Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040. 1
Primary Objective
Objective MS1.1: Categories of Bone Tumors. Describe examples of bone forming, cartilage forming, and other common bone tumors including the clinicopathological features, radiological features, treatment, and prognosis of each.
Competency 2: Organ System Pathology; Topic MS: Musculoskeletal System; Learning Goal 1: Bone Neoplasia
Patient Presentation
An otherwise healthy, 10-year-old boy presents to his physician with right thigh pain. The pain began as a dull intermittent ache, but it worsened significantly over the past few weeks. It is now constant and severe. The pain is worse in the evening and keeps him awake at night. On a pain scale of 10, the pain is 7/10 in the morning and 9/10 in the evening. He has tried taking nonsteroidal anti-inflammatory drugs (NSAIDs) for relief but that has not helped at all. He has no history of trauma to the affected area. Family history is noncontributory.
On physical examination, his vital signs are temperature 36°C, heart rate 90 beats per minute, blood pressure 110/70 mm Hg, and respiratory rate of 14 breaths per minute. His right thigh is swollen and extremely tender. A mass is palpated. Cardiac, pulmonary, and abdominal examinations are within normal limits.
Diagnostic Findings, Part 1
A conventional radiograph of the right femur demonstrates a cortical-based lesion (Figure 1). An ultrasound demonstrates a heterogeneous mass involving the soft tissues (Figure 2).
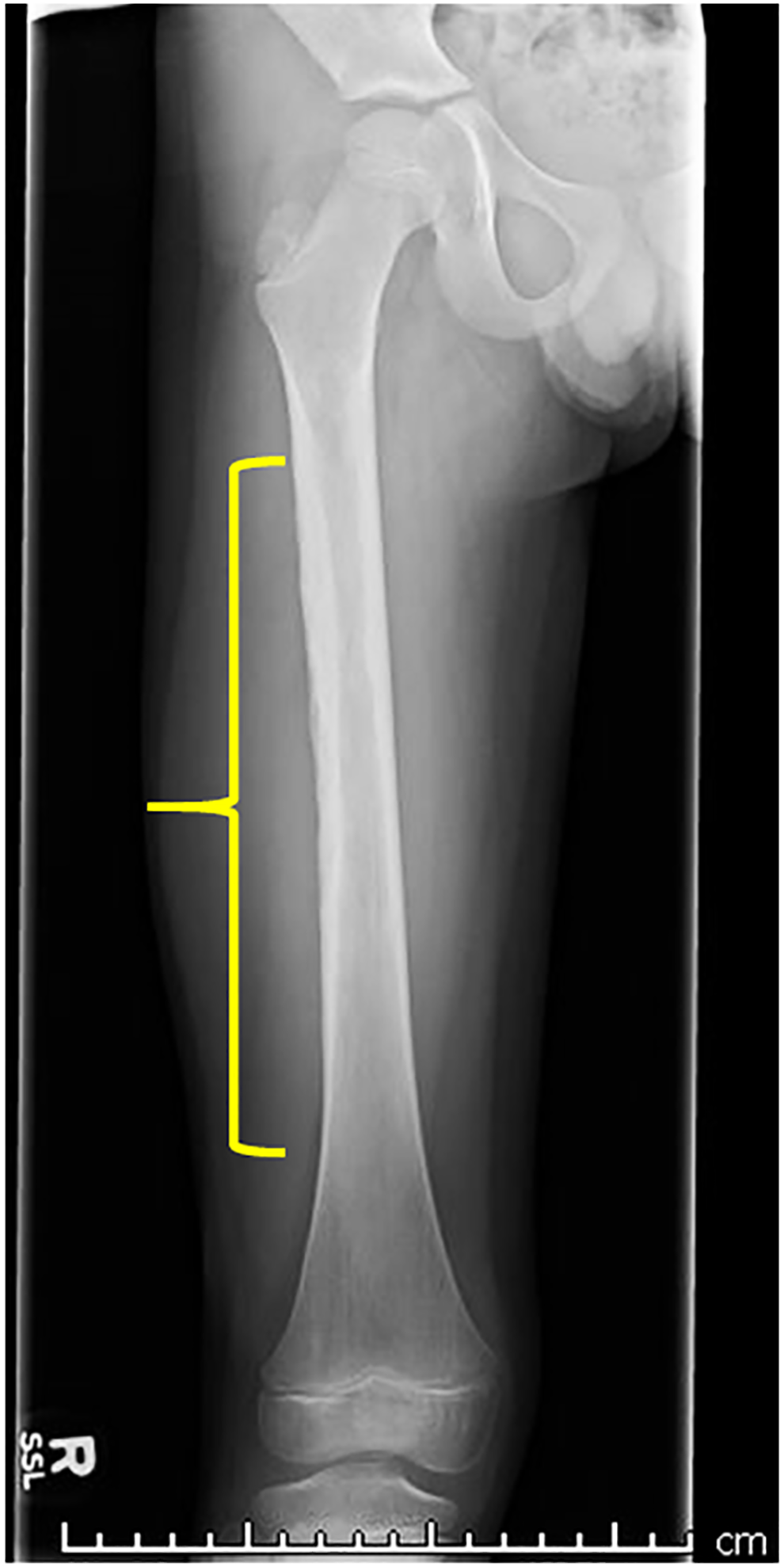
Frontal radiograph of the right femur obtained for swelling demonstrates a permeative cortically based lesion at the lateral cortex of the femur (yellow parenthesis). Wide zone of transition without periosteal reaction. Fullness of the soft tissues is noted laterally.
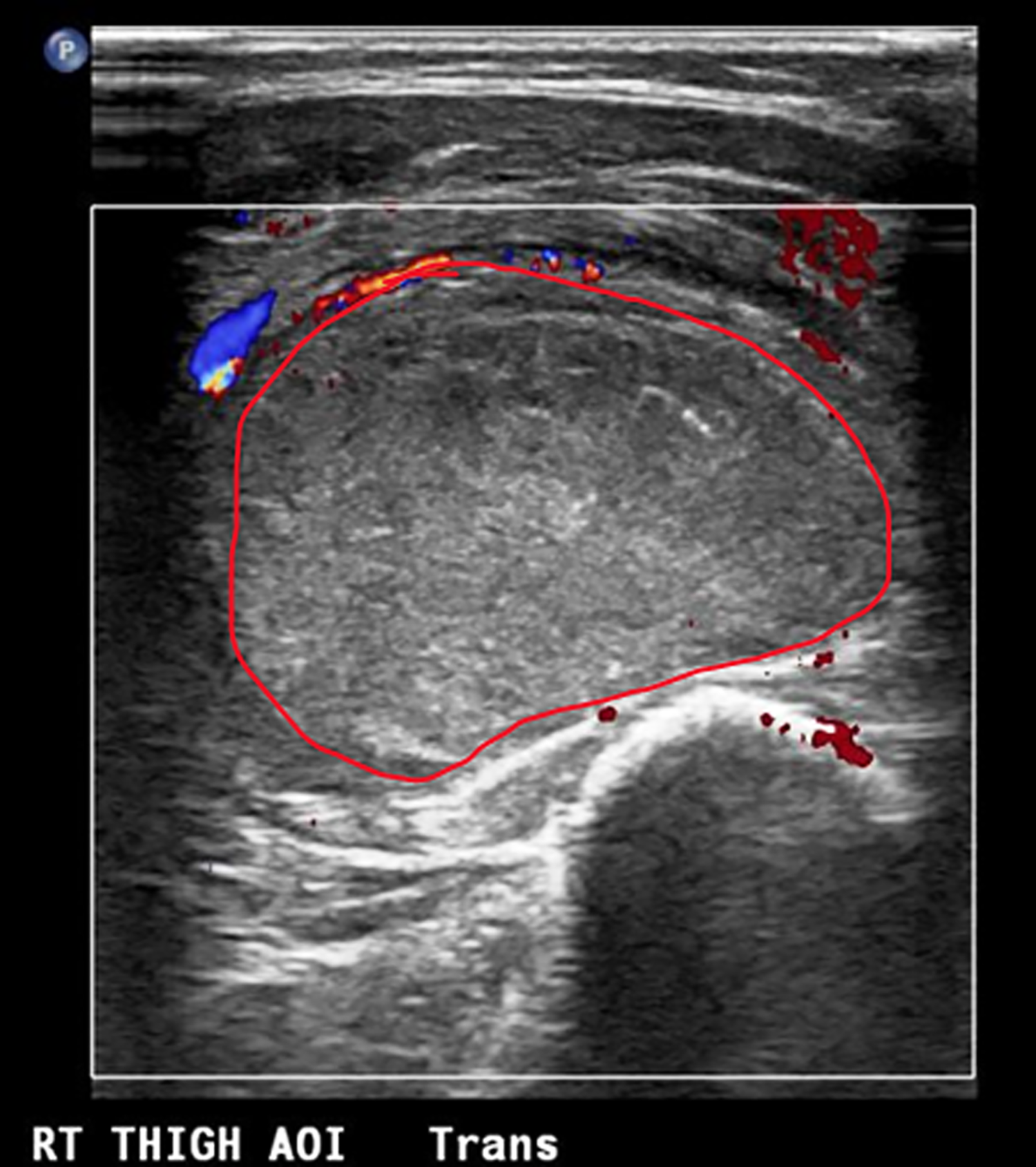
Single sonographic image with color Doppler demonstrates a large heterogeneous solid mass within the soft tissues of the lateral thigh. Internal vascularity is demonstrated. Borders are outlined in red.
Magnetic resonance imaging scan reveals an osseous lesion involving the femoral diaphysis with cortical erosion and a prominent periosteal reaction with soft tissue involvement of the lateral thigh (Figure 3).
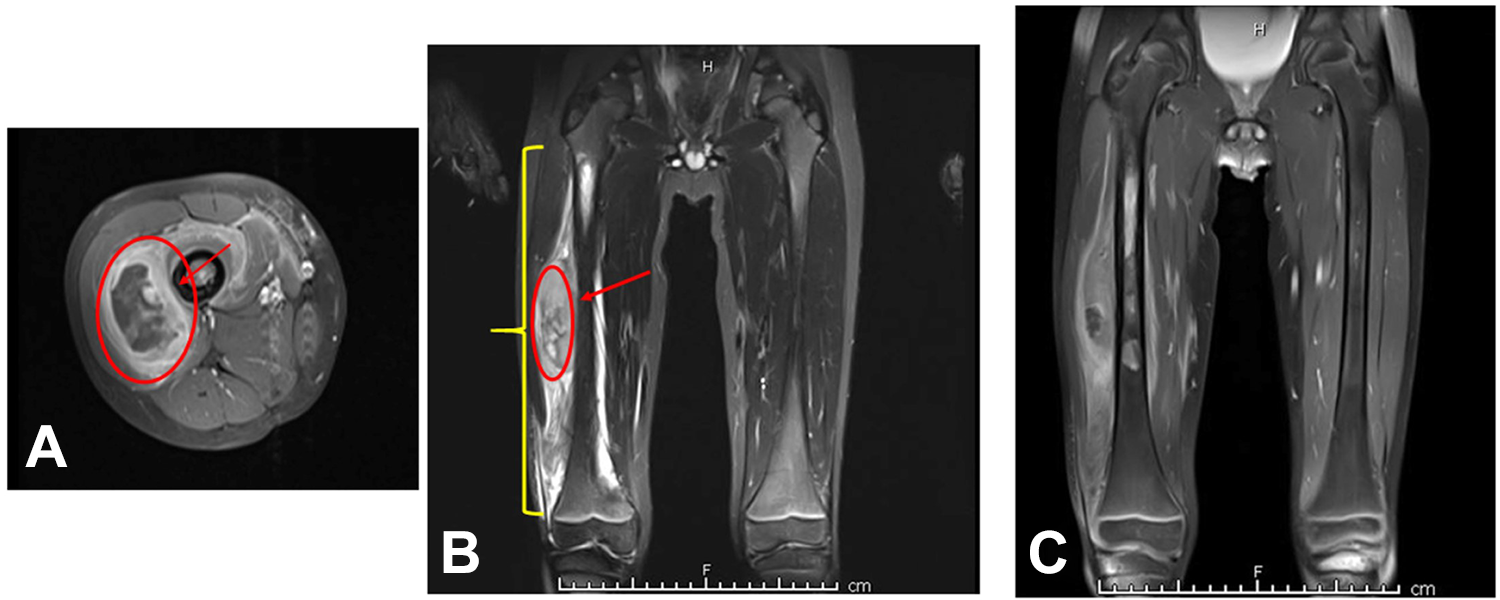
A-C, Magnetic resonance (MR) scan: (A) Axial, (B) coronal STIR, and (C) T1 post contrast images are shown. An osseous lesion spans most of the femoral diaphysis (yellow parenthesis), with cortical erosion (red arrow) and periosteal reaction. There is a large enhancing soft tissue component at the lateral thigh (red circle).
Questions/Discussion Points, Part 1
What Is in the Differential Diagnosis for a Diaphyseal Tumor?
The most common bone neoplasm is by far metastatic disease from other regions of the body. However, this is not likely in a child. Once metastasis has been ruled out, the differential diagnosis for a diaphyseal bone tumor includes osteoid osteoma, enchondroma, fibrous dysplasia, chondrosarcoma, and Ewing sarcoma. These diagnoses are in the differential when a patient presents with an osseous lesion. However, some of the above entities are more likely than others, and multiple factors must be considered. Age is an important factor. Most bone neoplasms occur in the metaphyseal area. Diaphyseal tumors are not nearly as common. A metadiaphyseal location is common for Ewing sarcoma. 2 The most common diaphyseal tumors in patients younger than 40 include Ewing sarcoma, enchondroma, and osteoid osteoma. 3 In older patients, chondrosarcoma is much more likely. 3
What Are Some Examples of Bone Forming, Cartilage Forming, and Other Common Bone Tumors?
In general, benign bone tumors are much more common than malignant ones. A few examples of benign, bone-forming tumors include osteoid osteomas and osteoblastomas. These tumors are less likely as the patient’s pain was not relieved by NSAIDs. Benign, cartilage-forming tumors include chondromas, osteochondromas, and chondroblastomas. Of these, osteochondromas are the most common. 3,4 Some of these tumors are found incidentally on imaging and are not life threatening. The location of the lesion and the involvement of soft tissues rules out most benign bone tumors in the case scenario.
The most common malignant, bone-forming tumor is osteosarcoma. It makes up 20% of all primary bone tumors. The second most common malignant bone tumor regardless of age is chondrosarcoma, it is cartilage forming with no osseous differentiation. 3
Other common bone tumors include giant cell tumor and Ewing sarcoma. Giant cell tumor is composed of osteoclastic multinucleated giant cells distributed in a background of mononuclear cells. 3,5 These tumors are locally aggressive, but they usually do not metastasize. They are also not very common in young individuals. They are more often seen in individuals between 20 and 40 years of age. 5 Ewing sarcoma is a primitive, neuroectodermal tumor composed of uniform, round cells with dark blue nuclei. 3,4,6 It represents 6% to 8% of primary malignant bone tumors, and over two-thirds of cases occur in children and adolescents. 3,5,6 In adults, the most common bone malignancy is distant metastases from other sites.
Diagnostic Findings, Part 2
Based on the clinical presentation and imaging findings, a biopsy was performed. 7 The diagnosis was confirmed. The patient was treated with neoadjuvant chemotherapy followed by a limb-sparing resection followed by adjuvant chemotherapy.
Questions/Discussion Points, Part 2
Describe the Pathological Features Observed in the Biopsy
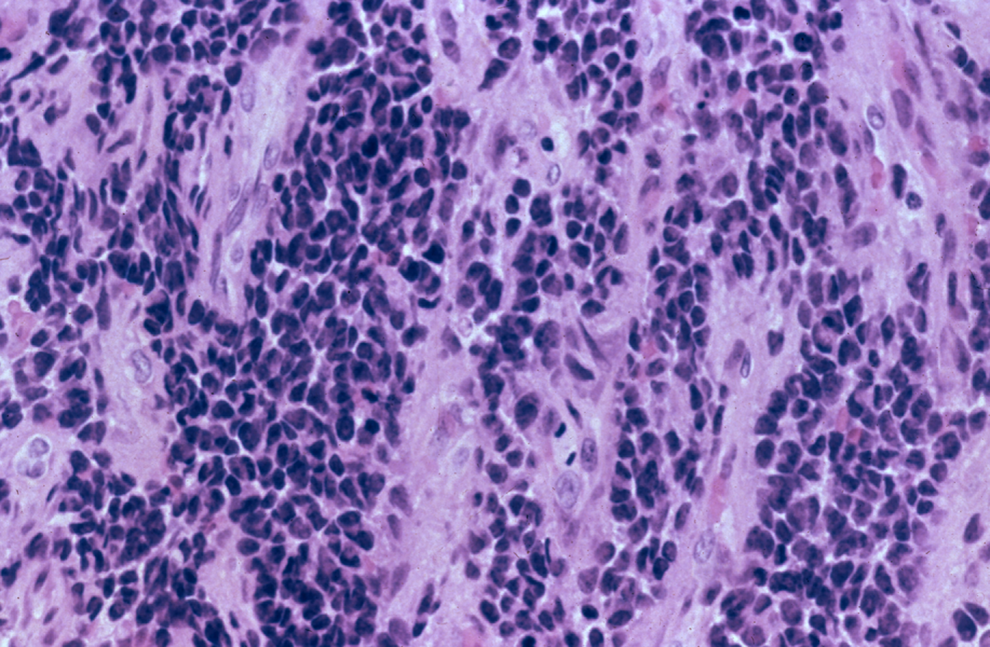
Small, uniform cells with large, dark nuclei and scant cytoplasm accompanied by mitotic figures are present (hematoxylin and eosin [H&E], intermediate power).
Small round blue cells arranged in sheets with indistinct cell outlines are present on histological examination. Mitotic figures are also present. The tumor cells were immunoreactive for cytokeratin and CD99. CD99 showed strong, diffuse membranous staining. Tumor cells were nonreactive for lymphoid markers and Myo D1 (Figure 4).
What Is the Diagnosis Based on Pathological Examination of the Lesion?
The differential diagnosis for Ewing sarcoma includes other tumors of similar origin, lymphoblastic lymphoma, small cell osteosarcoma, mesenchymal chondrosarcoma, and rhabdomyosarcoma. 2,4 The histological and immunochemical features confirm the diagnosis of Ewing sarcoma. The nonreactivity for lymphoma markers and Myo D1 exclude lymphoma and tumors of skeletal muscle differentiation from the differential of small round blue cell tumors. Finally, reverse transcriptase–polymerase chain reaction (RT-PCR) confirmed the presence of the EWSR1/FLI1 fusion gene, which was the final confirmation of Ewing sarcoma.
What Are the Pathologic Features of Ewing Sarcoma?
Ewing sarcoma tumors typically appear as a soft gray or tan-white mass (Figure 5). Tumors typically have focal areas of hemorrhage and necrosis. 3 -6 Necrotic tissue extracted from the intramedullary or subperiosteal tumor may be erroneously interpreted as pus by surgeons. 2,4,5 Microscopically, Ewing sarcoma cells are packed in sheets of small, round cells. Chromatin is evenly distributed. They have scant cytoplasm and are about twice the size of a normal lymphocyte. They are also much more cohesive than lymphocytes. 3 -6 The neoplastic cells are separated into nests by fibrous stands with little interstitial stroma. 3,4 Mitotic activity is variable. Rosettes may be present. 4,5
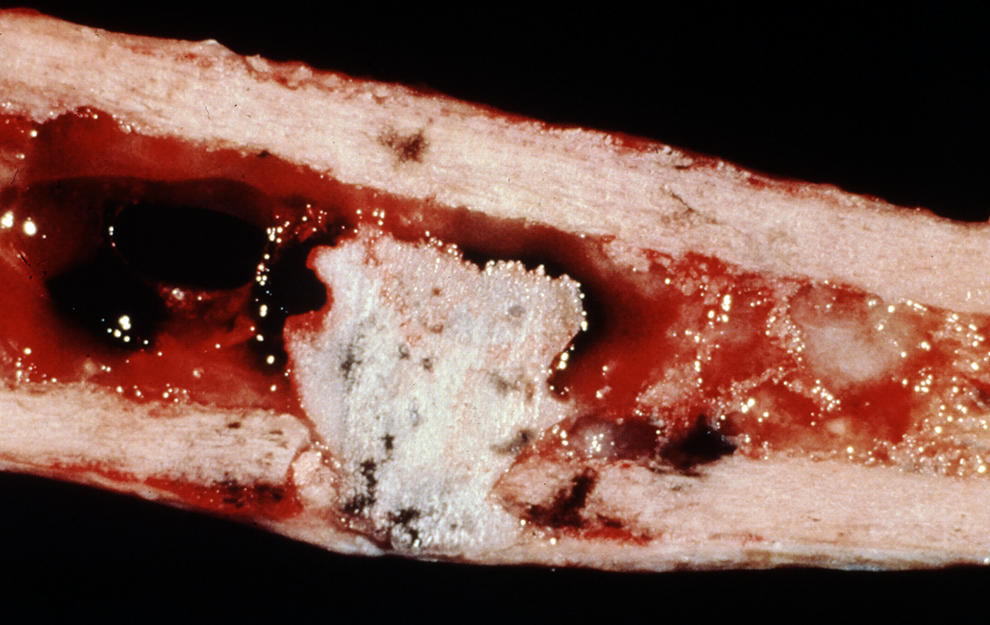
The medullary space within the femoral diaphysis demonstrates a gray tumor with hemorrhage and areas of necrosis. The tumor has penetrated the cortex and periosteum.
The cytoplasm present in these cells contains a substantial amount of glycogen, which can be visualized with periodic acid–Schiff stain. Ewing sarcoma cells also stain positive for FLI-1 and CD99. The EWSR1/FLI-1 fusion upregulates CD99 expression. 3 -6 Tumor cells may also stain positive for neuronal cell markers such as CD57, synaptophysin, S-100, and neuron-specific enolase. 3,4 If the tumor contains Homer-Wright Rosettes (grouping of cells with central fibrillary core), this indicates a greater degree of neuroectodermal differentiation. Homer-Wright Rosettes consist of round groupings of tumor cells gathered around a fibrillary core. 5,8,9
Identify the Populations at Risk for Ewing Sarcoma
Ewing sarcoma is the second most common primary malignant, bone-forming tumor in young patients after osteosarcoma. It typically arises in adolescents. 5,6 About 80% of patients are younger than 20 years; it is very uncommon in patients older than 30 years. 5,6 It is more common in males than females (1.4:1 ratio). 3,5,6 It has a strong predilection for whites. 6,8 It is extremely rare in Asians and African Americans. 2,3,5 Ewing sarcoma accounts for approximately 6% to 10% of primary malignant bone tumors. 5,6 In the Western Hemisphere, the frequency of Ewing sarcoma is 1 to 3 per million per year. 2,10,11 Currently, the only known risk factors for Ewing sarcoma are age younger than 20, male sex, and white race. 3,5,9
Describe the Etiology and Pathogenesis of Ewing Sarcoma
About 85% of Ewing sarcoma tumors contain a reciprocal translocation between chromosomes 11 and 22. 2,5,6,8,11,12 This translocation causes the amino terminus of the EWSR1 protein to fuse with the carboxy terminus of the FLI-1 protein. The FLI-1 gene encodes a transcription factor. When the 11;22 translocation occurs, it creates a fusion protein known as EWSR1/FLI-1. 3 -6 About 10% of Ewing sarcoma tumors arise from a 21;22 translocation. 2,4,8,10 This leads to the creation of an EWSR1/ERG gene fusion protein. 3,8,11 Other cases result from other fusion gene products containing part of the EWSR1 gene and one of the ETS family of transcription factors. 6,8,11 These gene products create aberrant transcription factors that alter target genes. 3,5,8,11 Even though their exact targets are yet to be fully determined, these translocations are extremely important when confirming the diagnosis of Ewing sarcoma. Translocation identification by cytogenetics or molecular diagnostics using fluorescence in situ hybridization or RT-PCR, respectively, are used for diagnosis. 8
The origin of Ewing sarcoma cells remains unclear, but the most likely candidates include neuroectodermal cells and mesenchymal stem cells. 5,6,8 The tumor typically arises in the medullary cavity of long bones. It invades the cortex, periosteum, and surrounding soft tissue. 5 Ewing sarcoma has also been known to arise in the flat bones of the pelvis and ribs. Other possible sites of involvement include the skull, vertebra, scapulae, and bones of the hands and feet. 6 A small minority of cases are extra skeletal arising in soft tissue. 4,6 Ewing sarcoma is an aggressive tumor displaying early hematogenous metastasis to the lungs, brain, and other bones, particularly the skull. 4
Describe the Common Clinical Features Associated With Ewing Sarcoma
Patients with Ewing sarcoma usually present with pain and swelling localized to one area. They usually notice mild pain at first, but it worsens rapidly. The area will be extremely tender to palpation. A mass can often be palpated. The mass will be firmly attached to the underlying bone, and the area will likely be swollen and erythematous. 2,3,5,6 The pain is often worse at night and exacerbated by exercise. It may also limit joint motion. When present, constitutional symptoms include unintentional weight loss, fatigue, anemia, and fevers. These symptoms are present in about 10% to 20% of patients at the time of diagnosis. 5,6 Patients may also present with pathologic fractures. Ewing sarcoma may be mistaken for osteomyelitis, clinically due to bone pain, fever, and leukocytosis. 2,4 -6
Conventional radiographs will show a lytic tumor extending into the surrounding soft tissue with an aggressive periosteal reaction. The mass is often described as having an onion skin appearance on plain film. 2,3
Compare the Variants of Ewing Sarcoma
Neoplasms in the Ewing sarcoma family of tumors include Ewing sarcoma of bone and extraosseous Ewing sarcoma. Ewing sarcoma of bone makes up about 80% to 90%, and it is most often found in long bones. 12 It has also been seen in the pelvis, ribs, vertebrae, skull, and hands and feet. 9 Extraosseous Ewing sarcoma seen in approximately 20% of patients with Ewing sarcoma includes tumors that grow in the soft tissues surroundings bones, most often the cartilage. 12 Even though these tumors grow in different areas of the body, they share similar histology, immunohistochemical characteristics, and chromosomal translocations. In addition to these microscopic and genetic similarities, these tumors also show similar clinical behavior. They all have a peak incidence between 10 and 20 years of age. They are all extremely rare after age 30. They all tend to metastasize early to the lungs and bone. 9 Finally, they are all very sensitive to radiation and chemotherapy. Due to these similarities, they are all classified as part of the Ewing sarcoma family of tumors. 2,4,9,12
Describe the Possible Treatments of Ewing Sarcoma and Associated Outcomes
Ewing sarcoma family of tumors is extremely aggressive, and historically, survival rates have been low, particularly for those with advanced disease. Fortunately, survival rates have increased significantly in the past 40 years due to chemotherapy which is a mainstay of treatment and radiation. 3,4 , Today, patients are treated with neoadjuvant chemotherapy followed by surgical resection or radiation therapy. 2,4 -7 The neoadjuvant chemotherapy was a very important addition to the treatment regimen because it is now believed that nearly all patients have subclinical metastatic disease at the time of diagnosis. 8 The most important prognostic factor is metastatic disease at the time of diagnosis. 12 The 5-year survival rate today in those without metastatic disease treated with multimodal therapy is greater than 70%. 4,5,9,11 -13 In contrast, the 5-year survival rate in patients with overt metastatic disease at diagnosis is less than 30%. 9,11,12 The amount of chemotherapy-induced necrosis of the tumor is an extremely important prognostic factor. 2,4 -6 Other prognostic factors include stage, tumor size, and proximal primary location. 4,9,12
Teaching Points
Ewing sarcoma is a primary bone tumor of neuroectodermal or mesenchymal stem origin. It is most commonly identified in the diaphysis or metadiaphysis of the distal femur.
Grossly, the tumor appears gray or tan-white with areas of hemorrhage and necrosis. On histology, Ewing sarcoma tumors are composed of small, round cells with scant cytoplasm.
Ewing sarcoma is a tumor of childhood. The average age at diagnosis is 15, and it is slightly more common in males and overwhelmingly more common in whites.
Most cases of Ewing sarcoma result from a translocation between chromosomes 11 and 22, creating an EWSR1/FLI1 fusion protein. This protein acts as an aberrant transcription factor.
Ewing sarcoma arises in the medullary cavity of long bones and invades the cortex, periosteum, and surrounding tissue.
Clinical signs of Ewing sarcoma include pain localized to the tumor site. Swelling and erythema may be present as well as constitutional symptoms such as fever and unintentional weight loss.
Conventional radiographs will show a destructive lesion in the distal femur with layers of reactive bone and involvement of surrounding soft tissues.
Ewing sarcoma of bone is part of a broader category known as Ewing sarcoma family of tumors. Ewing sarcoma family includes Extraosseous Ewing sarcoma.
Current treatment of Ewing sarcoma involves neoadjuvant chemotherapy followed by limb-sparing resection or radiation therapy. Chemotherapy is an essential component of treatment due to the presence of subclinical metastases in 80% to 90% of patients.
The overall 5-year survival rate for Ewing sarcoma without metastatic disease treated with multimodal therapy today is more than 70%.
Osteoid osteomas and osteoblastomas are common benign bone tumors. Aspirin classically relieves the pain from an osteoid osteoma.
Benign, cartilage-forming tumors include chondromas, osteochondromas, and chondroblastomas. Of these, osteochondromas are the most common.
The most common malignant, bone-forming tumor is osteosarcoma.
Footnotes
Author’s Note
Figures 4 and ![]() were obtained during the scope of US government employment for Dr Conran.
were obtained during the scope of US government employment for Dr Conran.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
