Abstract
Objective:
The aim of this study was to assess patient experiences when reporting symptoms of twin–twin transfusion syndrome (TTTS) to their health-care providers.
Methodology:
The study utilized an online, retrospective survey of women, over the age of 18, who were living in the United States at the time of their pregnancy and had completed a TTTS pregnancy.
Results:
Three hundred sixty-seven cases were included for analysis. Nearly half of the respondents (45.2%) reported experiencing maternal symptoms prior to TTTS diagnosis. The average number of symptoms experienced was 2.85. The average gestational week of symptom onset was 18.2. A total of 76.2% of respondents experiencing symptoms shared these concerns with their health-care provider; however, slightly more than half (51.2%) believed that the provider dismissed their complaints.
Conclusions:
Results suggest a disconnect between patients’ reporting TTTS symptoms and health-care providers responding attentively, as perceived by the patient. It would be advantageous for health-care providers to inform women pregnant with a monochorionic–diamniotic pregnancy to immediately report the presence of any symptom described in the present research, which may be associated with any number of twin pregnancy–related complications.
Keywords
Introduction
Twin–twin transfusion syndrome (TTTS) complicates 15% of monochorionic–diamniotic (MCDA) twin pregnancies and carries a high risk of morbidity and mortality (1). The syndrome is caused by unbalanced blood flow from the donor to the recipient through placental anastomoses (2,3). Twin–twin transfusion syndrome diagnosis is ultrasound-based and characteristically presents as an oligohydramnios–polyhydramnios sequence (4). When left untreated, TTTS is 80% to 100% fatal for fetuses; however, the increased use of interventions such as laser coagulation of the intertwin anastomoses (laser therapy) has resulted in significantly improved survival rates for both twins (69.5%) and at least 1 twin (89.5%) (5).
The Quintero staging system is widely used for identifying TTTS severity. Stage 1 requires an imbalance of fluid between the donor (<2 cm) and the recipient (>8 cm) and serves as a baseline requirement for all of the following stages. In Stage 2, the bladder of donor twin is no longer visible. Stage 3 includes critically abnormal Doppler velocimetry in either fetus. Stage 4 presents with hydrops in either fetus; and finally, stage 5 is the demise of one or both fetuses (6).
Twin–twin transfusion syndrome treatments such as laser therapy have been shown most effective when performed in earlier stages of disease progression (7). And while the Quintero staging system provides a helpful definition of this progression, TTTS is not always linear and can develop rapidly (8). For this reason, it is recommended that all MCDA twin gestations receive biweekly ultrasound surveillance starting in week 16 and continuing through delivery (4).
Although serial ultrasonography is the only definitive way to diagnose TTTS, initial efforts have been made to investigate the value of maternal symptomatology reporting practices. Due to the 2-week gap that accompanies the recommended biweekly ultrasound schedule for all MCDA gestations, patient reporting of physical symptoms may help indicate an impending diagnosis, potentially timelier than waiting for the next scheduled ultrasound.
Polyhydramnios comprises the oligohydramnios–polyhydramnios sequence in which the donor fetus has too little amniotic fluid (oligo) and the recipient fetus possesses too much (poly)—a complication directly caused by TTTS (4). Symptoms of polyhydramnios can include sudden abdominal swelling and weight gain (9). Correspondingly, anecdotal reports indicate that symptoms of TTTS include sensation of rapid growth of the womb; a uterus that measures large for dates; abdominal pain, tightness, or contractions; sudden increase in body weight; and swelling in the hands and legs in early pregnancy (10). In a case series, Sueters et al examined the value of serial ultrasound examinations in conjunction with instructing patients to report the maternal symptoms of rapidly increasing girth and/or premature contractions. In 2 of the 4 cases presented, patients reported feeling rapidly increasing girth which was predictive of a TTTS diagnosis (11).
Additional research suggesting the existence of TTTS symptoms was reported in a qualitative study that analyzed 45 families’ stories about their TTTS experience. From the publicly available online stories, it was discovered that nearly 32% described some type of TTTS symptom prior to diagnosis, including pain, gaining weight quickly, or shortness of breath. One mother reported that upon sharing her concerns with her health-care provider, she felt as though her symptoms were dismissed (12).
The work by Sueters et al and Fischbein et al suggests the existence of maternal TTTS symptoms prior to diagnosis; however, no research has examined the full range of symptoms that may be experienced by mothers prior to TTTS diagnosis. Given the high-risk nature of MCDA pregnancies, the full range of maternal symptoms during TTTS should be explored to assist with differential diagnosis. In addition, further information is required to understand the patient–health-care provider interaction when symptoms are shared by patients prior to TTTS diagnosis.
Methodology
A comprehensive, online survey with women who completed a TTTS pregnancy was created to explore multiple areas related to a TTTS pregnancy. The survey was targeted toward individuals who met 4 criteria: (1) female, (2) older than 18 years, (3) living in the United States at the time of their pregnancy, and (4) completion of a TTTS pregnancy. Responses were gathered over a 4-month period in 2016. Participants were recruited through social media groups including Facebook group sites devoted to TTTS, identical twins, MCDA pregnancies and twins, twin loss, and multiples loss. Participants were also recruited through advertisement on the Facebook pages of national foundations including the Twin to Twin Transfusion Syndrome Foundation and the Fetal Health Foundation. Finally, the survey was advertised with the Mother of Multiples of America/Mother of Twins national website and newsletters. Participants were informed that the survey was anonymous and were also provided with contact information for crisis services in the event of a mental or behavioral health crisis during or after participation in the survey. After reviewing the informed consent statement, participants who participated in the subsequent survey were then considered to have given consent. This research was approved by the institutional review boards of both D'Youville College and Kent State University.
The online survey asked participants to respond to questions related to their TTTS pregnancy in the following domains: demographics, TTTS diagnostic and treatment experiences, mental health and mental health treatment experiences, pregnancy outcomes, infertility treatment, social support, relationship outcomes, and perceptions of care. Questions related to symptomatology included “Did you experience any TTTS symptoms prior to the diagnosis?” “If you answered yes to the previous question, what symptoms did you experience (you may select more than 1)?: swelling, sudden weight gain, contractions, decreased fetal movement, pain, other,” “In which gestational week did these symptoms first appear?,” “Did you share these symptoms with your health-care provider?,” “With which health-care provider did you share your symptoms?,” and “Do you believe your health-care provider responded to your concerns appropriately?.” Participants were provided the option of “I don’t know” for each item and also the ability to write in details to describe other symptoms as well as their health-care provider’s response to their concerns.
Data were analyzed using Stata 14. Analyses included descriptive statistics. Two reviewers independently reviewed the open-ended responses and coded for themes and categories. Consensus-based discussion was used where necessary.
Results
Three hundred ninety-four women participated in the survey. Eight participants indicated they were not living in the United States at the time of their pregnancy and were removed from the analysis, for a total of 386 respondents. The majority of respondents were white (89.4%), had at least some college (87.7%), married (80.7%), with annual family income of at least US$60 000 (63.0%). The median year that the TTTS pregnancy occurred was 2012, and the average participant age during the TTTS pregnancy was 30.2. Most participants reported having private insurance during their TTTS pregnancy (72.6%).
Of the 386 patient-reported cases of TTTS available for review, an additional 19 (4.9%) participants failed to respond to symptomatology-related questions. A total of 367 cases are included for symptomatology analysis. Nearly half of these cases (45.2%) reported experiencing maternal symptoms prior to TTTS diagnosis. The average number of symptoms experienced was 2.85 (standard deviation [SD]: 1.30 and range: 1-8 symptoms). The average gestational week of first symptom experience was 18.2 (SD: 18.16 and range: 6-33 weeks).
The most common symptoms experienced were sudden weight gain (76.2%), swelling (58.8%), and pain (58.2%). However, 21.0% of respondents selected “other” and listed additional symptomatology. Within this voluntary “other” category, descriptions of “tight stomach” (22.9%), “rapid abdominal growth” (20.0%), and “measuring big” (20.0%) were reported most frequently (see Table 1).
Symptom Types.

aRespondents were able to select more than 1 symptom.
Among respondents who experienced maternal symptoms prior to a TTTS diagnosis, 76.2% shared these symptoms with their health-care provider. The most common type of health-care provider was an obstetrician (67.7%). When respondents were asked to report whether their provider responded appropriately to the symptoms, slightly more than half (51.2%) reported they believed the provider did not attend to their concerns (see Table 2). To help control for the possible effects of new clinical recommendations published in 2013, which outline screening for TTTS during MCDA pregnancies, a separate analysis was conducted restricting to years 2013 to 2016. However, this analysis did not yield meaningful changes in the number of women reporting that they believed that the health-care provider responded appropriately to their symptoms and complaints.
Symptom Sharing and Provider Response.

Respondents were allowed to write-in and describe what they believed was an appropriate or inappropriate response on behalf of their health-care provider. Of those who felt their provider responded appropriately, the most common descriptions were “immediate ultrasound” (36.4%) and “sent to MFM/specialist” (30.0%). Of those who felt their provider did not attend to their concerns, the most common descriptions were being told that they were experiencing “‘typical’ twin pregnancy symptoms” (50.1%) and being “dismissed” (16.9%). “Dismissed” was most often described as the health-care provider not engaging in discussion about the complaint (see Table 3).
Qualitative Patient-Reported Provider Responses to Symptoms.
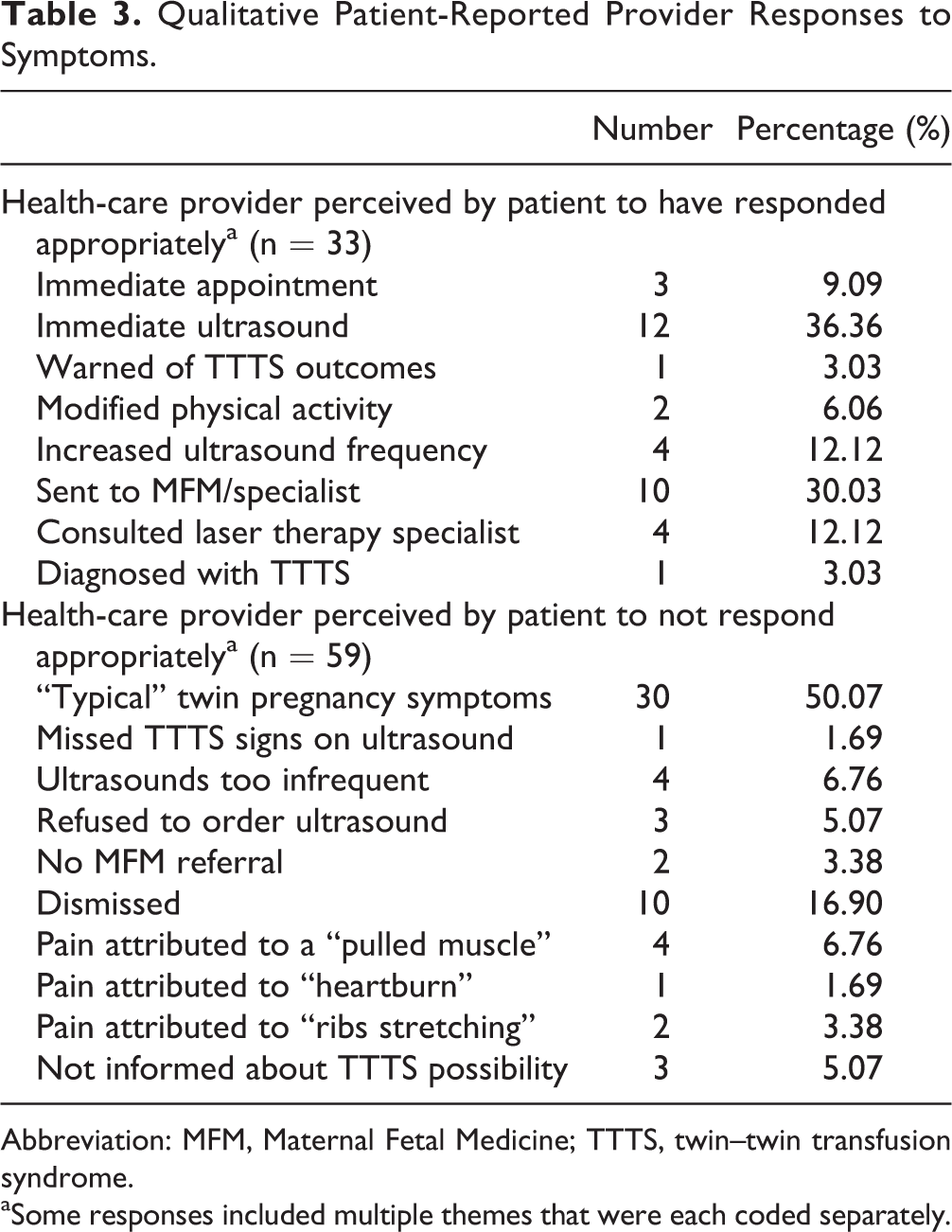
Abbreviation: MFM, Maternal Fetal Medicine; TTTS, twin–twin transfusion syndrome.
aSome responses included multiple themes that were each coded separately.
Discussion
The results of the present work reveal that symptoms including sudden weight gain, swelling, and pain were experienced among many women prior to their TTTS diagnosis. The symptoms reported are consistent with those described in the works by Sueters et al and Fischbein et al. In addition, many women experienced multiple symptoms, with the average number of symptoms experienced by respondents was 2.83. The majority of respondents who experienced symptoms reached out to their health-care providers to communicate their concerns, but more than half of those women felt that their concerns were dismissed by their health-care provider.
Twin gestations are more prone to complications, including preterm labor/birth, low birth weight, gestational diabetes, intrauterine growth restriction , and preeclampsia (13). Preeclampsia, specifically, is a serious pregnancy complication and if left untreated can result in maternal and infant illness and death (14). Notably, symptoms of preeclampsia include sudden weight gain, swelling, and abdominal pain (15)—symptoms that mirror those of TTTS (10).
However, preeclampsia is rarely seen prior to 20 weeks’ gestation and is most common in the late second and third trimesters (16). The average gestational week of symptom onset as reported in this study was 18.2 weeks, or before preeclampsia would be likely to occur, indicating that a thorough symptom investigation, to identify symptom causes and establish differential diagnosis, would become all-the-more pertinent. Unfortunately, many women reported not experiencing a thorough investigation of their symptoms.
As demonstrated in other disease domains, whether real or perceived, time constraints during exams can influence the extent to which symptoms are shared by patients and responded to by physicians (17,18). Nonetheless, effective physician–patient communication can lead to improved health outcomes (19). Specifically, communication that occurs during the history-taking portion of an exam and the discussion of management moving forward is vital (20). Conversely, inadequate history-taking and examination has been shown to influence delayed diagnosis (21).
Although this research is the first to report maternal symptomatology using a large sample of mothers who experienced TTTS, there are limitations to the present work. The current study used an online, self-reported, retrospective survey and asked patients to recall and report symptoms. Recall bias, or the inaccurate or incomplete memory of the TTTS experience, can be a threat to the internal validity of retrospective studies and may have influenced the quality of the data reported in the present study (22). However, concerns over the impact of recall bias may be reduced given our results support the findings presented by Sueters et al in their case series study (11). Additionally, maternal recall obtained within 4 to 6 years of the prenatal experience has been found to be 89% accurate when compared to patient charts (23). The median year that the TTTS pregnancy occurred was 2012, or a 4-year recall period.
An additional limitation of the present research was the use of a convenience sample with recruitment conducted through a variety of support groups, primarily using social media. This may have skewed our sample as these groups may be comprised of individuals who had better or worse TTTS experiences and outcomes. In particular, our findings regarding health-care provider responses to patient-reported symptoms may have overestimated the number of participants who reported feeling their concerns were not addressed. However, Baud et al reported that from 2010 to 2012 approximately 30% of women with TTTS presenting at a Canadian fetal treatment center for fetoscopic selective laser ablation surgery had experienced misdiagnosis. Further, only 40% of the anticipated TTTS cases in Canada were referred to a fetoscopic laser surgery treatment center during that time (24). Consequently, despite concerns related to convenience sampling, our research provides additional evidence of complex issues related to TTTS diagnosis.
Finally, it remains unclear whether the symptoms reported in this study that are indicative of polyhydramnios and other prenatal complications (preeclampsia) are also associated with uncomplicated MCDA twin pregnancies. The current research was a cross-sectional study of only women who had experienced TTTS and therefore was unable to assess whether women with MCDA pregnancies that do not ever become complicated by TTTS also experience the symptoms described here and with the same frequency. Future research should explore this issue to assist with differential diagnosis between the uncomplicated MCDA pregnancy, TTTS, and other twin pregnancy complications. Specifically, a prospective cohort study is needed to help determine whether or not common maternal symptoms of TTTS, like those presented here, can accurately predict an impending diagnosis, potentially prompting earlier, more effective treatment.
Conclusion
Because of the high-risk nature of the MCDA pregnancy, we suggest health-care providers inform women pregnant with an MCDA pregnancy to be alert for and immediately report to their health-care providers the presence of any symptom described in the present research, which may be associated with any number of complications. We further recommend health-care providers promptly investigate the concerns and physical complaints experienced by women during an MCDA pregnancy.
Footnotes
Authors’ Note
Lauren Nicholas received full IRB approval from D’Youville College’s institutional review board (approval #412016) and Rebecca Fischbein received full IRB approval from Kent State University’s institutional review board (approval #16-169) prior to all research beginning for purposes of this study.
Acknowledgments
The authors would like to acknowledge all of the study participants who made research possible by sharing their time and experiences. Also, the authors would like to thank all of the Facebook pages dedicated to identical twinning that allowed us to recruit participants. Finally, the authors would like to express their gratitude to the Twin to Twin Transfusion Syndrome Foundation, the Fetal Health Foundation, and the Mother of Multiples of America/Mother of Twins.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Lauren Nicholas and Rebecca Fischbein are current or former members of several surveyed networks and organizations.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
