Abstract
Serotonin (5HT) is involved in emotion and sleep regulation and the 5HT transporter (5HTT) regulates 5HT function. A common 44-base pair deletion (short allele) or insertion (long allele) polymorphism in the promoter region of 5HTT (5-HTTLPR) is differentially associated with 5HTT transcription efficiency. Under stressful conditions, the short allele of 5-HTTLPR has been associated with depression and sleep disturbance. Black women are at higher risk for preterm labor and depressive symptoms. Thus, this exploratory study aimed to examine whether depressive symptoms and sleep disturbance in Black mothers would vary as a function of the 5-HTTLPR genotype when they faced the stress of infant hospitalization after preterm birth at early postpartum. A total of 30 Black mothers filled out a battery of questionnaires, including the Perceived Stress Scale, Edinburgh Postnatal Depression Scale, and General Sleep Disturbance Scale. A wrist actigraph was used to assess total sleep time and circadian activity rhythms. Buccal cells from saliva were collected to test the 5-HTTLPR genotype. Results showed that about 38% of the mothers were heterozygous for the short (S/L) allele, and 62% were homozygous for the long (L/L) allele. Mothers’ perceived global stress, depressive symptoms, and circadian activity rhythms did not vary with their 5-HTTLPR genotypes. Unexpectedly, mothers with the L/L allele reported greater sleep disturbances than those with the S/L allele. Ethnic specificity in genetic susceptibility to stress was discussed.
Keywords
Having a low-birth-weight (LBW) preterm infant hospitalized in the neonatal intensive care unit (NICU) is a stressful experience for mothers (Holditch-Davis, Bartlett, Blickman, & Miles, 2003; Holditch-Davis et al., 2009; Lee & Hsu, 2012; Miles, Holditch-Davis, Schwartz, & Scher, 2007). Stress has been identified as the most common cause for both depressive symptoms (Kendler, Karkowski, & Prescott, 1999) and insomnia (Roehrs, Zorick, & Roth, 2000). As a group, mothers with a hospitalized LBW preterm infant experience elevated depressive symptoms and disrupted sleep during early postpartum (Lee & Hsu, 2012). However, marked individual differences are also observed. For example, while some mothers were extremely distressed during the first 2 years after a preterm birth, some mothers had a moderate level of distress, and others only experienced a low level of distress (Holditch-Davis et al., 2009). Genetic variability has been suggested to underlie individual differences in mothers’ responses to stress (Caspi et al., 2003; Risch et al., 2009). A “genetic plasticity” or “biological susceptibility” model further posits that some genotypes are highly susceptible to environmental influences, whereas others are not (Belsky et al., 2009; Ellis & Boyce, 2011). This model suggests that individuals with genetic susceptibility would have more negative outcomes than those without genetic susceptibility when the environment is poor and better outcomes when the environment is rich. Mothers’ reactivity to the stress associated with preterm birth may be differentially associated with genetic variations. Relative to Whites, Blacks are at higher risk not only for socioeconomic disadvantage, but also for preterm birth (18.5%) and depression (40.6%; Centers for Disease Control and Prevention [CDC], 2008, 2010). Thus, in this study we focused on a variable nucleotide repeat polymorphism in the promoter region of 5HTT (5-HTTLPR) to examine its association with depressive symptoms, sleep disturbance, and circadian rhythms among Black mothers who are facing the stress of infant hospitalization resulting from preterm birth at an early postpartum period.
Serotonin, localized in the raphe nuclei, is an inhibitory neurotransmitter in the central nervous system. Central nervous system serotonin (5HT) has been implicated in emotion regulation (Albert & Lemonde, 2004; Fortier et al., 2010) and sleep regulation (Brummett, Krystal, Siegler, et al., 2007), and increased activity of serotonin neurons is associated with wakefulness (Tabuchi et al., 2013). Dysfunctions in the serotonergic system have also been linked to psychopathology, including depressive or anxious disorders (Brown & Harira, 2006; van Goozen, Fairchild, Snoek, & Harold, 2007). A functional polymorphism in the promoter region of the serotonin transporter gene, 5-HTTLPR, involves a 44-repeat insertion or deletion (Contreras et al., 2009). In humans, the 5-HTTLPR polymorphism linked to the serotonergic system has two common alleles, the short (S) and the long (L). The S-allele variant of 5-HTTLPR has been found to increase reactivity to psychosocial adversity. Individuals who are either homozygous for the S-allele (S/S) or heterozygous (S/L) are at an increased risk for poor sleep quality (Brummett, Krystal, Siegler, et al., 2007) and mood disorders, such as depression, bipolar disorder, and seasonal affective disorder (Beevers, Gibb, McGeary, & Miller, 2007; Caspi et al., 2003), particularly when exposed to stressful environmental conditions. Specifically, among female caregivers caring for relatives with dementia, S-allele carriers had higher depressive symptom scores than the L-allele carriers (Brummett et al., 2008). Also, among women who experienced the stressor of low socioeconomic status in childhood, those with the S-allele had higher depression scores than those with the L-allele (Brummett et al., 2008).
In the general population, the prevalence of postpartum depression ranges from 11.7% to 20.4%. Mothers of LBW preterm infants are particularly vulnerable to depressive symptoms (Poehlmann, Schwichtenberg, Bolt, & Dilworth-Bart, 2009; Segre, Losch, & O’Hara, 2006). As a result of the combination of stress and sleep disturbances, the prevalence rate for depressed mothers of preterm infants is almost three times higher than the general population (Vigod, Villegas, Dennis, & Ross, 2010). As noted earlier, Black women are at high risk for preterm labor (CDC, 2010) and depression (CDC, 2008). Given that the S-allele of 5-HTTLPR has been associated with an increased likelihood of depression when a person is under significant life stress, it was expected that after a preterm birth, Black mothers with the 5-HTTLPR S-allele would experience greater depressive symptoms than those with 5-HTTLPR L-allele.
In addition to stress and fatigue, mothers with a hospitalized LBW infant are sleep disrupted and sleep deprived (Lee & Hsu, 2012). Even without infant care responsibilities, they still experienced disrupted sleep and reported poor sleep quality when sleeping at home (Lee, Aycock, & Moloney, 2013; Lee, Lee, Rankin, Weiss, & Alkon, 2005). Wrist actigraphy data further showed that their 24-hr biological and activity cycles, known as circadian activity rhythms (CARs), were irregular (Lee, Grantham, Shelton, & Meaney-Delman, 2012). CARs are closely linked to sleep and to health (Germain & Kupfer, 2008). At least two factors may explain the disrupted CAR in mothers of preterm infants: (a) circadian phase shift (Postolache & Oren, 2005) resulting from prolonged exposure to artificial dim light in the NICU and (b) social rhythms disturbance (Ehlers, Frank, & Kupfer, 1988) resulting from altered daily routines and changed social activities, including altered maternal roles due to separation from the infant.
Available research suggests that variations in the 5-HTTLPR genotypes underlie sleep-related phenotypes. For example, patients with insomnia disorder were more likely to have the S-allele than control individuals with normal sleep (Deuschle et al., 2010). A study with freshmen in an ivy-league university found that students with S/S allele had shorter total sleep time (TST) than those with the S/L and L/L alleles (Carskadon, Sharkey, Knopik, & McGeary, 2012). In particular, students who reported shorter nocturnal sleep and higher depressed mood were more likely to be S/S allele carriers than others. Previous research also found that the S-allele was associated with poorer sleep quality in adult primary caregivers of relatives with dementia, but such association was not found among non-caregivers (Brummett, Krystal, Ashley-Koch, et al., 2007). Together, these findings suggest that in stressful situations, the 5-HTTLPR S-allele is associated with poorer sleep quality. It was hypothesized that mothers with the S-allele would have higher levels of sleep disturbance and less synchronized CARs.
Methods
A purposive sampling was used in this cross-sectional exploratory study. A total of 30 Black mothers were recruited from two NICUs affiliated with a university in a southern metropolitan city in the United States. The inclusion criteria were that (a) mothers were at least 20 years of age and self-identified as an African American; (b) mothers whose infants hospitalized in the NICU had gestational age less than 37 weeks, birth weight less than 2,500 gm, and without other medical morbidities and were unlikely to be discharged within a 5-week postnatal age based on the medical discharge plan; (c) mothers were able to read and speak English as evidenced by their ability to read aloud the first paragraph of the consent form; and (d) mothers were willing to participate in sleep–wake activity monitoring for three consecutive days/nights during the second week postpartum. Mothers were excluded from participation for any of the following conditions: 1) if they had a history of depression, bipolar disorder, or other affective illness, 2) if they had a history of diagnosed sleep disorder such as sleep apnea or nocturnal myoclonus (which might result in a higher waking time from the overnight wrist actigraphy monitoring), (c) if they were shift workers whose circadian rhythms may already be affected, (d) if they were using medications that might alter sleep (e.g., central nervous system stimulants, depressants, or antidepressants), (e) if they required an extended hospitalization period (more than three days for vaginal birth mothers and more than seven days for cesarean birth mothers), and (f) if they had an infant rated 5 or higher on the Clinical Risk Index for Babies (CRIB; Network, 1993) by the attending physician indicating a high morbidity.
Protocol
The NICU admission logs were screened weekly to identify potential participants. The bedside nurse informed all eligible mothers about the study and referred those who expressed an interest to the research team. A research team member set up an appointment with interested mothers for informed consent and data collection. After signing the consent form, buccal cells from saliva were collected from mothers to test for the serotonin transporter polymorphism (5-HTTLPR). A wrist actigraph was used to collect TST and CARs. In addition, mothers filled out a battery of questionnaires to assess their sleep quality, stress, and depressive symptoms. Sociodemographic data were also collected.
Measurements
Genotyping
Saliva samples used for genotyping were obtained from mothers using Oragene DNA kits (Genetek, Calgary, Alberta, and Canada). Mothers rinsed their mouths with tap water and then deposited 4 ml of saliva in the Oragene sample vial. The vial was sealed, kept at room temperature, and shipped to a university laboratory where samples were assayed according to the manufacturer’s specifications. Extracted DNA samples were normalized to 5 ng/µl in ddH2O for the downstream assays. All samples were done in duplicate with eight negative controls in total. S- versus L-genotype of 5-HTTLPR was determined via polymerase chain reaction following the standard and validated protocol from KBiosciences (a division of LGC genomics).
Maternal sleep
Maternal sleep was measured using both objective (wrist actigraph) and subjective (General Sleep Disturbance Scale [GSDS]) measurements.
The
The GSDS was used to assess mothers’ subjective evaluations of their sleep quality and quantity over the past week (Lee, 1992). This 21-item instrument was rated on 8-point Likert scales ranging from 0 (
Maternal stress
The Perceived Stress Scale (PSS), a 10-item, 5-point scale, was used to assess global, everyday stress experienced by the mother in the past month. Higher scores indicated greater stress as experienced by mothers. The PSS possesses adequate psychometric properties (Cohen, Kamarck, & Mermelstein, 1983); the internal consistency was .85 in the current study.
Depressive symptoms
The Edinburgh Postnatal Depression Scale (EPDS; Cox, Holden, & Sagovsky, 1987), a 10-item widely used questionnaire, was administered to measure the severity of maternal depressive symptoms over the past week on 4-point scales from 0 (
Data Analysis
The variables were evaluated to ensure that they all met the assumptions for further data analysis. Bivariate correlations were computed to reveal the magnitudes of associations between continuous variables.
Results
Maternal Demographics and Study Variables (

Demographics and Characteristics of the Infants (

Maternal Stress, Depressive Symptoms, and Sleep
Descriptive Statistics for Sleep Measures (

EPDS = Edinburgh Postnatal Depression Scale; GSDS = General Sleep Disturbance Scale; PSS = perceived stress scale; TST = total sleep time.
Nocturnal TST, as recorded by the wrist actigraph, of less than 7 hr did not meet the recommendation from the National Sleep Foundation (NSF) of 7 to 9 hr TST for adults (Hirshkowitz et al., 2015). The daytime TST was more than 1 hr, which was longer than the maximum nap time (NSF, 2010). Compared with normal adults, mothers in this study had a statistically significant longer percentage of WASO, spending 5% more time awake after going to bed. With respect to circadian rhythms, the mothers’ CAR median was 0.69 (ranging from 0.42 to 1.03), and the group average was 0.68 which is statistically significantly lower than the ideal CAR of 1,
Association of 5-HTTLPR with Depressive Symptoms and Sleep
Although mothers with S/L and L/L alleles did not differ in their perceived stress in early postpartum after preterm delivery, more than one half of the mothers (55.5%) with the S/L genotype, in contrast to about one third of the mothers with L/L homozygotes, reported clinically significant depressive symptoms (EPDS ≥ 10). However, this group difference was not statistically significant.
Comparisons between Carriers of L/L and S/L Alleles.
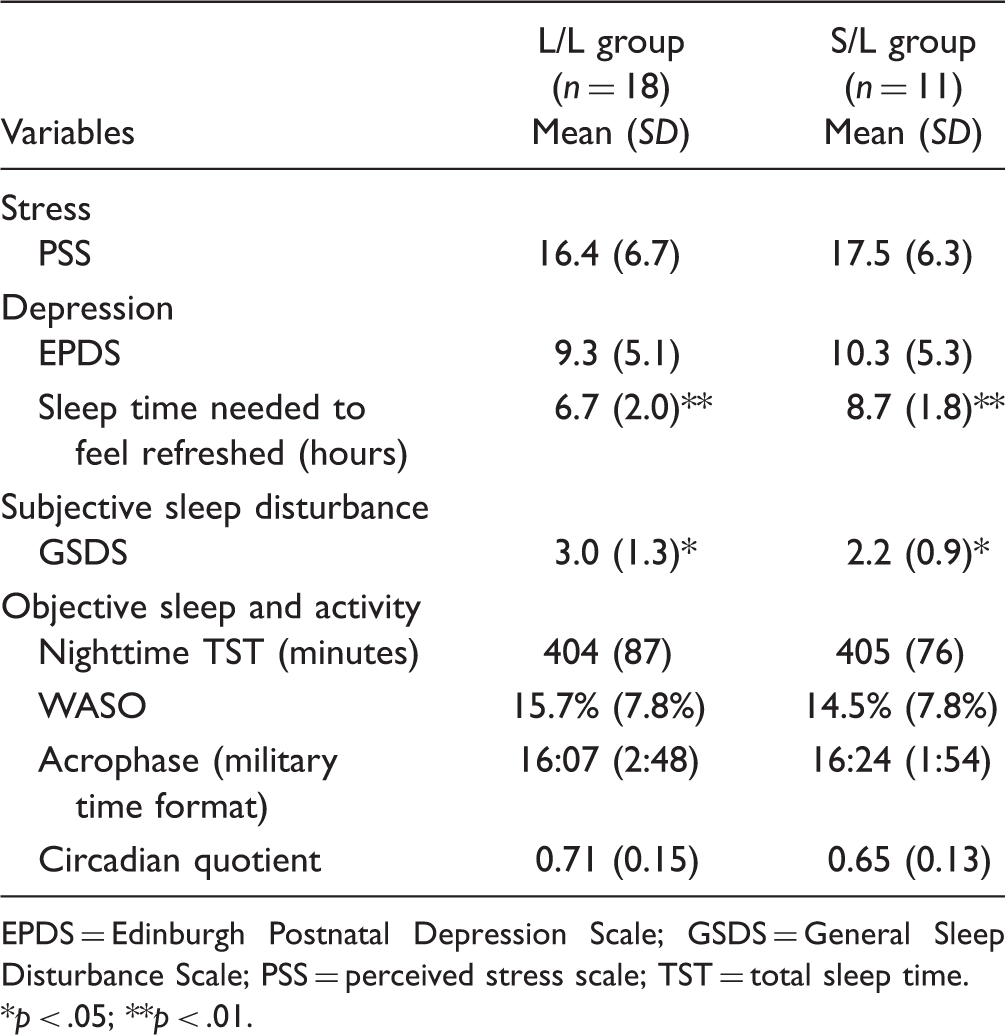
EPDS = Edinburgh Postnatal Depression Scale; GSDS = General Sleep Disturbance Scale; PSS = perceived stress scale; TST = total sleep time.
Discussion
The current study examined the association of the 5-HTTLPR gene with depressive symptoms, sleep, and CARs among Black mothers with a hospitalized preterm infant during their early postpartum period. It was found that after preterm delivery, Black mothers experienced heightened stress, elevated depressive symptoms, and poor sleep. This pattern was similar to previous findings in studies of mothers with a medically ill infant hospitalized in the ICU (Lee et al., 2013; Lee & Hsu, 2012). Poor sleep experienced by Black mothers was also similar to mothers with a well baby during the first month of postpartum (Gay, Lee, & Lee, 2004), although the mothers in the current study did not need to care for their infant at home. Poor sleep among these mothers with an infant in the ICU could be resulted from stress (Lee et al., 2005), as hyperarousal and sympathetic nervous system activation occur under stressful circumstances (Roehrs et al., 2000).
This study also found that more Black mothers with S/L alleles were classified with clinically significant depressive symptoms than those with L/L alleles in early postpartum after preterm delivery. Although not statistically significant (which is likely due to the small sample size), such a pattern is consistent with a previous study that showed when individuals experience chronic stress in early life, those with short alleles (S/L and S/S) reported higher depression scores than those with the long (L/L) allele (Hammen, Brennan, Keenan-Miller, Hazel, & Najman, 2010). Differential associations between S- and L-alleles with reactivity to stress may be because, relative to the L-allele, the S-allele is linked to reduce transcription efficiency, lower transporter levels, and diminished serotonin uptake. Allelic variation of 5-HTTLPR was also differentially associated with treatment effect for depressive mood. After receiving light therapy combined with total sleep deprivation, the treatment effect for mood amelioration was more marked in those with the L/L allele than in those with the S/L or S/S alleles (Benedetti et al., 2003). Moreover, the short allele of the 5-HTTLPR promoter polymorphism was also associated with the use of fewer problem-solving strategies (Wilhelm et al., 2007). Finally, multiple genes and gene–gene interactions may be involved in individuals’ depressive responses to stressful events. For example, although the L/L 5-HTTLPR is associated with significantly fewer symptoms of depression, the presence of interleukin-6 (IL-6) associated with the inflammatory response system may further alter its effect. The “protective” effect of the 5-HTTLPR polymorphism is evident only in the presence of the low IL-6 genotype and not in the presence of the high IL-6 genotype (Bull et al., 2009); therefore, further study should also examine the IL-6 genotype.
Furthermore, compared with those with the L-allele 5-HHTTLPR genotype, Black mothers with the S-allele required more nocturnal sleep time to feel refreshed. Specifically, mothers with S/L alleles slept almost 2 hr less than what they needed, whereas mothers with L/L alleles slept similar to what they needed, which might indicate that sleep plays a protective role in depressive symptoms. The sleep deviation index (SDI) was further examined; SDI is an absolute value of the ratio of the difference between nocturnal actual TST and needed sleep time to 24 hr (Lee & Hsu, 2012). The SDI should range between 0 and 1, and 0 indicates the TST exactly matches the individual preferred sleep time. There were statistically significant differences between the two groups, and the SDI in S/L group was less ideal than the L/L group,
With respect to CAR, Black mothers with the S/L allele were less synchronized as compared with those with the L/L allele (0.65 vs. 0.71), albeit not statistically significant. Desynchronized CAR was found to be associated with poor well-being in postpartum women (Lee et al., 2012; Lee, Lee, Aycock, & Decker, 2010). The decreased CAR may explain why Black mothers with the S/L allele were more likely to be classified with clinically significant depression than those with the L/L allele. Research is limited about CAR among postpartum women. Future research is needed to investigate exactly how the experience of having an LBW infant in the NICU may negatively impact mothers’ environmental cues, such as social activities and other daily life events that entrain circadian rhythms.
Limitations
Given that this study included a small sample from one region of the United States and that the data were collected at one point of time, the findings should be interpreted with caution. Further research should replicate this study with a larger and more ethnically diverse sample of women and examine maternal sleep and health outcomes over time, both during infant hospitalization and after hospital discharge. In addition, the recent recommendation for wrist actigraph monitor period is one week (Briscoe et al., 2014); therefore, 3-day monitoring may not be sufficient to explain circadian rhythms, and future study should prolong monitoring time to better validate circadian rhythms.
Conclusion
Findings from this small-sized study indicate that in early stage postpartum, mothers with heterozygous (S/L) or homozygous for the long (L/L) allele did not differ in their perceived global stress, depressive symptoms, nocturnal TST, and CAR synchronization. However, mothers with the L/L allele reported greater sleep disturbances and required less nocturnal TST to feel refreshed than mothers with S/L allele. Future research is needed to investigate, prenatally and postnatally, the role of disturbed sleep in depressive symptoms among Black mothers with the S-allele of 5-HTTLPR.
Footnotes
Acknowledgment
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute on Drug Abuse or the National Institutes of Health.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project described was supported by the Center for Contextual Genetics and Prevention Science (Grant Number P30 DA027827) funded by the National Institute on Drug Abuse.
