Abstract
Keywords
Introduction
Overcrowding in emergency departments (ED) has been a problem for decades. Many observers point out that a significant number of ED visits could be directed to primary care to prevent or treat low-acuity conditions. 1 It is generally believed that non-ED alternatives such as primary care practices and walk-in clinics offer good quality of care at a lower cost to low-acuity ambulatory ED patients. However, data supporting these assumptions are scarce and often contradictory.2,3 Moreover, to our knowledge, very few studies have assessed this issue from the patients’ perspective. 4
In a patient-centered system, assessing the patient's perspective is crucial to providing optimal care pathways and quality of care. Some evidence suggests that this practice can break down the barriers between health professionals and patients, ensure a more transparent system, shift emphasis onto patients’ priorities, and reduce healthcare costs.5,6 Moreover, the quintuple aim adopted by many healthcare organizations (improving population health, patient experience and care providers’ work life while reducing the cost of care and improving health equity),7,8 underscores the importance of the patients’ experience to successful organizations. It is therefore pivotal to measure patient experience using validated tools to effectively improve health services.
The aim of this study was to develop a patient-reported experience measure (PREM) in English and French suitable for assessing and comparing the experience of ambulatory care patients with acute unexpected needs presenting in EDs, walk-in clinics, and primary care practices.
Methodology
Study Design
The APEX (Ambulatory Patient EXperience) questionnaire was developed in five phases using a mixed-methods research design, in accordance with the Guidelines for Cross-Cultural Adaptation of Self-Report Measures.
9
Phase 1: Creation of the prototype; Phase 2: Forward–reverse translation; Phase 3: Pretesting; Phase 4: Pilot study; Phase 5: Synthesis of the above four phases.
In each phase, problems with the intelligibility, reliability, and structure of the questionnaire were identified and resolved before beginning the next phase. Modifications after each phase were made by a steering committee composed of the principal investigators, the patient partners, a linguist, and the research coordinator. Prior approval for the study was received from the CHU de Québec-Université Laval research ethics board (authorization number MP-20-2021-5224).
Phase 1: Creation of the Prototype
To create the first APEX prototype, two validated English PREMs were merged, one used in EDs (Ontario emergency department patient experience of care survey [EDPEC] - 48 questions 10 ), the other in primary care settings (Ontario Primary Care Patient Experience Survey [PCPES] - 39 questions 11 ). Since both surveys include questions specific to their particular care setting, neither is wholly suited to compare multiple care settings. A panel of 12 experts was therefore assembled to identify the questions from the two source PREMs that would be most relevant to assessing the experience of care in all targeted settings (ED, walk-in clinic, primary care practice). Panelists were asked first to rate each candidate question independently on a scale of 1 (not relevant) to 9 (very relevant) using an online questionnaire (REDCap). Questions with a median score between 1 and 3 were excluded, while those with a median score between 7 and 9 were included in the APEX prototype. Questions with a median score between 4 and 6 were discussed by videoconference. Panelists were asked to reach a consensus on the usefulness of these questions. They were also asked to propose minor modifications to the questions thus retained (eg, replace “emergency department” with “care facility”) to adapt them for use in any setting. If similar questions from the two source PREMs were retained, panelists were asked to merge the questions to avoid redundancy in the new questionnaire.
Phase 2: Forward–Reverse Translation
The APEX prototype was translated into French then back to English to identify potential inconsistencies or conceptual errors by comparing the original version with the translations. Two French native-speaker professional translators were first tasked to translate the prototype into French. After comparing the two translated versions, discrepancies and ambiguities were resolved in discussion with the translators, the linguist, and the patient partners to ensure the intelligibility of all questions. The APEX prototype was then translated back into English by two other different native English-speaking translators. The same process of reconciling discrepancies and eliminating ambiguities was carried out on this new English version.
Phase 3: Pretesting
In the third phase, a convenience sample of ambulatory adults was recruited in the CHUL (Centre hospitalier de l'Université Laval) ED, a teaching hospital in Québec City (Canada) receiving about 80,000 visits per year. Participants were asked to answer the APEX prototype and then rate the different items on a scale of 1 to 9 (1 = totally disagree to 9 = totally agree) to evaluate the clarity, utility, discriminability, face validity, content validity, and lack of redundancy 12 of the questionnaire. Respondents were also allowed to provide free-text comments on these domains.
Phase 4: Pilot Study
In the fourth phase, a prospective cohort study was conducted between June 8 and October 3 of 2021 to test the APEX prototype in a real-world clinical setting and compare the experience of care in the CHUL ED to that of two nearby walk-in clinics. Because recruitment took place during the COVID-19 pandemic and the availability of walk-in health services was limited, we included walk-in clinics designated for the management of patients with symptoms of respiratory infections. A convenience sample was recruited by research staff, between 10:00 and 20:00, Monday to Sunday, in the ED, and between 9:00 and 17:00, weekdays, in walk-in clinics. Patients were included if they met the following 4 criteria: (1) were at least 18 years of age; (2) were ambulatory throughout their visit at the participating care facility; (3) had a low-acuity acute respiratory condition, namely common cold, otitis media, acute tonsillitis or pharyngitis, acute laryngitis, acute sinusitis, COVID-19, influenza, pneumonia, bronchitis, acute exacerbation of chronic obstructive pulmonary disease or asthma; and (4) were able to provide written informed consent. Patients were excluded if they: (1) were transported by ambulance; (2) were not covered by provincial health insurance; (3) had consulted for a similar symptom in the previous 30 days; (4) were living in a long-term care facility or were incarcerated; and (5) were receiving palliative care. After giving on-site consent, participants were called back between 24 and 72 h later to answer the APEX questionnaire. We planned to recruit at least 30 participants, 15 from the ED and 15 from the clinics since previous studies have shown that 90% of the problems associated with using a questionnaire can be identified by testing it on a sample of 15 respondents. 13
Phase 5: Synthesis of the 4 Previous Phases
Following the pilot study, the expert panel re-evaluated the questionnaire and the notes taken during each phase (screening and selection of questions, translation, pretesting, and pilot study) to ensure that no issues relating to content, translation, or usability were overlooked.
Statistical Analysis
Changes to the APEX prototype in each phase were recorded and are reported as counts. The APEX pretest in phase 3 is reported as medians of participants’ responses for each of the six domains studied (clarity, utility, discriminability, face validity, content validity, lack of redundancy). A median score between 7 and 9 was determined a priori to be satisfactory for each domain assessed. In phase 4, descriptive statistics and frequencies were generated from the data to provide trends in patients’ experience of care. Wilcoxon–Mann–Whitney tests were used to compare continuous characteristics across care settings (ED and clinics), and Fisher's exact tests were performed to explore the presence of associations between categorical characteristics and settings. Multivariate generalized linear regression models were used to compare care settings adjusted by propensity score. The propensity score was estimated using a logistic regression model with care settings as outcome and the APEX responses for age, gender, sex, ethnicity, comorbidities (Charlson index score and number of regular medications), patient perception of illness severity, and length of stay as independent variables. All statistical tests have a significance threshold of 0.05. Analyses were performed using SAS (v9.4).
Results
The final APEX is composed of 61 questions and 7 sections: (1) “Upon your arrival,” (2) “During your visit,” (3) “At your departure from the healthcare setting,” (4) “Your overall experience during your most recent visit,” (5) “Your usual healthcare,” (6) “About you,” and (7) “Comments.” Of these questions, 2 use a 4-step Likert scale, 25 use a 5-step Likert scale, 29 offer multiple choice answer, and 5 are open-ended. The final APEX in French and English is available in the Web appendices. Figure 1 shows the workflow during the 5 phases of the APEX questionnaire development.

Workflow of the APEX questionnaire development and testing process.
Phase 1: Creation of the Prototype
We assembled a panel consisting of two patient partners, two clinicians, an expert linguist in information design, an expert in methodology, an emergency room nurse manager, an expert in patient experience measurement and improvement, the principal investigator and three co-investigators. From the 87 questions that made up the two PREM source questionnaires, the panel's selection of questions and the process of merging similar questions on the same dimension of experience resulted in a prototype with 63 questions. Questions about ambulance use, medication administered during the visit, ease of scheduling appointments, and the quality of the long-term relationship with the family physician were excluded since they were not applicable in all of the targeted care settings. Following a recommendation from the expert panel, two questions from the EDPEC questionnaire, asking separately if the doctor and the nurses had spent enough time with the patient, were merged into one question to evaluate the patient satisfaction with the time spent with all health professionals during the visit. This recommendation addressed the fact that not all clinics have the same staffing model, some having no nurses and others offering appointments with nurse practitioners rather than physicians. However, the panel decided also to evaluate physicians and nurses separately on several aspects of care quality, to detect possible differences in patient experiences of physicians and nurses. Seven additional questions were thus generated by separating seven PCPES questions for this purpose. Based on discussion of the demographic questions during the panel meeting, two anthropologists were consulted, which led to modifying section 6 (“about you”) to obtain a more inclusive questionnaire, one that could measure whether patient experience is related to demographic characteristics such as ethnicity, gender, and sexual orientation.
Phase 2: Forward–Reverse Translation
After forward and reverse translations, one question was split in two to assess separately patient experience of the advice received regarding medication and investigations. Two questions were excluded and two were merged to reduce redundancy (Figure 1). Some questions were rephrased without changing their meaning to make them more relevant and applicable to the three clinical settings. The modifications are reported in the Web appendices.
Phase 3: Pretesting
The questionnaire was pretested on 25 participants, of which 48% were over 40 years old and 52% were women. With median scores between 8 and 9 for all dimensions (Table 1), the APEX prototype was rated as clear, low in redundancy and useful, with good discriminatory properties and good face and content validity. Based on respondents’ comments and suggestions, the following three additions were made: (1) a question to evaluate the convenience of the waiting area (eg, access to restrooms, food, outlet for charging phone batteries); (2) a clarification (examples of the type of information the healthcare professional can give on a specific medication; question 2.5 in the Web appendices) to make one question more understandable; and (3) an introduction to the section “about you” to explain the purpose of potentially sensitive questions (Figure 1). After this phase, the APEX prototype had 61 questions and 7 sections.
APEX Pretest Results (n = 25 Respondents/Potential Users) (Phase 3).
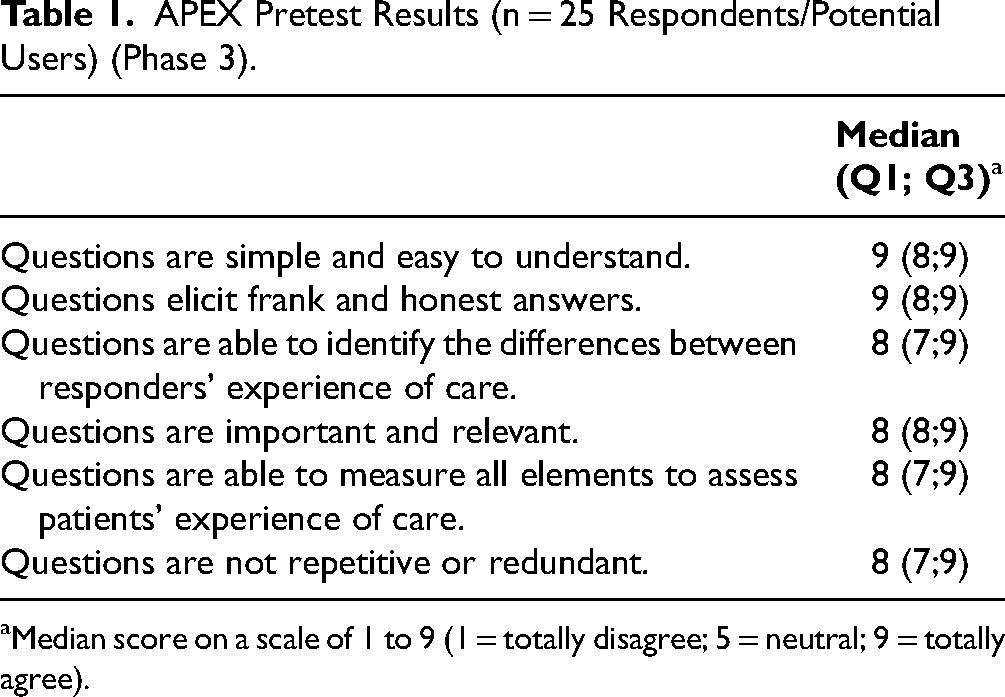
Median score on a scale of 1 to 9 (1 = totally disagree; 5 = neutral; 9 = totally agree).
Phase 4: Pilot Study
During phase 4, 91 eligible participants were approached: 23 declined participation, 2 ED patients were excluded because they were not ambulatory throughout their visit and 3 others because they did not fill out the APEX completely. Overall, 63 participants were included in the analysis, 34 from the walk-in clinics and 29 from the ED. The demographic characteristics of the participants are shown in Table 2. The mean length of stay was 55 min (SD: 96) in the clinics and 170 min (SD: 191) in the ED. More men were recruited from the clinic than from the ED (47% vs 21%). No participant reported a gender identity other than male or female. Comparing patient experience between settings, adjusted results show that access, cleanliness, and feeling treated with respect and dignity by nurses and physicians were significantly better in the clinics than in the ED (Table 3). However, the perception that the professional spent enough time with them was similar. No statistically significant difference in the overall care experience and the tendency to recommend the facility to family and friends was found between the two settings. Table A1 in the Appendix shows the crude results for each APEX question.
Demographic Characteristics of the Pilot Study Participants (Phase 4)a.

SD, standard deviation.
All results are reported as n(%), unless otherwise indicated.
Between-Settings Differences in Adjusted Means (95% CI) for Selected APEX Questions (n = 63).

Results are estimated by generalized linear regression models adjusted by propensity score. Propensity score was calculated using age, sex, gender, ethnicity, comorbidities, patients’ perception of illness severity, and length of stay.
Results with the 5-point Likert scale; 1 = poor, 2 = acceptable, 3 = good, 4 = very good, 5 = excellent.
Results with the following scale; 1 = definitely yes, 2 = probably yes, 3 = probably not, 4 = definitely not.
Results with the following scale; 1 = yes, definitely, 2 = yes, somewhat, 3 = no.
Phase 5: Synthesis of the 4 Previous Phases
The panel re-evaluated the four preceding phases. Only one point was raised regarding the accuracy of the vocabulary. The panel decided to replace the term “consultation” with “visit” because the latter encompasses the entire episode of care and not just the consultation with a specific care provider.
Discussion
Interpretation
In this study, we developed, adapted, and produced evidence supporting the validity of a PREM designed to assess the care experienced by ambulatory patients presenting with unexpected needs. Called APEX, this questionnaire can be used in EDs, primary care practices, and walk-in clinics. Consisting of 61 questions divided into 7 sections, it can be used to identify and evaluate differences in care experience between these care settings and to assess the quality of care provided to ambulatory patients with acute conditions.
Previous Studies
The development of a new PREM was made necessary because we found no robust, validated tool in the scientific literature to achieve our objective of comprehensively comparing the experience of care in EDs, walk-in, and primary care clinics14–20 Indeed, to our knowledge, in only one study was such a tool designed to compare the three settings from the patient's perspective. Hutchison et al 4 assessed patient satisfaction using three domains of questions: (1) Perception of patient-centered communication; (2) Doctor's attitude; and (3) Delay in the waiting room. 4 Conducted in Ontario (Canada), this study found that patients were more satisfied with the doctor's attitude and with the waiting time in walk-in clinics than in EDs. Our study showed similar results: significantly more participants felt that they were treated with respect and dignity by doctors in the clinics than in the ED, and more participants reported unacceptable waiting times in the ED than in clinics. However, Hutchison et al. only surveyed patient satisfaction with doctors. The tool they used was not designed to evaluate other important dimensions of the patient experience such as the relationship with non-medical staff (eg, nurses), continuity of care, medication administration, and support for self-care. 21 Our PREM tool addresses each of these dimensions to some extent.
Clinical Implications
The evaluation of quality of care provided in walk-in clinics represents a blind spot in many health jurisdictions since these establishments operate without formal monitoring by health authorities. Our PREM tool can be used to evaluate the patient experience in walk-in clinics and provide benchmarks against other facilities treating the same population.
Research Implications
Our goal in developing a new PREM was to allow a multicenter study that would compare the value of care provided in EDs, walk-in clinics, and primary care practices from the patient's perspective. This study is currently ongoing in 14 institutions in Canada. 22 The APEX will also enable other research initiatives intended to evaluate different care pathways offered to ambulatory patients with unexpected needs, such as redirection protocols whereby patients are redirected from the ED to outpatient clinics.
Limitations
This study has a number of limitations. First, although developed in English and French, our PREM tool was piloted in a primarily French-speaking community with less socio-cultural diversity than is found in other Canadian cities of similar size. However, we expect the English version of the APEX to perform equally for two reasons: (1) the tool was created by merging two existing PREMs validated in English; and (2) it has been reviewed by two professional translators whose native language is English. Another limitation is that the pilot study was conducted with a convenience sample that included only respiratory conditions, which may not be wholly representative of the population of ambulatory patients with acute conditions. However, the primary objective of the pilot study was to identify and correct usability problems. We are confident that our sample size was large enough to achieve this goal and secondarily to explore potential differences in the experience of care between the sites. A third limitation is that although the APEX assesses most of the dimensions of patient-centered care endorsed by the Institute of Medicine and other internationally recognized societies, it provides no evaluation of the involvement of patients, their families, and caregivers in the care received.23,24 Despite originating from two validated PREMs and benefitting from numerous opportunities for amendment by patients, the final tool contains no direct questions on this aspect of healthcare. Finally, in adapting the PREM for use in different facilities where emergency ambulatory patients may be treated, some setting-specific questions were excluded. While the final tool facilitates comparison across settings, it may not be the most appropriate tool if only one type of setting (eg, ED) is assessed.
Conclusion
We have developed the APEX questionnaire, a PREM to compare the care experience of ambulatory patients in EDs, walk-in clinics, and primary care practices. Through a 5-phase process, we have addressed several usability problems and accumulated evidence of validity such that the tool appears to be very usable in the ambulatory care context regardless of setting. The pilot study on a sample of patients has shown some differences in the experience of the care provided in one ED and two walk-in clinics. The APEX tool is currently being used in a multicenter study to provide robust evidence to determine which alternative offers the best value of care to ambulatory patients.
Key points
It is generally believed that non-ED (emergency department) alternatives such as primary care practices and walk-in clinics offer good quality of care at a lower cost to low-acuity ambulatory ED patients, but data supporting these assumptions are scarce.
We developed the Ambulatory Patient EXperience (APEX) questionnaire, a patient-reported experience measure designed to assess and compare the experience of care in EDs, walk-in, and primary care clinics.
The comparative data obtained from this questionnaire will help health authorities identify optimal care pathways for ambulatory patients with acute unexpected needs.
Supplemental Material
sj-xlsx-1-jpx-10.1177_23743735241229373 - Supplemental material for Development of a Patient-Reported Experience Measure Tool for Ambulatory Patients With Acute Unexpected Needs: The APEX Questionnaire
Supplemental material, sj-xlsx-1-jpx-10.1177_23743735241229373 for Development of a Patient-Reported Experience Measure Tool for Ambulatory Patients With Acute Unexpected Needs: The APEX Questionnaire by Myriam Nadeau, Dominique Chabot, Mylaine Breton, Jason R. Guertin, Laurie Harvey Labbé, Danièle Roberge, Gabrielle Lefebvre, Myriam Mallet, Sandrine Beaulieu, Éric Kavanagh, Nathalie Cloutier, Philippe Garant, Lynda Bélanger, Samuel Vaillancourt, Tarek Boumenna, Kathryn Bareil, Joanie Savard, David Simonyan, Mahukpe Narcisse Ulrich Singbo and Simon Berthelot in Journal of Patient Experience
Footnotes
Author Contributions
Each author substantially contributed to (1) the study’s conception and design, or acquisition of data, or analysis and interpretation of data; (2) drafting the article or revising it critically for important intellectual content; (3) final approval of the version to be submitted.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research [PJM 175404].
Ethical Approval
Prior approval for the study was received from the CHU de Québec-Université Laval research ethics board (authorisation number MP-20-2021-5224).
Supplemental Material
Supplemental material for this article is available online.
Appendix
Crude results of phase 4 for each APEX question.

| Clinic (n = 34) | ED (n = 29) | P-value | |
|---|---|---|---|
|
|
|||
| Poor | 1 (2.9) | 4 (13.8) | .007 |
| Acceptable | 1 (2.9) | 2 (6.9) | |
| Good | 7 (20.6) | 12 (41.4) | |
| Very good | 10 (29.4) | 9 (31.0) | |
| Excellent | 15 (44.1) | 2 (6.9) | |
|
|
|||
| Poor | 0 (0) | 8 (27.6) | < .0001 |
| Acceptable | 1 (2.9) | 11 (37.9) | |
| Good | 3 (8.8) | 3 (10.3) | |
| Very good | 7 (20.6) | 4 (13.8) | |
| Excellent | 23 (67.6) | 3 (10.3) | |
|
|
|||
| Poor | 0 (0) | 2 (6.9) | .11 |
| Acceptable | 3 (8.8) | 2 (6.9) | |
| Good | 3 (8.8) | 8 (27.6) | |
| Very good | 7 (20.6) | 6 (20.7) | |
| Excellent | 21 (61.8) | 11 (37.9) | |
|
|
|||
| Poor | 1 (2.9) | 3 (10.3) | .01 |
| Acceptable | 4 (11.8) | 8 (27.6) | |
| Good | 4 (11.8) | 7 (24.1) | |
| Very good | 8 (23.5) | 8 (27.6) | |
| Excellent | 17 (50.0) | 3 (10.3) | |
| Missing | 1 (2.9) | 0 (0) | |
|
|
|||
|
|
|||
| Poor | 0 (0) | 1 (3.4) | .41 |
| Acceptable | 0 (0) | 1 (3.4) | |
| Good | 5 (15.2) | 5 (17.2) | |
| Very good | 7 (21.2) | 9 (31.0) | |
| Excellent | 21 (63.6) | 13 (44.8) | |
| Missing | 1 (2.9) | 0 (0) | |
|
|
|||
| Poor | 0 (0) | 0 (0) | .48 |
| Acceptable | 0 (0) | 0 (0) | |
| Good | 2 (6.1) | 4 (13.8) | |
| Very good | 6 (18.2) | 7 (24.1) | |
| Excellent | 25 (75.8) | 18 (62.1) | |
| Missing | 1 (2.9) | 0 (0) | |
|
|
|||
| Poor | 0 (0) | 1 (3.4) | .35 |
| Acceptable | 0 (0) | 1 (3.4) | |
| Good | 3 (9.4) | 6 (20.7) | |
| Very good | 5 (15.6) | 5 (17.2) | |
| Excellent | 24 (75.0) | 16 (55.2) | |
| Missing | 2 (5.9) | 0 (0) | |
|
|
|||
| Poor | 0 (0) | 1 (3.4) | .27 |
| Acceptable | 0 (0) | 3 (10.3) | |
| Good | 5 (15.2) | 4 (13.8) | |
| Very good | 6 (18.2) | 6 (20.7) | |
| Excellent | 22 (66.7) | 15 (51.7) | |
| Missing | 1 (2.9) | 0 (0) | |
|
|
|||
| Poor | 0 (0) | 0 (0) | .24 |
| Acceptable | 0 (0) | 2 (6.9) | |
| Good | 2 (6.3) | 2 (6.9) | |
| Very good | 4 (12.5) | 7 (24.1) | |
| Excellent | 26 (81.3) | 18 (62.1) | |
| Missing | 2 (5.9) | 0 (0) | |
|
|
|||
| Poor | 0 (0) | 0 (0) | .70 |
| Acceptable | 3 (9.1) | 1 (3.4) | |
| Good | 5 (15.2) | 3 (10.3) | |
| Very good | 6 (18.2) | 8 (27.6) | |
| Excellent | 19 (57.6) | 17 (58.6) | |
| Missing | 1 (2.9) | 0 (0) | |
|
|
|||
| Poor | 0 (0) | 0 (0) | .39 |
| Acceptable | 0 (0) | 2 (6.9) | |
| Good | 5 (15.2) | 6 (20.7) | |
| Very good | 7 (21.2) | 7 (24.1) | |
| Excellent | 21 (63.6) | 14 (48.3) | |
| Missing | 1 (2.9) | 0 (0) | |
|
|
|||
|
|
|||
| Poor | 0 (0) | 0 (0) | .80 |
| Acceptable | 1 (7.1) | 3 (10.3) | |
| Good | 1 (7.1) | 5 (17.2) | |
| Very good | 3 (21.4) | 7 (24.1) | |
| Excellent | 9 (64.3) | 14 (48.3) | |
| Missing | 20 (58.8) | 0 (0) | |
|
|
|||
| Poor | 0 (0) | 0 (0) | .51 |
| Acceptable | 1 (6.7) | 1 (3.4) | |
| Good | 0 (0) | 2 (6.9) | |
| Very good | 4 (26.7) | 12 (41.4) | |
| Excellent | 10 (66.7) | 14 (48.3) | |
| Missing | 19 (55.8) | 0 (0) | |
|
|
|||
| Poor | 1 (7.1) | 0 (0) | .34 |
| Acceptable | 0 (0) | 2 (6.9) | |
| Good | 1 (7.1) | 3 (10.3) | |
| Very good | 2 (14.3) | 9 (31.0) | |
| Excellent | 10 (71.4) | 15 (51.7) | |
| Missing | 20 (58.8) | 0 (0) | |
|
|
|||
| Poor | 1 (7.1) | 2 (6.9) | .46 |
| Acceptable | 0 (0) | 2 (6.9) | |
| Good | 0 (0) | 2 (6.9) | |
| Very good | 3 (21.4) | 10 (34.5) | |
| Excellent | 10 (71.4) | 13 (44.8) | |
| Missing | 20 (58.8) | 0 (0) | |
|
|
|||
| Poor | 0 (0) | 1 (3.4) | .94 |
| Acceptable | 0 (0) | 1 (3.4) | |
| Good | 1 (7.7) | 3 (10.3) | |
| Very good | 3 (23.1) | 8 (27.6) | |
| Excellent | 9 (69.2) | 16 (55.2) | |
| Missing | 21 (61.8) | 0 (0) | |
|
|
|||
| Poor | 0 (0) | 2 (8.7) | .72 |
| Acceptable | 0 (0) | 1 (4.3) | |
| Good | 2 (20.0) | 2 (8.7) | |
| Very good | 2 (20.0) | 7 (30.4) | |
| Excellent | 6 (60.0) | 11 (47.8) | |
| Missing | 24 (70.6) | 6 (20.7) | |
|
|
|||
| Poor | 1 (5.9) | 1 (3.4) | .91 |
| Acceptable | 0 (0) | 1 (3.4) | |
| Good | 3 (17.6) | 5 (17.2) | |
| Very good | 4 (23.5) | 10 (34.5) | |
| Excellent | 9 (52.9) | 12 (41.4) | |
| Missing | 17 (50.0) | 0 (0) | |
|
|
|||
| Yes, definitely | 24 (70.6) | 19 (65.5) | .91 |
| Yes, somewhat | 8 (23.5) | 8 (27.6) | |
| No | 2 (5.9) | 2 (6.9) | |
|
|
|||
| Yes | 1 (2.9) | 14 (48.3) | <.0001 |
|
|
|||
| Yes | 16 (47.1) | 22 (75.9) | .02 |
|
|
|||
| Yes | 29 (85.3) | 25 (86.2) | 1.00 |
|
|
|||
| Yes | 27 (79.4) | 24 (82.8) | .76 |
|
|
|||
| Yes | 15 (44.1) | 20 (69.0) | .07 |
|
|
|||
| Yes, definitely | 12 (80.0) | 14 (70.0) | .70 |
| Yes, somewhat | 3 (20.0) | 6 (30.0) | |
| No | 0 (0) | 0 (0) | |
| Missing | 19 (55.9) | 9 (31.0) | |
|
|
|||
| 13 (38.2) | 12 (41.4) | 1.00 | |
|
|
|||
| Yes | 7 (53.8) | 6 (50.0) | 1.00 |
| Missing | 21 (61.8) | 17 (58.6) | |
|
|
|||
| Poor | 0 (0) | 3 (10.3) | .004 |
| Acceptable | 0 (0) | 1 (3.4) | |
| Good | 3 (8.8) | 7 (24.1) | |
| Very good | 5 (14.7) | 8 (27.6) | |
| Excellent | 26 (76.5) | 10 (34.5) | |
|
|
|||
| Poor | 0 (0) | 1 (3.4) | .76 |
| Acceptable | 0 (0) | 0 (0) | |
| Good | 6 (17.6) | 4 (13.8) | |
| Very good | 6 (17.6) | 7 (24.1) | |
| Excellent | 22 (64.7) | 17 (58.6) | |
|
|
|||
| Poor | 0 (0) | 1 (3.4) | .10 |
| Acceptable | 0 (0) | 2 (6.9) | |
| Good | 1 (2.9) | 2 (6.9) | |
| Very good | 5 (14.7) | 8 (27.6) | |
| Excellent | 28 (82.4) | 16 (55.2) | |
|
|
|||
| Poor | 0 (0) | 2 (6.9) | .43 |
| Acceptable | 1 (2.9) | 2 (6.9) | |
| Good | 6 (17.6) | 6 (20.7) | |
| Very good | 8 (23.5) | 8 (27.6) | |
| Excellent | 19 (55.9) | 11 (37.9) | |
|
|
|||
| Yes, definitely | 3 (8.8) | 12 (41.4) | < .0001 |
| Yes, somewhat | 2 (5.9) | 8 (27.6) | |
| No | 29 (85.3) | 9 (31.0) | |
|
|
|||
| Definitely yes | 28 (82.4) | 18 (62.1) | .29 |
| Probably yes | 5 (14.7) | 8 (27.6) | |
| Probably not | 1 (2.9) | 2 (6.9) | |
| Definitely not | 0 (0) | 1 (3.4) | |
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
