Abstract
Co-design provides a meaningful way to engage patients in research. However, there is limited practical guidance. We used our co-design project to identify strategies for other researchers. An ethnographic case study design was used. Data included participant observation of co-design meetings, meeting minutes, analytic fieldnotes, qualitative patient interviews, and research team member self-reflections. Additionally, we got external feedback. We analyzed data iteratively. Our team included 5 patients and 6 researchers. We identified 3 strategies to include patients in co-design: (1) Deliberately build the team, from recruiting patients to specifying roles. (2) Tailor the meeting format to thoughtfully use patients’ time and expertise. (3) Disrupt traditional hierarchies, to empower patients to actively participate. Researchers seeking to include patients as team members should consider: team composition and roles, leveraging meeting formats to optimize contributions and purposefully creating a culture of collaboration, so patient expertise informs the end product. Our work provides practical guidance for researchers to incorporate patient expertise in the co-design process and meaningfully involve them in their work.
Introduction
Co-design provides a meaningful way to involve patients in research. 1 Through co-design, end-users are engaged as equal partners in the development of a product. 2 However, detailed guidance is needed; as Slattery notes, “vague description of co-design makes it very difficult for researchers to undertake co-design activities.” 3 Our co-design study had 2 overarching goals: (1) co-design patient-facing materials to promote engagement in shared decision-making (SDM) for lung cancer screening; 4 (2) draw from our process to identify strategies for other researchers. Below we describe our study co-designing patient-facing materials to improve SDM for lung cancer screening, with the goal of identifying strategies from our work that others can use.
Methods
Setting
Our parent organization, the Department of Veteran Affairs (VA), is the United States' largest nationally integrated healthcare system. We engaged VA patients (veterans) in co-designing patient-facing materials to promote SDM for lung cancer screening. Veterans have unique communication and information needs that warrant tailored materials to support SDM. First, Veterans eligible for lung cancer screening are older (ages 50-80). Older adults are more likely to accept providers’ authority, a known barrier to SDM. People who smoke experience stigma-associated guilt and shame, hindering conversations with providers. 5 Finally, deference to providers may be especially true of Veterans due to military hierarchy.6,7
Veterans Participatory Action Research To Co-design Innovative Patient Activation Tools for Engagement (Vets PARTiCIPATE) was conducted from October 2020 to September 2021. All procedures were conducted per VA Bedford Institutional Review Board (1654427) approved protocols. They determined co-design activities did not constitute human subjects research and approved research interviews with co-designers about their experiences. Co-designers provided verbal informed consent for research interviews. This article draws from those interviews.
Design
We used an ethnographic case study design to identify our co-design strategies.8,9 This approach provided an “emic,” insider perspective on our real-world co-design process. 10
Data Collection
Data included qualitative interviews, documents, research team self-reflection and analytic fieldnotes. We conducted interviews with patient team members to learn about their co-design experiences. Patients were interviewed at the study's beginning and conclusion. The interview guide included questions evaluating their expectations of and experiences with co-design, including enjoyable and challenging aspects.
All team meetings had agendas and minutes. Study documents consisted of meeting minutes, which GF annotated with analytic fieldnotes. Research team members were also participant observers of co-design sessions. We each wrote self-reflections of our experiences to provide reflective, autoethnographic accounts of researcher experiences of co-design.
Analysis
Our goal was to identify strategies for other researchers. Research team members systematically reviewed deidentified patient interview transcripts and researcher self-reflections; the lead author reviewed the meeting minutes and field notes. The research team analyzed data iteratively through discussion, including their own experiences at team meetings as participant observers. Findings were organized into broad categories which lent themselves to actionable steps. Finally, we gathered “etic,” external feedback through informal discussions with advisory board members with expertise in engaging patients in SDM for lung cancer screening. The 3 categories presented below were then refined by the research team.
Results
We identified 3 strategies to include patients in co-design.
Strategy 1. Build a Co-design Team
Our final co-design team included 5 patients and 6 researchers. We overrecruited patients to allow for attrition, initially recruiting 8. Over the study year, 3 patients withdrew (2 for health reasons; one because the focus of our research, which was determined by the researchers before co-design commenced, was a poor fit for his interests). Overrecruiting prevented us from having too few patients throughout the full co-design process—which would have led to an imbalance between research and patient co-designers, as well as limiting the number of individuals contributing to the end product. One (of the initial 2) patient co-designers was a woman; all were military veterans. The research team was entirely women, including anthropologists, a clinical psychologist, a public health researcher, a pulmonologist, and a health communications researcher. Patients were invited to participate in manuscript writing; one coauthor (DC) is a patient.
The patients were demographically like the target population (ie, older veterans with a history of smoking). Flyers, information sessions and individual interviews were used to identify and onboard patients. The information session helped patients decide whether to join the team and allowed the researchers to observe team dynamics.
Interested candidates were interviewed, reinforcing the message that patients would be joining the team as professionals. During the interview, 2 researchers described the study, the role of patients, and expectations. Afterwards, the researchers discussed and ultimately invited everyone interviewed to join our team.
Before starting, we met one-on-one to prepare patients for their roles. These early steps oriented patient co-designers to the research process, set expectations and prepared them to be equal team members. Each patient had a different work, military, and health history, providing a diverse range of experiences and contributions, which facilitated relationships across the team: I want to know that the people I’m dealing with are worth dealing with; not from a monetary standpoint but from a consultant one; on a human level, not a hard level. I want to know everyone- including me- has something to contribute. (Patient-04)
One researcher was dedicated to planning and facilitating meetings; another was a patient liaison. The facilitator balanced warmth and humor, and ensured we started and ended on time, but was flexible. The liaison routinely checked in with patients; monitored an anonymous, virtual suggestion portal; addressed technical issues; and tracked patients’ hourly compensation. Research team members described: A good balance of using group consensus and individual roles/responsibilities. Having a co-design lead was really helpful, with someone at the end of the day being responsible for our plan for each meeting. (Researcher-05)
Another reflected: All of the work that (the liaison) does to stay in touch with the veterans, helping them with the technology, getting them paid. (Including endless conversations with “Fiscal.”) And so many other things! No matter how well the co-design sessions go, the project just won’t work without a strong commitment to the veterans and attention to these details. (Researcher-01)
Strategy 2: Tailor Co-design Meetings to Patients’ Needs
Co-design meetings were the primary way the researchers and patients collaborated. We had 2 meeting types: one for researchers and one for the full co-design team (researchers and patients). The research team met weekly to develop the co-design agenda, track tasks, debrief, and problem-solve. The full co-design team met virtually, with cameras on, for 2 h, biweekly, after the workday to accommodate patients’ schedules.
In creating meeting agendas, we researchers wanted to be cognizant of the patients’ time, maximizing their feedback, while also building community. Co-design meetings, with their emphasis on generative discussion, were distinct from typical research team meetings. We carefully planned each agenda to allow all ideas to be heard.
We used slides to guide the agenda, illustrate key points, and notate blank slides to capture patient input. We synthesized this input in our weekly research team meeting, then returned to the same slide at the next co-design meeting, noting “This is what you told us.” Another co-designer reflected: We keep building on what we’ve done as each week goes on. It's really quite interesting to start off with nothing and see the materials and the excitement of the group. It's really cool. (Patient-03)
The lead facilitator developed activities including ice breakers, codeveloping ground rules, and starting each meeting by sharing a personal story from 1 of 10 prompts related to SDM for lung cancer screening (see Figure 1). Activities emphasized honesty and vulnerability and helped build trust. This in turn facilitated candid conversations, such as fears about cancer screenings or distrust in clinical recommendations.

A prompt for co-design team members to share a story about themselves.
Strategy 3. Disrupt Existing Hierarchies to Empower Patients
Traditional health research culture is predicated on hierarchies and power differentials.11,12 This can be seen in how teams are structured, like having a principal investigator with an advanced degree being the leader, with master’s and bachelor’s degree holders in supportive roles. Similarly, military service has a chain of command, through which orders are transmitted from higher-ranking members in leadership roles to lower-ranking members in supportive roles.
Our research team sought to disrupt these hierarchies to empower patients to participate equally. By reflecting on typical research team meetings, we researchers purposefully reduced our jargon and speaking time to increase patients’ time and space to speak. This sharing of power was noticed: When the doctor talks, she's the chief. When another veteran talks, he's the chief. Everyone gets a chance to lead. When you’re speaking, you have 12 people listening. No one says you’re wrong. Everyone is trying. It's a good thing we’re trying to do. (Patient-02)
A research team member reflected: You need to put a lot of conscious effort into breaking down the power dynamics between researchers and veterans and fostering an environment where similarities between team members are more salient than differences. This will help build trust and it will create a more comfortable and encouraging environment in which to contribute. (Researcher-03)
Co-design meetings were dedicated to working on the end product and building a collaborative culture. An early meeting focused on jointly creating ground rules; later meetings developed messages and materials. Brief, structured presentations covered the scientific concepts, which then facilitated group discussion. These activities gave everyone permission to speak, regardless of their typical places in the hierarchy.
The patients described feeling like equals: When I first was doing this work … I was seriously intimidated because of all the brain power that was in the room. I was like, I’m not going to open my mouth and say anything … (But) I figured out that you people actually want my opinion and what I have to say. I think you people value it. My opinion has got some value. (Patient-06)
The initial meetings included a “my story” activity to facilitate knowing each other, beyond being researchers or patients, as unique people—a concept central to both SDM and patient empowerment. This facilitated discussion, in a way relevant to personal experiences. It necessitated all of us to be open and vulnerable, which enhanced trust, sharing, and listening.
Discussion
Our authorship team of researchers and a patient co-designer, identified strategies for engaging patients in co-design by deliberately building the team; tailoring meetings to patients’ needs; and empowering them to actively participate. These strategies align with best practices such as creating deliberative spaces to share experiences, broadening power, being flexible, and having clear roles and objectives. 13
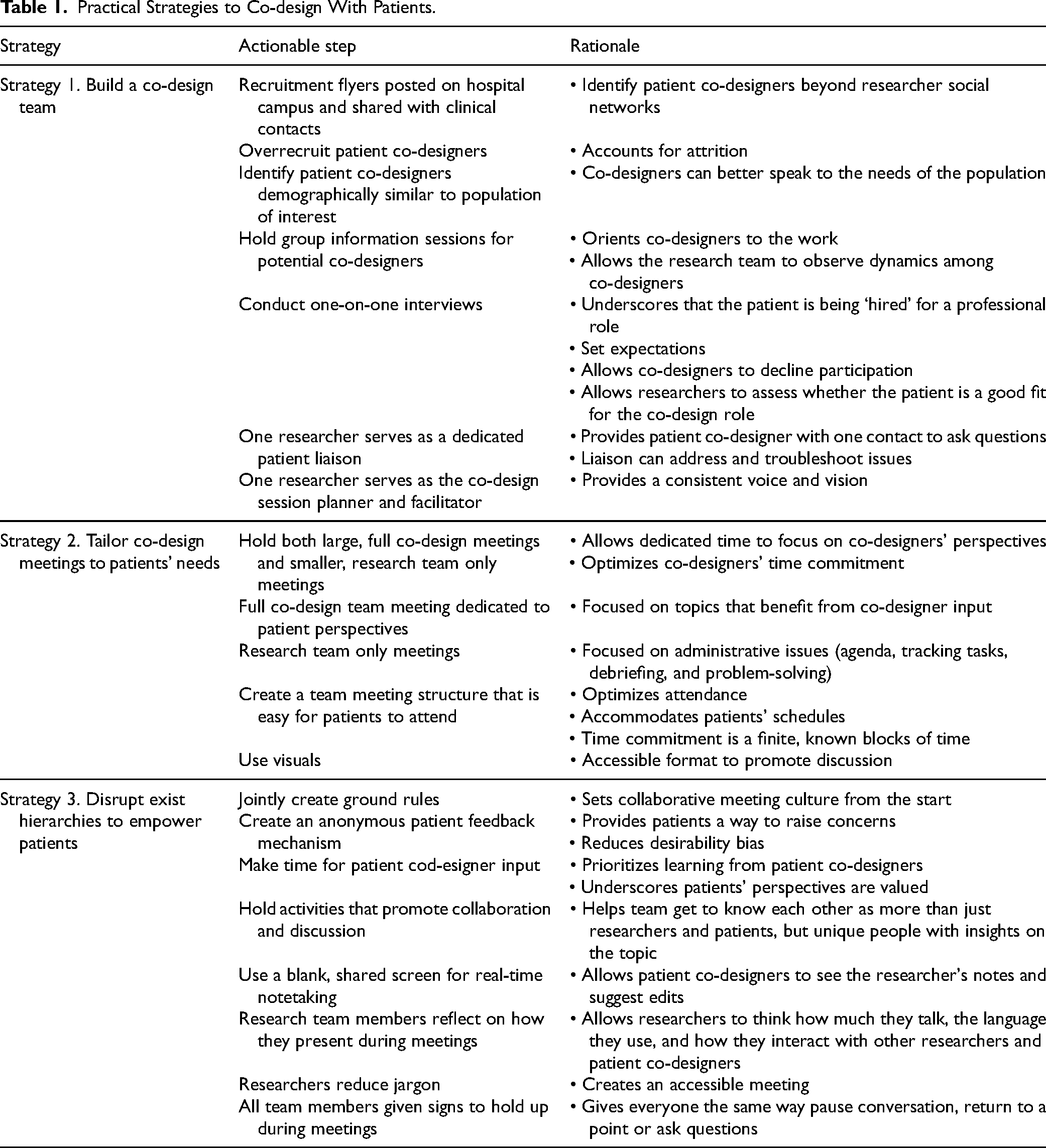
We used several practical steps to include patients in co-design that may be useful to others (see Table 1). The strategies reinforced each other. Together, they created a relationship of trust, which was foundational to our work. Trust was necessary for the team to feel free to participate in co-design, which is required for full participation in SDM, and the resulting booklet was designed by the team as a trust-building tool.
Practical Strategies to Co-design With Patients.
Limitations
Our co-design process was not without challenges. The patients had varied communication styles and preferences. Some were quiet, needing to be drawn out by the facilitator. Others were frustrated with the focused, slow pace of our research, given the research team stressing the urgency of addressing lung cancer. Further, we did not collect outcomes about these co-design strategies but did assess the resulting product. 4
Including patients in research occurs on a continuum from having them involved in generating the research area, to only providing feedback on a late-stage product. 13 We had patients involved in developing an end-product to support SDM for lung cancer screening. 4 While the research team set this objective, the end product itself was selected and envisioned by the patient co-designers. Our co-designers could have been engaged earlier in the process, like identifying the focus of our work, developing our meeting agendas, or chairing meetings.
Conclusion
Having practical guidance can increase researchers’ use of co-design, improve the experiences of researchers and patients, and may result in improved outcomes. Our co-design strategies provide an actionable tool for researchers to meaningfully involve patients and can be applied to a variety of contexts, conditions, and populations.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This material is based upon work supported by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development and Health Services Research and Development (Grant No: SWIFT 2020; RVR 19-491).
