Abstract
Keywords
Primary Learning Objective
Competency 2: Organ System Pathology; Topic: Skin (SK); Learning Goal 3: Immune-Related Disorders of the Skin.
Patient Presentation
A 38-year-old Caucasian male presents to the dermatologist concerned about “white” spots on his arms and legs. The spots have been present for many years and are asymptomatic. The patient has no other relevant past medical history and takes no medications.
Diagnostic Findings, Part 1
Physical examination shows symmetrically located, well-demarcated 6- to 10-mm depigmented macules and patches on his forearms, dorsal hands, and distal legs. The lesions show no surface scale. No other lesions are seen on full-body examination. Potassium hydroxide (KOH) preparation test is negative for fungal organisms.
Questions/Discussion Points, Part 1
What Is Your Differential Diagnosis Based on the Clinical Presentation and History?
The patient is presenting with a depigmenting disorder of his skin. The differential diagnosis would include vitiligo, postinflammatory hypopigmentation, tinea versicolor, and idiopathic guttate hypomelanosis (IGH). Vitiligo is an asymptomatic eruption of depigmented macules and papules, often symmetrically distributed. Patients may or may not have a family history of vitiligo or a history of autoimmune disease. Vitiligo is the leading differential diagnosis in this case. Postinflammatory hypopigmentation is a result of a preceding inflammatory lesion or eruption, disrupting the melanin pigment in the epidermis, and causing pigment dropout into the dermis. The clinical result is that of a pigment change. Given the lack of history of a preexisting eruption, a postinflammatory process is unlikely. Tinea versicolor is a cutaneous fungal infection and can cause pigment alteration in the skin. A cutaneous fungal infection should still be considered, although the lack of scale on examination and negative KOH prep argues against this. Idiopathic guttate hypomelanosis is a sun-related condition, often seen in older adults, appearing as small 1- to 3-mm macules of depigmentation on actinically damaged skin. The lesions in this patient are larger, though are on sun-exposed areas. A skin biopsy can help to differentiate IGH from vitiligo as melanocytes are retained in IGH.
Diagnostic Findings, Part 2
A Skin Biopsy of the Dorsal Hand Is Performed for Diagnosis and Is Shown in Figure 1. How Would You Describe the Microscopic Findings in the Hematoxylin and Eosin–Stained Skin Biopsy?
Junctional melanocytes are not readily identified. There is overlying orthokeratosis. There is no appreciable inflammation in the dermis. Overall, these features are nonspecific and are of near “normal” skin for site.
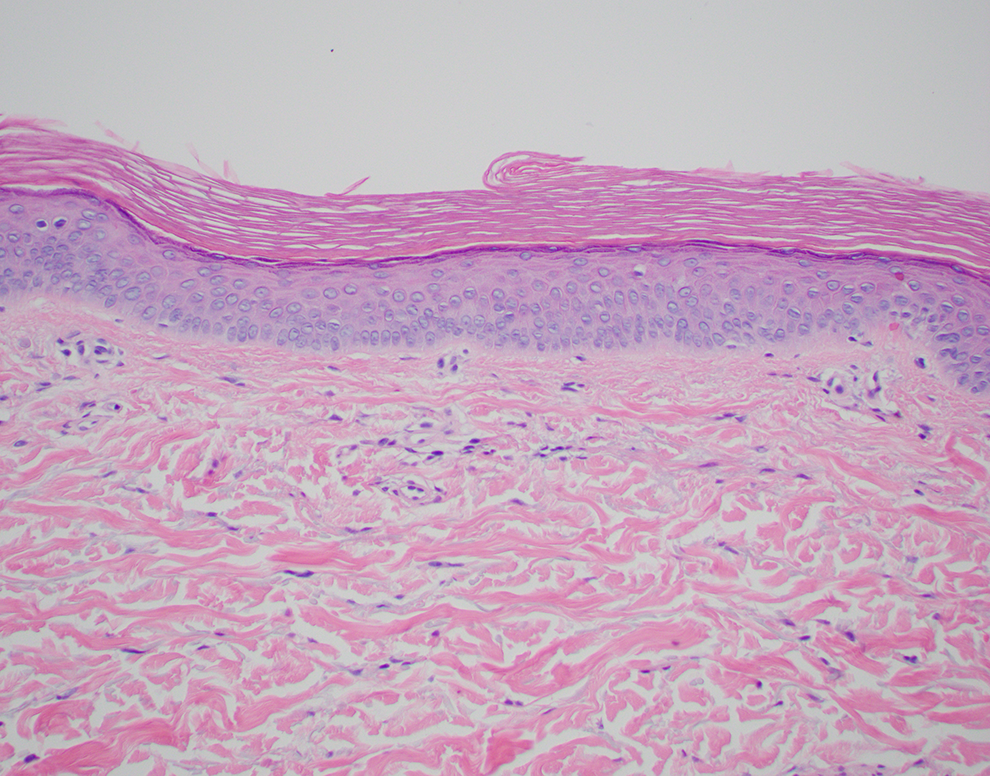
The patient’s skin biopsy from the dorsal hand. Melanocytes are not readily identifiable along the junction. There is no appreciable inflammation within the dermis (hematoxylin and eosin, ×20).
A panel of stains is performed to further characterize the process, including to detect melanin pigment, identify any fungal infection present, and highlight the melanocytes within the epidermis.
A Fontana Masson Stain Is Performed and Is Shown in Figure 2. What Does a Fontana Masson Stain Detect? How Would You Interpret the Fontana Masson Stain on This Case?
A Fontana Masson stains melanin pigment. Any cell containing melanin will stain black. The Fontana Masson stain in this case showed no positive staining (no black melanin pigment seen). The inset photo shows an example of “normal” skin with basilar pigment seen.
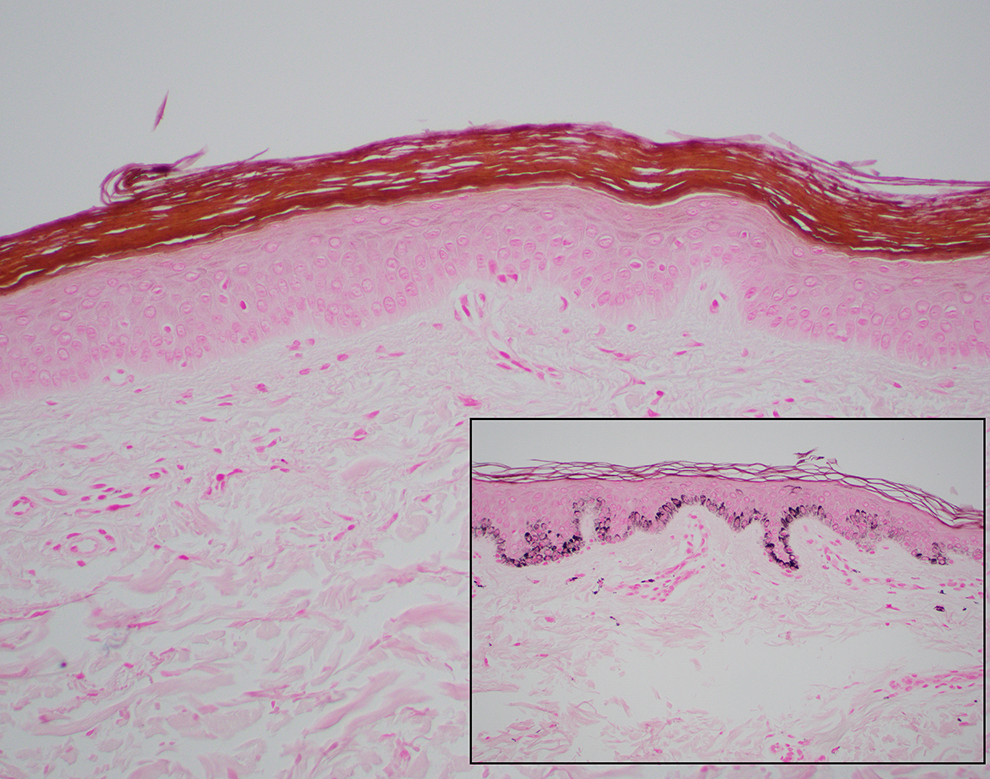
The patient’s skin biopsy stained with Fontana Masson, a melanin pigment stain, showing no appreciable melanin in the epidermis or dermis. Inset, A Fontana Masson stain of “normal” skin showing melanin pigment (black cytoplasmic pigment) in keratinocytes along the dermal–epidermal junction (Fontana Masson, ×20).
A Periodic Acid–Schiff Stain Is Done and Is Shown in Figure 3. Why Was a Periodic Acid–Schiff Stain Performed? How Would You Interpret the Periodic Acid–Schiff Stain on This Case?
A periodic acid–Schiff (PAS) is performed in this case to detect fungal organisms present in the stratum corneum (skin surface). The PAS stain in this case was negative—there were no fungal organisms seen. A positive example of a PAS stain is shown in Figure 4 to illustrate superficial fungal hyphae and yeast forms as would be found in tinea versicolor.
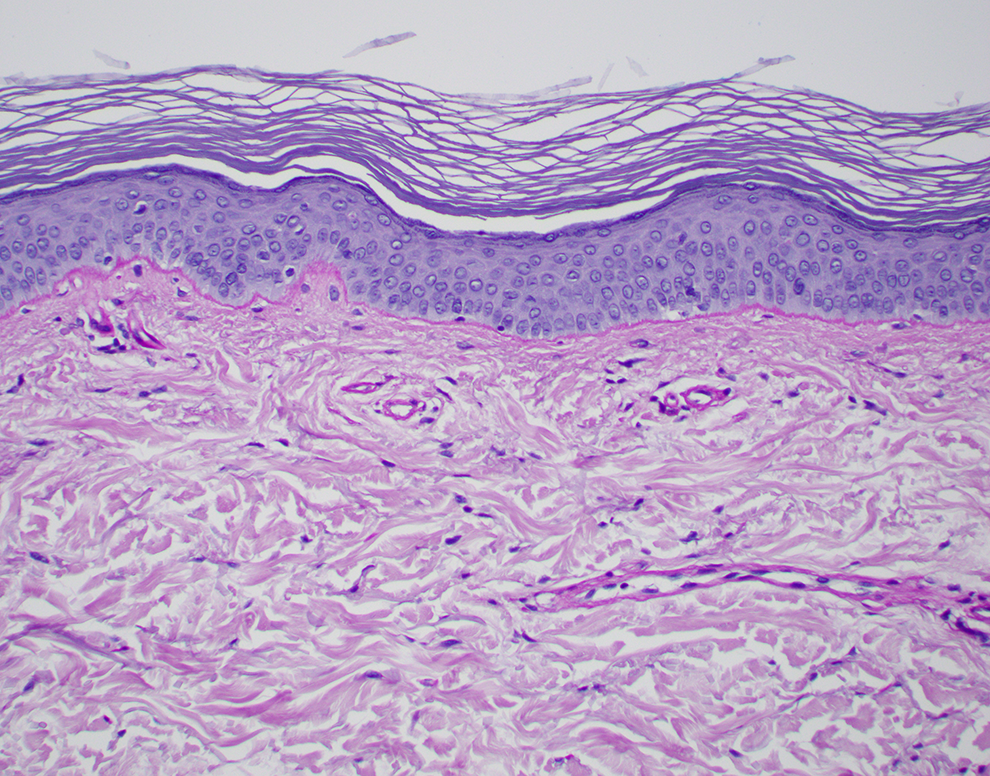
The patient’s skin biopsy stained with period acid–Schiff (PAS), a fungal stain, showing no evidence of fungal hyphae in the stratum corneum, the uppermost layer of the epidermis (PAS, ×20).
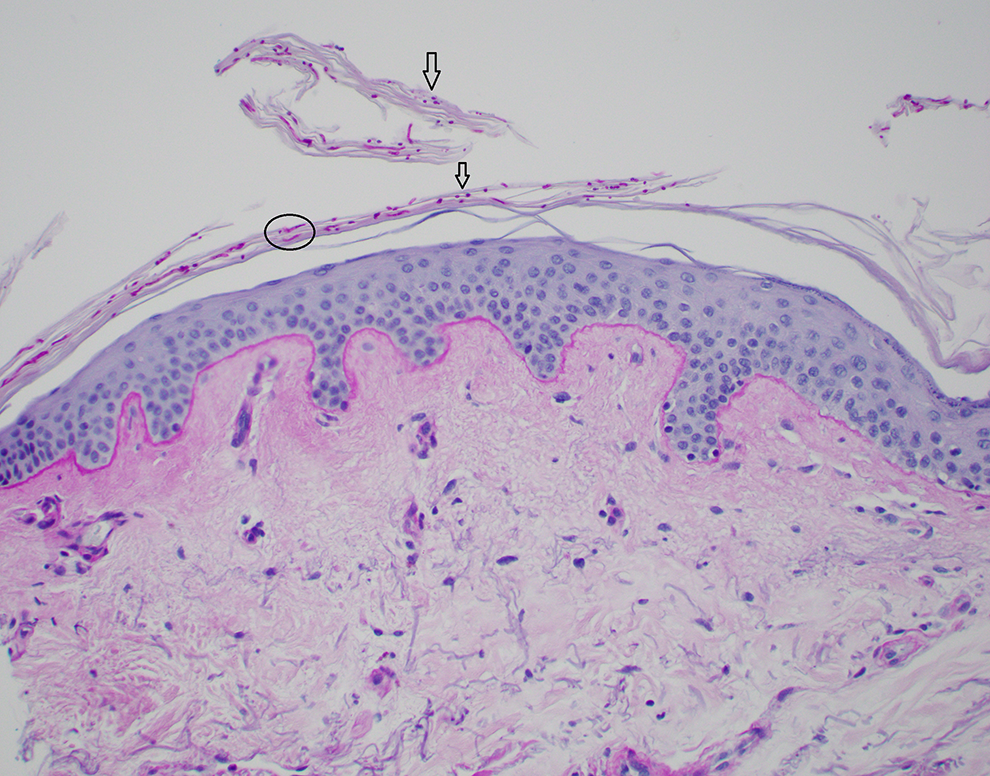
An example case of Tinea versicolor. Pictured is a skin biopsy with superficial fungal hyphae (circled) and yeast (arrows) forms seen within the stratum corneum, stained with period acid–Schiff (PAS). This is often referred to as a “spaghetti and meatballs” appearance (PAS, ×20).
A SOX-10 Stain Is Performed and Is Shown in Figure 5. What Cells Are Highlighted by SOX-10 Immunohistochemical Stain? How Would You Interpret the SOX-10 Stain in This Case?
SOX-10 is a transcription factor found in neural crest cells and melanocytes. It is a nuclear stain and would be positive in melanocytes found within the epidermis. The SOX-10 stain is negative in this case, confirming a lack of melanocytes along the junction. The inset photo is of “normal” skin and highlights scattered, evenly distributed melanocytes along the junction (red nuclear stain).
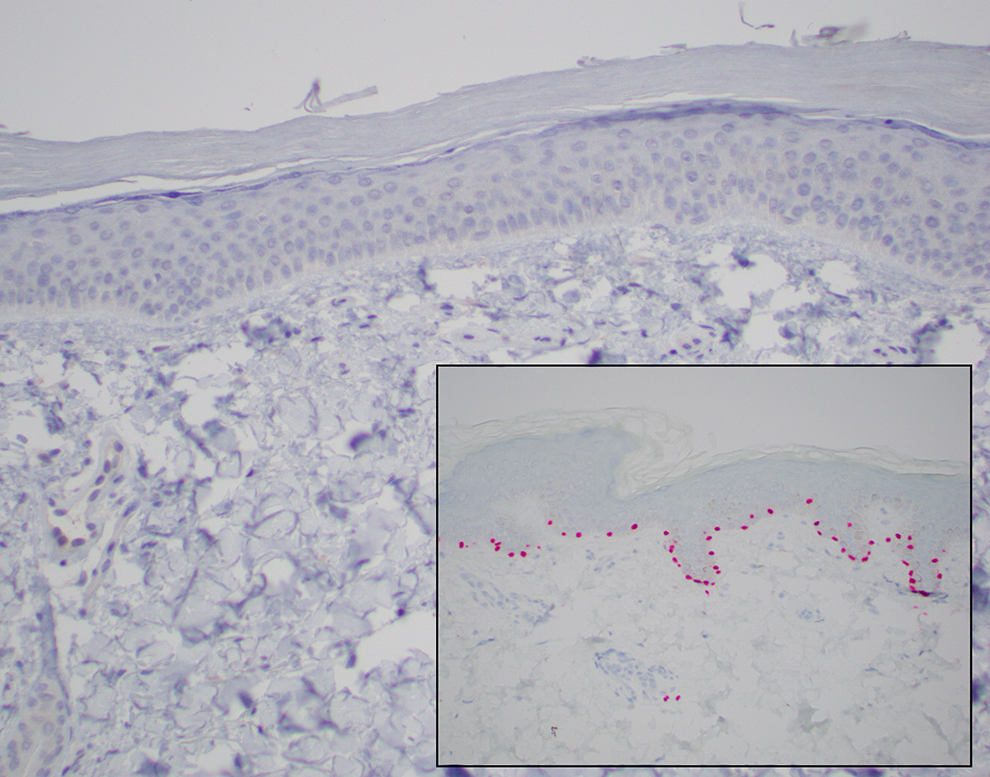
The patient’s skin biopsy stained with SOX-10, an immunohistochemical stain for melanocytes, showing no melanocytes along the dermal-epidermal junction. Inset, A SOX-10 stain of “normal” skin showing evenly distributed melanocytes (red nuclear stain) along the dermal–epidermal junction (SOX-10 ALK PHOS (EP268, Epitomics), ×20).
How Does the Microscopic Examination Narrow Your Differential Diagnosis?
A near “normal” skin biopsy with absent melanocytes and lack of melanin pigment with a clinical history of symmetrically located, depigmented macules and patches on arms and legs is most consistent with vitiligo. Postinflammatory hypopigmentation is the result of a resolved/resolving inflammatory dermatitis. The loss of pigment is due to the inflammation in the primary process disrupting the epidermal keratinocytes, which contain melanin pigment. The keratinocytes “lose” their pigment into the dermis, causing the clinical impression of pigment alteration. The biopsy will often reveal melanophages (histiocytes containing melanin) in the upper dermis—a sign of prior inflammation. Stains are not useful in making the diagnosis of postinflammatory pigment alteration. Tinea versicolor is a superficial cutaneous fungal infection caused by
Differential Diagnosis for Skin Disorders Showing Depigmentation.
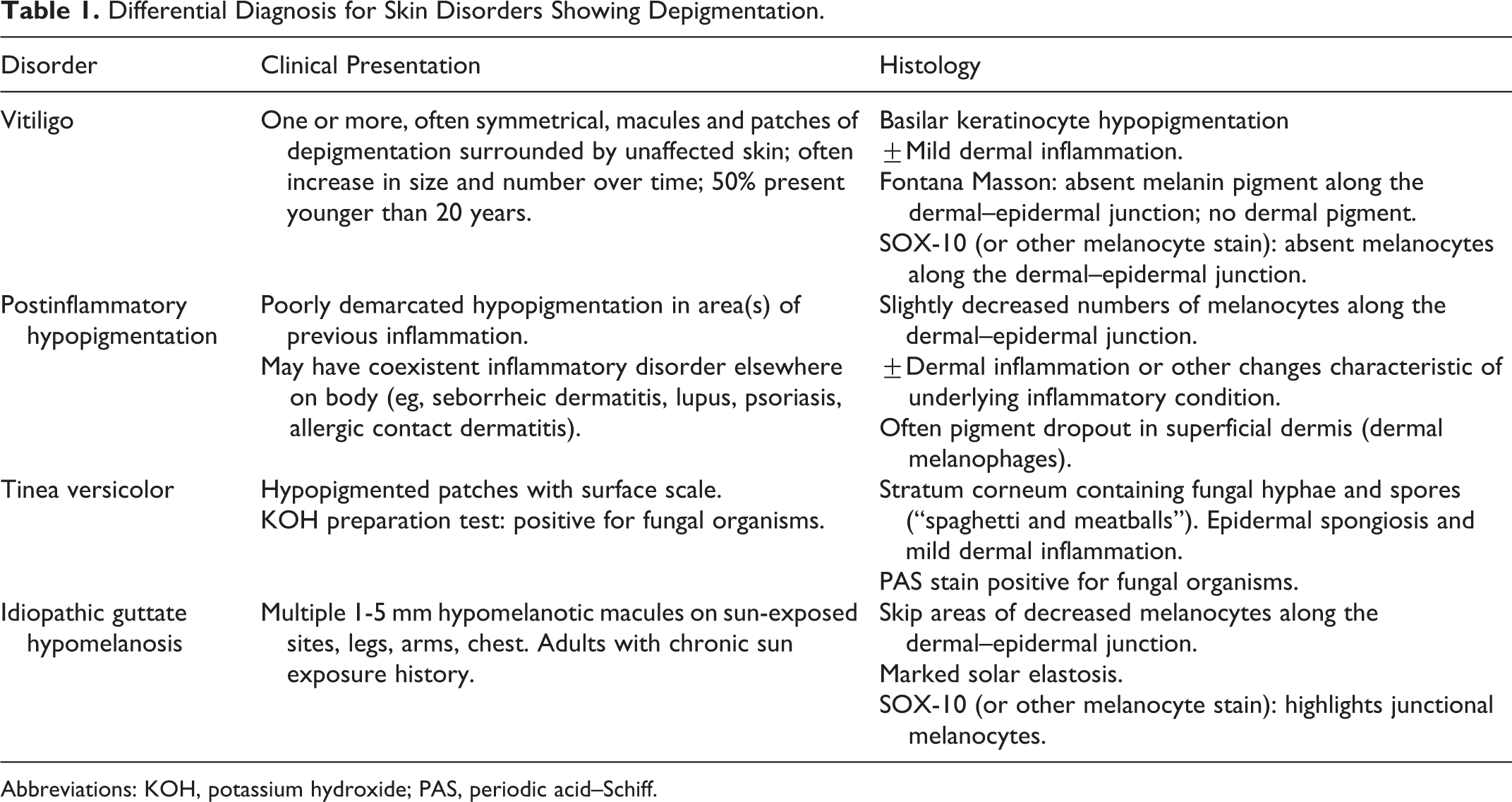
Abbreviations: KOH, potassium hydroxide; PAS, periodic acid–Schiff.
Questions/Discussion Points, Part 2
What is Vitiligo?
Vitiligo is a skin disorder characterized by depigmentation of the skin and hair follicles resulting in white, often symmetrical macules and patches on the skin surrounded by normal skin. They often increase in number and size over time. Vitiligo is classified as localized, generalized, and universal, affecting different regions of the body and with variable extent. 2 Over 50% of patients present before age 20, with many of these before age 10. Vitiligo is common, affecting 0.5% to 2.0% of the world’s population. There is no race or sex predilection. 3 Interestingly, patients can experience the “Koebner phenomenon” in which new lesions of vitiligo appear in areas of trauma. The clinical course of vitiligo is difficult to predict. 4
What Causes Vitiligo?
The exact cause of vitiligo is unclear but what results is damage to melanocytes and their subsequent disappearance in the affected skin. There are several theories regarding the pathogenesis of vitiligo; the most prominent is an autoimmune condition. Other theories include a neural, self-destructive, and inherent defect—all of which are not mutually exclusive. Vitiligo may in fact represent a group of heterogeneous pathophysiologic disorders with a similar phenotype. Although most cases of vitiligo are sporadic, familial clustering is not uncommon, and human leukocyte antigen (HLA) haplotypes may contribute to vitiligo susceptibility; HLA-A2 has been identified as a high-risk haplotype. In addition, there are numerous candidate genes and genetic loci associated with vitiligo. These genes have been implicated in a number of autoimmune diseases and likely function as a general autoimmune or autoinflammatory susceptibility loci similar to HLA. 2 Epidemiologic studies indicate that vitiligo is inherited in a non-Mendelian, multifactorial, and polygenic pattern with incomplete penetrance. 2 Different phenotypes are associated with different genetic susceptibility genes and environmental exposures. Intrinsic defects in melanocytes may initiate disease through innate inflammation, including recruitment of natural killer cells and inflammatory dendritic cells. Cytotoxic CD8+ T cells have been shown to be involved in melanocyte destruction. 3,5 Environmental factors also contribute, including exposure to phenolic compounds found in household products. 3
How Is Vitiligo Diagnosed?
Vitiligo is a clinical diagnosis, meaning clinicians can recognize the disease and, by clinical presentation only, make the diagnosis without further testing. Wood’s lamp examination helps to confirm the diagnosis and extent of the disease by delineating lesions on the skin and is often performed in the clinic. Biopsy may also be performed to confirm the diagnosis. Histologic examination will show basal keratinocyte hypopigmentation, mild dermal inflammation, and absent melanocytes. 6,7
Are There Systemic Changes in Someone With Vitiligo?
Patients with vitiligo may present with systemic changes including inflammation of the ear and eye. Melanocytes are found in the uveal tract, retinal pigment epithelium, and membranous labyrinth of the inner ear. Therefore, any area with melanocytes may be affected. Patients with vitiligo and their first-degree relatives have an increased prevalence of autoimmune thyroid disease, type 1 diabetes mellitus, pernicious anemia, rheumatoid arthritis, Addison disease, lupus, Guillain-Barre syndrome, and others. 2 Thyroid disease is the most common condition found in patients with vitiligo.
Teaching Points
Vitiligo is an acquired, idiopathic condition favored to be an autoimmune disease characterized by destruction of melanocytes, the pigment producing cells in the skin.
Patients with vitiligo present with macules and patches of depigmentation of the skin. The clinical course is difficult to predict.
Melanocytes are also found in the eye and ear, and therefore, patients with vitiligo may have inflammation affecting these sites.
Vitiligo is associated with other systemic autoimmune diseases, most commonly thyroid disease.
Histologically, vitiligo shows loss of melanocytes along the dermal–epidermal junction and lack of melanin pigment in the basilar keratinocytes. Stains can be used to confirm the findings and rule out other causes of clinical depigmentation.
The differential diagnosis of vitiligo includes other disorders of hypopigmentation. Differentiation can be made by a combination of clinical findings and histologic features.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article processing fee for this article was funded by an Open Access Award given by the Society of ‘67, which supports the mission of the Association of Pathology Chairs to produce the next generation of outstanding investigators and educational scholars in the field of pathology. This award helps to promote the publication of high-quality original scholarship in
