Abstract
Anti-spike monoclonal antibodies emerged as effective early treatment of high-risk individuals with mild-to-moderate COVID-19. Although their clinical and safety outcomes have been reported, patient perspectives of these experimental therapies have not been evaluated. In this survey participated by 644/2412 (26.7% response) individuals evaluated for anti-spike monoclonal antibody therapies, the majority of 523 patients who received the antibody infusion were very satisfied with their overall patient experience, the quality of care provided, and various aspects of medical care. They voiced satisfaction with the communication with providers before and during treatment, including education provided about monoclonal antibody treatment, the potential benefits and adverse effects, detailed instructions on the process of infusion, and safety protocols employed at the infusion facilities. Nearly a quarter (23.6%) of 121 patients who declined therapy indicated they would accept treatment should it be offered again. These patient perspectives may be used to guide healthcare facilities and providers in optimizing the care provided to high-risk outpatients with COVID-19.
Introduction
Patients with mild-to-moderate coronavirus disease-2019 (COVID-19) are generally managed outside hospital settings with supportive measures to relieve symptoms including fever, cough, and body aches (1,2). However, patients with high-risk comorbidities are at increased risk for progression to severe COVID-19 which may lead to hospitalization, need for intensive care, and may even result in death (3).
In November 2020, the United States (US) Food and Drug Administration (FDA) granted emergency use authorization (EUA) for bamlanivimab and casirivimab-imdevimab monoclonal antibodies as experimental treatment of mild to moderate COVID-19 among high-risk persons to prevent progression to severe disease (4,5). This EUA was based on results from clinical trials (6,7) and was supported by reports of real-world outcomes (3,8,9). The use of anti-spike monoclonal antibody products was associated with up to 70% relative risk reduction in hospital admission within 28 days after treatment (3,8,9).
Although the clinical outcomes were favorable, patient experience with the process of experimental monoclonal antibody infusion, at such a difficult and anxious time, has not been measured. Patient-centered care, an indicator of healthcare quality, is defined as the provision of health care that engages patients in decision-making processes and empowers them to engage in healthcare that is consistent with their individual values and preferences (10). The uncertainty related to COVID-19 treatment and outcomes emphasizes the need to design healthcare systems for COVID-19 patients that engages them in decision-making and cognizant of the values and preferences of patients.
Here, the study team sought to understand the perspectives of the patients about the monoclonal antibody infusion program through satisfaction surveys. This understanding of the nonclinical aspects of patient experience may assist providers in further improving the clinical care of patients with COVID-19 during a stressful time.
Methods
Practice Setting
The study was conducted in a large, integrated healthcare delivery system serving more than 1 million unique patients each year across the US Midwest. In anticipation of the FDA EUA of anti-spike monoclonal antibodies in the outpatient setting, the healthcare system established its Monoclonal Antibody Treatment (MATRx) Program in November 2020 (11). MATRx providers began administering bamlanivimab monotherapy on November 19, 2020, and casirivimab-imdevimab on December 1, 2020. The details of the MATRx program, procedures, and outcomes are described in greater detail elsewhere (11).
Survey Population and Methods
Patients who tested positive for COVID-19 at the facility were automatically identified and assessed for eligibility for monoclonal antibody therapy (11). Patients tested for COVID-19 outside our testing facilities were referred by their providers for consideration of monoclonal antibody therapy. All automatically screened and referred patients were evaluated for eligibility for anti-spike monoclonal antibody treatment using an algorithm created based on the FDA EUA criteria by the MATRx clinical team (11).
All eligible patients were provided detailed education about monoclonal antibody products through telephone conversations (lasting on an average of 20 min) and via patient video education, when possible (11). The patient education video has been published (11). Upon determination of eligibility, patients were offered opportunity for treatment, and those who consented were immediately scheduled for anti-spike monoclonal antibody infusion. Median time to infusion was 2 days after diagnosis of COVID-19 (8,9).
The patients approached during the initial 6 weeks of the MATRx program (November-December 2020) comprised the sampling population for this electronic survey (12). During this period, 59% of patients approached for monoclonal antibody therapy consented and received treatment (12). Sociodemographic and clinical factors that influenced their decision to consent for monoclonal antibody treatment have been reported (12). Clinical outcomes of monoclonal antibody treatment have also been reported (3,8,9,12).
The study team developed a survey instrument tool to assess for patient decision-making to obtain the monoclonal antibody therapy should the situation present itself again, along with their experience and satisfaction with communication and quality of care provided. The survey was deployed electronically via Qualtrics to patient-provided electronic (email) addresses. An initial invitation to participate in the survey was sent to all 2412 patients within the sampling population on May 25, 2021, with 2 follow-up reminder emails. The survey closed to responses on June 7, 2021.
Measures
Demographic and Clinical Information. Patient age, sex, and race were assessed using the patient registration system prior to survey deployment. Monoclonal Antibody Screening Score (MASS) was developed by the MATRx program as a mechanism to screen eligible patients and stratify their risk of hospitalization (3,12). Details of this score are shared elsewhere (3,12). The study team included MASS as a measure of medical complexity and risk profile. That score was obtained from the MATRx program registry.
Patient-Reported Information. To assess for patient satisfaction, the study team created a set of 8 questions. The study team was interested in communication with patients about the monoclonal antibody therapy, explanations that were provided about potential risks of the experimental therapy, perceived quality and safety of care, and overall satisfaction. Questions were set on a 5-point Likert scale (very satisfied, satisfied, neither satisfied or dissatisfied, dissatisfied, very dissatisfied). To understand patient-reported outcomes following COVID-19 diagnosis, questions were asked to report on time to resolution of COVID-19 symptoms and whether patients needed to reach out to a healthcare provider following COVID-19 diagnosis.
Statistical Analysis
The study team utilized descriptive statistics of the survey response population by patient choice to accept or decline anti-spike monoclonal antibody therapy. Responses to the clinic experience questionnaire are listed for those who accepted anti-spike monoclonal antibody therapy. Median and interquartile ranges for continuous and index variables are reported. Categorical variables are summarized as frequency (n) and proportion. The number of incomplete/unanswered is reported for each survey question. Differences in response distribution between patients who accepted or declined monoclonal antibody therapy were estimated using the Mann-Whitney U test for continuous variables, and the χ2 test of independence for categorical variables. If assumptions of the χ2 test were unmet, the Fisher exact test would be utilized. All data management and statistical analyses were performed using Statistical Analysis Software (SAS) Version 9.4.
Results
Patient Population
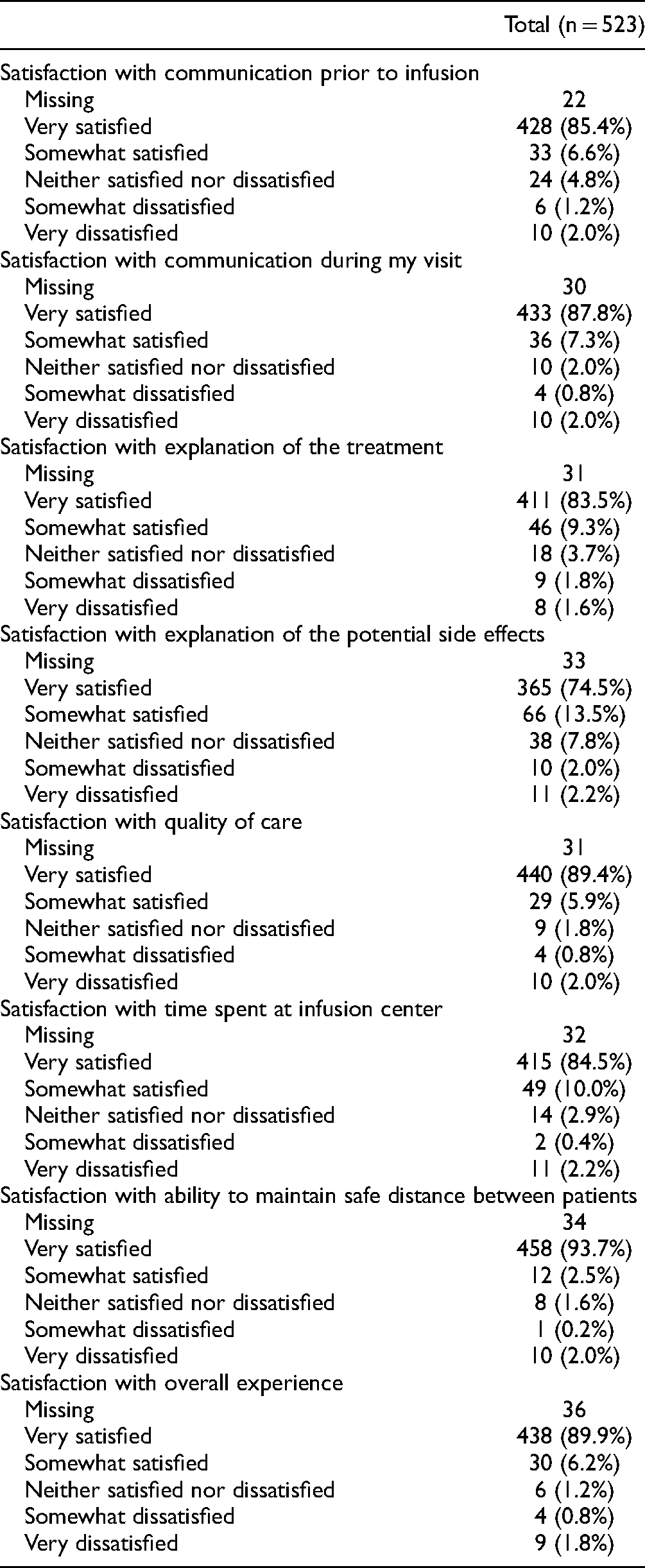
The survey was sent by electronic mail to 2412 patients, including 1436 high-risk patients (59.5%) who consented and received anti-spike monoclonal antibody therapy and 976 (40.5%) who declined treatment. Overall response rate was 26.7% (644/2412). Response rate among patients who accepted monoclonal antibody therapy (523/1436 = 36.4%) was greater than patients who declined therapy (121/976 = 12.4%). Mean age of all survey respondents was 61 years, nearly evenly split between male and female sex, and were primarily of white race. All had at least one medical comorbidity or characteristic that qualified them as high risk of clinical disease progression (Table 1).
Characteristics of Survey Respondents by Decline/Acceptance of Monoclonal Antibody Therapy.

The majority of responders had reported resolution of clinical symptoms by the time of survey in late May to early June 2021. Median time to clinical resolution of symptoms was similar between those who received and those who did not receive therapy (median of 10 days). Notably, 9% to 10% of responders indicated the persistence of clinical symptoms by the time of survey, and this was similar for those who accepted or declined monoclonal antibody therapy. The majority did not seek further medical care after anti-spike monoclonal antibody infusion or after their diagnosis of COVID-19 among those who declined treatment.
Patient satisfaction with monoclonal antibody therapy infusion
The majority of 523 responders reported they were very satisfied (89.4%) or somewhat satisfied (5.9%) with the overall quality of care. The majority were very satisfied with the different aspects of medical care provided (74.5%-93.7% across different measures; Figure 1 and Table 2). Responders voiced satisfaction with communication with the MATRx providers before and during treatment, including education provided about anti-spike monoclonal antibody treatment and its potential benefits and adverse effects. Among the various aspects of care, the explanation of the adverse effects scored the lowest, although 74.5% of all responders were still very satisfied with this aspect.
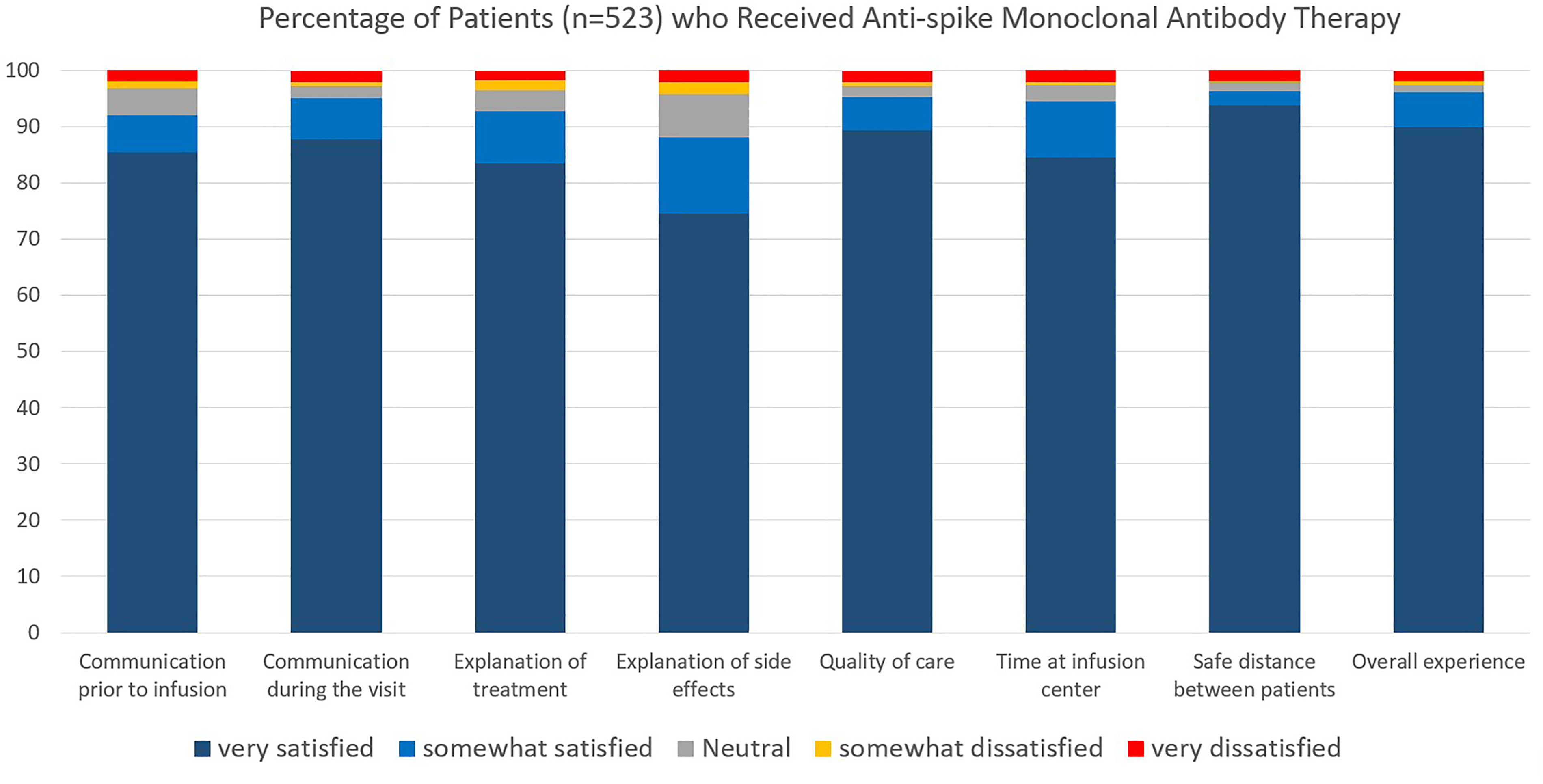
Patient perspectives and experience on anti-spike monoclonal antibody infusion therapy.
Satisfaction With Monoclonal Antibody Infusion Clinic.
Responders were very satisfied with the time spent in infusion centers (appointment slots of 2-3 h, allowing for check-in, 30-60 min infusion time, and mandatory 60-min post-infusion monitoring for adverse effects) and safety protocols, including physical distancing. The overall patient experience was rated highly with the majority being very satisfied (89.9%) or somewhat satisfied (6.2%).
Patients who declined the offer for monoclonal antibody therapy
Nearly a quarter of 121 patients who initially declined (23.6%) therapy indicated they would accept treatment should it be offered again. An almost equal proportion exists between those who will continue to decline (37.3%) or who are undecided (39.1%) should experimental treatment be offered again.
Discussion
The best measure of health care initiatives is the satisfaction experienced by patients. This survey shows an overall positive experience among high-risk patients with COVID-19 who received anti-spike monoclonal antibody therapy. These patient-reported satisfaction outcomes complement clinical and safety outcomes reported with these experimental therapies (3,8,9,12) and support the deployment of patient-centered healthcare for patients offered anti-spike monoclonal antibody therapy in our outpatient settings.
Patients reported they were very satisfied with the overall quality of care provided. This is reflected by the positive feedback provided regarding the team's communication with them prior to monoclonal antibody infusion. Upon automatic screening for eligibility, multimodal approaches were employed to engage patients eligible for monoclonal antibody therapy, which is consistent with best practices of shared decision making to facilitate patient-centered care (13). MATRx providers proactively contacted eligible patients to inform them of the experimental monoclonal antibody therapies, educated them of potential benefits and adverse effects, and the logistics about the infusion therapy. A standardized script was used by team members in order to provide similar information to all eligible patients. Additionally, an education video was provided to patients to discuss monoclonal antibody therapies. We hypothesize that as a result of these practices, even though none of the MATRx providers have established relationships with the patients contacted for therapy, nearly 60% of patients consented to monoclonal antibody therapy. We believe that the time and attention paid by our clinical teams in contacting and educating the patients, along with the educational videos created to answer and reinforce patient questions helped to facilitate high consent rates due to patient-centered care delivery.
After consent was obtained, patients were scheduled for infusion at one of the infusion sites nearest their home residence. The MATRx team established 7 infusion locations across the region, which allowed for easy access by patient populations served (11). There was rapid time to infusion—median time of 2 days after COVID-19 diagnosis (and within one day after education and consenting) (8,9). The infusion sites are within 30 to 60 min by vehicle for most patients. The consenting team made extra effort to schedule patients from same households at the same infusion times to allow them to be in the same infusion room.
Patients were satisfied with the infection control measures and the safety protocols. Patients were advised to show up only during appointment times to limit the potential for congestion in infusion units. They were instructed to remain in their vehicles until a staff member arrived to escort them safely to a private infusion room. Single rooms were used for infusion (unless members of the same household were infused at the same time) and neither patients nor their companions were allowed to mingle or linger in common areas. Personal protective equipment and physical distancing were enforced minimizing exposure to uninfected persons (14).
Patients expressed high satisfaction during the process of infusion. Upon nursing assessment confirming the patient meets eligibility for mild to moderate COVID-19, monoclonal antibodies were infused over 30 to 60 min, and thereafter, patients are monitored clinically for adverse effects. The infusion therapy nursing staff communicated each step of the process, ensured patients were well cared for, provided them with get well soon cards and other treats to uplift their spirits during this anxious time. After infusion, patients were escorted back to their private vehicle. Direct phone line to the MATRx Infusion Center was also provided, with instructions on what to expect and what to do if issues were encountered. High-risk patients were also enrolled in a remote monitoring program to ensure virtual monitoring of symptoms after infusion (15,16).
Almost a quarter of responders who declined the offer for monoclonal antibody therapy indicated they would accept the treatment should this become available to them again. Although data were not collected on the reasons for this, it could be postulated that patients may have had persistent symptoms or progressed to severe disease. Clinical trials demonstrated that anti-spike monoclonal antibody therapies resulted in faster time to viremia clearance and symptom resolution (6,7). However, there was no significant difference among survey responders in time to resolution of symptoms, with up to 10% of patients indicating presence of symptoms at time of survey. This may represent those who have long hauler COVID-19 syndrome. Evaluation of the impact of anti-spike monoclonal antibody on the incidence of long hauler COVID-19 syndrome is in process.
Limitations
Some limitations of the study need emphasis. The overall response rate of the survey was only 27%, but this is consistent with responses of survey research within our community populations (17). Nonetheless, the satisfaction rates reported in this study among the responders of the survey may not necessarily reflect the level of satisfaction among the nonresponders. A cross-sectional design was used, which allowed for quick assessment of patient perception, but this may suffer from recall bias in reporting duration of COVID-19 symptoms, particularly among those who were treated early in the study period. Likewise, comparison between the 2 groups may have inherent biases related to the decision to accept or decline treatment. This is illustrated by more responders among those who had consented to therapy—this group may be more amenable to agree to medical interventions compared to those who declined. It will also be important to assess reasons for accepting or declining therapies, particularly among high-comorbidity groups. Despite high satisfaction outcomes among those who accepted treatment, there are areas that needed to be improved upon. In a separate study, health disparities were identified with regards to the decision to consent for monoclonal antibody therapies (12). Although these individuals represent a small percentage of the overall population (which was predominantly Caucasian), underrepresented populations were much less likely to accept treatment (12). In response, language interpreters were made available to help address this, as well as engagement with local community leaders to encourage acceptance of these experimental therapies. Likewise, partnerships were formed with local primary providers who have established relationships with these patients and encouraged them to recommend treatment should their patients seek input on these therapies (11).
Conclusions
Substantial evidence points to a positive association between various aspects of overall patient experience, such as good communication between providers and patients, and clinical outcomes through patient-centered care (18–20). These outcomes include patient adherence to medical advice (acceptance of the offer for experimental therapies), better clinical outcomes (patient survival), and lower utilization of health care services (reduced need for hospitalization and intensive care unit use). The proactive approach by this program translated to a higher acceptance rates for experimental monoclonal antibody therapy, a rapid time to infusion of potentially lifesaving therapies, and have resulted in significant reduction in severe disease that would have warranted utilization of inpatient services (8,9). In our view, the provision of high-quality care that revolved around communication and establishment of trust was instrumental to ensuring high satisfaction among patients.
Supplemental Material
sj-docx-1-jpx-10.1177_23743735221105673 - Supplemental material for Patient Perspectives on Anti-spike Monoclonal Antibody Therapy for Mild to Moderate Coronavirus Disease-2019
Supplemental material, sj-docx-1-jpx-10.1177_23743735221105673 for Patient Perspectives on Anti-spike Monoclonal Antibody Therapy for Mild to Moderate Coronavirus Disease-2019 by Raymund R Razonable, Ravindra Ganesh, Rachel K Bishop, Tracy L Culbertson, Molly Destro Borgen, Michelle C Hedin, Laura H Hopkins, Tammy A Jackson, Jennifer J Larsen, Jennifer A Matoush, Darcie E Moehnke, Shelly M Olson, Kellie M Peterson, Jordan Rosedahl and Lindsey M Philpot in Journal of Patient Experience
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Mayo Clinic.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
