Abstract
High cardiovascular and cortisol reactivity to stressful tasks are predictors of increased future cardiovascular risk. Few studies have investigated the impact of shift work on cardiovascular reactivity, and none have examined cortisol reactivity. The purpose of this study was to compare cardiovascular and cortisol stress reactivity in female shift workers (SW) versus non-shift workers (NSW). Nineteen SW (40 ± 11 years) and 19 NSW (42 ± 11 years) participated. Heart rate, systolic blood pressure, and diastolic blood pressure (DBP) were measured at rest, and during each minute of a speech (5 min) and mental arithmetic (5 min) stress task. Serum cortisol was measured pre- and poststress task (immediately and 15 min post). Values are means ± SD. Peak increases in DBP during the task did not differ between groups (p = .261), however, analyzed over time there was an interaction such that DBP increased significantly more in SW during the fifth minute of the speech task (p = .035). There were no group differences in heart rate or systolic blood pressure responses. The increase in cortisol also did not differ between groups (ΔSW: 5.5 ± 7.5 µg/dL, ΔNSW: 1.8 ± 2.9 µg/dL, p = .165). However, when compared separately, the increase in cortisol from baseline to peak poststress was significant in SW (p = .013) but not in NSW (p = .125). In conclusion, these preliminary data suggest that shift work exposure may have a modest influence on cardiovascular and cortisol reactivity. Further research is necessary to fully characterize and explore the importance of stress reactivity in this population.
Keywords
Introduction
Shift work is an increasingly common work pattern with approximately 20% to 25% of the North American and European workforce employed in shift work positions (Costa et al., 2004; Williams, 2008). The proportion of shift work positions in the health-care work force is higher, with women assuming the majority of these positions (Canadian Center for Occupational Health and Safety, 2010). A growing body of evidence supports associations between shift work and increased risk for chronic diseases, including cardiovascular disease (CVD; Puttonen, Harma, & Hublin, 2010; Wang, Armstrong, Cairns, Key, & Travis, 2011). Given health priorities to delay CVD development and to promote heart-healthy environments, we need to identify potentially modifiable pathways through which shift work contributes to increased cardiometabolic risk and related disorders. It is hypothesized that shift work influences cardiometabolic risk directly through the circadian disruption of physiological-metabolic pathways, or indirectly through mediation with psychological-behavioral pathways (Puttonen et al., 2010).
Cardiovascular and cortisol reactivity to acute mental stress (characterized as the magnitude of increase in heart rate [HR], blood pressure [BP], and cortisol in response to an acute stressor) are predictors of future cardiovascular risk and may also play a mechanistic role in the development of CVD (Chida & Steptoe, 2010; Hamer & Steptoe, 2011). Shift work is associated with a reduction in HR variability (Togo & Takahashi, 2009; Wehrens, Hampton, & Skene, 2012), altered catecholamine responses (Hansen, Larsen, Rugulies, Garde, & Knudsen, 2009; Scheer, Hilton, Mantzoros, & Shea, 2009), and disrupted cortisol production patterns (Manenschijn, van Kruysbergen, de Jong, Koper, & van Rossum, 2011; Mirick et al., 2013; Van Dycke et al., 2015), which collectively indicate altered sympathetic nervous system and hypothalamic pituitary adrenal (HPA) axis activity. While shift work is associated with disruption of these physiological stress response systems, the effect of shift work history on stress reactivity has not been extensively investigated (Kario et al., 2002). However, individuals who work in shift work patterns report high psychological job stress and have difficulties with work-life balance (Boggild, Burr, Tuchsen, & Jeppesen, 2001; Jansen, Kant, Nijhuis, Swaen, & Kristensen, 2004), and prolonged exposure to chronic stress may amplify cardiovascular responses to acute stressors (Chida & Hamer, 2008; Low, Salomon, & Matthews, 2009). Collectively, this evidence suggests that shift work may alter the cardiovascular and cortisol reactivity to acute mental stress.
Therefore, the objective of this pilot study was to compare the cardiovascular and cortisol stress reactivity of long-term (>6 years) shift workers (SW) and non-shift workers (NSW) in response to an acute laboratory mental stress task. We hypothesized that SW would have greater cardiovascular and cortisol reactivity to acute mental stress. If high reactivity contributes mechanistically to CVD progression, this could help to further elucidate the causal pathways linking shift work experience to CVD.
Materials and Methods
Participants
Participant Characteristics.
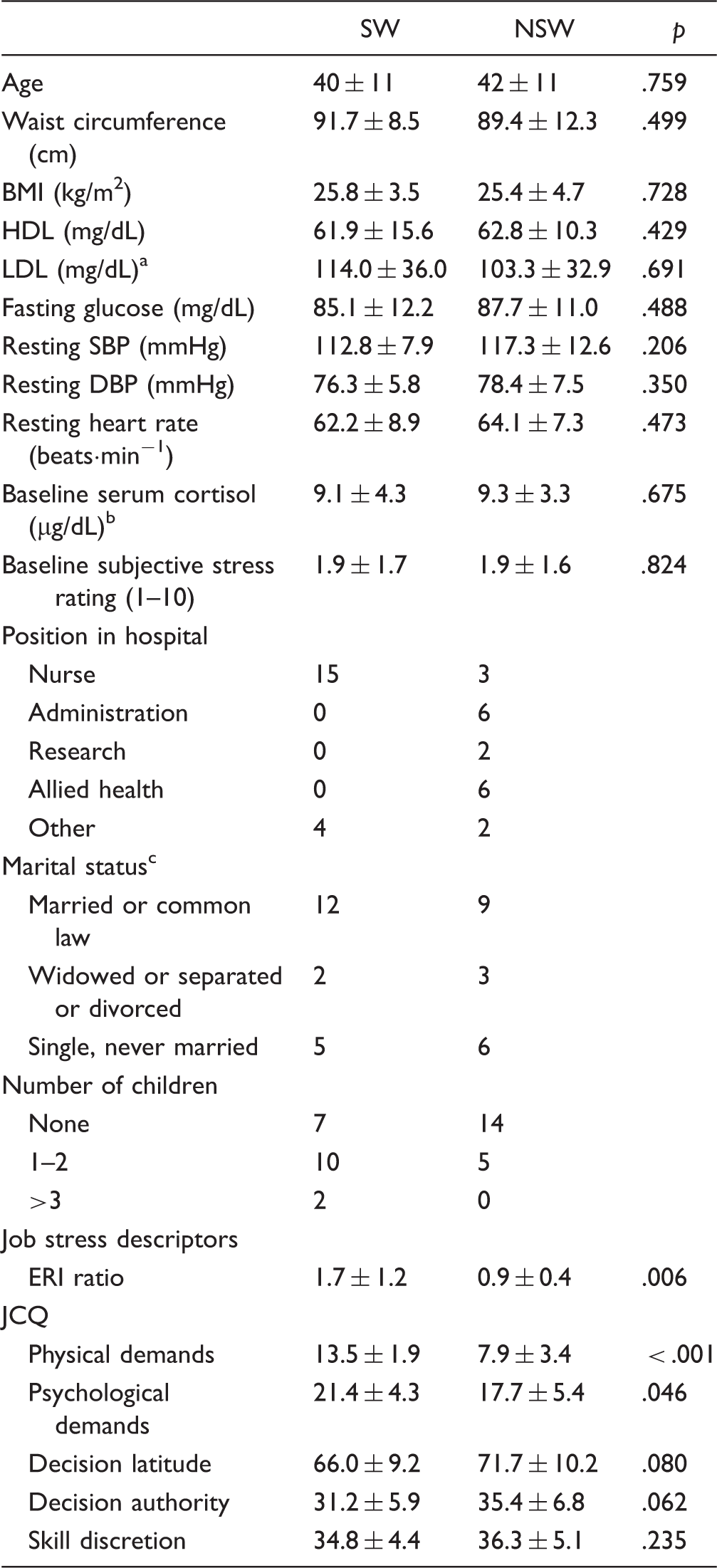
Note. An ERI ratio > 1 indicates that effort exceeds reward, and a ratio < 1 indicates that reward exceed or equals effort. Values are means ± SD.
SW: n = 14, NSW: n = 13.
SW: n = 15, NSW: n = 8.
NSW: n = 18.
BMI = body mass index; ERI = effort-reward imbalance; HDL = high-density lipoprotein; JCQ = job content questionnaire; LDL = low-density lipoprotein; SBP = systolic blood pressure; DBP = diastolic blood pressure; SW = shift workers; NSW = non-shift workers.
Participant Monitoring
At the beginning of the visit, baseline blood pressure was assessed using an automatic blood pressure monitor (BPM-100, BpTru, Coquitlam, Canada). HR and BP were monitored continuously throughout the experiment with a 3-lead electrocardiogram and finger photoplethysmography (Finometer PRO, Finapres Medical Systems, Amsterdam, The Netherlands), respectively. Electrocardiogram and Finometer signals were recorded and stored with the program LabChart (AD Instruments, Colorado Springs, USA) for future analysis.
Experimental Procedure
All testing was done at the same time of day between 0800 and 1000 h with the exception of one NSW participant who began testing at 1615 h. Testing was done in a quiet room with a maintained temperature of 21℃. Participants were instructed to fast for 12 h prior to visiting the lab and to avoid caffeine, alcohol, and vigorous exercise for 12 h before testing. All SW participants were tested at least 12 h following the most recent night shift, NSW were tested at least 8 h post shift with the exception of one who was tested 45 min after work. At the beginning of the visit, a 22-gauge catheter (BD Nexivia, BD Medical Supplies, Mississauga, Canada) was inserted in the right antecubital vein. The blood sample for prestress baseline cortisol determination was obtained approximately 60 min before initiation of the stress task. Poststress task samples were taken immediately and 15 min following task completion (Figure 1). HR and BP were recorded before and during the stress task (Figure 1). Subjective assessment of stress (rated on a scale of 0–10) was obtained before and immediately poststress task.
Protocol timeline. The vascular testing protocol to be reported elsewhere.
Blood Lipids and Glucose
Prior to initial rest and instrumentation, a small sample of blood was used for analysis of blood lipids and fasting glucose using a Cholestech LDX System (Alere Inc., Ottawa, Canada). If a venous catheter could not be successfully inserted, blood samples were taken by finger-prick. Low-density lipoprotein values were not obtained in five SW and six NSW as the values fell outside of the range of the device.
Mental Stress Task
The mental stress task protocol was based on the Trier Social Stress Test (Kirschbaum, Pirke, & Hellhammer, 1993) and consisted of instructions, preparation time, a speech task, and a mental arithmetic task; all administered by a researcher with whom the participant had not interacted previously. In the speech task, the participant was asked to defend herself against a false accusation of shoplifting (Rosengren, Tibblin, & Wilhelmsen, 1991). Participants were given 10 min to prepare their speech followed by 5 min of speaking time. The researcher provided a standard prompt if the participant stopped speaking at any point. Immediately following the speech task, participants were given instructions for the 5-min mental arithmetic task. The participant was asked to serially subtract 17 from a four-digit prime number. At planned intervals during the mental arithmetic task, participants were told that their answers were incorrect even when correct. Participants were informed that their speech and mental arithmetic responses were being video recorded for facial expression analysis.
Serum Cortisol Sampling
Venus blood samples were collected into silicone coated serum tubes (BD Vacutainer, BD Medical Supplies, Mississauga, Canada). Samples were kept at room temperature for at least 30 min following collection in order for clotting to occur as per BD Medical Supplies instructions. After clotting, samples were spun for 10 min at 21℃, at a rate of 2680 r·min−1 and 1200 relative centrifugal force (IEC-Centra MP4R; International Equipment Company, Mass, USA). Once spun, the samples were aliquoted and stored at −80℃ until analysis. It was only possible to collect and analyze full sets of samples (prestress, immediately poststress task, and 15-min poststress task [total of three samples]) in 15 SW and 8 NSW due to technical difficulties with catheter insertion, maintenance of patency, and sample processing. Only participants with all three samples were included in the analysis.
Job Content Questionnaire and Effort-Reward Imbalance Ratio
Work stress was assessed using the job content questionnaire (Karasek et al., 1998) and the effort-reward imbalance (ERI) ratio (Siegrist, 1996), respectively. The job content questionnaire consists of 56-items, including subscale measurements for decision latitude (9 items), decision authority (3 items), skill discretion (6 items), psychological demands (5 items), and physical demands (5 items). For each question, the participant rated on a scale—1 = strongly disagree to 4 = strongly agree—how she felt the statement described her job and job environment. The ERI ratio questionnaire uses questions about effort, reward, and overcommitment at work. This tool consists of 52-items, including 6 effort and 11 reward subscales. To calculate the degree of imbalance, the following formula was used: Effort-reward Ratio = e/(r * c), where e is the sum of the effort score, r is the sum of the reward score, and c is the correlational factor (number of items in the numerator divided by the number of items in the denominator). An ERI ratio > 1 indicates a poor work situation where effort exceeds reward, and a ratio < 1 indicates an optimal work situation where reward exceeds or equals effort (Siegrist, 2010). Surveys were given to participants following data collection to be completed on the participant’s own time. Surveys were returned within approximately one week of data collection.
Data Analysis
Systolic blood pressure (SBP), diastolic blood pressure (DBP), and HR were analyzed offline in 3-s average time bins. The prestress SBP, DBP, and HR baseline was reported as the average of the last 3 min of the prestress task baseline rest (Figure 1). The response to the stress tasks was characterized as the change in SBP, DBP, and HR from the prestress baseline to the value during each minute of the stress task (10 min: 5 min speech and 5-min mental arithmetic). A single reactivity value for each of HR, SBP, and DBP was also calculated for each participant as the change from the prestress baseline to the peak 1-min average during the stress task (Szijgyarto, Poitras, Gurd, & Pyke, 2014). Due to poor Finometer signals, SBP and DBP reactivity were not collected in one NSW. Subjective stress reactivity was characterized as the change in subjective rating from prestress baseline to immediately poststress task.
Serum samples were thawed on the day of the assay. Serum cortisol was determined in duplicate, according to the manufacturer’s instructions, using an enzyme-linked immunosorbent assay kit (cat. no. 11-CORHU-E01, ALPCO Diagnostics, Salem, United States). The sample intraassay coefficient of variation was 6%, and the sample interassay coefficient of variation was 13%. Cortisol reactivity to the stress task was characterized as the change in cortisol from prestress baseline to the highest of the two poststress samples (Szijgyarto et al., 2013).
Statistical Analysis
Independent sample t tests (Mann–Whitney rank sum test in the case of nonnormal distribution or unequal variance) were used to compare participant characteristics. Independent t tests were used to compare stress reactivity (peak minus prestress baseline) between SW and NSW for HR, SBP, DBP, stress ratings, and cortisol. A paired t test was also used to compare baseline cortisol with peak poststress cortisol in each group. A repeated measures analysis of variance was used to compare changes in HR, SBP, and DBP over time between groups. The factors were group (SW and NSW) and time (each minute of the stress task [10 min total]). Linear regression analysis was performed to assess the association between peak physiological reactivity and psychological variables. The level of significance was set at p < .05. Effect size was estimated with Cohen’s d using the following equation: Cohen’s d = mean1 – mean2 ÷ √((SD12 + SD22)/2). All statistics were calculated using SigmaPlot 11.0 or SPSS 24, and all data were expressed as means ± SD.
Results
Participant Characteristics
Physiological and anthropometric characteristics were not significantly different between groups (p > .05). ERI was significantly higher in SW (p = .006), and SW indicated significantly higher physical and psychological work demands (p < .001 and p = .046, respectively). There was a trend for lower decision latitude and decision authority in SW. A greater proportion of NSW were taking medications (SW: 1 (statin) versus NSW: 9 (single or combination of lipid lowering, anti-depressant, anti-inflammatory, or diuretic medications). A greater proportion of the SW were nurses while NSW worked in a variety of positions in the hospital.
Cardiovascular Baseline Measures and Stress Reactivity
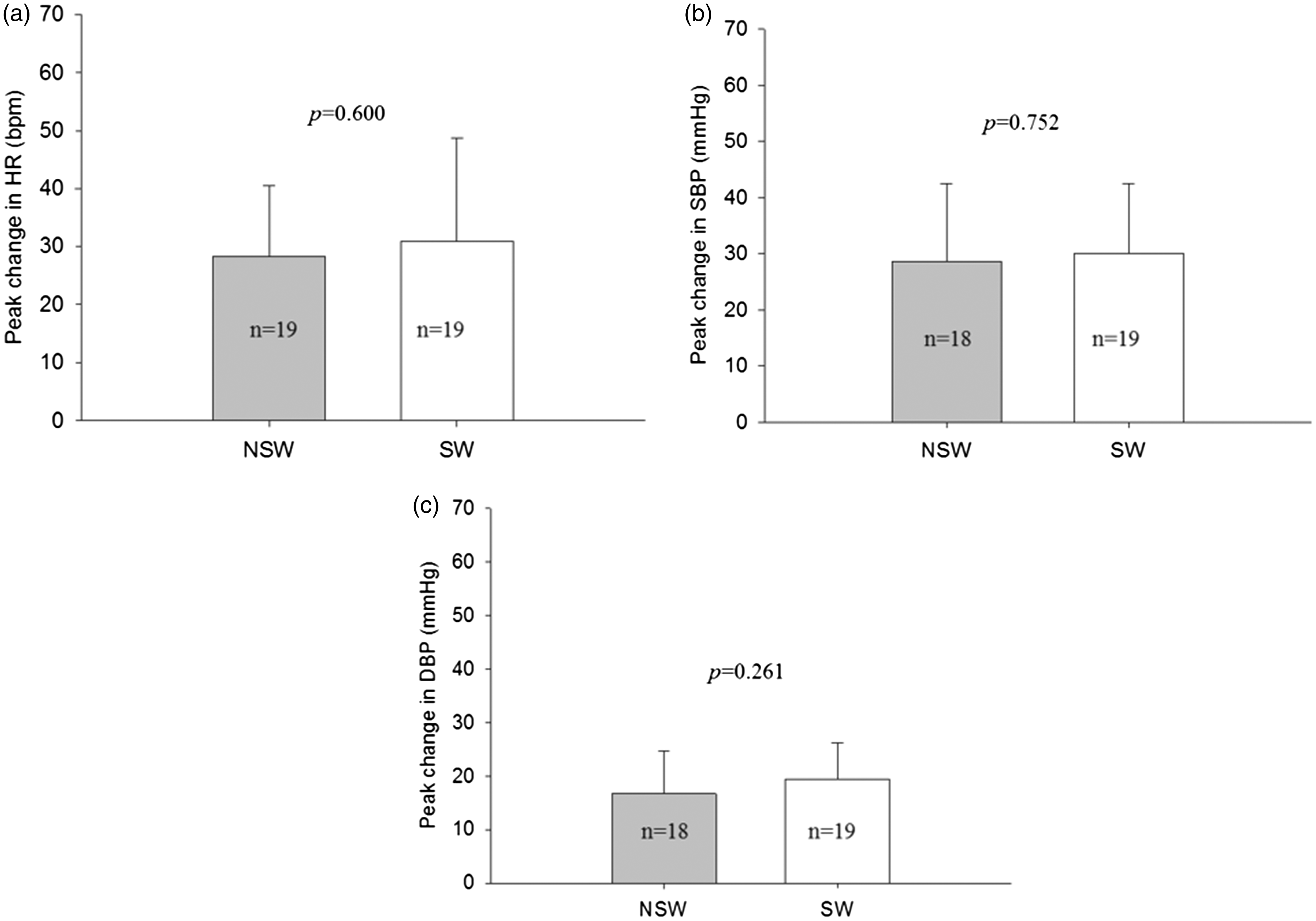
There were no significant differences between groups in baseline HR, SBP, or DBP (Table 1). HR and SBP changes over time during the stress task did not differ between groups (Figure 2(a) and (b)). There was an interaction between group and time for DBP reactivity (p = .043). Post hoc comparisons at each time point identified a significantly greater increase in DBP in SW compared with NSW during the fifth minute of the speech task (p = .035). The group difference approached significance in minutes 2, 3, and 4 of the speech task (p = .06, .057, and .059, respectively; Figure 2(c)). There were no differences in peak reactivity (i.e., peak 1 min average during stress minus prestress baseline) between groups for HR (p = .600), SBP (p = .752), or DBP (p = .261; Figure 3(a–c)). No significant relationships were detected between cardiovascular reactivity variables and the variables assessed in the ERQ and job content questionnaire (data not shown).
Change from the prestress baseline during each minute of the stress task (each data point represents a 1-min average). The p values are derived from two-way ANOVA analysis (T = time, G = group). (a) Change in heart rate (HR). (b) Change in systolic blood pressure (SBP). (c) Change in diastolic blood pressure (DBP). All values are means ± SD. Change from the prestress baseline to the peak 1-min average during the stress task. (a) Peak change in heart rate (HR). (b) Peak change in systolic blood pressure (SBP). (c) Peak change in diastolic blood pressure (DBP). The p values reflect t-test comparisons between groups. All values are means ± SD.
Subjective Stress Rating and Cortisol Baseline Measures and Stress Reactivity
There were no significant differences between groups in baseline subjective stress ratings or serum cortisol concentrations (Table 1). SW and NSW did not differ significantly in subjective stress rating changes or cortisol stress reactivity (peak poststress minus prestress baseline; Figure 4(a) and (b); cortisol reactivity was also not significantly different between groups when both poststress time points were included in a group by time analysis of variance—data not shown). Although the larger cortisol reactivity in SW did not reach significance (p = .165) with the present comparison of 8 NSW and 15 SW, the difference in cortisol reactivity between groups had a moderate effect size (d = 0.65). In addition, when comparing SW and NSW separately, cortisol increased significantly from pre- to poststress in the SW group (p = .013) but not in the NSW group (p = .125; Figure 4(c) and (d)). Considering the variation of the cortisol response, approximately 39 SW and NSW (78 participants total) would be required to achieve a power of 0.8 to detect the observed group difference in cortisol reactivity (Figure 4(b)) as significant (alpha = 0.05). When only cortisol responders were considered (defined as those who experienced an increase in cortisol from baseline to poststress; n = 5 NSW and 11 SW), the difference between groups was more pronounced, although still did not reach significance (3.7 ± 1.4 µg/dL vs. 8.6 ± 5.9 µg/dL, p = .089; d = 1.1). No significant relationships were detected between cortisol reactivity and the variables assessed in the ERQ and job content questionnaire (data not shown).
(a) Change in subjective stress rating from pre- to poststress. (b) Change in serum cortisol from prestress to the highest of the two poststress samples. (c) Pre- and poststress serum cortisol concentration in non-shift workers (NSW). (d) Pre- and poststress serum cortisol concentration in shift workers (SW). The p values in (a) and (b) reflect t-test comparisons between groups. The p values in (c) and (d) reflect paired t-test comparisons within group, over time (pre- to peak poststress). All values are means ± SD.
Discussion
This pilot study was designed to compare cardiovascular and cortisol reactivity to an acute laboratory mental stress task in female SW and NSW. The primary findings are twofold. First, while the peak increase in DBP during the stress task did not differ between groups, DBP reactivity was higher in SW during the speech portion of the stress task. Second, although the group difference in cortisol reactivity was not significant, the increase in cortisol from baseline to peak poststress reached significance in the SW but not in the NSW. These results extend previous evidence supporting the effects of shift work on cardiovascular regulatory and HPA-axis function and support further investigation of a potential connection between stress reactivity and CVD risk in relation to shift work.
Cardiovascular Stress Reactivity
This study used a mental arithmetic and public speaking task to elicit a cardiovascular stress response. Over the combined 10 min of the stress task (speech and arithmetic portions), we observed an increase in HR of approximately 19 bpm, and a rise in SBP and DBP of approximately 20 and 12 mmHg, respectively. These magnitudes of change are in agreement with previous studies that have examined HR, (Heponiemi et al., 2007; Kario et al., 2002), SBP (Carroll et al., 2001; Kario et al., 2002), and DBP (Carroll et al., 2001) stress reactivity in men and women using mental arithmetic, public speaking, anger recall, and cognitive stress tasks.
Interestingly, in the present study, we found that SW had higher DBP responses when analyzed over time during the speech portion of the stress task only (∼5 mmHg higher in SW), and this was not accompanied by a significantly larger peak DBP reactivity. There is variability in the literature regarding how reactivity is quantified including both peak changes (Kario et al., 2002) and changes averaged over multiple fixed time points during the task (Carroll et al., 2001; Heponiemi et al., 2007). The present findings highlight that the way in which reactivity is assessed (peak vs. fixed time point averages) can impact the result of group comparisons.
In a meta-analysis examining the relationship between acute cardiovascular reactivity and negative cardiovascular outcomes, DBP reactivity was found to be predictive (Chida & Steptoe, 2010). However, when split by task type, cardiovascular reactivity to cognitive tasks alone (e.g., mental arithmetic) was found to significantly predict negative outcomes while reactivity to public speaking tasks alone was not (Chida & Steptoe, 2010). This could suggest that the higher reactivity in the speech portion of the task has lower health-related significance; however, the studies included in the Chida and Steptoe (2010) meta-analysis were not specific to a shift working population.
Although all participants in the present study worked in a hospital setting, the positions of the SW and NSW differed. Fifteen SW and only three NSW were working as nurses at the time of the study while several of the NSW were in administrative type positions (e.g., administrative assistant, manager, education coordinator; Table 1), and it is possible that this different work experience contributed to a lower perception of stress during the speech task, although posttask subjective stress ratings did not differ between groups. We cannot rule out that other nonoccupational group differences may have also influenced responses (marital status and number of children reported in Table 1). Taken together, the significance of the speech specific greater DBP reactivity in the SW group is unclear; however, this finding provides evidence that work characteristics may influence cardiovascular reactivity and highlights a need for a better understanding of the implications of different strategies to determine cardiovascular reactivity.
To the author’s knowledge, only one other study has examined cardiovascular stress reactivity in a shift-working population (Kario et al., 2002). Kario et al. (2002) examined 54 female night-shift nurses and 33 day-shift nurses and were unable to identify a significant difference between SW and NSW in HR or SBP reactivity using similar tasks to elicit cardiovascular responses as the present study (a mental arithmetic task and a coping task; public speaking or anger recall). These authors did not report DBP stress task reactivity, although clinic DBP (but not ambulatory) was significantly higher in night-shift nurses. Kario et al. (2002) reported cardiovascular reactivity as the difference between the prestress baseline and the peak value during the stress task, and as the area under the curve during the stress task. It is possible that this prevented the detection of group differences at specific time points during the task, although in the present study we did not detect group differences over time for HR or SBP.
While baseline participant characteristics including age, body mass index, resting BP, and work environment (hospital employees) were similar between studies, Kario et al. (2002) did not characterize work stress. A number of studies, including this pilot, consistently report that SW report higher levels of psychological job stress (Esquirol et al., 2009; Wong, Ostry, Demers, & Davies, 2012). However, we did not find that physiological stress reactivity was associated with the job stress parameters.
The Importance of Cardiovascular Reactivity
Although there is evidence that elevated cardiovascular reactivity is associated with poor future cardiovascular health status (Chida & Steptoe, 2010), it is difficult to determine the existence or the direction of causality. However, several causal pathways have been suggested and seem likely (Chida & Steptoe, 2010). It is possible that in hyper-reactive individuals frequent daily stress responses stimulate vascular changes in response to the bursts of high BP that eventually contribute to an increase in their resting BP. This is supported by evidence showing that high cardiovascular reactivity only predicted elevated future BP in individuals who reported high life stress (Light et al., 1999). Under these circumstances, SW might be especially vulnerable to high reactivity-induced damage as they report high job stress (the present study and [Esquirol et al., 2009; Wong et al., 2012]).
Cortisol Stress Reactivity
To the author’s knowledge, this is the first study to compare cortisol stress reactivity in SW and NSW. Baseline serum cortisol levels were found to be 9.1 ± 4.3 µg/dL and 9.3 ± 3.3 µg/dL in SW and NSW, respectively. These values fall within the normal range (5–25 µg/dL) for morning serum cortisol in women (Guber & Farag, 2011). There was wide variability in the cortisol response to the mental stress task. Approximately 30% of participants had no cortisol response to stress (defined as a net decrease in serum cortisol from baseline), while individuals who had responses showed increases in cortisol ranging from 10% to 190% from baseline. Previous studies have also observed large variability in cortisol reactivity (Hamer, O’Donnell, Lahiri, & Steptoe, 2010; Hamer & Steptoe, 2012; Kirschbaum et al., 1995; Szijgyarto et al., 2013). Most commonly, cortisol concentrations for stress reactivity have been assessed using salivary cortisol (Al Absi & Wittmers, 2003; Hamer et al., 2010; Hamer & Steptoe, 2012; Kirschbaum et al., 1995). While this study used serum cortisol, salivary and serum cortisol concentrations are known to be well correlated (Poll et al., 2007).
Cortisol stress reactivity was not significantly higher in SW compared with NSW (p = .165). However, the group difference had a moderate effect size (d = 0.65), and tested independently, only SW experienced a significant increase in cortisol from baseline to poststress. While no previous studies have compared cortisol reactivity to acute stress in a SW versus NSW sample, there is evidence that shift work affects HPA axis activity. Reports have shown changes in diurnal cortisol secretion patterns with SW experiencing increased (Lindholm et al., 2012) and decreased (Fekedulegn et al., 2012) waking cortisol concentrations compared with day workers. Hair cortisol, a technique used to assess long-term cortisol levels, has also been shown to be elevated in SW (Manenschijn et al., 2011). Similarly, high-reported stress, a characteristic of shift work that was observed in the current study, has been correlated with increased waking cortisol after dexamethasone (Pruessner, Hellhammer, & Kirschbaum, 1999). Future research is required to determine the existence and nature of association between these diurnal and chronic cortisol levels and acute reactivity in SW.
The Importance of Cortisol Reactivity
Compared with cardiovascular stress reactivity, there has been significantly less research regarding the ability of cortisol reactivity to predict future risk. Nevertheless, studies have shown associations between heightened cortisol reactivity and increased risk of hypertension (Hamer & Steptoe, 2012) and significant coronary artery calcification (Hamer et al., 2010). Furthermore, hypertension-prone men and women (high resting systolic BP) have demonstrated elevated cortisol responses to public speaking tasks (Al Absi & Wittmers, 2003). While, as described with respect to cardiovascular reactivity, there is uncertainty regarding direction of causality, some evidence suggests that cortisol reactivity could play a mechanistic role in CVD development. For example, infusion of cortisol in normotensive individuals has been reported to cause increases in BP (Connell et al., 1987), and the very high cortisol concentrations seen in Cushing’s syndrome patients are thought to be a likely cause of hypertension (Whitworth, Williamson, Mangos, & Kelly, 2005). There is also evidence that high cortisol has a negative impact on endothelial function (Akaza, Yoshimoto, Tsuchiya, & Hirata, 2009; Broadley et al., 2005). A reduction in endothelial function may result in attenuated vasoprotection and potentiate atherosclerosis progression because the endothelium is essential for controlling vascular tone, anticoagulation, and smooth muscle proliferation (Furchgott & Zawadzki, 1980; Gewaltig & Kojda, 2002; Lefroy, Crake, Uren, Davies, & Maseri, 1993). Given the potentially detrimental effects of excess cortisol, exposure to daily mental stress-associated high cortisol concentration bursts over an extended period of time may contribute to the development of hypertension and atherosclerosis in high-reacting individuals.
Limitations
A limitation of this study was the small sample size overall and in particular in the cortisol stress reactivity analysis. Due to technical challenges or lack of participant consent, it was not possible to obtain blood samples for all participants. We used blood sampling because other unrelated blood variables were measured for a separate vascular protocol in these participants. Use of saliva sampling for cortisol determination in future studies would have less potential for sampling difficulty. While this small sample size indicates that the results should be regarded as preliminary, a moderate group effect size was detected.
A greater number of NSW reported taking medication. Although the cholesterol and BP levels were not different between groups, these variables were controlled by medication in two NSW participants, which may have implications for cardiovascular risk. In addition, one NSW participant for whom we have cortisol data reported taking a serotonin reuptake inhibitor. There is some evidence that this medication may reduce cortisol reactivity (Vermetten et al., 2006), and this could have contributed to our observation of nonsignificantly lower cortisol reactivity (p = .165) in the NSW group. Further research in a larger sample will be required to resolve any influence of the medication. In addition, we did not gather information regarding nonprescription vitamins or supplements and it is possible that this might have influenced our results.
This study only included women working a single shift work pattern making the results less generalizable to male SW or different shift schedules. In addition, our study did not permit identification of the mechanisms or factors within shift work that may have contributed to trends for group differences. For example, sleep quality and physical activity levels were not assessed in the study population. This is a limitation because there is evidence that these factors (Bassett, Lupis, Gianferante, Rohleder, & Wolf, 2015; Klaperski, von, Heinrichs, & Fuchs, 2014; Rimmele et al., 2009) influence stress reactivity.
Conclusion
These preliminary data suggest that SW may result in elevated cortisol and cardiovascular reactivity to acute stress. This is important because high reactivity is associated with future cardiovascular risk and may be a potential mechanistic pathway that leads from shift work to CVD. Future research is required to confirm if significantly heightened cortisol and cardiovascular reactivity exist in larger samples of SW, groups that contain men, and include different shift work patterns. Shift work is a necessary work pattern, particularly for the health-care sector. If we can better elucidate harmful pathways then we are better positioned to develop workplace and individual strategies, such as reduction of the physical and psychological stress associated with circadian disruption, to buffer and minimize these harmful effects.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a Natural Sciences and Engineering Research Council of Canada (NSERC) Discovery Grant to K. E. Pyke and a Garfield Kelly Cardiovascular Research Development Fund Grant to J. E. Tranmer and K. E. Pyke. I. N. Carson was supported by a NSERC CGS M scholarship.
