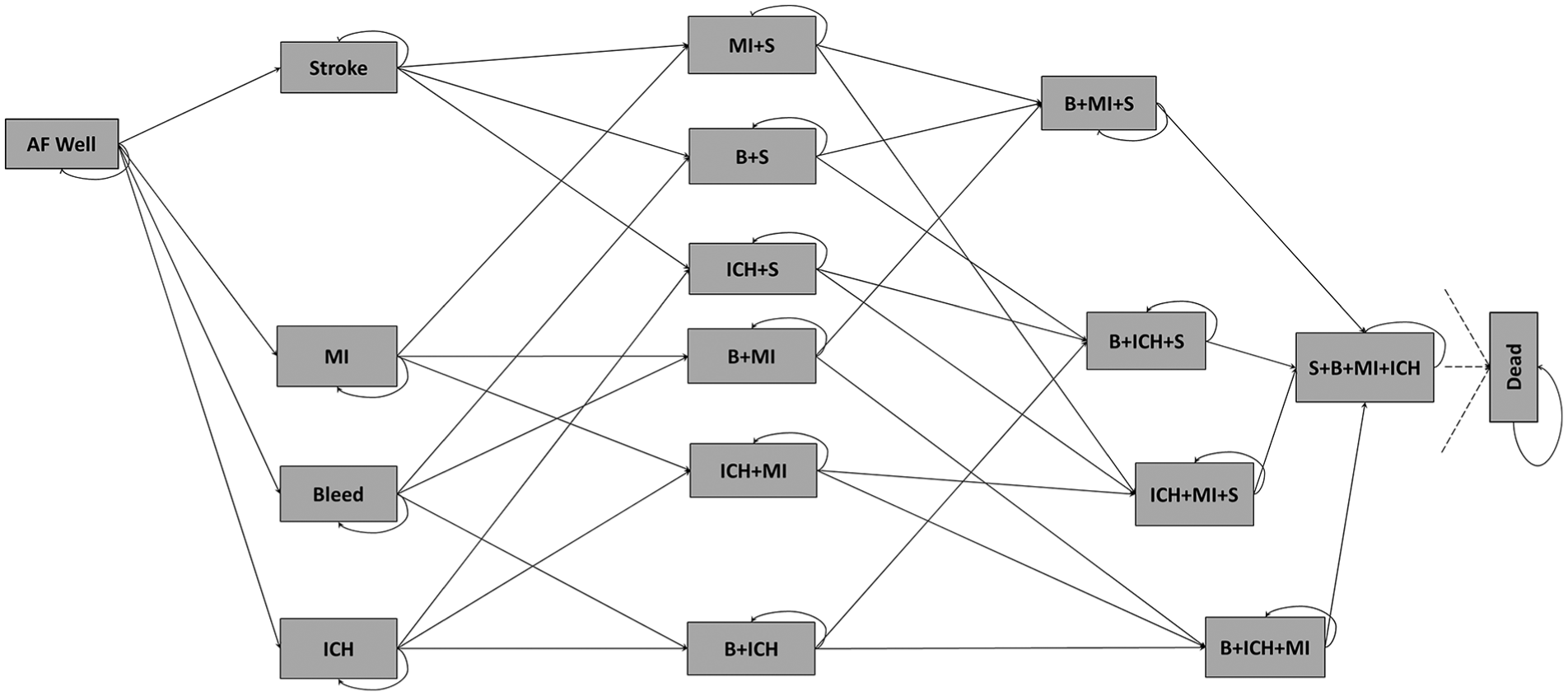
Illustration of the Markov model for atrial fibrillation. Patients can experience transient events (TIA or SE) but stay in same health state, with possibly changed treatment, thereafter. B, clinically relevant bleed; ICH, intracranial hemorrhage; MI, myocardial infarction; S, ischemic stroke. Adapted from Sterne et al. (2017) 15 under Creative Commons Attribution (CC BY 4.0) license.
The patient cohort represented an “average” patient with age 70 (SD = 8) years and 60% male, with characteristics chosen to match average patients in the RCTs identified in the efficacy and safety review.15,16 These RCTs were considered to be representative of the AF population in England and Wales. Different ages at treatment initiation were explored in sensitivity analyses discussed below. These model assumptions are summarized in Table 1.
Main Assumptions in the Atrial Fibrillation Model

AF, atrial fibrillation; DOAC, directly acting oral anticoagulant; ICH, intracranial hemorrhage; MI, myocardial infarction; NICE, National Institute for Health and Clinical Excellence; QALY, quality-adjusted life-year; RCT, randomized controlled trial; SE, systemic embolism; TIA, transient ischemic attack.
For full justification, please see main text.
Treatment Strategies and Treatment Switching
The reference first-line treatment strategy was warfarin with international normalized ratio (INR) target range of 2 to 3 with the option to switch to no treatment. The DOACs comparator strategies were apixaban 5 mg twice daily, dabigatran 150 mg twice daily, edoxaban 60 mg once daily, and rivaroxaban 20 mg once daily, doses recommended by NICE, 17 all with the option to switch to warfarin INR 2 to 3 and then no treatment, or directly to no treatment following serious adverse events. Aspirin is not recommended by NICE so is not included as a first-line strategy for comparison. 17 Our model only incorporated treatment switching as a result of an acute event. Switching from DOAC to warfarin or no treatment was modelled as a probability of switching following acute events, with details below. We were unable to include betrixaban due to lack of evidence.
The same model structure is used for the different first-line treatment options. If a patient switches from a DOAC to warfarin, they maintain their event history but their remaining lifetime is estimated assuming event risks, costs, and utilities corresponding to the warfarin model. Various treatment switching rules and probabilities were assumed. Due to the higher risk of MI on dabigatran, patients on dabigatran who experience an MI are assumed to always switch to warfarin,18,19 while patients on other treatments do not switch following an MI. Patients who experience an ICH, whether on warfarin or a DOAC, will always switch to no treatment. Following a stroke or bleed, patients have a treatment switching probability, specified in Table 2, of switching from a DOAC to warfarin or from warfarin to no treatment. Patients experiencing SE and TIA make the same transition with a lower treatment switching probability. We used Beta distributions to best represent clinical advice on the uncertainty in these probabilities.
Summary of Estimates of Treatment Switching Probabilities, Costs, and Utilities Used in the Economic Model

BNF, British National Formulary; CI, confidence interval; CRB, Clinically relevant bleed; CrI, credible interval; DOAC, directly acting oral anticoagulant; ICH, intracranial hemorrhage; MI, myocardial infarction; NHS, National Health Service; ONS, Office of National Statistician; SE, systemic embolism; TIA, transient ischemic attack.
Capped above at 1 for quality of life and 0 for disutility.
Following stroke, bleed, SE, or TIA, patients may switch from DOAC to warfarin or from warfarin to no treatment with the specified probabilities.
We inflated to 2013/2014 values using the ONS Consumer Price Inflation index for medical services (DKC3)58 and placed a Uniform distribution ∼(52.57, 157.70) and (210.26, 630.79) (on the cost per 3 months and yearly cycles, respectively).
We inflated to 2013/2014 values using the ONS Consumer Price Inflation index for medical services (DKC3). 58
Average of gastrointestinal and non-gastrointestinal bleed.
Disutilities assumed to last for 3 months.
Table 3, year mean EQ-5D score.
Table 2 in source article, weighted average EQ-5D score for ischemic stroke.
Table 3 in source article, EQ-5D for hemorrhagic stroke.
Model Inputs
A systematic literature review was used to identify RCTs comparing DOACs to warfarin and reporting on the events of interest in our model,15,16 This search identified 23 RCTs for inclusion, which were synthesized in a competing risks NMA. The hazard ratios estimated from the NMA for the seven events relative to warfarin are summarized in Table 7 of the appendix. Note that evidence on TIA was weak, as represented by very wide credible intervals that indicate no evidence of a difference between treatments on this outcome. The absolute hazard of events on warfarin were estimated using a competing risks single-treatment meta-analysis based on data from RCTs that included a warfarin arm. 20 Because our model allows treatment switching to “no treatment,” we require event hazard estimates under no treatment. We estimated these hazard ratios of events on warfarin compared to placebo/no treatment using a meta-analysis of studies identified in a published systematic review.5,15,16 We assumed there was no effect of taking warfarin on MI rates due to a lack of evidence.
A baseline all-cause mortality rate for patients on each DOAC was estimated using NMA but Office of National Statistician (ONS) lifetables were used to estimate the increase in underlying all-cause mortality as the model cohort ages, assuming a 60/40 male/female gender split. Non-mortality event rate estimates represent an average over the characteristics of the RCT populations. The effect of prior events on future risks are reported in the appendix (Table 6). A Swedish cohort study of AF patients 21 estimated hazard ratios for stroke, ICH, bleeds, and MI in patients with a history of each of these events compared to patients with no history of these events. A Danish registry study of AF patients 22 reported the effect of history of MI or stroke on mortality. We assumed the impact of a history of clinically relevant bleed or ICH on mortality would be the same as that for stroke, but explored this in sensitivity analysis. The effect of prior events on future risks was assumed to be additive on log hazard scale.
Acute event costs and costs per cycle are summarized in Table 2; when these were based on NHS reference costs, uniform distributions were assumed due to the absence of evidence on uncertainty. The cost for a DOAC was calculated using the BNF (British National Formulary). 23 It was assumed that DOACs incurred no monitoring or administration costs. Warfarin costs were estimated from a NICE report and, due to lack of evidence on uncertainty, assumed to follow a uniform distribution with upper and lower bounds 50% and 150% of this estimate, respectively. 24 No long-term management costs were assumed to be incurred by patients experiencing an MI or extracranial bleed. Following clinical advice, the post-ICH management cost was assumed to be the same as the post-stroke management cost, itself an average cost of management of mild, moderate, and severely disabling stroke reported by Luengo et al. 25 The cost per cycle for patients with a history of both stroke and ICH was assumed the same as for a history of only one of these events.
Acute event disutilities and utilities per cycle are summarized in Table 3. The disutilities for TIA and SE were assumed to be the same and estimated using a UK standard gamble study of AF patients. 26 The same study was used to inform the disutility of a stroke or clinically relevant (non-ICH) bleed. A computerized preference survey of healthy US patients was used to estimate the disutility of an ICH. 27 A longitudinal study of English patients discharged from hospital following acute MI provided an EQ-5D-based estimate of MI disutility. 28 The utility for a cycle spent in the post stroke, MI, and ICH states was based on a German cohort study of post stroke patients using the German EQ-5D 29 ; we used a beta distribution for the post-ICH utility as this was based on only five patients. The utility for post extracranial bleed was assumed to be the same as for post stroke. Utilities for chronic health states are assumed to be multiplicative. For example, the utility of a patient who has experienced both an ischemic stroke and a myocardial infarction will be the product of the two utility scores. The state utilities were assumed to reduce with age by factors estimated relative to a reference age (65–75), based on general population EQ-5D utility estimates. 30 Utilities were weighted by the 60/40 gender split in gender so differences between genders were not explicitly modelled.
Cost Effectiveness of First-Line Treatment Strategies for Atrial Fibrillation Patients a

INR, international normalized ratio; QALY, quality-adjusted life-year.
Expected (mean) values are reported with 95% confidence intervals. Incremental values are relative to warfarin (INR 2–3). Incremental net benefit is the difference in QALYs and costs for willingness to pay per QALY thresholds of £20,000 and £30,000.
Our model is fully probabilistic, and we will present mean and 95% credible intervals (CrI) for the estimated total and incremental costs, quality adjusted life-years (QALYs), and net benefits. The upper and lower limits of the 95% CrI correspond to the 2.5% and 97.5% percentiles of the probability sensitivity analysis samples. If a 95% CrI for incremental costs, QALYs, or net benefits excludes zero, it indicates at least 97.5% Bayesian probability that the comparator has a higher value than the reference on that outcome.
Sensitivity Analyses
Sensitivity analyses were conducted to test the robustness of the analysis to our assumptions. Two sensitivity analyses were conducted analyzing cohorts initially aged 60 or 80 years old. Additionally, we explored the sensitivity to warfarin monitoring costs, effects of ICH and bleed on future mortality, treatment switching rules, the data for our meta-analysis comparing warfarin and no treatment, and the assumed hazard ratio of ICH on warfarin relative to no treatment. The base case assumes no long-term management costs for MI so this was increased to £142 per year, in line with the Bayer single technology assessment for rivaroxaban. 10 The lower doses of 2.5 mg for apixaban and 110 mg for dabigatran were explored as treatment strategies in a further sensitivity. As the DOACs are due to come off-patent and thus reduce in price, threshold analyses were conducted to explore the price at which each DOAC becomes the optimal strategy, assuming other DOAC prices remain fixed. Monitoring costs, priced at 50% those of warfarin, were added for the DOACs. Changed all beta and uniform distributions to Normal. Reran the model comparing DOACs and warfarin for patients with a history of stroke. Reran assuming patients do not switch treatment following MI on dabigatran, as for other treatments. Table 9 in the appendix provides more detail on these sensitivity analyses.
Value of Information Analyses
To assess the impact of uncertainty on decision making and measure the value of further research, expected value of perfect information (EVPI) and expected value of partial perfect information (EVPPI) were computed. 31 The EVPI and EVPPI measure the value of removing all uncertainty around all or partial subsets of parameters, respectively. If EVPPI for parameter subsets are close to the total EVPI, it suggests the most important subsets have been identified. Parameter subsets considered were all costs, only event costs, only state costs, all utilities, only event utilities, only state utilities, treatment switching probabilities, impact of past events on future risks, baseline (warfarin) log hazards, and DOAC treatment effects relative to warfarin. In addition, we calculated EVPPI for the relative effects parameters that would be informed by a two-arm RCT comparing the top two cost-effective treatments, a three-arm RCT comparing the top three cost-effective treatments, and so on for four-arm and five-arm RCTs. Table 4 provides more details on investigated parameter sets. EVPPI was estimated using up to 128 inner samples and 1024 outer samples of Monte Carlo simulation, chosen to provide low bias and variance in estimates.31,32 Assuming an incidence of 1%, 33 and that there are 500,000 70-year-olds in England and Wales, 34 there are 5000 new cases of AF every year. Population EVPI and EVPPI were therefore calculated assuming 5000 new AF patients per year, discounting at 3.5% per year, and summing over a 10-year technology lifetime. As a sensitivity we also estimate using a 2-year technology horizon to represent the impact of patent expiry from 2020 onwards.
Expected Value of Partial Perfect Information Results a
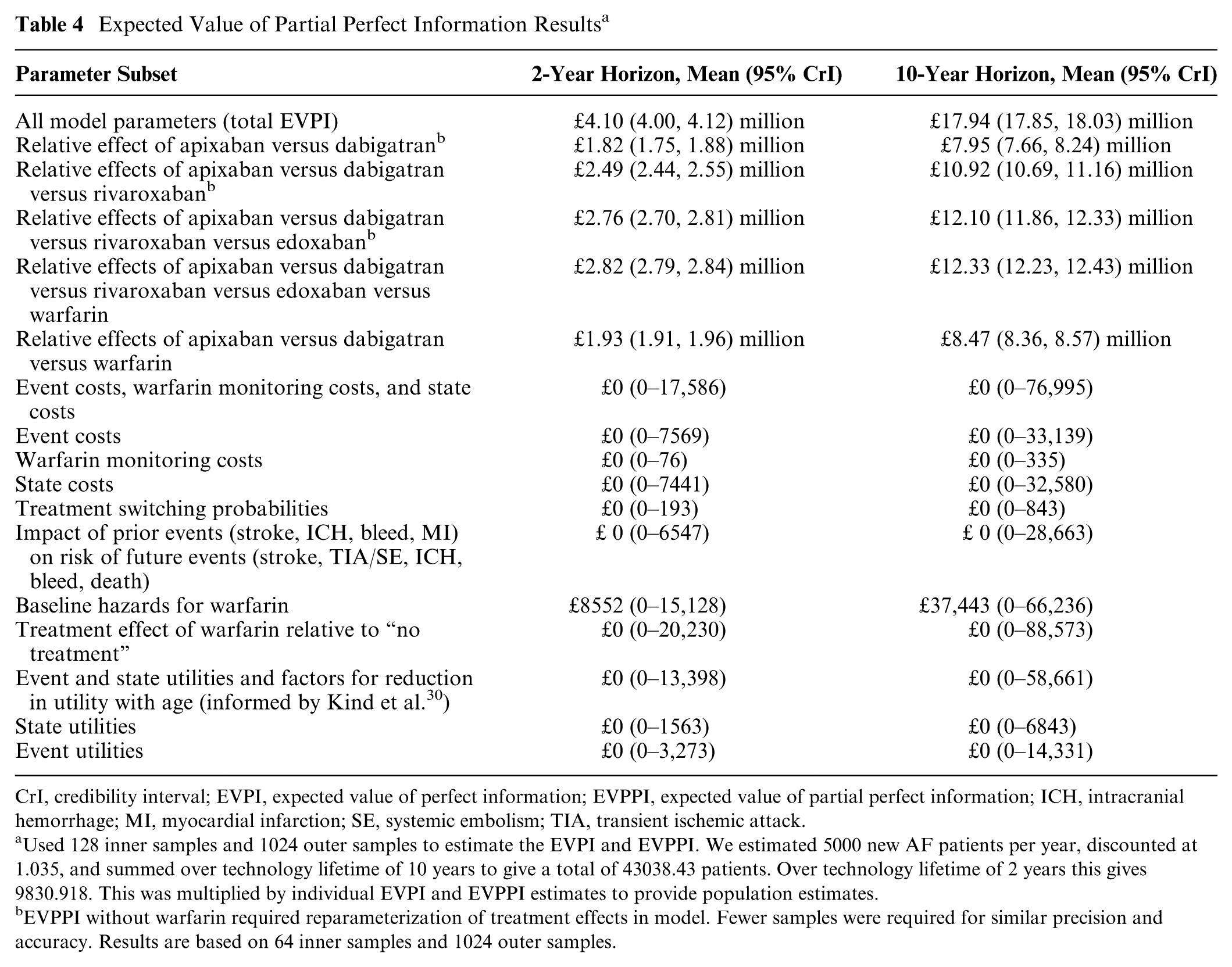
CrI, credibility interval; EVPI, expected value of perfect information; EVPPI, expected value of partial perfect information; ICH, intracranial hemorrhage; MI, myocardial infarction; SE, systemic embolism; TIA, transient ischemic attack.
Used 128 inner samples and 1024 outer samples to estimate the EVPI and EVPPI. We estimated 5000 new AF patients per year, discounted at 1.035, and summed over technology lifetime of 10 years to give a total of 43038.43 patients. Over technology lifetime of 2 years this gives 9830.918. This was multiplied by individual EVPI and EVPPI estimates to provide population estimates.
EVPPI without warfarin required reparameterization of treatment effects in model. Fewer samples were required for similar precision and accuracy. Results are based on 64 inner samples and 1024 outer samples.
Results
The total and incremental costs, QALYs, and net benefits at both a £20,000 and £30,000 willingness-to-pay threshold, for a cohort with initial age 70, are presented in Table 3. Cost-effectiveness acceptability curves are presented in Figure 2. All the DOACs have higher expected incremental net benefits than warfarin at both thresholds but only apixaban 5 mg had a 95% CrI that excluded zero. Apixaban 5 mg had the highest expected incremental net benefit and its probability of having the greatest net benefit was close to 60% at both thresholds. Dabigatran 150 mg had the second highest incremental net benefit and a probability of having greatest net benefit above 20% for both thresholds. The probability that warfarin had highest net benefit was 0% at both thresholds, indicating there is little uncertainty that a DOAC is optimal. All DOACs except rivaroxaban 20 mg had lower expected costs than warfarin but all estimates of incremental costs were uncertain with 95% CrI crossing the zero line. All DOACs also had higher expected QALYs but again all 95% CrI indicated high uncertainty. Dabigatran 150 mg had the lowest expected costs while rivaroxaban 20 mg had the highest expected costs of all anticoagulants, including warfarin. Apixaban 5 mg had highest expected QALYs while edoxaban 60 mg had lowest out of the DOACs and warfarin had lowest overall.
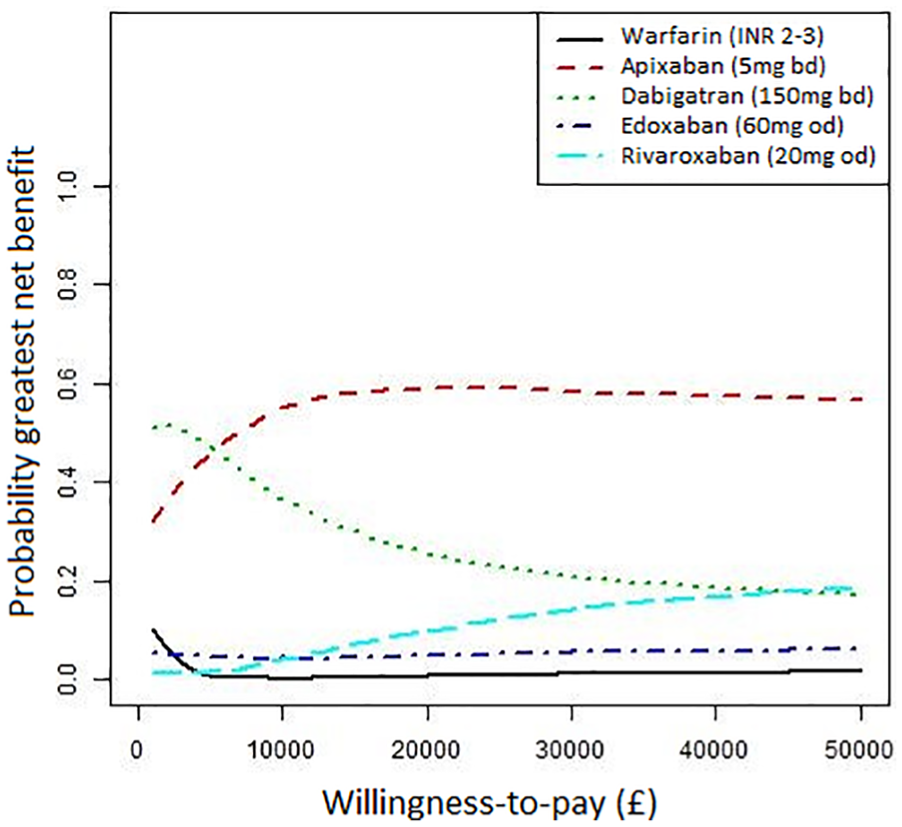
Cost-effectiveness acceptability curves. The probability each first-line treatment is has highest net benefit against willingness to pay per QALY threshold. Adapted from Sterne (2017) 15 under Creative Commons Attribution (CC BY 4.0) license.
The estimated hazard ratios of events and their expected impacts on costs and utilities explain these findings. Table 8 in the appendix presents the proportion lifetime spent with history of each of the events with lifetime consequences; these indicate stroke and ICH are less common on DOACs than on Warfarin but extracranial bleeds are more common. On the two most optimal treatments, dabigatran has less common stroke and ICH than apixaban but this is offset by more frequent MI and extracranial bleeds.
Sensitivity analyses, results of which are summarized in Table 9 of the appendix, confirmed that the conclusion of apixaban 5 mg being optimal was robust to assumptions in the model. Apixaban 5 mg was also optimal in cohorts with initial age 60 or 80 years old. As lower doses of apixaban and dabigatran are recommended in older patients, 2.5 mg and 110 mg, respectively, we conducted a sensitivity analysis with these doses but apixaban remained optimal. However, warfarin became optimal when it was assumed that patients would always switch after a stroke, bleed, SE, TIA, ICH, and MI (if on dabigatran), due to patients spending less time on a DOAC. Sensitivity analysis demonstrated that, holding prices of other treatments fixed, warfarin would have to reduce from £404.52 to −£1393.79, dabigatran would have to reduce in annual price from £801.76 to £487.86, edoxaban from £801.76 to £222.93, and rivaroxaban from £766.52 to £201.47, to have greater net benefit than apixaban. This represents a substantial discount from their current prices; further details are provided in Table 8 and Figure 4 of the appendix.
Results of the value of information analysis are summarized in Table 4. Using a 10-year time horizon, the total estimated population EVPI was £17.94 million (17.85 million, 18.03 million), suggesting high potential value in research reducing uncertainty about the cost-effectiveness of DOAC treatment. The EVPPI was effectively zero for all cost, utility, switching parameters, and impact of previous events on future risk parameter subsets, and was only £37,443 (0, 66,236) for event hazards on warfarin. This minimal impact of cost uncertainty on the results supports our threshold analysis that the conclusion of apixaban being optimal is relatively insensitive to price reductions in other DOACs. The key uncertainty contributing to EVPI is from the relative effectiveness of the different anticoagulants. EVPPI for the relative effect of apixaban versus dabigatran (the treatments with highest and second highest expected net benefit, respectively) was £7.95 million (7.66 million, 8.24 million) but adding the relative effect of rivaroxaban raises EVPPI to £10.92 million (10.69 million, 11.16 million) and further adding edoxaban raises EVPPI to £12.10 million (11.86 million, 12.33 million). Further adding the relative effect of warfarin (giving a five-arm RCT) only increases EVPPI to £12.33 million (12.23 million, 2.43 million), suggesting little additional value from including warfarin. Using only 2-year time horizon, the population EVPI was £4.10 (4.08, 4.12) million and the EVPPI for an apixaban versus dabigatran trial was £1.82 (1.75, 1.88) million, still suggesting potential value, although adding rivaroxaban increases it to £2.49 (2.44, 2.55) million, which may be more convincing. That the EVPPI are approaching the total EVPI suggests the most important parameters, namely, the relative effects, have been identified.
Discussion
At a willingness-to-pay threshold of £20,000 per QALY, all DOACs had higher expected net benefit than warfarin. Warfarin was dominated, with lower effects but higher costs, than all DOACs except rivaroxaban and had a 0% probability of having highest net benefit at £20,000. There is considerable uncertainty between the DOACs, but apixaban 5 mg twice daily had the highest expected net benefit and the highest probability (60%) of having highest net benefit as first-line anticoagulant for the prevention of stroke in AF. This conclusion held for cohorts of initial age 60, 70, and 80. Conclusions were robust to all sensitivity analyses except when assuming patients switch to warfarin following any adverse clinical event, when warfarin is optimal. The differences in lifetime total cost, QALYs, and net benefit, compared with the totals, were very small and the 95% credible intervals were overlapping. Our results therefore highlight uncertainty as to which DOAC is optimal.
Value of information analysis suggested there was potential value in reducing uncertainty in relative efficacy of the DOACs, but limited value in further warfarin comparisons or investigating other model inputs such as costs. In addition to directing future research, EVPI and EVPPI quantify the sensitivity of the decision to parameter uncertainty by combining the probability that a decision is wrong with the consequences of a wrong decision. 35 However, EVPPI assumes all uncertainty is eliminated, while in practice the uncertainty reduction will depend on sample size and follow-up time. Given the low event rates and chronic nature of AF, a very large trial may be required and the costs of such a trial may well not outweigh the benefits. Of the research designs we explored, a two-arm RCT comparing apixaban versus dabigatran is most likely to be value for money. An expected value of sample information (EVSI) and expected net benefit of sampling (ENBS) analysis is required. 36 However, the nonlinearity of the net benefit function and high dimensionality and correlation of the input parameters for our model makes nested Monte Carlo simulation infeasible. Efficient approximation methods for EVSI exist but rely on the suitability of a linear approximation, which is not appropriate for our model.37,38 This is an area for further research. 39
That apixaban 5 mg had the greatest expected net benefit was driven by its higher expected QALYs than dabigatran. As stroke and MI have similar chronic health utilities and acute disutilities (Table 2), these higher QALYs were explained by the advantage of apixaban over dabigatran on hazard of MI being greater than its disadvantage on stroke reduction. That dabigatran 150 mg had the lowest expected costs is driven by its low hazard of ICH and stroke, the events with highest acute costs (Table 2), which overcame its greater hazard of MI, which had a lower acute cost. However, apixaban 5 mg also had low expected costs due to its low hazard of ICH, so the finding that it has the highest expected net benefits is still driven by the advantage on QALYs. Conversely, rivaroxaban 20 mg had the highest expected cost primarily because of its limited reduction in ICH risk, which drove its low incremental net benefit over warfarin. All DOACs had similar reduction in risk of death compared to warfarin and so this was not a key driver of the model results.
On the efficacy and safety parameters, there were no direct head-to-head RCT comparisons between different DOAC drugs—all such comparisons were based on indirect evidence derived from the networks. Also, the profile of patients entering trials may not be the same as those treated in practice, who may be older and have more comorbidities. Considering AF is a lifetime chronic condition, the trials have also been of relatively short duration. We relied on warfarin arms of the trials to estimate absolute event rates but reliable estimation of cost-effectiveness in different clinical scenarios requires high-quality data on absolute event rates. NHS health record data could provide further evidence on absolute event rates. Rare adverse effects that remained undetected during drug development may come to light with high-volume use post licensing.
Our model made various simplifying assumptions. We assumed that nonmortality event rates do not depend on age. We do not distinguish between minor and major stroke and assume the split of minor and major stroke does not differ across treatments. Some previous models split stroke by severity40–42 but we found insufficient evidence to estimate rates differentially. Following an MI, we assumed patients on dabigatran switch to warfarin, but those on other DOACs do not switch. We only considered simple treatment sequences of first-line DOAC followed by warfarin followed by no treatment or first-line warfarin followed by no treatment; wider comparison of sequences of DOACs and warfarin could be interesting for future research. We assumed that SE and TIA are transient events with no long-term consequences. However, severe consequences such as limb loss are extremely rare, so are unlikely to affect results. We did not account for costs due to unrelated diseases, which can reduce the cost-effectiveness of interventions that extend life; however, there is not yet consensus on how to account for unrelated costs and including them is not standard practice. 43 Finally, we do not distinguish between types of AF despite increasing evidence that stroke risk increases as AF progresses from paroxysmal to persistent to permanent.44,45
In addition to using RCTs to estimate relative effects, we also used their warfarin arms to estimate baseline event risks. Baseline characteristics across RCTs were similar, suggesting our first-line population of interest could be similar across populations. In practice, however, patients may be older and have more comorbidities. Time in therapeutic range for warfarin varied considerably in the RCTs and may be lower in practice. 15 Although registry studies in Danish and Swedish populations have been published, we could not identify studies in the United Kingdom.21,22 These limitations prevent us from making rigorous patient-specific recommendations; the similarity in net benefits across DOACs suggest the choice be left to physicians for individual patients. If the necessary RCT data could be obtained, NMA using individual participant data to identify optimal patient populations could be useful further research. 46
The use of DOACs may be associated with a number of issues including class- and drug-specific cautions and contraindications, the potential for subtherapeutic dosing, reduced adherence due to lack of regular monitoring, absence of, or limited experience with antidotes, as well as the added cost of maintaining stocks of numerous different anticoagulants and the potential for prescribing errors due to unfamiliarity. We have not accounted for these in this model because the impact of some of these potential problems is untested within RCTs and will only emerge once uptake of the drugs is greater. Despite NICE estimates that 29% and 22.5% of AF patients received no treatment or only aspirin, respectively, we did not model these as they are no longer recommended. 7 Previous studies have demonstrated cost-effectiveness of warfarin over aspirin.47,48 We can therefore infer that DOACs would be cost-effective compared with aspirin and no treatment. Patients currently on aspirin or no treatment due to difficulties on warfarin may now be offered DOACs.
Several cost-effectiveness analyses of DOACs compared to warfarin for the prevention of stroke in AF in the UK context have been published.10–14 These found individual DOACs to be cost-effective compared to warfarin, in line with our finding that all DOACs have greater expected net benefits than warfarin. The NICE technology appraisals (TA) also found DOACs to be cost-effective and recommended dabigatran (TA249), rivaroxaban (TA256), apixaban (TA275), and edoxaban (TA355). Three published cost-effectiveness analyses compared apixaban, rivaroxaban, dabigatran, and warfarin.12–14 Of these, only Verhoef et al. 13 disagreed with our finding that apixaban was the optimal; these authors found dabigatran 150 mg to be the optimal. This difference is likely because their model had only a single post-event disability state, so did not include the impact of MI on future event risks. Dabigatran’s higher risk of MI is lessened if the consequent increase in stroke, death, and TIA/SE risk is omitted. In addition, all previous analyses used only single pivotal trials on each DOAC and did not use NMA. They also did not include edoxaban. Multiple treatment assessments of the DOACs conducted in other countries support our finding that apixaban was optimal, including in the United States,49,50 Belgium,51,52 France, 53 and Taiwan. 54
Our study is the first to simultaneously compare the cost-effectiveness in the UK context of all licensed anticoagulants, including edoxaban 60 mg, and to make use of all available evidence through NMA. Ours is also the first to include comprehensive value of information analysis. Our findings, in particular that apixaban 5 mg twice daily is optimal, will be useful for guidelines and practice, while our value of information analysis may direct future research.
Supplemental Material
Appendices_-_rev1_-_clean.rjf_online_supp – Supplemental material for Directly Acting Oral Anticoagulants for the Prevention of Stroke in Atrial Fibrillation in England and Wales: Cost-Effectiveness Model and Value of Information Analysis
Supplemental material, Appendices_-_rev1_-_clean.rjf_online_supp for Directly Acting Oral Anticoagulants for the Prevention of Stroke in Atrial Fibrillation in England and Wales: Cost- Effectiveness Model and Value of Information Analysis by Howard H. Z. Thom, Will Hollingworth, Reecha Sofat, Zhenru Wang, Wei Fang, Pritesh N. Bodalia, Peter A. Bryden, Philippa A. Davies, Deborah M. Caldwell, Sofia Dias, Diane Eaton, Julian P. T. Higgins, Aroon D. Hingorani, Jose A. Lopez-Lopez, George N. Okoli, Alison Richards, Chris Salisbury, Jelena Savović, Annya Stephens-Boal, Jonathan A. C. Sterne and Nicky J. Welton in MDM Policy & Practice
Footnotes
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HHZT reports personal fees for consultancy work from Pfizer, Novartis Pharma, Hoffman La-Roche, and Eli Lilly and company, all outside this work. NJW and SD report an MRC funded project in collaboration with Pfizer Ltd. Pfizer part-fund a researcher. The project is purely methodological using historical data unrelated to this work. DE reports support from Boehringer Ingelheim, Pfizer, Bayer, Leo Pharmaceuticals, and Bristol-Myers Squib, all outside this work. The authors declare no support from any other organization for the submitted work; no financial relationships with any other organizations that might have an interest in the submitted work in the previous three years; and no other relationships or activities that could appear to have influenced the submitted work
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided in part by a grant from the UK government agency National Institute for Health Research (HTA Grant 11/92/17), the UK Medical Research Council Network of Hubs for Trials Methodology Research (MR/L004933/2- N79), and the UK Medical Research Council ConDuCT-II Hub for Trials Methodology Research (MR/K025643/1). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report. The following authors are employed by the sponsors: WF and ZW (MR/L004933/2- N79); HHZT, WH, and NJW (MR/K025643/1). JACS was partly funded by a National Institute for Health Research (NIHR) senior investigator award (Ref NF-SI-0611-10168). PAD and JS were partly funded by the NIHR Collaboration for Leadership in Applied Health Research and Care West (CLAHRC West).
AR was supported by the NIHR CLAHRC West. The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR, or the Department of Health and Social Care. HT was partly supported by the National Institute for Health Research (NIHR) Biomedical Research Centre at University Hospitals Bristol NHS Foundation Trust and the University of Bristol.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
