Abstract
Keywords
Over the past decade, there has been increasing support for more active patient involvement in medical decision making.1–6 Shared decision making (SDM) is a process in which clinicians and patients work together to arrive at a mutually agreed upon decision after considering evidence and patient preferences, and it has been touted to be imperative for true informed consent. 7 The SDM process lies between paternalistic decision making, in which doctors make decisions for the patients, and autonomous decision making, in which patients are asked to make decisions on their own and without inputs from doctors. SDM is particularly relevant for decisions where there is clinical equipoise, that is, no “right” or “wrong” answer based on a universally agreed upon good outcome. These decisions are referred to as preference-sensitive/value-laden (decisions for which there is clinical equipoise, which therefore depend on patient preferences) 6 and, for true informed consent, require: 1) the exchange of information about baseline disease prognosis, treatment choices, and likely outcomes; 2) the exchange of information about patient goals and preferences in light of these treatment choices; and 3) providers helping patients to “weigh” what choice is best for them based on their preferences.8–12
As doctors are being urged and incentivized to employ SDM for value-laden decisions, it is unclear whether this style of decision making is associated with improved outcomes. Furthermore, if there are positive associations, it is unclear which components of SDM are necessary to achieve these outcomes. Although most patients want to be informed of their disease diagnosis, preferences for decision making style and degree of input from the doctor vary between patients and may depend on the type of decision being made. For example, patients with life-threatening conditions and older patients may be more likely to want to leave the decision making to the doctor. 13 Other factors influencing preference for decision making style may be race, socioeconomic status, and country of origin.14,15 In a recent survey of patients at an inner-city clinic in New York City, which provides care for largely economically disadvantaged patients, many of whom are recent immigrants and do not speak English as a first language, we found that most participants described some elements of SDM with their doctors, including discussing the risks and benefits of potential treatments. 16 Most patients expressed the desire to be involved in decision making with their doctor, but many deferred final treatment decisions to their doctors.
Although decision aids designed to support SDM have been shown to improve patient-level outcomes, as compared to no decision aid, there are few data on whether SDM itself, as compared to other styles of decision making, is associated with these improved outcomes (as opposed to, for example, the provision of information). Recently, Shay and Lafata performed a systematic review on the empirical evidence regarding the effectiveness of SDM for improving patient outcomes. 17 In their work they identify only 32 studies in which the use of SDM was explicitly measured with a SDM tool, and concluded that there was insufficient evidence for the association between empirical measures of SDM and patient outcomes.
In this study, we performed a systematic review to describe how SDM has been compared to other forms of decision making for the particularly value-laden and preference-sensitive end-of-life (EOL) decisions. We sought to determine the components of SDM tested, the EOL decisions being assessed, the outcomes used to measure the effectiveness of SDM, and what other styles of decision making SDM has been compared to.
Methods
Study Selection Criteria
Studies were included if they were published in the English language, reported primary data collected, had participants/subjects who were 18 years of age or older, evaluated SDM for an EOL decision, compared SDM to another decision making style, and reported an outcome. Studies were included even if the main research question was not whether SDM improved outcomes. Studies were determined to be evaluating SDM if the term shared decision making was used to describe the study, or if other terms were used to describe patient/caregiver participation with their health care providers in medical decision making. To this end, broad search terms were used in conjunction with “decision making” including shared, participatory, collaborative, cooperative, mutual, and bilateral, to ensure we captured studies that evaluated SDM that may not have been labeled as such (see Figure 1 for full search strategy). We included SDM that occurred between the patient and health care provider as well as between the caregiver (family member) and health care provider. EOL decisions were those related to mechanical ventilation, left ventricular assist device, cardiopulmonary resuscitation, dialysis, salvage cancer treatment, or life-sustaining treatment/life support in general. All study designs were eligible, all outcomes of SDM were of interest, and there were no restrictions on publication date. Studies were excluded if they included participants/subjects less than 18 years of age, were published in a language other than English, were cancer-related other than terminal-stage cancer, did not describe the measurement of an outcome, and were case studies or publications not reporting primary data such as comments, opinions, and letters to the editor.

Search strategy.
Search Strategy
Medical librarians (SWH and JL) conducted literature searches in PubMed, Web of Science, Cochrane Central Register of Controlled Trials, EMBASE, PsycINFO, and CINAHL databases during the month of April 2014. The librarians and other authors identified a comprehensive list of search terms and constructed the final search strategy (Figure 1). No restrictions were set on publication date, language, or age. An adapted search strategy that included MeSH terms was used for the PubMed search (see Supplementary Appendix).
Study Selection
Titles and abstracts, and then full text articles, were screened for exclusion or possible inclusion by one reviewer (LU), with a second reviewer (NH) screening a subset to check for accuracy. Disagreement over inclusion of any article was resolved by discussion between the reviewers. Of those selected for inclusion, data were extracted by one reviewer (LU) with the other reviewer (NH) checking for accuracy by extracting data from half the articles.
Data Extraction
Data extracted included research questions, sample, setting, study design, the EOL decision being studied, the interventions and components of SDM in each intervention, the comparators, outcomes, analysis, and findings (Tables 1 and 2).
Summary of Studies Selected for Inclusion

Note: Shared patient-physician and patient-controlled decision making were combined for the analysis. CHF = congestive heart failure; ESRD = end-stage renal disease; CPR = cardiopulmonary resuscitation; DM = diabetes mellitus; EOL = end of life; ICU = intensive care unit; DNR = do not resuscitate.
Components of Shared Decision Making in Included Studies

Data Synthesis
Results were analyzed using narrative synthesis, a technique that involves developing preliminary synthesis of findings and then exploring relationships between studies.18,19 This process was aided by content analysis, tabulation, description, summarization, and groupings. No data transformation was undertaken because the included studies looked at different EOL decisions, different SDM interventions, and different outcomes. Instead, we aimed for a largely descriptive review in which we summarized what has been studied with regard to SDM for EOL decisions. We did, however, translate data by identifying and integrating common themes across different studies. 19
Main features of the included studies are tabulated in Table 1. Vote counting of the SDM components included in each study was used in Table 2, allowing for visual comparison of the studies. We then further synthesized this information using textual description by comparing and contrasting studies. 19 Because outcomes assessed in each study were so heterogeneous, we did not attempt to do any analysis of the strength of evidence for a particular outcome (see Appendix Table A1, online); however, we included a risk of bias table for each individual study (see Appendix Table A2, online).
Studies were divided based on whether they were experimental studies of an intervention involving some components of SDM, or observational studies reporting whether outcomes were affected by patients’ or their caregivers’ participation in SDM. Studies were examined based on our a priori questions, including the following: which components of SDM were included; what the EOL decision being assessed was; the comparators used to assess the effectiveness of this particular form of decision making; and which outcomes were measured for evaluating SDM.
Results
Results of Search
Database searches yielded 4931 articles (including 3518 unique articles). Based on the inclusion criteria, 148 articles were selected for full text review, and of these, 7 articles met all the inclusion criteria. Figure 2 shows the screening and eligibility flowchart, including reasons for exclusion at every step. The primary reason for exclusion after full text review was that the study did not include SDM.
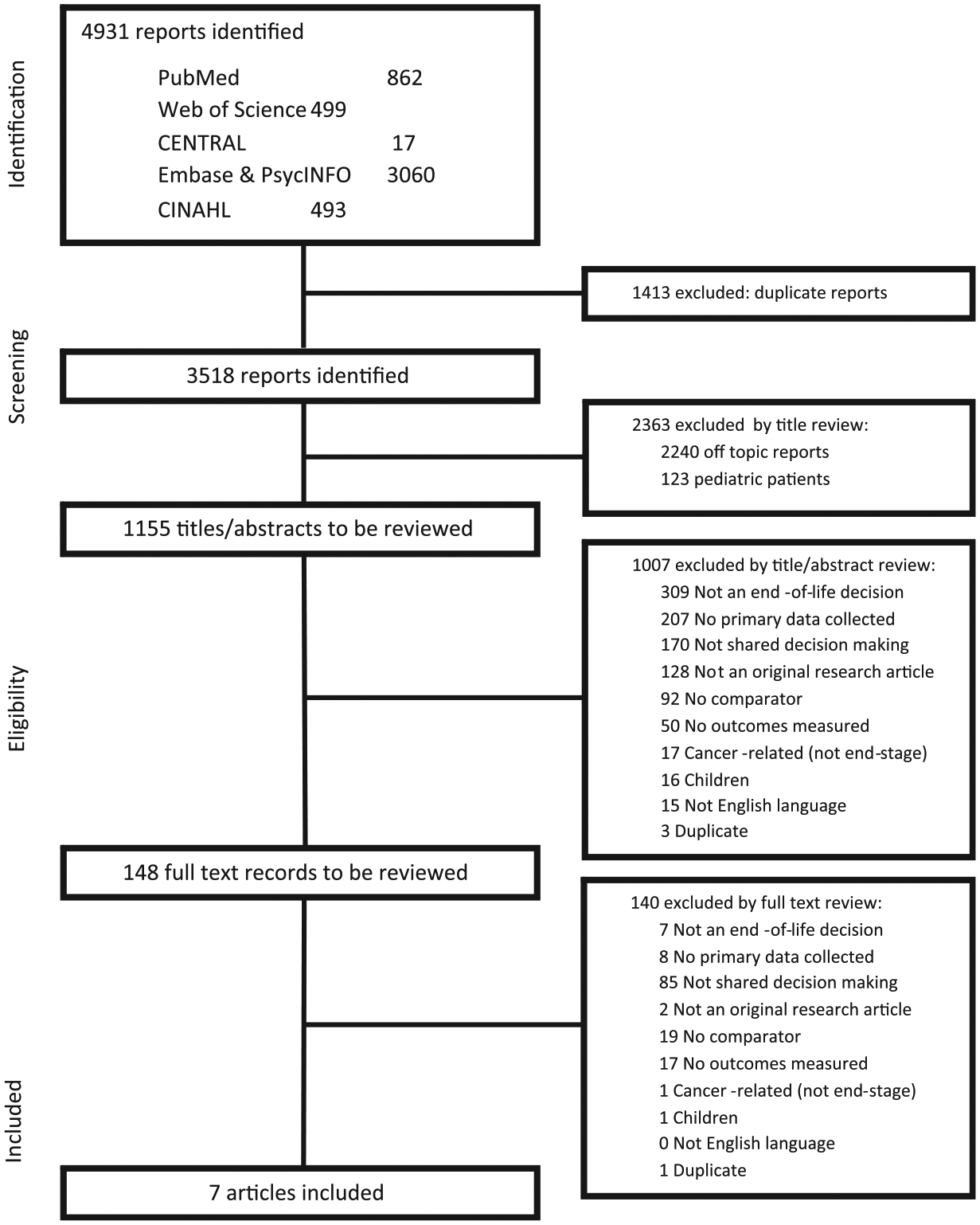
PRISMA flowchart of screening and eligibility evaluation.
How Shared Decision Making Has Been Evaluated in the Context of End-of-Life Decisions
Study Design
Of the seven studies included, three were experimental and four were observational. Two studies were nonrandomized controlled trials,20,21 one was a controlled before/after study, 22 and four were cross-sectional studies,12,23–25 (Table 1).
Sample and Setting
Four studies looked at SDM between patients and health care providers, two looked at SDM between surrogates and health care providers, and one study looked at both. Studies were split between outpatient and inpatient settings.
Components of SDM Used in Each Study
Although there is no consensus in the literature about the necessary components for SDM to have occurred, we needed a framework to describe which components of SDM were implemented or observed in each study. We therefore chose to summarize the components outlined in several articles attempting to define SDM10–12 (Table 2) and used this to query the articles.
In two of the seven studies included, the components of SDM were not specified or measured (Table 2). In these studies, participants were asked how medical decisions were made. One of those studies asked the question, “How were the decisions about your care actually made?” 24 Patients’ responses were categorized as active, shared, or passive decision making. The authors then reported the outcomes they assessed separately by each of these three types of decision making. Outcomes were reported for patient-controlled or shared decision making (combined) compared to physician-controlled decision making. The other study asked family members of a deceased patient whether that patient had participated sufficiently in EOL decisions. 23 In the studies in which components of SDM were specified, three were studies in which the intervention being tested was a tool to guide clinician-patient communication based on SDM concepts.20–22 These experimental studies did not, however, include external measures of whether SDM actually occurred in the intervention or comparator arms. Another study asked patients about the clinicians’ communication to create an IDM score (informed decision making score). 25 Only one study coded audiotapes of clinician-patient conversations to determine whether SDM occurred using a validated SDM measurement tool. 12
The SDM component most frequently described in the studies was presenting the patient/family with information about the risks and benefits of treatment choices (included in five studies). Other common SDM components were providing the patient/family with information about treatment choices and eliciting values and goals.
Interventions
Interventions in the three experimental studies included a 1-h advance care planning (ACP) interview, 20 a decision aid for surrogates of patients on prolonged mechanical ventilation, 22 and information-sharing in a cognitive model for ACP meetings (Table 1).20,21 The patient-centered ACP interviews 20 were designed to allow the patients to examine their belief systems as well as new information provided by the interviewer using the representational approach to patient education. The study evaluating a decision aid for surrogates of patients on prolonged mechanical ventilation 22 was based on SDM principles, and on evidence that other decision aids have led to more preference-congruent decisions. Intervention surrogates received training in the decision aid and were allowed to keep it for the duration of the study. The third experimental study tested a multifaceted intervention that included education for nurses and doctors on ACP and ACP discussions developed from SDM models. 21
End-of-Life Decisions Assessed
Among the experimental studies, one study aimed to facilitate ACP in general for decisions around mechanical ventilation and cardiopulmonary resuscitation 20 ; one study aimed to facilitate creation of advance directives specifying either full code or any limit to life-sustaining treatments 21 ; and one study aimed to help caregivers specify goals of care for their family member on prolonged mechanical ventilation (maximize comfort v maximize survival) 22 (Table 1).
Among the nonexperimental studies, EOL decisions assessed included the overall decision making process for cancer care (among patients with advanced cancer), 24 initiation of dialysis, 25 withdrawing life support or creating a do not resuscitate order, 12 and general decision making in the last 24 hours of the patient′s life. 23
Outcomes Assessed and Measures Used for Evaluating SDM
Appendix Table A1 (online) shows the main outcomes assessed in each study and the instruments used to measure them. The outcome measure most frequently assessed was the effect of SDM on communication. Different aspects of communication were measured across studies: “quality of communication” was assessed in two studies—one using the Quality of Patient-Clinician Communication About End-of-Life Care scale20,26 and one using the Quality of Communication scale22,27; “family satisfaction with communication” was assessed in the third study, using a 6-item questionnaire adapted from McDonagh and others.12,28 The studies using the ACP interview and the decision aid20,22 measured many of the same outcomes, such as communication, knowledge, congruence of treatment preference or of expected survival, and decisional conflict. Two studies used the Decisional Conflict scale, 29 which was used to assess decisional uncertainty after participation in both the ACP interview 20 and the decision aid for prolonged mechanical ventilation. 22 Otherwise, outcomes differed widely across studies, from trust of physicians 22 to satisfaction with care 24 to aspects of the decision making process such as “feeling rushed.” 25 Overall “quality of dying” was measured in one study, 23 using a questionnaire with items from previously developed scales including the Quality of Death and Dying questionnaire.
Comparators Used to Assess Effectiveness of SDM
Among the experimental studies, all three interventions were compared to usual care. For the study on patient-centered ACP interviews, 20 patients in the control group were asked if they had an advance directive or if they would like more information about advance directives. They were given an information card about ACP and advance directives. Control patients also received referrals to trained ACP facilitators, and if a patient had completed an advance directive, the documents were placed in a specific place in the medical record (per institutional policy). Finally, predialysis patients were invited to a class about dialysis. In the study of the decision aid for surrogates of patients on prolonged mechanical ventilation, 22 control surrogates did not receive any additional information. 22 The study of a cognitive model for ACP meetings 21 was implemented on two separate wards of a hospital, one of which served as the control ward and other as the intervention ward. Control participants received routine care. 21 There were no descriptors of usual care, including whether participants had other decision making conversations with providers before the study.
Among nonexperimental studies, one study compared patients who experienced SDM with those who described their experience as active or passive decision making. 24 Two studies compared the association of different levels of SDM with outcomes,12,25 and another study compared SDM to no SDM (assessed via surveys). 23
Risk of Bias
We used both the Cochrane Risk Assessment tool 30 and the EPHPP 31 (Effective Public Health Practice Project) risk assessment tool to allow for evaluation of the interventional and observational studies (Appendix Tables 2.1 and 2.2; online). All three interventional studies had a high risk of bias for the detection of the association between SDM and outcomes, due to lack of allocation concealment and blinding. In addition, there was no measurement of whether SDM actually occurred with the intervention (application of the intervention was not observed), and there may have been other confounding interventions such as prior EOL decision making conversations. Of the observational studies, most had a high risk of bias due to the cross-sectional study design, the low response rate with risk for selection bias, and the possible confounding effect of other interventions.
Discussion
Few studies have attempted to evaluate the effect of SDM for EOL decisions as compared to other forms of decision making. Most of these studies loosely defined SDM as representing patient/family participation in decision making. All but one study used interviews with patients/surrogates to assess whether SDM had occurred, rather than observation. It has been shown that the association between SDM and patient outcomes varies based on the SDM measurement and the perspective from which it is measured (patient v provider). 17 In addition, several studies suggest that there is variation between patient self-reports of SDM as compared to provider reports and/or objective ratings of SDM.32,33
Almost all of the seven studies included in our analysis had results suggesting improved outcomes when compared to usual care, or compared to more “passive” decision making. The one study that did not find an improved outcome with SDM (measured satisfaction with decision making) suggested that this may have been due to the “excellent communication skills among palliative care teams.” In fact, despite the style of decision making (active v shared v passive), more than 87% of patients expressed a high level of satisfaction.
We found heterogeneity in the outcomes measured and the tools used to measure a given outcome such as communication. One explanation for this heterogeneity may be that the definition of a “good decision” is not clear. Outcomes that suggest good decision making include knowledge and decisional conflict and satisfaction with decision making, and these are the outcomes most commonly measured when evaluating a decision making process.
Our study is the first systematic review that has evaluated the outcomes of SDM for EOL decisions. Frank 34 performed a literature review to evaluate the role of SDM in EOL care with a particular focus on studies exploring the role of a staff nurse in decision making, and the author found an association between patient participation in decision making and a “good death.” However, whether SDM in its entirety as a process (as opposed to individual components such as information exchange, or “excellent communication skills” as suggested by one of the included studies) is associated with a “good death” was not evaluated. Royak-Schaler and others 35 convened focus groups to explore family perspectives on communicating with health care providers during EOL care and found that perceived quality of EOL care was related to high-quality communication, which was defined as information being readily available and “compassionately shared.” These results raise the question of whether compassionate communication alone—as opposed to the entire process of SDM which involves information exchange and values clarification—is sufficient for family perceptions of high-quality care. Furthermore, is this a sufficient outcome for good-quality decision making?
There is increasing support for SDM as a central component for high-quality, patient-centered care.36,37 Ethical and legal considerations require that patients be provided information about their options in order to make informed decisions. It is clear that clinicians need to inform patients about their choices and engage them in decision making. However, SDM is more than an exchange of information and also requires patients to weigh their options with their provider, in light of their values and goals to arrive at a decision. Patients may prefer not to engage in this type of process with their providers, and it may depend on the clinical decision. In fact, Robinson suggests that patients may prefer not to talk about advance care plans with their medical providers. 38 The question remains whether SDM leads to better outcomes, and if so, which components of SDM are associated with improved outcomes. Is it the provision of high-quality information with checks to ensure comprehension? Is it the patient empowerment and relationship with their provider that is strengthened during the process of SDM? Is it compassionate communication? Unfortunately, our analysis was not able to determine which components of SDM are the most effective and whether particular components are sufficient to achieve improved patient outcomes.
To start to answer these questions, studies need to first be clear about which components of SDM they are including in their interventions. Second, studies need to explain how the presence of SDM was measured. Scales that could be used to assess the degree to which clinicians involve their patients in decision making and which components of SDM are being implemented include the OPTION scale 39 and the Informed and Shared Decision Making Instrument. 40 Third, studies also need to be clear about which outcomes are assessed and the reason for the selection of these outcomes. Finally, studies need to explore the baseline communication skills of clinicians and whether participants had prior conversations about the decision being made, in order to explore potential confounding or effect modification.
Limitations
We could not perform an analysis of evidence strength for different outcomes due to the heterogeneity of reported outcomes. However, we include an assessment of risk of bias for each individual study (see Appendix Table 2; online). An additional limitation is that our description of the components of SDM that were studied is limited to what was outlined in the articles rather than an assessment of the protocols/tools applied.
Conclusion
In summary, although there are several studies of SDM for EOL decisions, there are very few studies that have specified the components of SDM tested and compared the effectiveness of SDM relative to other types of decision making. As such, there is insufficient evidence to support SDM being associated with improved outcomes for EOL decisions as opposed to other forms of decision making. Future studies should clearly outline which components of SDM are being studied and compare this to other forms of decision making, such as the provision of high-quality information, compassionate communication, or promoting patient participation.
Footnotes
Financial support for this study was provided entirely by Grant 1R21AG048196-01 from the National Institutes on Aging (Dr. Hajizadeh). The funding agreement ensured the authors’ independence in designing the study, interpreting the data, writing, and publishing the report. The funding sources had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication. The authors had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data review. The authors of this article have no financial disclosures to report.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
