Abstract
Background
Relaxation techniques can reduce sympathetic nervous system activation and stress, potentially improving heart failure patients’ physical and psychological outcomes.
Purpose
To examine the effects of biofeedback-assisted relaxation (BFAR) therapy in patients with heart failure.
Methods
A prospective randomized control study was conducted. Participants in the treatment group received BFAR therapy, while participants in the control group received standard of care. Short-term outcomes were physical symptoms and psychosocial variables measured at baseline and 3 months; long-term outcomes were cardiac events and mortality assessed at 12 months.
Results
Fifty-two heart failure patients participated in the study: 23 (mean age 60.0 ± 13.7 years; 60.9% male; 39.1% New York Heart Association III/IV) in the treatment group and 29 (mean age 59.2 ± 12.2 years; 72.4% male; 48.3% New York Heart Association III/IV) in the control group. Short-term effects of BFAR on outcome variables were not significantly different between treatment and control groups. However, longer event-free survival was found in the treatment group compared with the control group (p = .019).
Conclusions/Implications for Practices
BFAR therapy is effective to improve cardiac event-free survival of heart failure patients and can be applied to clinical setting.
Introduction
Because developments in the management of heart disease have decreased mortality and thus the population is living longer, heart failure incidence and prevalence are expected to grow worldwide (Mozaffarian et al., 2015). As a consequence, the global impact of this condition is expected to dramatically intensify within the next 10 years (Mosterd & Hoes, 2007; Norton, Georgiopoulou, Kalogeropoulos, & Butler, 2011). Patients with heart failure experience debilitating physical symptoms, emotional distress, impaired quality of life, frequent hospitalizations, and premature death (Atherton et al., 2012; Ramani, Uber, & Mehra, 2010). To surmount the increasing heart failure epidemic, effective and innovative intervention strategies that improve outcomes for patients with heart failure must be developed.
Previous studies have not focused on nonpharmacologic intervention in heart failure management (Ramani et al., 2010). The use of relaxation therapy to induce the relaxation response is one potentially beneficial nonpharmacologic therapy in heart failure (Richardson, 2001; Woltz et al., 2012). Herbert Benson, professor and cardiologist of Harvard University, coined the term Relaxation Response in the early 1970s and tried to designate a physical status, including reduced sympathetic nervous system (SNS) tone, increased parasympathetic activity, decreased metabolism, decreased heart rate and blood pressure, and reduced oxygen consumption (Benson & Klipper, 1976). The use of biofeedback is a scientifically supported method of helping patients achieve the relaxation response and could potentially be used to lessen the negative sequelae of heart failure (Moser, Dracup, Woo, & Stevenson, 1997; Yu, Lee, & Woo, 2010).
The SNS initiates and sustains a series of intensive neurotransmitter activation, which further leads to marked vasoconstriction in heart failure. (Agarwal, Briasoulis, & Messerli, 2013; Braith & Edwards, 2003). Except for the disease-related physical manifestations or incapability, heart failure patients are often involved in different kinds of psychological distress, including feelings of depression, anxiety, and loss of control (Dekker, Peden, Lennie, Schooler, & Moser, 2009; Lee et al., 2010). Both neurohumoral activation and psychological consequences caused by heart failure can lead to decreased quality of life, frequent heart failure hospitalizations, and increased mortality. Biofeedback-assisted relaxation (BFAR) therapy is an existing intervention that has been shown in multiple previous studies to both diminish psychological distress and improve physical symptoms and quality of life in diverse populations (Evetovich, Conley, Todd, Rogers, & Stone, 2007; Yu, Lee, & Woo, 2007; Yu, Lee, Woo, & Hui, 2007).
BFAR therapy includes three complementary, but distinct, strategies to improve outcomes. When done successfully, BFAR (a) can decrease SNS activation, (b) produce stress reduction, and (c) result in vasodilatation (Moser et al., 1997; Yu et al., 2010). Through these synergistic effects, BFAR may have a powerful effect in patients with heart failure, but the long-term effects of this nonpharmacological intervention are unknown. The specific aim of this study was to examine both the short-term and long-term effects of BFAR therapy for patients with heart failure.
Methods
Study Design
A two-group, randomized prospective experimental design was used in this study with assessments at baseline, 3, and 12 months. Three- and 12-month follow-ups were chosen to assess the short- and long-term effects of the intervention, respectively. To examine the effect of BFAR and avoid the influence of angiotensin-converting-enzyme inhibitor (ACEI) or angiotensin II receptor antagonist (ARB) to outcomes, both within- and between-subject comparisons were made to determine the effect of BFAR therapy, with stratification to control for ACEI or ARB use. Subjects were recruited using a stratified randomization table so that half of participants had been prescribed an ACEI or ARB. Patients were randomly assigned to a BFAR therapy group and a usual care control group (Figure 1).
The flow diagrams of the participants through each stage of the study.
Sample and Data Collection Procedure
To be included in this BFAR study, patients (a) were adults (older than 18 years) and (b) had a definite diagnosis of heart failure by a cardiologist. Patients with a history of cerebral vascular accident, recent myocardial infarction (within the preceding 6 months), a major comorbid terminal illness, or receiving treatment such as hemodialysis or concurrent cancer treatment, were excluded. Patients with cognitive impairment or residing in institutional settings were also excluded. Patients were recruited from several medical centers located in southern Taiwan. The approval to conduct the study was obtained from the appropriate hospital institutional review boards (97-010B and NTUH-REC 200710001R) and informed consent was obtained from each patient. After identifying patients with heart failure at the outpatient clinics, nurse research assistants and cardiologists invited eligible subjects to participate in the study. Patients completed the questionnaires in the presence of nurse research assistants who were available to answer any question. In some cases, if necessary, the nurse research assistant read for patients to help them to respond to questionnaire. In general, it took the patients approximately 30 minutes to complete the questionnaires.
Measurement via Validated Questionnaires
Physical symptoms and psychosocial variables (i.e., depression, anxiety, perceived control, perceived social support, and health-related quality of life) were measured using the Modified Pulmonary Function Status and Dyspnea Questionnaire (PFSDQ-M; Huang et al., 2008a), Brief Symptom Inventory (BSI)-Depression & Anxiety Subscale (Derogatis, 1993), Control Attitudes Scale-Revised (Moser et al., 2009), Multidimensional Scale of Social Support Scale (MSPSS; Bruwer, Emsley, Kidd, Lochner, & Seedat, 2008), and Minnesota Living with Heart Failure Questionnaire (MLHFQ; Ho, Clochesy, Madigan, & Liu, 2007), respectively. Demographic variables and New York Heart Association (NYHA) functional class were obtained by patient interview. Left ventricular ejection fraction (LVEF) was obtained from the medical record.
Physical symptoms of dyspnea and fatigue
The PFSDQ-M was used to measure heart failure patients’ physical symptoms in this study. The PFSDQ-M was developed for measuring symptoms of patients with chronic pulmonary obstructive disease in 1994 and modified in 1998 (Lareau, Meek, & Roos, 1998). Caroci then used the PFSDQ-M to distinguish symptom differences between patients with heart failure and chronic pulmonary obstructive disease, and confirmed validity and reliability in heart failure patients (Caroci Ade & Lareau, 2004). The PFDSQ-M is a 40-item questionnaire that measures dyspnea and fatigue based on patients’ subjective perception of the symptoms. It consists of questions which assess the main components of the symptoms: experience, frequency, and intensity of symptom. The PFSDQ-M consists of general questions concerning the patient’s experience of dyspnea and fatigue, the frequency of symptom occurrence over the past month, and the overall intensity of dyspnea and fatigue (i.e., on most days, today, and with usual activity levels). Higher scores indicate more severe physical symptoms induced by heart failure. The Chinese version of the PFSDQ-M has been validated and is reliable (Huang et al., 2008b). In the current study, the Cronbach’s α of PFSDQ-M was .92.
Depression and anxiety
The BSI Depression and Anxiety Subscales (Derogatis, 2006) were used to measure heart failure patients’ depression and anxiety levels in the current study, and each contains six items (Derogatis, 1993). Patients rated their distress level related to a given item on a scale from 0 to 4 (0 = not at all and 4 = extreme). The mean of these six items produces the score. The score ranges from 0 to 4 for each subscale, and a higher score indicate higher levels of depression and anxiety. The Cronbach’s α of the BSI-depression and BSI-anxiety subscale was .82 and .73 in the current study.
Perceived social support
MSPSS which contains 12 items is a self-reported instrument used to measure social support (Bruwer et al., 2008). The total score and subscale scores for family, friends, and significant others indicate the level of perceived support. The MSPSS used a 7-point Likert scale where 1 = very strongly disagree and 7 = very strongly agree; therefore, a higher total score indicates more perceived support. The Cronbach’s α of the MSPSS questionnaire in the current study was .90.
Perceived control
The Control Attitudes Scale-Revised (CAS-R; Moser et al., 2009) is an 8-item scale modified from the original Cardiac Attitudes Index and Control Attitudes Scale. The CAS-R developed by Moser et al. (2009) measures the degree to which patients feel they have control (and, conversely, a sense of helplessness) related to their cardiac disease and can evaluate an individual’s perspective of their ability to control their heart condition Moser et al., 2009. Patients rate their level of agreement with statements in the instrument on a 5-point Likert scale, where a higher score indicates a higher level of perceived control. The Cronbach’s α of the CAS-R in the current study was .77 in the current study.
Health-related quality of life
The MLHFQ is a 21-item, disease-specific measure of health-related quality of life for patients with heart failure. Each item ranges from 0 to 5 and total scores range from 0 to 105. Higher scores indicate a more negative perception of quality of life. The MLHFQ has excellent psychometric properties, and the Chinese version has been validated previously (Ho et al., 2007).
Endpoint of cardiac event-free survival
Cardiac event-free survival was defined as living with heart failure without an emergency department visit for heart failure exacerbation or hospital admission or death attributed to cardiac events. Patient or family interview and medical records were used to collect these data.
BFAR Intervention
The BFAR intervention in the current study was conducted once a week for 6 weeks and consisted of skin temperature BFAR. Sessions were individualized based on the needs of the patient and included teaching, demonstration, practice, and reinforcement of BFAR. During each session, temperature sensors are attached, and the display is placed where it can be seen by the subject. Patients also hear an auditory signal that changes pitch differentially as temperature increases or decreases. In the first session, patients are given instructions to assist them to increase their skin temperature and they practice these for up to 20 minutes. All remaining sessions begin with a review of the practice diary and discussion of patients’ practice experiences and any difficulties they had. Patients are then attached to the biofeedback sensors and asked to increase their skin temperature without coaching. Sessions end with reinforcement to practice. The interventionist was a trained nursing specialist with expertise in the care of patients with heart failure and psychiatric counseling. All patients received the standard clinical care. Only patients in the treatment group received the BFAR.
Statistics
In the associated results section and tables, categorical data are presented as number (percentage) and continuous data are presented as mean ± standard deviation. To assess the effect of the intervention on short-term outcomes, one-way analysis of covariance was used to compare the adjusted posttest score (adjusted for pretest score) between experimental and control groups. The first event-free survival curves were depicted by the Kaplan–Meier method, with the log rank test being used to assess whether the treatment group (intervention effect) was associated with longer time to first event during 12-month follow-up. The difference in first event-free survival between study groups was further compared by a multivariate Cox proportional hazard model with the adjustment of gender, age, LVEF, NYHA functional class, and control attitudes scale (CAS-R). All data analyses were conducted using SPSS software version 22 (SPSS Inc., Chicago, IL).
Results
Baseline
Comparison of Characteristics Between the Treatment and Control Groups.

Note. NYHA = New York Heart Association functional classification; EF: ejection fraction; HF: heart failure; MLHFQ = Minnesota Living with Heart Failure Questionnaire. Categorical data were presented as N (%) and was compared using Fisher’s exact test. Continuous data were expressed as mean ± SD and was compared using independent sample t-test.
There were no differences in the psychosocial variables at baseline between the study groups, except for control attitudes scale (t = 2.65, p = .011). Heart failure patients in the treatment group had higher levels of perceived control at baseline than heart failure patients in the control group (Table 1).
Short-Term Psychosocial Outcomes
Summary Statistics for Comparison of 3-Month Outcomes Between Patients in the Treatment and Control Groups.
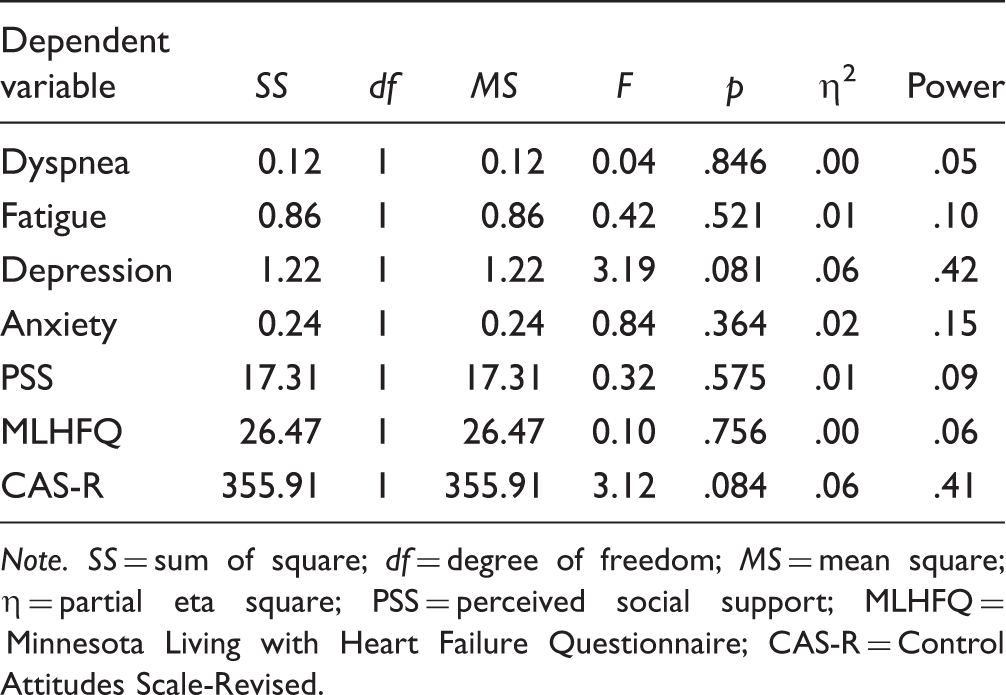
Note. SS = sum of square; df = degree of freedom; MS = mean square; η = partial eta square; PSS = perceived social support; MLHFQ = Minnesota Living with Heart Failure Questionnaire; CAS-R = Control Attitudes Scale-Revised.
Descriptive Statistics by Group of Adjusted Scores at 3 Months for All Short-Term Outcomes.

Note. The posttest score was adjusted by pretest score; SE = standard error; PSS = perceived social support; MLHFQ = Minnesota Living with Heart Failure Questionnaire; CAS-R = Control Attitudes Scale-Revised.
Long-Term Outcomes
The mean patient follow-up period was 294 days (SD = 113 days). During this period, 18 patients (34.6%) suffered a cardiac event. The incidence rate in the treatment and control groups was 17.4% (4 out of 23) and 48.3% (14 out of 29), respectively.
The mean event-free survival time in the treatment and control groups was 331 ± 17 days and 264 ± 23 days, respectively. The log-rank test from the Kaplan–Meier analysis was statistically significant, χ2(1) = 5.47, p = .019, indicating that prognosis in the two groups was different based on study group (Figure 2). In a further multivariate Cox proportional hazard model controlling for gender, age, LVEF, NYHA functional class, and perceived control score from the CAS-R, the control group had a higher risk of events when compared with treatment group (adjusted hazard ratio = 4.42, 95% CI of hazard ratio = 1.30 ∼ 14.99, p = .017).
Kaplan–Meier curves demonstrating time to first event by group.
Discussion
In this study of a nonpharmacologic BFAR intervention for management heart failure, we demonstrated that event-free survival was significantly better in patients in the treatment compared with the control group. There was no short-term effect of the intervention seen at 3-month follow-up as measured in the psychosocial or symptom variables.
Although only a few investigators have examined the effects of interventions similar to the BFAR intervention tested in this study, most demonstrated that BFAR can improve heart failure patients’ quality of life (Chang et al., 2005; Yu et al., 2010) and psychological distress (Yu et al., 2007). In these studies, outcomes were only assessed within a time frame of 3 months after the intervention. Our inability to demonstrate a short-term effect of the intervention could be a result of too short a follow-up period after the end of the intervention. It is possible that testing at 6 or 9 months of psychosocial and symptom variables would have demonstrated a significant intervention effect. Our inability to find an effect on quality of life, symptoms, and psychosocial variables also may be related to an historical threat to internal validity that occurred during our study.
The Typhoon Morakot landfall in southern Taiwan affected around two thirds of our subjects during the time period when posttest data were being collected. Many participants in our study suffered from property loss and unanticipated trauma from the disaster, which may have impacted the short-term effects on heart failure patients’ outcomes. Although our study was done with scrupulous attention to protocol, the natural disaster and resulting complications of this event were unavoidable. A disaster such as this Typhoon likely overwhelmed coping responses in the short-term.
Despite the lack of an intervention effect in the short-term, a significant effect of the BFAR intervention was found on event-free survival at the 12-month follow-up. Our original hypothesis was that as a result of its physical and psychological effects, BFAR could have a clinically meaningful impact on rehospitalizations, survival, and quality of life. To produce a physical effect, BFAR may be an effective adjunct to pharmacologic therapy in the management of heart failure. Patients may also receive psychological benefits from BFAR by achieving stress reduction. Stress reduction may diminish activation of SNS system (Woltz et al., 2012; Wu, Corley, Lennie, & Moser, 2012). We speculate that given the short follow-up after the intervention and the acute overwhelming threat of the typhoon, it took longer for the positive effects of the intervention to become apparent. Therefore, continuous enforcement and evaluation when applying the BFAR to real clinical situation may be necessary to assure this intervention has an impact.
Given the effects of the intervention, there is still a gap between clinical practice and the use of evidence from empirical studies such as this one. BFAR takes time to become familiar with and practice to exert an effect. Within this context of the intervention, each patient required about 350 minutes for the total BFAR therapy. In the present health-care system, such an intervention would not be affordable as a routine service. Pal, Ganesh, Karthik, Nanda, and Pal (2013) found that young medical students who practiced daily 1 hour for 6 weeks could improve their autonomic balance and promote cardiovascular health; heart failure patients even need more time and practice to achieve the same effects. Thus, further research to appropriately modify the tested intervention is warranted.
Limitations
The study has a number of limitations. First, the generalization of the results of this study is limited due to its single center study. Second, any face-to-face encounter is difficult to standardize. Intervention fidelity was enhanced by training sessions with all interventionists and consistence between them were confirmed before real intervention session. However, the BFAR intervention is complex, and there may have been some differences between interventionists. Finally, because the BFAR require 6 weeks of engagement, patients who were willing to receive the intervention could have unique characteristics that made them more susceptible to such intervention. Therefore, heart failure patients in the treatment group had higher levels of perceived control at baseline than heart failure patients in the control group even using randomization allocation in the study. To control the treatment covariates, larger sample size or different randomization method are needed for the future study.
Conclusion
Our findings demonstrate that BFAR can improve the end point of survival free of cardiac rehospitalization and death in patients with heart failure. Although our sample was small and unexpected internal validity issues occurred, the positive effect of BFAR intervention emerged.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Science Council in Taiwan (NSC 97-2314-B-255-006-MY3), Sigma Theta Tau International Honor Society of Nursing (2008–2009 Sigma Theta Tau International Small Grant), and Chang Gung Medical Foundation.
