Abstract
Keywords
Primary Objective
Competency 2: Organ System Pathology; Topic: Skin (SK); Learning Goal 5: Skin Neoplasia.
Secondary Objective
Competency 1: Disease Mechanisms and Processes; Topic: Neoplasia (N); Learning Goal 1: Genetic Basis of Neoplasia
Patient Presentation
A 61-year-old white man presents to his primary care physician with concerns about a dark lesion on his upper right arm. He reports that he has always had many freckles, including this one, but believes it may have changed in shape and size over the past few months. He denies any pain, itchiness, or dryness associated with the lesion. He also denies other systemic symptoms such as fever, rash, and shortness of breath. He reports that when he was younger he worked for many summers as a lifeguard. His mother had a melanoma removed at age 65, and he has no other family history of cancer.
Diagnostic Findings, Part 1
The patient is fair-skinned and appears healthy. Physical examination reveals numerous small nevi and lentigines on his back, chest, and arms. The lesion of concern on his right arm is noticeably larger and darker than the other nevi. There are no other suspicious cutaneous lesions and no palpable lymphadenopathy. The rest of the physical examination is unremarkable.
Questions/Discussion Point, Part 1
What Is the Differential Diagnosis Based on the History and Physical Examination?
The differential diagnosis for a pigmented lesion is broad and includes nevus, lentigo, dysplastic melanocytic nevus, melanoma, seborrheic keratosis, pigmented basal cell carcinoma (BCC), and pigmented actinic keratosis. However, a pigmented lesion that is changing in size and shape and appears different than other lesions is particularly concerning for a melanoma.
The other 2 most common neoplasms of the skin include squamous cell carcinoma and BCC. Basal cell carcinoma arises on areas of skin subjected to chronic sun damage such as the face and head. Unlike melanoma it is typically an amelanotic lesion. The classic clinical features are a pink, pearly papule with raised or “rolled” borders and telangiectasias. Squamous cell carcinoma is also often seen in areas of sun exposure and typically appears as an erythematous lesion with scale or ulceration. 2
What Are the Next Steps in Management of This Patient?
The next step in management is a full body skin examination to inspect the patient’s other pigmented lesions and for context as to whether the current lesion of interest looks different from other lesions. Further workup of suspicious lesions includes a closer visual inspection followed by a dermoscopic examination, a noninvasive method that increases visualization of the skin by utilizing visible light to illuminate and magnify microscopic structures not visible to the naked eye. Sensitivity and specificity of thorough visual examination of the skin has been reported to be as high as 92% and 80%, respectively. 3
Diagnostic Findings, Part 2
The patient’s lesion in vivo is shown in Figure 1.
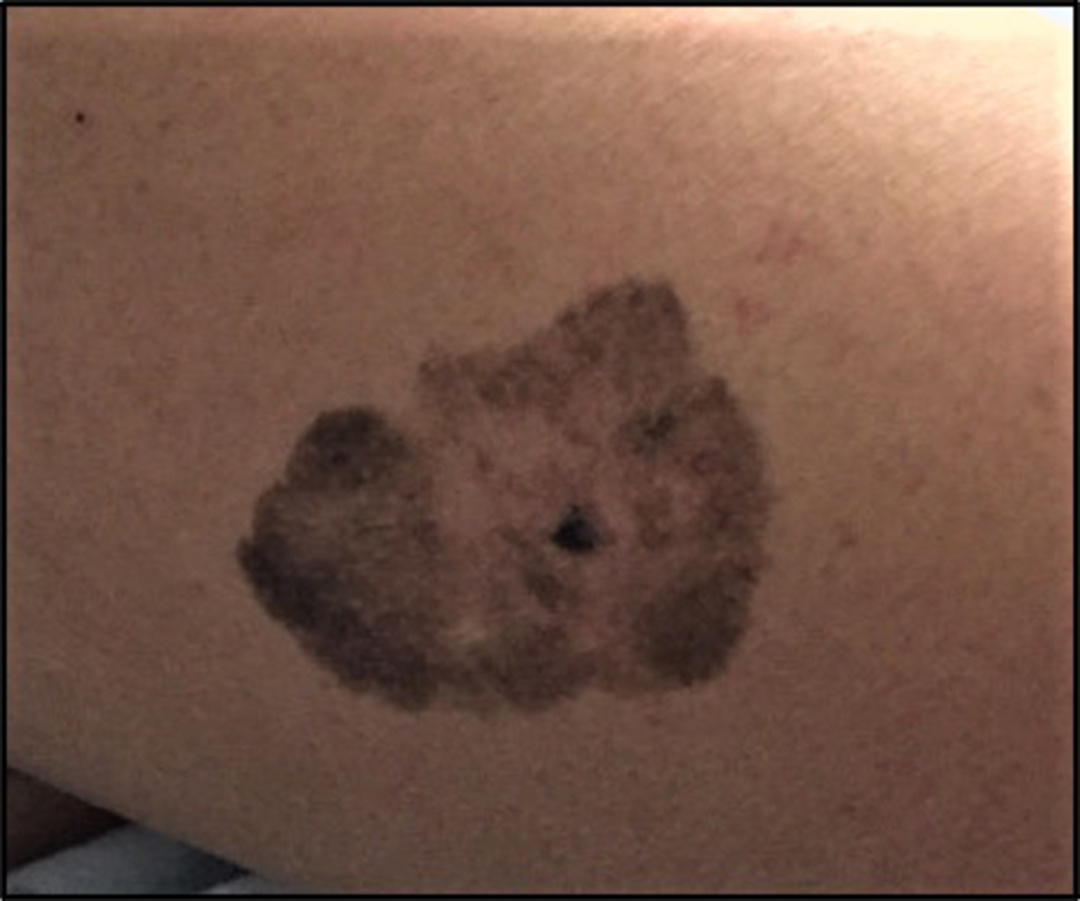
Pigmented lesion in vivo: This pigmented lesion is asymmetrical and has irregular borders and color variation throughout. The lesion measures 3.5 cm (ruler not shown).
Questions/Discussion, Part 2
What Does the Visual Examination Show?
The lesion is dark brown, asymmetrical, and has irregular borders and color variation throughout. The lesion measures 3.5 cm (ruler not shown in image).
Melanomas can be described using the ABCDEF checklist, a modification of the original ABCDE checklist. “ABCDEF” describes 6 variables that are concerning for malignancy: asymmetry, border irregularity, color, diameter, evolution, and “funny-looking” or “ugly-duckling” sign. In an asymmetric lesion, one half does not match the other. Border irregularity shows ragged, notched, or blurred borders. The color (pigmentation) is concerning if it is not uniform, with shades of brown, tan, and black present. The diameter of the lesion is concerning if greater than 6 mm. Evolution of the lesion describes changes in the size, shape, surface, symptoms (such as itching, dryness, or bleeding), or color, any of which are concerning. A history of a changing mole is an important sign that carries a significant relative risk of melanoma of about 400%. 4 “Funny looking” or the “ugly duckling” sign means the lesion does not look like the rest of the patient’s nevi. 5
In this patient, the lesion shows all of these characteristics: it is asymmetric, has irregular borders, variable color, diameter of 35 mm, and per report has changed over time. In addition, the lesion is noticeably darker and larger than the patient’s other nevi.
Another concerning physical finding is called the “little red riding hood sign”: a ring of erythema surrounding a pigmented lesion. This sign can be a clinical clue to an atypical melanocytic process. The ring of erythema corresponds to an inflammatory response typically located around the periphery of the melanoma and is sometimes present on histopathologic examination of the melanoma.
What Is the Next Step for This Patient?
Although the clinical setting and gross appearance of a lesion may suggest melanoma, definitive diagnosis requires histologic confirmation. The next step in the workup of a suspicious pigmented lesion is to obtain a full-thickness excisional biopsy to the subcutaneous fat with 1 to 2 mm lateral margins. 6 A full-thickness biopsy permits full visualization of the entire lesion, while a superficial or partial biopsy can lead to diagnostic uncertainty and staging errors.
Diagnostic Findings, Part 3
The patient undergoes a full-thickness excisional biopsy (Figure 2A-D).
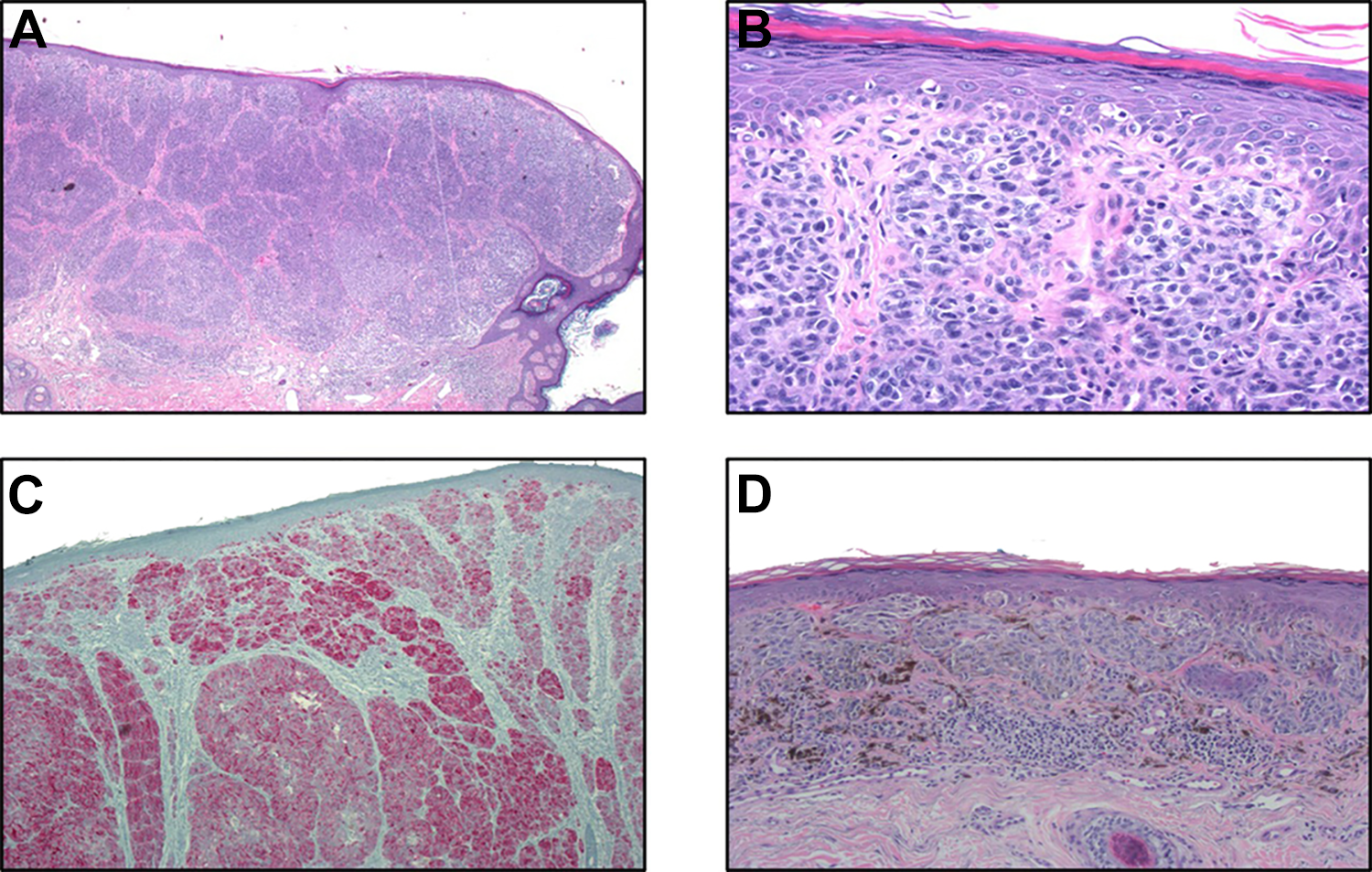
Biopsy: At low power, the biopsy shows cells invading the dermis in sheets and nests (A, H&E, ×20). At higher power, the atypical cells have irregular nuclei and prominent nucleoli (B, H&E, ×200). The cells are immunoreactive for Mart-1, consistent with melanocytic differentiation (C, Mart-1, ×40). A peripheral portion of the lesion shows nests of atypical pigmented cells (D, H&E, ×100).
Questions/Discussion, Part 3
What Are the Biopsy Findings?
The biopsy shows atypical cells present in the basal layer of the epidermis and invading the dermis in sheets and nests. The cells have irregular nuclei and prominent nucleoli. The cells are immunoreactive for Mart-1, confirming they are melanocytes. At the edges of the lesion, there are areas showing large nests of atypical pigmented cells.
What Is the Diagnosis?
This lesion was diagnosed as malignant melanoma.
What Are the Diagnostic Histopathologic Features of Melanoma?
The hallmark cells are atypical melanocytes, with hyperchromatic nuclei, high nuclear to cytoplasmic ratio, mitotic figures, and pleomorphism. Melanomas also have characteristic architectural features, including a broad asymmetric lesion, irregularly spaced nests of melanocytes that are not confined to the tips of rete ridges, a confluence of melanocytes between nests within the epidermis, Pagetoid spread within the epidermis (in which melanocytes from the lower levels of the epidermis begin to migrate upward to the superficial levels of the epidermis), lack of maturation of melanocytes as they move deeper in the dermis, and mitoses within the dermal component. 7 In some cases, immunohistochemical stains for melanocytes to include Melan-A, S-100 protein, Sox10, and HMB45 are used to facilitate the diagnosis. 8
What Are the Important Prognostic Features of Melanoma?
The most important prognostic factor is the Breslow depth, the depth of invasion of the malignant melanocytes as measured from the top of the granular layer within the epidermis to the base of the tumor, or if the lesion is ulcerated, from the base of the ulcer to the deepest malignant melanocyte. A deeper Breslow depth is associated with a worse prognosis. Additional poor prognostic histopathologic factors include mitoses, regression, ulceration, and absent or nonbrisk tumor-infiltrating lymphocytes. Male gender and tumor location somewhere other than an extremity also portend a worse prognosis. 9
List and Describe the Most Common Subtypes of Melanoma
The most common subtypes of primary cutaneous melanoma are superficial spreading (60%), nodular (20%), lentigo maligna (10%), and acral lentiginous (ALM; 5%). Most melanomas are diagnosed in patients in the fourth and fifth decade of life, while lentigo maligna and acral subtypes are more common in the sixth and seventh decade of life. The most common locations of melanomas are the head, trunk, and limbs; they most frequently present on the trunk in men and on the lower extremities in women. 6
Superficial spreading melanoma (SSM) is the most common variant in fair-skinned individuals and typically presents with many of the critical ABCDEF features. Nearly half arise from a preexisting nevus. As the name implies, the distinguishing feature of SSM is the initial horizontal growth limited to the epidermis, followed by vertical growth which can cause formation of a papule or nodule. Up to 67% show signs of regression in part of the lesion.
Nodular melanoma is characterized by a de novo vertical growth phase without an antecedent horizontal growth phase. This subtype has a worse prognosis than SSM, likely due to the advanced Breslow depth.
Lentigo maligna melanoma is seen more commonly in older patients, with a median age of 68. The precursor lesion to this variant is lentigo maligna, also known as melanoma in situ, with an estimated risk of transformation at 3.5% per year. 10 The key feature for this subtype is that it commonly develops in areas of the skin that have been subjected to chronic sun damage.
Acral lentiginous melanoma is unique in that it more commonly appears on the palms and soles, rather than the common sun exposed areas. Acral lentiginous melanoma is the most common melanoma seen in more darkly pigmented individuals, usually of African or Asian descent. Additional less common variants of melanoma include amelanotic melanoma, mucosal melanoma, ocular melanoma, and malignant blue nevus.
What Are the Risk Factors for Melanoma?
Much like other cancers, the main risk factors for melanoma include both hereditary and genetic factors, as well as environmental exposures. Fair skin, white race and a positive family history of skin cancer are important risk factors for melanoma. Additional environmental risk factors such as UV-B rays and tanning bed exposure can play a role in development of many skin lesions. UV light exposure is an important modifiable risk factor in the development of melanoma 11 ; intermittent high-intensity UV exposure before age 35 and a history of skin-damaging sunburns throughout life has the potential to double the risk of developing melanoma. 12,13 UV radiation is a factor in both the initiation and progression of melanoma, most likely due to DNA damage and subsequent errors in repair. 14
There is a strong link between a positive family history of melanoma and personal risk of developing melanoma. About 10% of melanoma patients have a positive family history, which may be due to similar environmental exposures or rarely an inherited familial syndrome.
15
Having one first-degree relative diagnosed with melanoma corresponds to a 1.7 times increase in risk, while having 2 first-degree relatives increases the risk 9-fold.
11
There are many familial syndromes that can increase the risk of developing melanoma such as xeroderma pigmentosum,
Describe the Pathogenesis of Melanoma, Including Precursor Lesions and Molecular Events
Dysplastic nevi are a precursor lesion to melanoma. These lesions show some atypical features beyond usual benign nevi but are insufficient for a diagnosis of melanoma. Not all dysplastic nevi progress to melanoma, and it is unclear which ones are more likely to do so. Approximately 20% of melanomas arise from dysplastic nevi, while the majority of melanomas arise de novo. Atypical and benign melanocytic nevi can be precursor lesions to melanoma, but the progression seems to be unclear and the transformation rate has been shown to be very low. 18
Mutations in BRAF (a proto-oncogene) and/or
In contrast to activating mutations in proto-oncogenes, additional inactivating mutations in tumor suppressor genes are usually evident in melanoma.
Describe General Principles of Treatment and Management of Melanoma
Management of melanomas varies depending on the stage of the lesion, determined by the Breslow depth. For low-staged melanomas without signs of metastasis, the main treatment is re-excision with appropriate margins that are based on the original stage. For example, an in-situ lesion requires smaller margins on re-excision compared to a 3-mm thick invasive melanoma. The goal of uniformly removing additional cells from the areas around the primary lesion is to remove all of the cancerous cell and prevent local recurrence of the disease.
Sentinel lymph node biopsy may be performed for staging and prognostication for lesions greater than 1 mm thick. Radioactive material combined with blue dye is injected into the primary area of concern, and the primary draining lymph nodes (usually 1 or 2) are determined by monitoring the radioactivity and color changes. Only these select or “sentinel” nodes are removed.
For melanomas with nodal or distant metastasis, systemic therapies are first-line options. Most of the available therapies target proteins like BRAF in the MAPK signaling cascade or are immune system modulators. Monoclonal antibodies like vemurafenib bind to the V600E BRAF and inhibit its constitutive activity limiting MAPK cascade activity. Ipilimumab and nivolumab target CTLA4 and PD1 proteins on the surface of T-cell lymphocytes, respectively, preventing the tumor cells from inactivating our adaptive immune response. Another systemic therapy is interferon alpha, which is a type I interferon produced by the immune system to increase the body’s ability to survey and detect abnormal cellular activity. Specific mechanisms like decreasing gene expression and protein synthesis, increasing MHC surface expression and increased activation of NK cells help the immune system to detect and destroy specific cells. 9
Are There Disparities Relevant to Melanoma in People With Different Races/Ethnicities?
People with fair skin are considered at highest risk for melanoma due to the protective nature of melanin against UV damage, and white people have the highest incidence of primary cutaneous melanoma relative to people of color: age-adjusted incidence rates are 21.6 per 100 000 among non-Hispanic whites versus 4.5 per 100 000 among Hispanics and 1.0 per 100 000 among blacks. 22 However, white people also have better survival outcomes, as well as better outcomes at each stage, relative to nonwhite people. 23
There are differences in the prevalence of melanoma subtypes as well. A greater proportion of melanomas diagnosed among non-Hispanic black people are ALM than are melanomas diagnosed among non-Hispanic whites. Acral lentiginous melanoma is associated with poor survival rates relative to other types. In addition, non-Hispanic black people are often diagnosed with melanoma at later stages. 24
These disparities are likely multifactorial. They may in part be due to the difficulty and potentially the lower likelihood of easily detecting lesions on the acral skin. It has also been postulated that there is a false perception among both the general public and medical community that people of color have essentially no risk of melanoma (as opposed to a lower risk). This may have dangerous consequences, including inadequate screening and counseling by the medical community as well as lack of risk-avoidant behaviors by patients. 22
Teaching Points
Melanoma is a malignancy of melanocytes and is the fifth most common cancer in both men and women in the United States.
Classic clinical characteristics of melanoma are described using “ABCDEF”: Asymmetry, Border (irregular), Color (variegated), Diameter (large), Evolution in size and/or shape, and “Funny looking” (different than the patient’s other pigmented lesions).
The 4 most common types of primary cutaneous melanoma are superficial spreading, nodular, lentigo maligna, and acral lentiginous (ALM).
Fair skin, white race, and family history of melanoma are the most important nonmodifiable risk factors for melanoma.
UV light exposure is an important modifiable risk factor; patients should be counseled to minimize UV light exposure to reduce their risk of skin cancer.
Dysplastic nevi are a precursor lesion in many cases of melanoma. However, many melanomas arise de novo, and not all dysplastic nevi become malignant.
Depth of invasion (“Breslow depth”) is the single most important prognostic factor when evaluating a melanoma.
Treatment for low-stage melanomas is excision with appropriate margins, while systemic therapies such as immune system modulators are effective in higher staged melanomas.
There are racial disparities relevant to melanoma: White people have the highest prevalence of primary cutaneous melanoma overall, while ALM make up a greater proportion of the melanoma cases in non-Hispanic black people. Melanomas are often diagnosed at higher stages in non-Hispanic black people, which may contribute to their worse survival outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article processing fee for this article was funded by an Open Access Award given by the Society of ‘67, which supports the mission of the Association of Pathology Chairs to produce the next generation of outstanding investigators and educational scholars in the field of pathology. This award helps to promote the publication of high-quality original scholarship in Academic Pathology by authors at an early stage of academic development.
