Abstract
Introduction
Low birth weight (LBW) remains a global health concern, especially in sub-Saharan African countries with fewer resources, such as Sudan. Despite ongoing preventive measures, LBW is still one of the main health problems and it is a leading risk factor for several adverse perinatal and neonatal outcomes.
Objective
To assess the prevalence of and factors associated (mainly maternal anemia) with LBW at Rabak Maternity Hospital, White Nile, Central Sudan.
Methods
A cross-sectional study was conducted involving live singleton newborns and their mothers who presented to Rabak Maternity Hospital from September to December 2021. Questionnaires were completed via face-to-face interviews to gather maternal information (maternal age, parity, history of miscarriage, educational status, and level of antenatal care [ANC]). Maternal hemoglobin levels were measured using an automated hematology analyzer. The neonate's birth weight and sex were recorded.
Results
Two hundred and fifty-three newborns were enrolled in this study, 40 (15.8%) of whom were LBW neonates. The median (interquartile range) maternal age and parity were 25 (21.0–30.0) years and 2 (1–4), respectively. While gestational age, maternal hemoglobin, and maternal anemia were associated with LBW, maternal age, parity, a history of miscarriage, education level, and level of ANC were not associated with LBW in the univariate analysis. Multivariate analysis showed that gestational age (adjusted odds ratio [AOR] = 0.80, 95% confidence interval [CI] = 0.66–0.96) and maternal hemoglobin (AOR = 0.76, 95% CI = 0.52–0.86) were inversely associated with LBW. Maternal anemia was associated with LBW (AOR = 4.70, 95% CI = 2.06–10.94).
Conclusion
LBW is a major health concern in White Nile, Sudan. Maternal age and maternal anemia are associated with LBW. Preventive measures for managing maternal anemia may help reduce the incidence of LBW.
Introduction
The World Health Organization (WHO) defines low birth weight (LBW) as newborns weighing <2.5 kg at birth, regardless of gestational age (International Statistical Classification of Diseases and Related Health Problems, n.d.). It has been estimated that there are 20.5 million LBW babies in the world, nearly a quarter (25%) of which are in Africa (WHO Recommendations for Care of the Preterm or Low-Birth-Weight Infant—World | ReliefWeb, n.d.). In sub-Saharan Africa, the number of LBW deliveries was estimated to increase from 4.4 million to 5 million in 2015 (Blencowe et al., 2019). LBW can lead to several adverse perinatal and neonatal outcomes, such as birth asphyxia, stillbirth, perinatal mortality, and neonatal death (Adam et al., 2008; Ahmed et al., 2022; Kamala et al., 2018), common childhood diseases (Class et al., 2014), other diseases in adulthood (Wang et al., 2022), and economic implications for the families and the health systems as well (Marzouk et al., 2017).
Literature Review
Despite continuing preventive measures, there is still a high prevalence of LBW in different countries in the sub-Saharan African region, including Sudan (Adam et al., 2008; Ahenkorah et al., 2022; Ahmed, 2022; Barut and Mohamud, 2023; Elhassan et al., 2010; Haggaz et al., 2010; Kamala et al., 2018; Kargbo et al., 2021; Odongkara et al., 2022; Tadesse et al., 2023; Wondemagegn et al., 2022). Several maternal factors, such as age (Odongkara et al., 2022), parity, level of antenatal care (ANC; Kamala et al., 2018), anemia (Barut and Mohamud, 2023; Kargbo et al., 2021; Odongkara et al., 2022; Tadesse et al., 2023), malarial infection (Odongkara et al., 2022), and malnutrition (Ahmed, 2022; Wondemagegn et al., 2022), as well as the neonate's gestational age (Ahenkorah et al., 2022; Ahmed, 2022), are reportedly associated with LBW in sub-Saharan Africa.
Sudan is the third largest African country. Several studies have reported a high prevalence of LBW in different regions of Sudan (Adam et al., 2008; Ahmed, 2022; Ali et al., 2011; Bilal et al., 2022; Elhassan et al., 2010; Haggaz et al., 2010; Omer et al., 2019); however, there is a paucity of epidemiological data regarding LBW in the state of White Nile. It is well established that the prevalence and chances of developing LBW are not homogenously distributed within a county (Musau et al., 2023). Likewise, the modifiable risk factors for LBW vary not only between countries but also among different regions within the same country (Banerjee et al., 2020).
LBW is not only an indicator of neonatal health but also an indicator of factors affecting maternal health, such as maternal malnutrition, anemia, malaria, and health service utilization (Menéndez et al., 2007). Therefore, reducing the incidence of LBW is a public health priority, especially in countries with fewer resources, and is part of the WHO's global commitment (Global Nutrition Targets) to reduce LBWs by 30% by 2025 (UNICEF-WHO Low Birthweight Estimates: Levels and Trends 2000–2015 | UNICEF, 2019)
Assessing the prevalence and factors associated with LBW is a fundamental component for generating valuable data that can be used to design effective and efficient public health strategies. Determining the epidemiology of LBW in White Nile, Sudan, will enable both national and local health agencies to direct their resources toward specific settings with poor health conditions. Accordingly, the current study assessed the prevalence and risk factors (mainly maternal anemia) associated with LBW at Rabak Maternity Hospital in central Sudan.
Methods
Study Design
This cross-sectional survey was conducted at Rabak Maternity Hospital, White Nile, in Central Sudan, from September to December 2021.
Research Question
Research questions for the current study: (a) What is the prevalence of LBW at Rabak Maternity Hospital? and (b) Is anemia associated with LBW?
Study Setting
Rabak Maternity Hospital is a tertiary hospital located in Rabak City, which is one of the largest cities in Sudan. Rabak lies at 362 m above sea level, and its geographical coordinates are between 13°9′0″ N and 32°44′0″ E (GPS Coordinates of Rabak, Sudan, DMS, UTM, GeoHash—CountryCoordinate.com, n.d.).
Sampling Procedure
Live newborns whose mothers were aged between 18 and 40 years were included in the study; all participating mothers signed a written informed consent form. Newborns delivered before 28 weeks of gestational age, twins, newborns with congenital malformations, and those delivered to mothers with antepartum hemorrhages or systematic diseases, such as diabetes mellitus, hematological disorders, thyroid disease, and hypertension, were excluded from the study.
A systematic random sampling technique was used to enroll the desired sample size. According to the hospital's records, 796 live newborns were delivered in the hospital during the 6 months that preceded the study. Therefore, a sampling interval of 3 was calculated by dividing the expected number of deliveries (796) by the calculated sample size (796/253 ≈ 3). Eligible newborns were therefore included in the study at three intervals during the study period until the required sample size (253) was reached.
Sample Size Calculation
A sample size (n) of 253 newborns was estimated, with an assumed prevalence of LBW of 12% (Elhassan et al., 2010) previously reported for central Sudan, using the single proportional formula, n = Z2pq/d2, where q = (1 − p), Z1−α (95% confidence interval [CI]) = 1.96, and d (margin of error of 5%) = 0.05.
Data Collection
After obtaining informed consent, the mothers of all included newborns were interviewed using a questionnaire to extract their basic sociodemographic, clinical, and obstetric data (age, parity, education level, history of miscarriage[s], and ANC level). The questionnaire was completed by two female medical officers via face-to-face interviews. Gestational age at birth was calculated from the last menstrual period and confirmed by ultrasound examinations. Next, the mother's weight and height were measured using standard measurement procedures, and their body mass index (BMI; in kg/m2) was computed (weight [in kg] divided by the square of the height [in m]). Two milliliters of venous blood were drawn from each woman by venipuncture and collected in an ethylenediaminetetraacetic acid tube and the sample was analyzed to provide hemoglobin levels using an automated hematology analyzer (Sysmex KX-21, Japan). Anemia during pregnancy was defined as a hemoglobin level of <11 g/dL (World Health Organization, 2011). Maternal, placental, and cord blood samples were taken to prepare blood smears for malaria. The blood films were stained using Giemsa stain and analyzed by an expert microscopist.
The newborn's weight at birth was measured immediately using the hospital's standard equipment, a digital floor scale (Seca, Hamburg, Germany), and recorded to the nearest two decimal points. The sex of the newborn was also recorded.
Ethical Approval
This study received ethical clearance from the ethical committee of the Faculty of Medicine at El Imam El Mahdi University, Kosti, Sudan (No. 2021, 08). Women signed an informed consent. A method was performed in accordance with the Declaration of Helsinki.
Statistical Analysis
All data were analyzed using Statistical Package for the Social Sciences (SPSS) for Windows version 22.0 (IBM, Armonk, NY, USA). Participant characteristics with continuous data were checked for normality using the Kolmogorov–Smirnov test; accordingly, the median (interquartile range [IQR]) was used to describe continuous variables and frequency and percentage for categorical variables. The Mann–Whitney U test was used to compare gestational age and maternal hemoglobin between newborns with and without LBW. The chi-square test was used to compare the proportions between the groups. Further, univariate analyses were performed with LBW as the dependent variable, and confounding variables which were, maternal age, parity, education, history of miscarriage, ANC level, BMI, and anemia/hemoglobin level as the independent variables. Thereafter, multivariable analysis was performed if the p-value was <.2 in the univariate analysis. The adjusted odds ratios (AORs) and 95% CI were also computed; a p-value of <.05 was considered statistically significant.
Results
Patient Characteristics
Two hundred and eighty-seven newborns were initially screened for this study. Thirty-four neonates were excluded from the analysis because they did not fulfill the inclusion criteria; accordingly, 253 newborns were enrolled. The median (IQR) age and parity of the mothers included were 25 (21.0–30.0) years and 2 (1–4), respectively. In total, 148 women (58.8%) had less than a secondary level of education, 54 (21.3%) had a history of miscarriage, and 42 (16.6%) women had received ≤2 ANC visits (Table 1). None of the maternal or umbilical cord blood samples tested positive for malaria, whereas five placental samples were found to be positive for Plasmodium falciparum. The median (IQR) maternal hemoglobin level was 10.9 (10.0–11.8) g/dL, and 132 (52.2%) women were anemic (hemoglobin <11.0 g/dL).
General Characteristics of Mothers of Neonates Enrolled in the Study at Rabak Hospital, Sudan in 2021 (n = 253).

The median (IQR) gestational age of the included neonates was 38 (37–39) weeks. Of the 253 neonates, 122 (48.2%) were male and 131 (51.8%) were female. The mean ± standard deviation of birthweight was 3138.3 ± 613 g, and 40 neonates (15.8%) were found to be LBW. Thus, the prevalence of LBW was 15.8%.
Factors Associated with LBW
The median (IQR) maternal hemoglobin (10.8 [9.0–10.9] g/dL vs. 11.2 [10.1–11.9] g/dL; p < .001) and gestational age at birth (38.0 [36.0–39.0] weeks vs. 38.0 [37.0–39.0] weeks; p = .027) was significantly lower in women who delivered LBW neonates (Figure 1). Furthermore, compared with neonates born with normal birthweight, a greater number of LBW newborns were born to anemic mothers (32 out of 48 [33%] vs. 16 out 120 [46.9%]; p = .001). Notably, the five placental samples that tested positive for malaria belonged to LBW newborns. However, when these factors were entered into the model, the model was distorted.

Comparing maternal hemoglobin between normal birthweight and low birthweight deliveries.
While gestational age, maternal hemoglobin, and maternal anemia were found to be associated with LBW, maternal age, parity, a history of miscarriage, education level, level of ANC, and BMI were not associated with LBW, as per the univariate analysis (Table 2). Furthermore, the multivariate analysis showed that gestational age (AOR = 0.80, 95% CI = 0.66–0.96) and maternal hemoglobin (AOR = 0.76, 95% CI = 0.52–0.86) were inversely associated with LBW and maternal anemia was associated with LBW (AOR = 4.70, 95% CI = 2.06–10.94; Table 3).
Results of the Univariate Analysis to Determine Factors Associated With Low Birth Weight in Neonates Born at Rabak Hospital, Sudan in 2021.
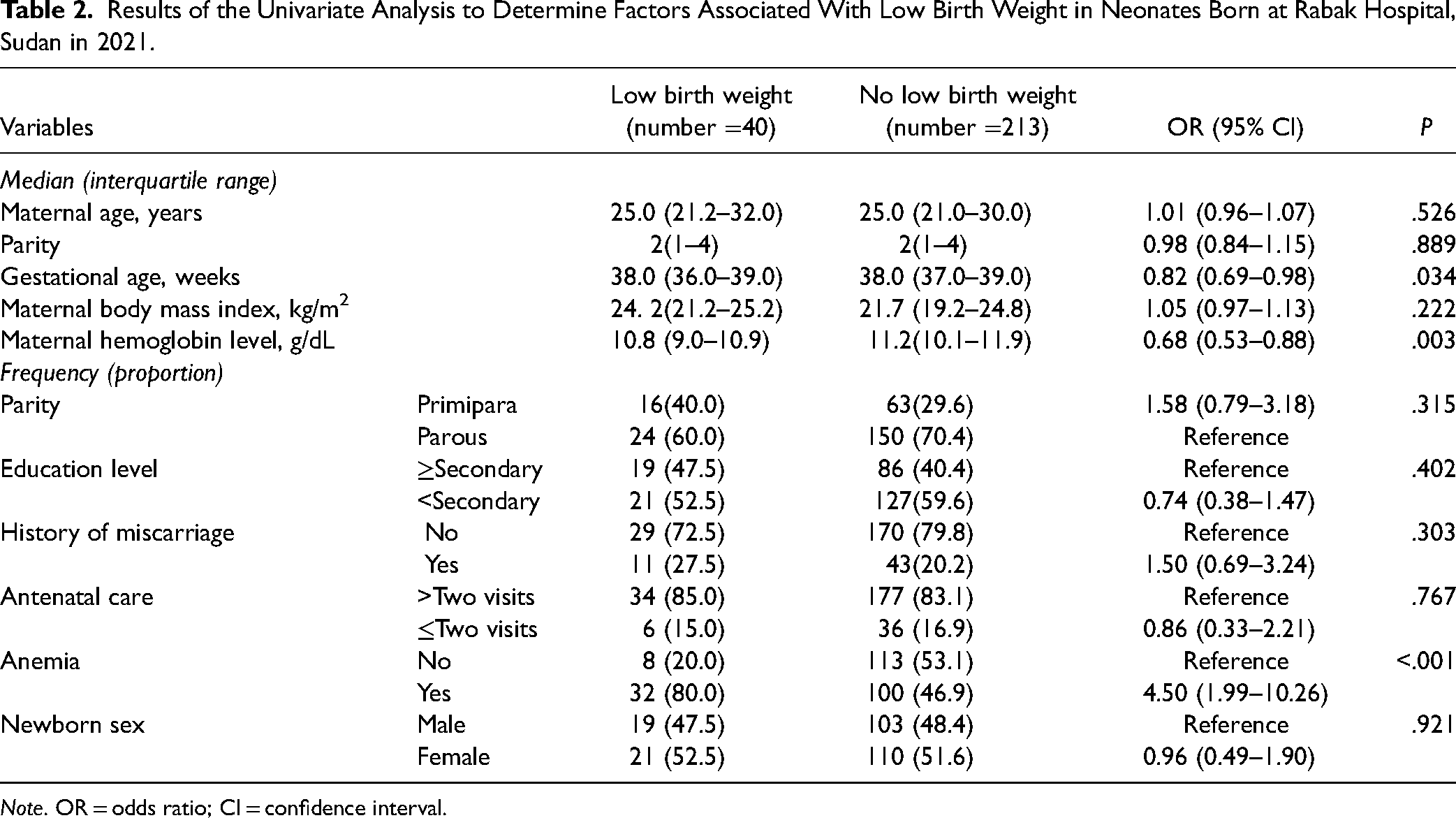
Note. OR = odds ratio; CI = confidence interval.
Results of the Adjusted Logistic Regression for Factors Associated With Low Birth Weight in Neonates Born at Rabak Hospital, Sudan in 2021.

Were entered one by one in the model.
Discussion
The prevalence (15.8%) of LBW in the current study is comparable to the prevalence of LBW reported for Eastern Sudan (15.3%; Adam et al., 2008) and Darfur, Western Sudan (14.9%; Haggaz et al., 2010). However, the prevalence was slightly higher than that reported for Khartoum, Sudan (12.1%; Bilal et al., 2022), Wad Medani (Central Sudan: 12.6%; Elhassan et al., 2010), Addis Ababa, Ethiopia (13.06%; Tadesse et al., 2023), Ghana (13.5%; Ahenkorah et al., 2022), and Tanzania (21%; Kamala et al., 2018). A pooled prevalence of 5.7% for LBW was recently reported in the Demographic and Health Survey of Saharan African countries, which enrolled 33,585 newborns (Weyori et al., 2022). Presumably, the differences in the prevalence of LBW in different regions of Sudan and Africa could be attributed to the diversity of sociodemographic characteristics across different regions and the resultant factors that may influence the prevalence of LBW itself.
The current study showed that gestational age was inversely associated with LBW (AOR = 0.80), which means that every one-week increase in the gestational maturity of the neonate can decrease the chances of LBW by 20%. These findings concur with previous studies conducted in Khartoum, Sudan (Ahmed, 2022; Bilal et al., 2022) and other African countries, such as Ghana (Ahenkorah et al., 2022). Nevertheless, it is difficult to isolate the effect of gestational age alone on LBW in the presence of other poor maternal and perinatal factors, such as preterm birth, which is frequently observed in sub-Saharan countries and constitutes a major health problem (Young et al., 2023). Therefore, preventive measures are warranted to reduce the incidence of preterm births and prolong gestation to avert LBW deliveries.
Another notable finding of the current study was that maternal anemia increased the risk of LBW by 4.7 times. Several studies have also reported similar findings for other regions of Sudan and Africa—Wad Mani in Central Sudan (Elhassan et al., 2010), Kassala in Eastern Sudan (Ali et al., 2011), Darfur in Western Sudan (Haggaz et al., 2010), Ethiopia (Deriba and Jemal, 2021; Tadesse et al., 2023), Somalia (Barut and Mohamud, 2023), Sierra Leone (Kargbo et al., 2021), Uganda (Odongkara et al., 2022), and Ghana (Mohammed et al., 2019). This association was further corroborated by several meta-analyses that have shown that anemic pregnant women are at a higher risk of delivering newborns with LBW (Rahman et al., 2016; Young et al., 2019, 2023). Anemia during pregnancy is a global health problem; it is estimated that 36.8% of pregnant women in the world have anemia (Karami et al., 2022), 57% in sub-Saharan Africa (World Health Organization, 2021), and 53% in Sudan (Adam et al., 2018). The exact mechanisms underlying the association between anemia and LBW have not been fully elucidated; however, maternal nutritional deficiencies (including iron, vitamin D, and folic acid) and inflammation (Di Renzo et al., 2015) possibly decrease oxygen delivery to the fetus (Zimmermann and Hurrell, 2007), resulting in poor placental function (Hare et al., 2013), which may lead to LBW.
Similar to previous studies from Sudan (Elhassan et al., 2010; Haggaz et al., 2010), the current study did not report any association between certain maternal factors, such as age, parity, education level, and anthropometrics, and LBW. On the other hand, a maternal age of ≥35 years in Uganda was reportedly associated with LBW (Odongkara et al., 2022). Likewise, parity and LBW were found to be associated in studies from Ghana (Mohammed et al., 2019) and Tanzania (Kamala et al., 2018). Differences in sociodemographic characteristics, nutritional status, and the difference in the prevalence of diseases, for example, anemia between countries could explain the difference between the results of the current study and the results of the other studies.
Strengths and Limitations
This study included several important risk factors (covariates) that are known to affect birth weight to analyze their effect on the prevalence of LBW; however, this study did not account for the mother's nutritional status in influencing the neonate's birth weight.
Implications for Clinical Practice
The findings of the current study have significant implications for improving LBW screening and monitoring. Preventive measures to manage antenatal anemia should be employed by local and national agencies to reduce the incidence of anemia-related complications, such as LBW.
Conclusion
LBW is a major health concern in White Nile, Sudan, which may be worsened by concomitant maternal factors, such as anemia, and low gestational age at birth. Therefore, adequate ANC is essential to manage maternal anemia and ensure a healthy term gestation, which may help reduce the incidence of LBW in this part of Sudan.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
