Abstract
Recent evidence suggests that adding protein convertase subtilisin/kexin type 9 inhibitors to current lipid-lowering therapies may result in unprecedented reductions in low-density lipoprotein cholesterol. The objective of this study was to assess the potential pharmaceutical financial impact of including the protein convertase subtilisin/kexin type 9 inhibitor, alirocumab, in the Italian National Health Service budget. The budget impact of adding alirocumab as a lipid-lowering therapy treatment option was assessed among the population defined by the AIFA reimbursement criteria. Data from the IMS/CEGEDIM national electronic database were used to estimate the size of the potentially eligible population. Drug costs were estimated according to dosing schedule and published prices. Alirocumab annual uptake was assumed at 7%, 9% and 10% for years 1, 2 and 3, respectively, considering a 20% treatment drop-out rate. Sensitivity analyses tested the impact of model inputs on the results. The annual estimated incremental cost of alirocumab treatment was €5106/patient. Treating an eligible population of 843 patients with alirocumab in the first year resulted in a total National Health Service budget impact of €4.30 million. Assuming growth of the alirocumab-treated population of 2105 and 2819 patients in the second and third years, the budgetary impact would be €10.75 and €14.10 million, respectively. Sensitivity analysis suggested that financial impact is most sensitive to the uptake of alirocumab in the population. The uptake of alirocumab results in an overall modest budgetary impact to the Italian National Health Service when added to conventional lipid-lowering therapies in a carefully selected population compared to the total lipid-lowering therapy pharmaceutical expenditure in Italy (€1 billion).
Introduction
Cardiovascular disease (CVD) is the leading cause of death in Europe. 1 It is estimated that 42% of all deaths in women and 38% of all deaths in men, below 75 years of age, are CVD-related. 2 In Italy, ischaemic heart disease and cerebrovascular events were the two leading causes of death in 2012, with 12% and 10% of all registered death cases, respectively. 3 The prevalence of CVD among Italian adults was 4% for heart diseases and 1.5% for stroke with an incidence of over 135,000 acute strokes annually.4,5
CVD is associated with substantial cost to the national health care system with a pharmaceutical expenditure of €3.6 billion spent on CVD treatment and prevention by the Italian National Health Service (INHS) in 2015. 6 A recent Italian analysis based on the ARNO Observatory (large national administrative database for the epidemiological and economic surveillance), estimated direct medical costs associated with treatment of CV events of €9537/year for CVD, €12,004 for peripheral arteriopathy and €14,871/year for the treatment of acute myocardial infarction. 7 Over 80% of the costs were associated with hospitalizations and the frequent re-admissions among these patients. 7 Lowering low-density lipoprotein cholesterol (LDL-C) plasma levels is universally recognized as one of the key objectives in the primary and the secondary prevention of CVDs.2,8–10
Pharmacological treatment of hypercholesterolaemia and dyslipidaemia is recommended in the primary and secondary CVD prevention in patients with familial hypercholesterolaemia and in those at high and very high risk of CVD. 8 Despite an appropriate adherence to pharmacological treatment, approximately 30% of patients with very high and high CV risk and around 20% of patients with familial hypercholesterolaemia do not reach target LDL-C levels and are in need of alternative therapies.11–14
Current evidence suggests that an addition of anti-protein convertase subtilisin/kexin type 9 (PCSK9) monoclonal antibodies to current lipid-lowering therapies (LLTs) lead to a significant reduction in LDL-C. 15
The efficacy and long-term safety of PCSK9 inhibitors have been evaluated in the phase III ODYSSEY programme for alirocumab and the PROFICIO programme for evolocumab. Data from the ODYSSEY and PROFICIO clinical programmes suggest sustained LDL-C reductions of up to 61% after 12 weeks associated with alirocumab 16 and evolocumab. 17
In Italy, in the context of primary hypercholesterolaemia (heterozygote familial and non-familial) or mixed dyslipidaemia, PCSK9 inhibitors (alirocumab and evolocumab) are reimbursed according to the AIFA prescribing criteria:18–21 in adult patients below 80 years of age who do not reach pre-defined LDL-C levels despite at least 6 months of prior combination therapy with maximum tolerated dose of high-potency statin agent and ezetimibe or ezetimibe monotherapy in case of stating-intolerant patients, more specifically alirocumab is reimbursed in
Primary CVD prevention in patients with familial heterozygote hypercholesterolaemia (HeFH) and LDL-C ⩾ 130 mg/dL;
Secondary CVD prevention in patients with HeFH or non-familiar hypercholesterolaemia or mixed dyslipidaemia with LDL-C ⩾ 100 mg/dL.
The aim of this study was to estimate the pharmacy budgetary impact of the PCSK9 inhibitor alirocumab within the Italian National Health Service (INHS).
Methods
Model overview
The analysis here in was developed in Microsoft Excel 2007 and according to the ISPOR principles of Good Practice for Budget Impact Analysis. 22 The model compared the total pharmacy costs among an Italian population in scenarios with and without various market uptake thresholds of alirocumab as an option to add-on to high-potency statins and/or ezetimibe over a 3-year time period. To account for the uncertainty, a sensitivity analysis was performed on the first year. Key parameters including alirocumab price, drop-out rate and alirocumab utilization rate were varied between plausible ranges.
Target population
The target population considered in the analysis was defined according to AIFA prescribing criteria for PCSK9 inhibitors.20,21 The number of eligible patients was derived from estimates of the overall Italian population aged 18–80 years from the Italian 2014 IMS/CEGEDIM national electronic database; 23 an Italian primary care database in place since 1998 including data of more than 1.1 million patients with clinical and laboratory information, treated by over 900 general practitioners (GPs). Patient demographic details included in the database are linked through the use of an encrypted patient code with medical records (diagnoses, tests, tests results, therapeutic procedures and hospital admissions), information on drug prescriptions, lifestyle information (alcohol, body mass index and smoking habit) and date of death. In this database, all diseases are classified according to the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM). To be considered for participation in the database, GPs must meet up-to-standard quality criteria pertaining to the levels of coding, prevalence of diseases, mortality rates and years of recording. Quality and consistency of data collected in the database have been demonstrated and validated through several studies in which the retrieved information has been compared with other current data sources or findings from national surveys. 24
Based on the data from the 2014 IMS/CEGEDIM database, there are approximately 5.3 million adult patients treated with statins and/or fibrates and omega-3 in Italy (Figure 1). Out of those treated with statins and/or fibrates and omega-3, there are 3.02 million patients with a definite profile in line with AIFA prescribing criteria for familial heterozygote hypercholesterolaemia. Among them, a 2.2% was previously treated with a maximum tolerated dose of high-potent statin agents plus ezetimibe or was intolerant to statin therapy (defined as therapy with ezetimibe or at least one statin therapy in the last 5 years). Based on the available data on LDL-C measurements in the preceding 2 years from the 2014 IMS/CEGEDIM database, there were 30,092 patients who matched above described criteria and who did not reach recommended LDL-C levels. Patients with no LDL-C measurement within preceding 2 years (21.6%) were not considered in the scope of this analysis.

Patient flow – population eligible for alirocumab treatment.
Alirocumab utilization rate and cost
PCSK9i were estimated to take market shares of, respectively, 14%, 18% and 20% for years 1, 2 and 3 considering the prescribing limitations set by AIFA (PCSK9 inhibitors are reimbursed only upon specialist prescription in hospitals/centres locally identified and authorized by each regional authority) and timing of regional access. Considering equal penetration for both PCSK9 inhibitors, the assumed utilization rate of alirocumab would increase from 7% in the first year to 9% in the second year and 10% in the third year. It was assumed these assumptions reflect the current trends of PCSKi in Italy.
The total pharmacy costs were calculated for the number of full-year-treated (FYT) patients for each drug or drug combination, considering assumptions on alirocumab monthly uptake, and a discontinuation rate of 20% in the 3 year period.
Drug costs (Table 1) were calculated based on reported dosing schedules in package inserts and the public prices (for those drugs reimbursed in Class A), excluding VAT, net of the compulsory discounts. 25 The cost of generic drugs was determined based on the NHS’ reference price list, 26 and the cost of alirocumab (reimbursed in Class A-PHT (A-PHT is a special class of reimbursement by which a drug (such as the case of alirocumab) can be distributed by both hospitals and Local Healthcare Units (LHU) and in some instances even retail pharmacies, with reduced distribution margins)), was calculated as the ex-factory price net of compulsory discounts (€13.99 per day). 18 Confidential discounts are not publicly available and therefore could not be considered in this analysis (Table 1).
Drug unit cost and daily cost of therapy.
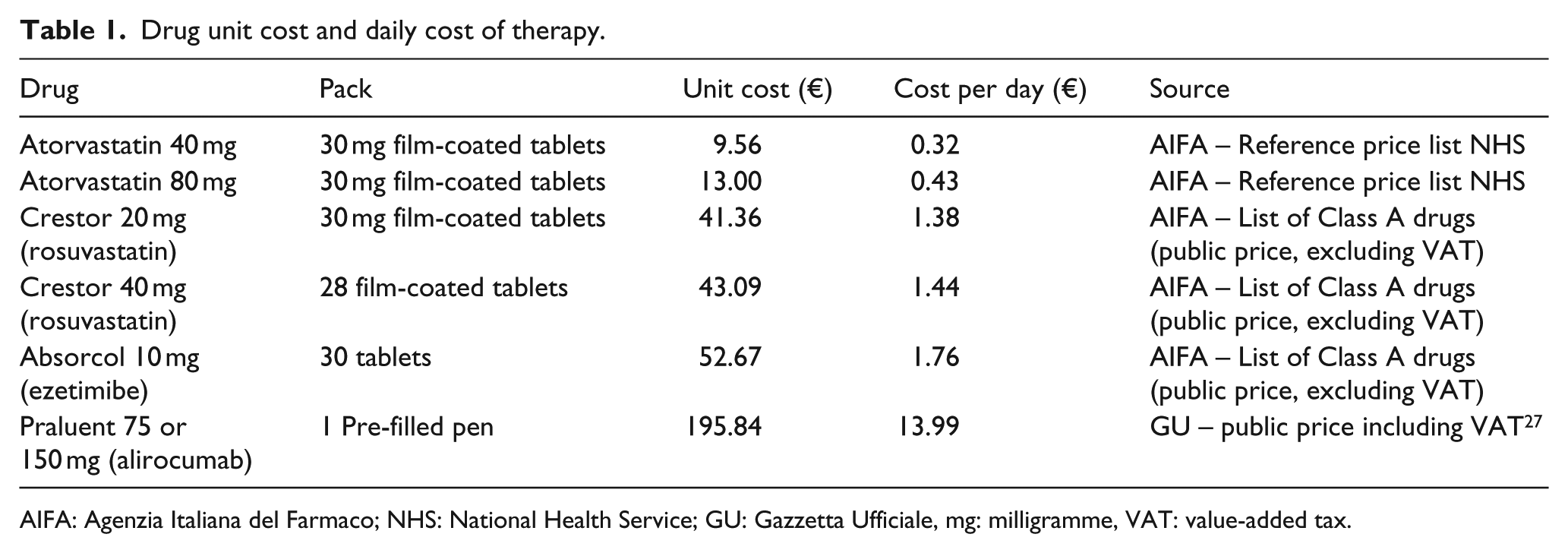
AIFA: Agenzia Italiana del Farmaco; NHS: National Health Service; GU: Gazzetta Ufficiale, mg: milligramme, VAT: value-added tax.
The cost of CV events, as financial implication of alirocumab therapy, was not included in this analysis because the results of the OUTCOME study from the ODYSSEY clinical development programme were not available at the time of this analysis.
The expected pharmacy budgetary impact was calculated as the incremental difference in the cost to the National Payer with varying degrees of uptake of alirocumab over the next 3 years.
To account for the uncertainty in model parameters, sensitivity analyses were completed varying the price of alirocumab (±5%; ±10%), drop-out rate (±10%) and alirocumab utilization rate (±25%).
Results
Patient population
Of the 47.1 million Italian adults (aged 18–80 years), 5.4 million were treated with statins and/or fibrates and/or omega-3 (Figure 1), and of these, 3 million (56%) have a risk profile matching the AIFA prescribing criteria for alirocumab (described earlier in the article). Out of the 3 million of patients, 67,135 (2.2%) were treated with high-potency statins (as defined by atorvastatin 40–80 mg and rosuvastatin 20–40 mg) and ezetimibe, or ezetimibe in monotherapy in patients considered as statin-intolerant. All patients on treatment with ezetimibe alone and with evidence of a previous treatment with at least one statin in the preceding 5 years were considered as intolerant to statin therapy. Approximately 20% of the statin-intolerant patients did not have any LDL-C measurement in the preceding 2 years, so were excluded from the eligible population. Of all patients with LDL-C measurement, 30,092 patients (45%) did not reach LDL-C goal and were considered as eligible for alirocumab treatment (Figure 1).
Projected cost
The base case analysis, from the national perspective, estimated a total number of 843 alirocumab FYT patients in the first year after alirocumab introduction with a budgetary impact of €4.30 million on the Italian NHS. The treated population is estimated to increase to 2105 and 2819 FYT patients in the second and third years, with a pharmacy budgetary impact of €10.75 million and €14.40 million, respectively (Table 2). The availability of alirocumab for treatment of patients of age 18–80 years that do not reach pre-defined LDL-C levels, despite at least 6 months of prior combination therapy with maximum tolerated dose of high-potency statins and ezetimibe, or in patients intolerant to statin therapy, would generate a total mean incremental budgetary cost of €5106 per patient per year.
Budget impact for alirocumab add-on to lipid-lowering therapy – NHS perspective.
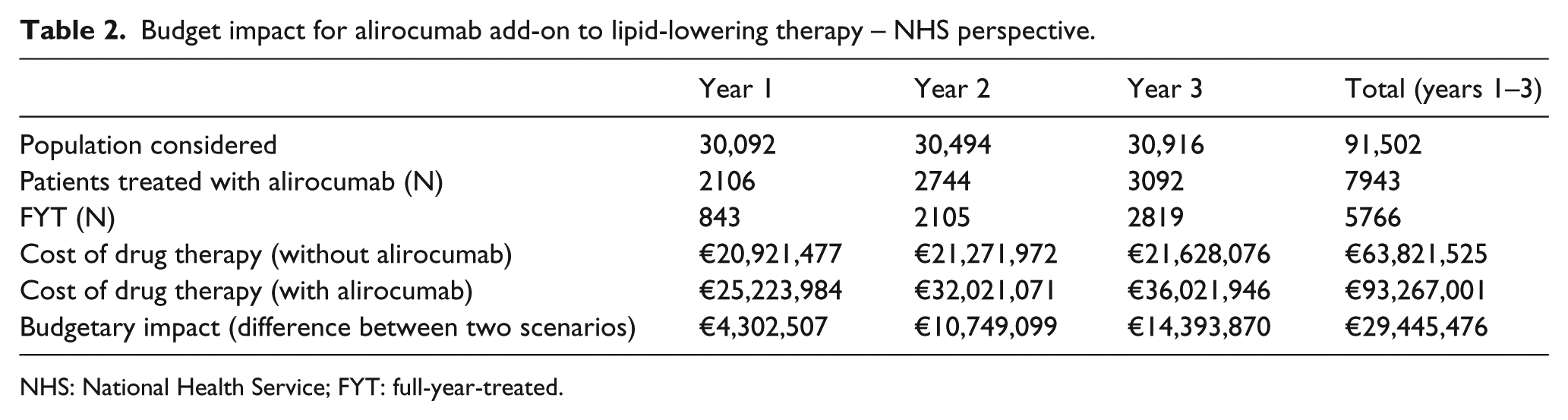
NHS: National Health Service; FYT: full-year-treated.
Sensitivity analysis showed that the model is sensitive to changes in the input parameters, that is, price of alirocumab, drop-out rates and market shares of PCSK9 inhibitors. Market shares of treatment uptake showed the highest financial impact with potential variations in the range of €3.23 to €5.38 million in the first year after treatment introduction (Table 3).
Sensitivity analysis – NHS perspective.
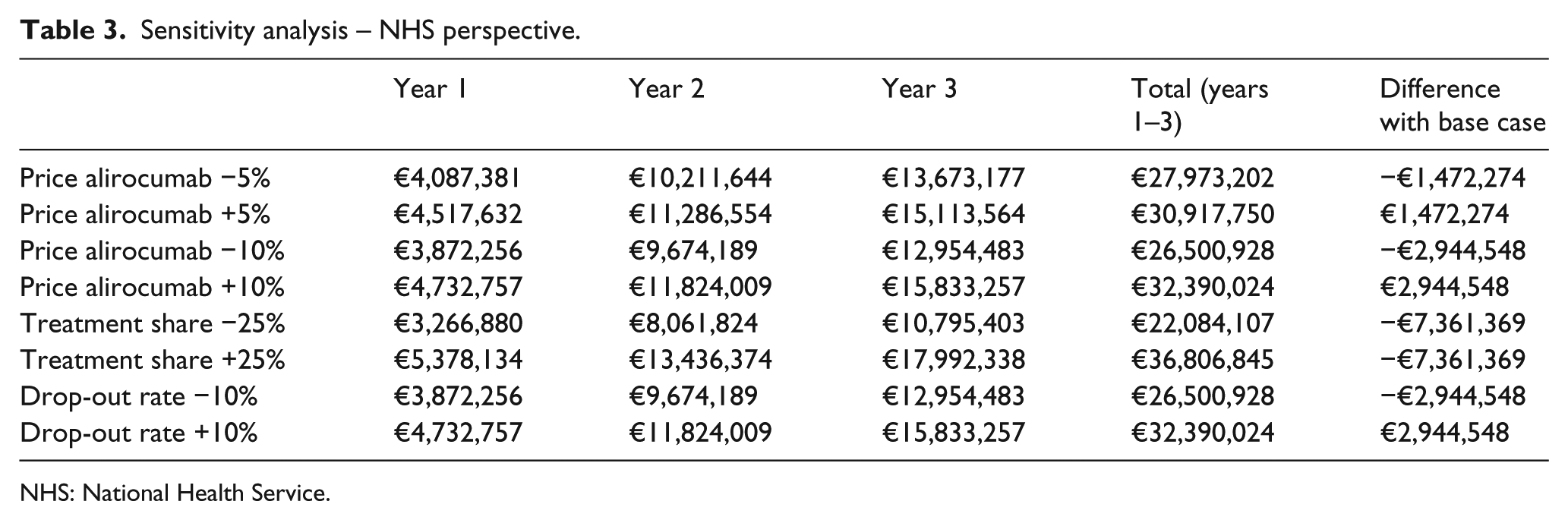
NHS: National Health Service.
Discussion
PCSK9 inhibitors are reimbursed in Italy in a strictly defined population of patients with hyper/dyslipidaemia at high or very high CV risk.18–21 Despite adequate pharmacological treatment and adherence, CV risk in this patient group remains up to five times higher compared to patients with lower CV risk, and thus, there is a high unmet need for additional treatment options. 13 The PCSK9 inhibitor, alirocumab demonstrated efficacy and safety in the comprehensive clinical investigation programme including more than 24,000 patients. It showed a favourable safety profile with common adverse events such as local injection site reactions. 16
In the environment of limited health care resources, PCSK9 inhibitors have sub-optimal market access in Europe where, similar to the Italian context, positive recommendations were restricted to patients without alternative treatments and thus with high unmet need (LDL-C concentrations persistently above locally recommended LDL-C target thresholds despite maximal tolerated lipid-lowering therapy or intolerant to statin therapy) and under special economic agreements. The UK’s National Institute for Health and Care Excellence (NICE) has issued technology appraisal (TA) guidance documents on the use of evolocumab and alirocumab in high-risk patient groups and with discounts agreed under patient access schemes.28,29 The Canadian Drug Expert Committee (CADTH) recommended the reimbursement of alirocumab at a reduced price in patients with confirmed HeFH or atherosclerotic CVD and LDL-C levels persistently above locally recommended threshold despite treatment with a maximum tolerated state dose or ezetimibe. In Canada, a price reduction of at least 57% would be required for alirocumab to be considered a cost-effective treatment option in a mixed population of HeFH patients and in patients with high risk of CV events. 30 Some health technology assessment (HTA) bodies across Europe showed concern about the high budget impact of these costly drugs. Considering the high prevalence of CVD and the current lack of comprehensive evidence on the clinically relevant long-term outcomes, concerns are likely associated to potential use of costly therapies in patients where long-term benefits are still not substantially supported with hard end-points. It is anticipated that availability of evidence of prolonged use of PCSK9i therapies will allow robust economic analyses that may further inform HTA bodies and ensure efficient use of local health care resources.
In Italy, PCSK9 inhibitors are reimbursed in strictly defined patient population and under price and volume managed entry agreements. PCSK9 inhibitors are anticipated to have limited penetration in Italy due to various hurdles immediately after launch. One of the most relevant would be the time needed for individual regional access, that is, inclusion in the local regional drug formularies as well as identification of the prescribing specialists/centres at the regional level. Indeed, another reason for relatively low expected market uptake is that PCSK9 inhibitors are reimbursed only upon prescription by specialists at hospital centres identified by the autonomous regions and provinces and dispensed directly the designated centres (i.e. the group of high-risk CV patients are not able to obtain a PCSK9i under GP prescription, but only after a referral to the specialist at designated regional centres and dispensed directly by respective centres).
Given no official sources to estimate the number of PCSK9i eligible patients in Italy, this analysis used national statistics and the IMS/CEGEDIM database to estimate the number of subjects who could be treated with PCSK9 inhibitors (approximately 30,000 patients). The results of the budgetary impact analysis showed a moderate budgetary impact of the PCSK9 inhibitors on the Italian National Healthcare System. The incremental annual cost of alirocumab treatment was estimated to be €5106 per patient, with a total incremental cost of €4.30, €10.75 and €14.39 million in the years 1, 2 and 3, respectively. Sensitivity analysis suggested potential impact to be the most sensitive to variation of treatment shares with the lowest financial impact of €3.24 million in year 1 at the lower uptake shares of PCSK9 inhibitors (which could be the case, due to the slowness of regional access).
Given the significant improvements in lowering the LDL-C levels in patients refractory or intolerant to statin therapy, its favourable safety profile, and moderate budgetary impact, alirocumab may represent an optimal treatment choice in this high-risk Italian patient population.
This analysis is the first attempt to evaluate the pharmacy budgetary impact of introducing PCSK9 inhibitors in Italy. While some studies can be found which evaluated the cost-effectiveness, studies that assess the budgetary impact on a national health system are very limited in the literature, and none has been published specifically for the Italian setting.
As with any research, this budget impact analysis bears some limitations. According to AIFA recommendations, the model assumed 100% adherence to hypolipemic treatments; yet, treatment adherence exerts a crucial role, especially on treatments for chronic diseases. Evidence shows that full hypolipemic treatment adherence in both primary prevention and secondary prevention is associated with significant risk reduction for fatal and non-fatal CV events, hospital admissions and all-cause mortality. 13 Adherence to statin treatment in Italy is low. In 2015, the patients fully adherent to their assigned statin treatment schedule was 45.8%. 6 Yet, only patients with 100% adherence to hypolipemic intensive statin therapy are eligible for treatment with PCSK9 inhibitors as per AIFA recommendations, and thus, this analysis applied this assumption. Financial implications of alirocumab therapy in relation to the number of CV events avoided were not assessed in this analysis as evidence on CV outcomes for alirocumab (OUTCOME study from ODYSSEY programme) was not available at the time of this analysis. The first results were presented only recently (March 2018) at the American College of Cardiology Congress. 31 This international study included 18,924 patients randomized to alirocumab or placebo in centres across 57 countries. After a median follow-up of 2.8 years, treatment with alirocumab showed to reduce primary efficacy outcome, major cardiac adverse events (MACE), by 15% (hazard ratio (HR) = 0.85 (95%confidence interval (CI): 0.75; 0.93), p = 0.0003) in the overall study population. In the population with LDL-C ⩾ 100 mg/dL at baseline MACE were reduced up to 24% (HR = 0.76 (95%CI: 0.65; 0.87)).
Alirocumab, a new PCSK9 inhibitor prescribed as add-on therapy to statins and ezetimibe (or ezetimibe monotherapy), showed an overall modest budgetary impact of €4.3 million in the first year, €10.8 million in the second year and €14.4 million in the third year on the Italian NHS. The expenditure for the PCSK9i Alirocumab is projected to have a small impact on the Italian NHS expenditure compared to the total pharmaceutical LLT expenditure (€1 billion reported in 2015 6 ).
Footnotes
Acknowledgements
A.M. and S.S. are employees of Analytica LASER, a Certara company. F.D. and F.F. are employees of Sanofi S.p.A., Italy. A part of this research was presented at the ISPOR 20th Annual European Congress in Glasgow, 4–8 November 2017.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported through unrestricted grant by Sanofi S.p.A., Milano, Italy.
