Abstract
Phantom tumour, vanishing tumour, and pseudotumour of the lung are terms used to describe the tumour appearance on a chest radiograph of a decompensated heart failure patient, which typically disappears after appropriate treatment of their underlying condition. It is a loculated pleural effusion in the fissures that gives rise to the tumour appearance on the chest radiograph of a patient with heart failure. Its existence might mislead the diagnosis, resulting in unnecessary invasive diagnostic investigations done on the patient. We present a case of a 64 year-old male with multiple co-morbidities presenting with dyspnea, loss of weight, and loss of appetite. A chest radiograph showed homogenous opacity over the right lower zone with cardiomegaly, raising the suspicion of a lung mass apart from heart failure. It disappeared after appropriate diuretic treatment. The computed tomography (CT) thorax, which was planned by the primary team, subsequently showed no lung mass. Hence, a high index of suspicion of phantom tumour is important when a lung mass is noted on a chest radiograph of a patient clinically presented with heart failure. This is because exposure to ionising radiation from CT is completely avoidable with just appropriate treatment of fluid overload. However, CT is still the best modality for assisting physicians in a doubtful case.
Introduction
Phantom tumour, vanishing tumour, and pseudotumour of lung are terms used to describe a tumour appearance on a chest radiograph of a decompensated heart failure patient which typically disappears after appropriate diuretic treatment. It was first reported in 1928 by Stewart et al. as an occurrence of encapsulated interlobar effusions in cases of cardiac failure.1,2 The tumour appearance on a chest radiograph is actually caused by localised transudative interlobar pleural fluid collection in a patient with congestive heart failure. 3 Incidence is rare in that it only comes from a small number of reported cases in literature, but it is a well-known finding in heart failure patients. Its existence might cause a diagnostic error resulting in unnecessary invasive diagnostic investigation for the patient. 3 Hence, it is important to be aware of the existence of such a form of pleural effusion when a patient with congestive heart failure presents with a pulmonary mass on a chest radiograph.
Case report
We present a case of a 64 year-old gentleman who is an ex-smoker with Type II Diabetes Mellitus, Hypertension, Chronic Kidney Disease stage 3A and dilated cardiomyopathy, presenting to the emergency department with dyspnea for 4 days, associated with reduced effort tolerance for 1 week. Further history reveals that the patient had a loss of appetite and lost approximately 10 kg in the previous year.
He was diagnosed with dilated cardiomyopathy in 2019 under cardiology follow-up. His echocardiography (ECHO) in 2019 showed poor ejection fraction (ejection fraction: 25%–30%, global hypokinesia). However, the patient opted for medical therapy. Subsequently, he had missed all his follow-up and medication since his last discharge in 2019.
Upon arrival to the emergency department, the patient was mildly tachypnoeic with a respiratory rate of 24 breaths per minute. Oxygen saturation was 91% under room air. The patient was noted to have bilateral pitting edema up to mid-shin with auscultation of the lungs showing bilateral lower zone fine crepitations. A chest radiograph on arrival (Figure 1) showed homogenous opacity over the right lower zone with cardiomegaly, raising suspicion of a lung mass apart from heart failure Chest X-ray upon presentation - red arrow showing right lower zone opacity, suspicious of lung mass.
He was put on nasal prong with oxygen flow of 3 L/min (NpO2 3 L/min) and treated for fluid overload. The patient was started on intravenous frusemide 80 mg stat, then 40 mg, three times a day. Clinically, diuresis improved his symptoms, and he was able to wean off NpO2 3 L/min the next day. His oxygen saturation showed 98% under room air. Arterial blood gas showed no respiratory failure (pH: 7.438, pCO2: 30.1, pO2: 77.9, HCO3: 22.3, SaO2: 95%). The chest radiograph was repeated the next day post diuresis, showing a similar appearance.
In view of the chest radiograph raising suspicion of a lung mass apart from heart failure, the patient was referred to the chest team for further evaluation of lung malignancy and it was decided to proceed with computed tomography (CT) Thorax in 2 weeks’ time. The CT thorax (Figure 2) showed cardiomegaly with minimal right pleural effusion without evidence of a lung mass. Computed tomography thorax after 2 weeks optimal diuresis - red arrow showing right pleural effusion, no lung mass.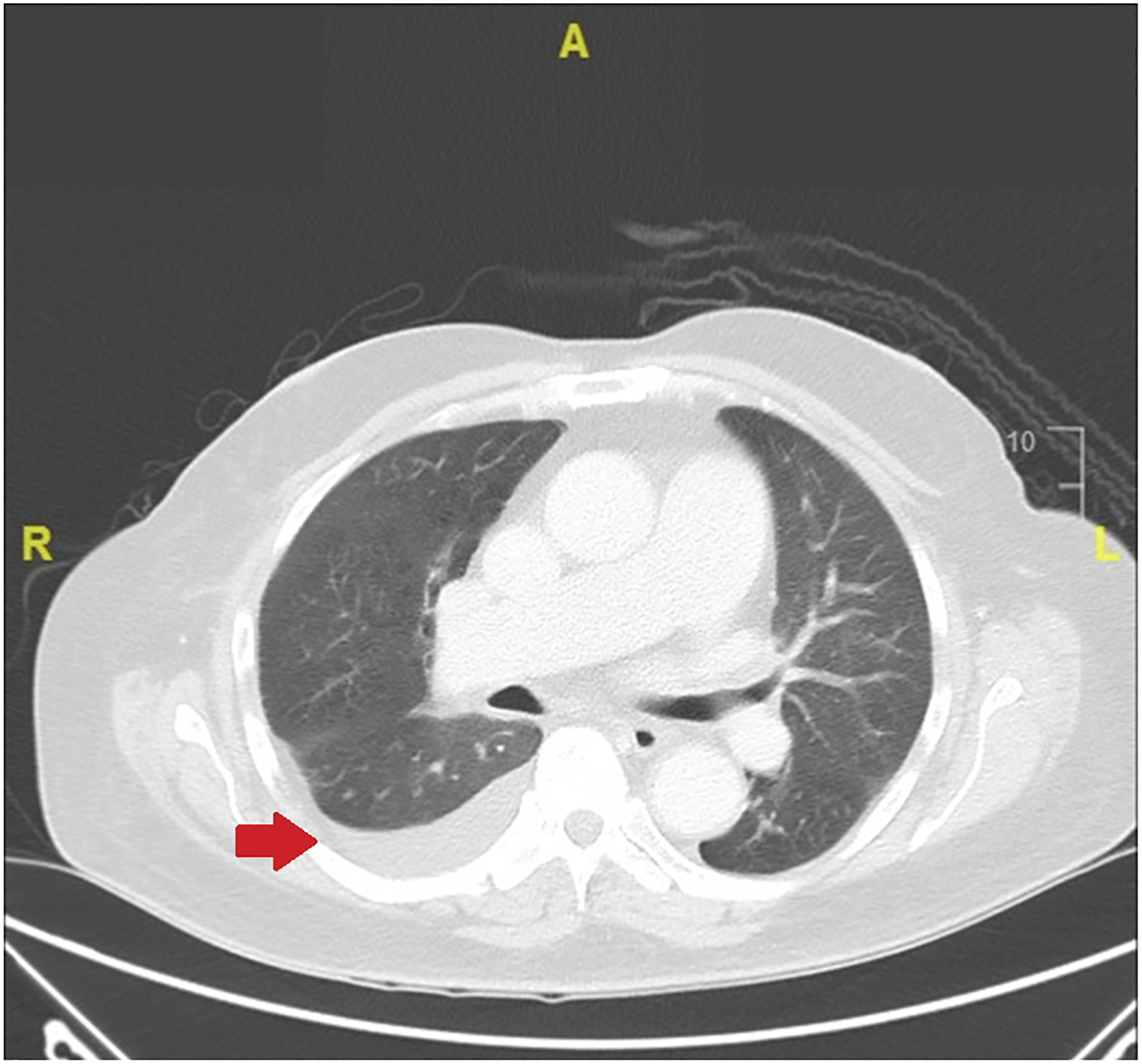
The patient was discharged after a CT thorax and referred to the cardiology team for further management of his condition.
Discussion
The vanishing or phantom tumour is a well-recognized entity in a patient with fluid overload, although the incidence is rare.3,4 Various literature suggests that the pathogenesis of this atypical fluid distribution is caused by pre-existing pleuritis, which in turn obliterates the pleural space around the edge of the fissure. Circulatory overload leads to fluid transudation due to an increase in hydrostatic pressure of the pulmonary vasculature. When fluid transudation exceeds the fluid reabsorption via pleural lymphatic, it causes the fluid to accumulate in the affected fissure and forms a “lung mass” appearance on a chest radiograph. Another pathogenesis proposed is related to the “suction cup” effect of a partially atelectatic lung adjacent to the effusion. It leads to a local increase in elastic recoil, which in turn favours localised pleural fluid collection.1,5
Due to its underlying pathophysiology as described, it is common that those loculated pleural effusions are found in the horizontal fissure in about three-quarters of the reported cases, followed by the oblique fissure, and very rarely multiple collections in both fissures.5,6
One should not deduce that such loculated pleural effusion only occurs in patients with severe heart failure, but it can occur in any patient with congestive heart failure presenting with fluid overload. Apart from heart failure, it is important to remember that phantom tumours can also occur in patients with overwhelming fluid overload due to other diseases as long as they share similar pathophysiology. Hence, it is good to have a few differential diagnoses in mind when encountering patients with such loculated pleural effusion. Causes are either transudative or exudative in origin, similar as per differentials of pleural effusion, as ultimately this loculation results from the accumulation of fluid in the pleural cavity. Transudative causes include chronic kidney disease, congestive or left heart failure, chronic liver disease, and hypoalbuminemia. Exudative causes include parapneumonic pleural effusions, pleural empyema, malignant pleural effusion, haemothorax, and chylothorax. 1
As for this patient, he was initially referred for further investigations due to the suspicion of lung malignancy. A CT thorax was performed due to the presenting constitutional symptoms and the chest radiograph showed no resolution post diuresis. A publication proposed that recognising such a chest radiograph might save the patient from being exposed to unnecessary ionising radiation from CT Thorax. 6 A lateral chest radiograph can help with diagnosis because it is very sensitive and can detect effusions as small as 5 mL in experimental studies. 7 However, assessment via CT thorax is vital in doubtful cases to assist the physician in making an accurate diagnosis.
Conclusion
It is key that one should not only suspect lung malignancy from the initial chest radiograph presentation but also possess a high index of suspicion of phantom tumour in patients with heart failure presenting with a “lung mass” on chest radiograph. This is due to the fact that recognising the phantom tumour on the chest radiograph in the appropriate clinical setting is prudent in order to avoid unnecessary ionising radiation and healthcare costs from CT with just appropriate fluid overload treatment. However, CT is still the best modality for assisting the physician in a difficult case.
Footnotes
Acknowledgements
None.
Author contributions
Kuan Yau Yeh: obtained patient's consent, prepared and drafted manuscript. Asyif Fairus Ahmad: assisted in patient's data collection and review of current available publication. Arvindran Alaga: Supervised the manuscript preparation and assisted in proof read and further correction on the manuscript. All authors provided critical feedback, reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval not applicable as per Malaysia National Medical Research Register (NMRR) recommendation. However, registration of case report had been approved under NMRR with research ID: RSCH ID-22-00261-TXR, NMRR ID: NMRR ID-22-00904-UVB.
Informed consent
Written informed consent was obtained from the patient for their anonymised information to be published in this article.
Availability of data and materials
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
