Abstract
Background:
Pleural fibrosis and restriction are well-known complications of tuberculous pleurisy, which is often accompanied by respiratory distress and limitation of daily life activities.
Objectives:
Current evidence suggests that pleural drainage offers little benefit over and above antituberculous treatment in improving pulmonary function. Our study aims to define the role of additional pleural drainage in the management of tuberculous pleural effusions.
Methods:
We enrolled 21 patients with tuberculous effusions and performed therapeutic drainage in 10 randomly selected cases. Pulmonary function testing, exercise capacity, chest radiography, and ultrasonography were performed at baseline, 7 to 10 days, and at 3 months.
Results:
Complete therapeutic drainage was achieved in 4 of 10 patients randomized to undergo drainage. Immediate benefit in total lung capacity (TLC) was achieved in the 10 patients assigned to intervention. Intervention group showed significant changes compared to the non-intervention group in several functional parameters at 3 months: change in forced vital capacity (ΔFVC 1.40 L, 95% confidence interval [CI] 1.08-1.71 vs Δ0.34 L, 95% CI 0.01-0.67, P < .000), change in forced expiratory volume in 1 second (ΔFEV1 1.08 L, 95% CI 0.79-1.37 vs Δ0.38 L, 95% CI 0.08-0.68, P = .001), change in TLC (ΔTLC 1.45 L, 95% CI 1.05-1.85 vs Δ0.56 L, 95% CI 0.00-1.12, P = .009), and change in diffusion capacity for carbon monoxide (ΔDLCO 6.43 mL/min/mm Hg, 95% CI 3.73-9.12 vs Δ0.57 mL/min/mm Hg, 95% CI 2.31-3.34, P = .005). Significant improvement after 3 months was not observed in the 6-minute walking distance as well as oxygen saturation before and after walking.
Conclusion:
Therapeutic drainage may offer additional short-term functional benefits to patients with pleural tuberculosis.
Introduction
Tuberculous pleural effusion accounts for approximately 5% of diseases due to Mycobacterium tuberculosis and is the second most common form of extrapulmonary tuberculosis (TB) after lymphatic involvement. 1-2 In the developing world, numbers are much higher, especially in the setting of HIV coinfection, where up to 80% of cases with TB may be associated with pleural effusions. 3,4 Pleural fibrosis (PF) or fibrothorax is a well-described complication of TB pleurisy 3,5 and causes typical clinical symptoms such as chronic chest pain, dyspnea, and impairment in lung function. 6,7 Treatment of TB pleural effusion is similar to treatment of pulmonary TB using standard anti-TB medication. With appropriate therapy, fever usually resolves within 2 weeks, and pleural fluid is resorbed within 6 weeks. However, some patients take up to 2 months to defervescence, and fluid resorption may take up to 4 months. In severe cases, surgical intervention is required. 8 -11 Uncertainty remains as to the prevalence of fibrothorax and permanent pleural thickening, and this has been reported to be between 5% and over 50%. 12 -15
Therapeutic thoracentesis or initial complete drainage in addition to standard anti-TB drugs has been advocated to reduce residual pleural thickening (RPT) and facilitate symptomatic recovery. However, the results have been inconclusive, 6,16 -19 and possible complications such as pneumothorax may limit its use. 20 Lai et al could not show the benefit of pleural drainage in the prevention of PF while performing a randomized trial. 16 In a subsequent study, Chung et al showed that effective drainage lowers the risk of fibrosis and accelerates pulmonary function recovery when compared to partial drainage of TB pleural effusion. 17 In a recent randomized trial by Bhuniya et al, patients were randomized to thoracentesis and anti-TB drugs or anti-TB drugs alone. They found significant improvement in forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC) with thoracentesis and lower percentage of RPT. 18
The aim of our study was to investigate the benefits of once-off pleural drainage in addition to medical management in the prevention of short-term sequelae of TB pleural effusion. In our study design, we established a control group, included an evaluation of drainage success, and focused on changes within a 3-month follow-up.
Methods
Study Design and Population
This randomized controlled study was performed at Tygerberg Academic Hospital. This institution is a 1200-bed academic hospital in Cape Town, South Africa. It is 1 of the 2 referral centers and renders a tertiary service to a population of approximately 1.5 million people. The incidence of pulmonary TB in South Africa is 1000 cases per 100 000 persons, one of the highest recorded incidences in the world according to the World Health Organization. 21 Between October 2012 and April 2013, we were able to enroll 21 patients with proven TB pleural effusion. All patients with radiological evidence of a pleural effusion (at least 30% of 1 hemithorax) and with at least 2 clinical indicators of active TB were invited to participate in the study. Indicators of a high clinical suspicion of TB included (1) known HIV infection, (2) persistent cough lasting >3 weeks, (3) hemoptysis, (4) weight loss >4 kg, (5) intermittent fever >3 weeks, and (6) drenching night sweats >2 weeks. 7 Patients who were subsequently found to have alternative diagnoses were excluded from the analysis (n = 6). Exclusion criteria at the outset included an age <18 years, former incomplete TB treatment, a recent history of invasive procedures within the pleural cavity, or recent penetrating chest wall trauma. Ethical approval was obtained from the Stellenbosch University Health Research Ethics Committee 2 (Ethics Reference #: N12/07/040), and all patients had to sign informed consent. Clinical trial registration was gained by the Pan African Clinical Trials Registry (identification number: PACTR201411000938131).
Initial Evaluation and Management
A baseline chest X-ray (CXR) with posterior–anterior and lateral films along with spirometric assessment according to guidelines by the American Thoracic Society (ATS) was performed on all patients.
22
-25
Initial CXR effusion size was graded as moderate (<50% of a hemithorax), moderate–large (50%-75% of a hemithorax), or larger (>75% of a hemithorax). Spirometry (MasterScreen Jaeger, Würzburg, Germany, Version 02.00, 2011) included a flow-volume loop, diffusion capacity for carbon monoxide (DLCO), and plethysmography. To evaluate patient’s functional exercise capacity, we performed a 6-minute walking test (6MWT) according to ATS criteria and graded symptoms according to Borg, including dyspnea and exhaustion with a visual analogue scale (VAS).
26,27
Additionally, we measured patients’ oxygen saturation before and after each 6MWT. Participants were randomized to 1 of the 2 parallel groups (control or intervention group) at an equal allocation ratio of 1:1. For randomization, a simple randomization schedule (number table) was used. Diagnostic thoracocentesis (50 mL) and pleural biopsy (≥4 samples) with an (Abrams Needle, Meditronix Corporation, New Delhi, India),
Pleural fluid was analyzed by means of routine biochemistry, including adenosine deaminase, cytology, and cell counts. Liquid TB cultures of pleural fluid and tissue biopsy were performed with a BACTEC MGIT 960 System (Becton, Dickinson and Company, Sparks, Maryland, USA)
Follow-Up
Participants were followed up after 1 week in order to review laboratory results and continued on (if relevant) standard 4-drug anti-TB treatment for at least 6 months. All patients with confirmed pleural TB were subsequently followed up at 3 months. The CXR, 6 MWT, and spirometric assessments were repeated at each visit. The RPT was assessed on follow-up chest radiographs by measuring the greatest linear width of the pleural opacity and estimating the overall pleural thickening of the hemithorax (<1/3, 1/3-2/3, and >2/3). Pleural thickening of 3 to 9 mm was defined as abnormal, and thickening ≥10 mm was considered to be a fibrothorax. 12,28
Outcome
For short-term sequelae, the primary outcome parameter we defined was the change in FVC in liter after 3 months. Key secondary outcome parameters included changes in FEV1, total lung capacity (TLC), DLCO, 6 MWT, and the incidence of fibrothorax after 3 months of treatment.
Statistical Analysis
For data analysis (IBM SPSS Statistics 20), only patients with completed 3-month follow-up data were evaluated which reduced patient numbers to N = 20. Change in FVC after 3 months was compared to baseline value, and an unpaired t test at an α level of 5% (2-sided) was used to compare the mean change in FVC between the intervention and the control groups. Respective 95% confidence intervals (CIs) were calculated. Further secondary end points were analyzed in line with the primary end point (change in FEV1, TLC, DLCO, and 6MWT after 3 months). Baseline characteristics were analyzed using mean and standard deviation for continuous and absolute frequencies for categorical variables.
Results
The study was terminated after 20 patients completed 3-month follow-up, as complete drainage (per protocol) was achieved only in 4 of 10 patients randomized to the intervention, and once-off complete drainage (to achieve <0.5 cm of pleural fluid visible in the posterolateral recess) was therefore not considered a realistic goal.
Baseline Observations and Interventions
Twenty-seven patients fulfilled the inclusion (screening) criteria, and 21 of these were ultimately diagnosed with pleural TB. Ten cases were randomized to therapeutic pleural drainage; and in the control group, 1 patient was lost to follow-up after the initial visit, leaving 10 patients with completed data per study arm (Figure 1). The mean age of the 20 patients was 32.70 (±9.98) years with 12 patients being HIV positive. None of the patients reported coeffecting conditions such as chronic obstructive pulmonary disease or diffuse parenchymal lung diseases. The general characteristics of the group are summarized in Table 1 and the results of the diagnostic thoracocentesis in Table 2. The mean volume drained during therapeutic drainage was 1139 ± 711 mL (range 250-2700 mL). Aspiration was abandoned in 6 patients due to suspected reexpansion pulmonary edema and/or patient discomfort.

Flow diagram of all patients screened and enrolled.
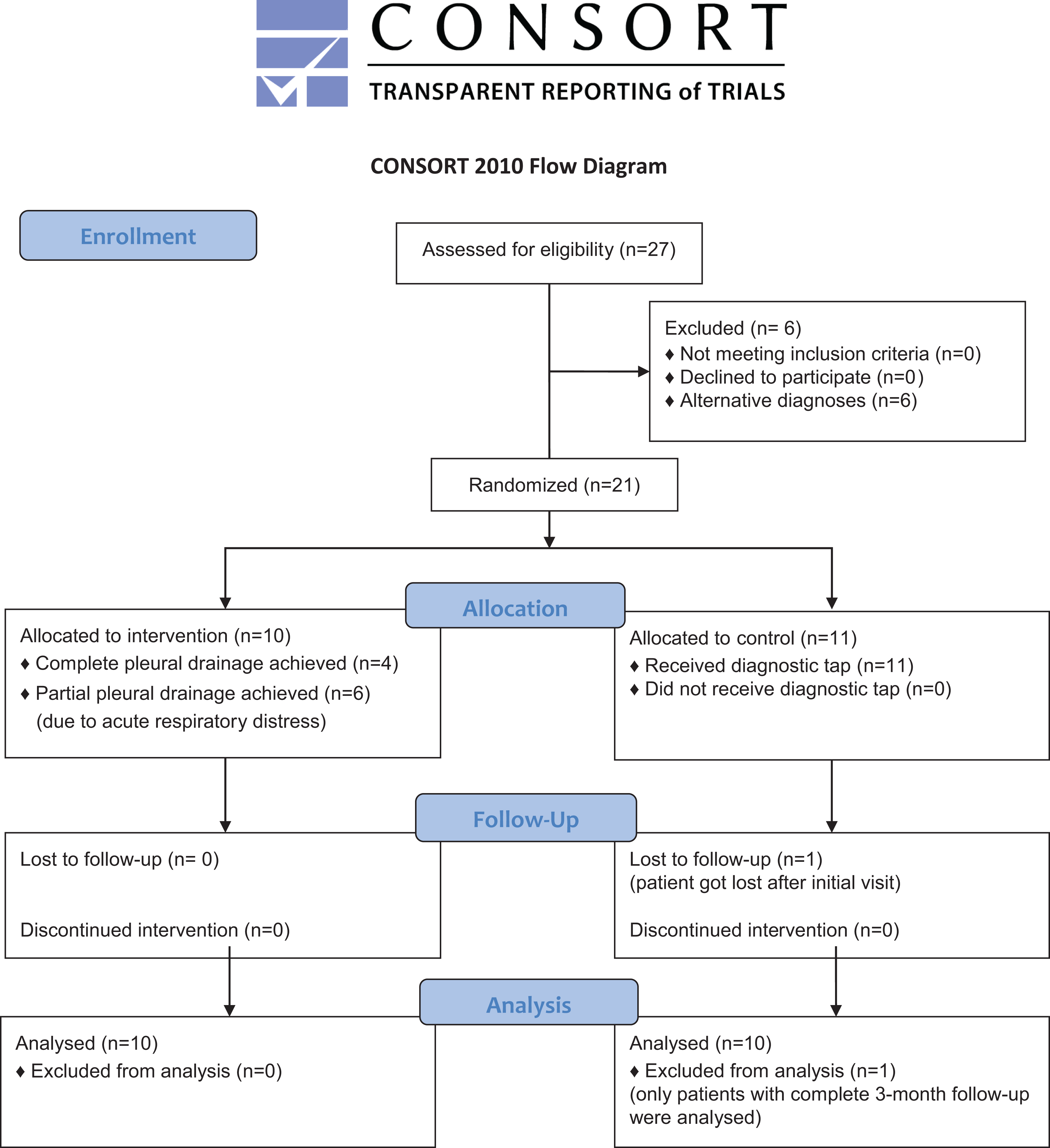
Participant flow through the study.
Clinical and Radiological Characteristics of the Study Population at Baseline.a

aN = 20.
Pleural Fluid and Blood Results of Study Population at Baseline.a

Abbreviations: ADA, adenosine deaminase; LDH, lactate dehydrogenase.
aN = 20.
Primary and Key Secondary Outcomes
All lung function parameters measured at baseline, 1 week, and 3 months are summarized in Tables 3 and 4. As regards the primary outcome variable, patients randomized to therapeutic drainage experienced a significantly greater improvement in FVC compared to the control group at 3 months (mean difference Δ1.40 L, 95% CI 1.08-1.71 in the intervention group vs Δ0.34 L, 95% CI 0.01-0.67 in the control group, P < .000). Mean percentage predicted FVC of the intervention group was significantly higher (79.27, 95% CI 72.27-87.27 vs 64.66, 95%CI 57.82-71.49, P = .006) after 3 months compared to the control group. The FEV1 in the intervention group was improved by a mean change of Δ1.08 L (95% CI 0.79-1.37) versus Δ0.38 L (95% CI 0.08-0.68, P = .001) in the control group. Patients randomized to therapeutic drainage also experienced significantly greater improvement in TLC (Δ1.45 L, 95% CI 1.05-1.85 vs Δ0.56 L, 95% CI 0.00-1.12, P = .009) and DLCO after 3 months (Δ6.43 mL/min/mm Hg, 95% CI 3.73-9.12 vs Δ0.57 mL/min/mm Hg, 95% CI 2.31-3.34, P = .005). The 6MWT improved in both groups (intervention group Δ113.50 m, 95% CI 67.28-159.72 vs control group Δ85.90 m, 95% CI 36.29-135.51, P = .369), and improvement in the intervention group was not significantly superior to the control group. After 3 months, the incidence of significant RPT in the control group was twice as high as in the intervention group. Pleural effusion of 1 patient in the control group increased during the 3-month follow-up. In all other cases, pleural effusion was treated successfully and showed moderate size after 3 months. In both groups, patients improved clinically during treatment, and surgery was not considered in any patient as no one showed severe restriction (FVC < 50%) after 3 months.
Lung Function Parameters (Mean ± Standard Deviation) of all Study Patients at Baseline and Immediately Following Drainage.a

Abbreviations: DLCO, diffusion capacity for carbon monoxide; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; NA, not available; TLC, total lung capacity; 6MWT, 6-minute walking test.
aAbsolute values in liter, and 6MWT values in meter.
Lung Function Parameters (Mean ± Standard deviation) of all Study Patients after 1 Week and 3 Months.

Abbreviations: DLCO, diffusion capacity for carbon monoxide; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; NA, not available; TLC, total lung capacity; 6MWT, 6-minute walking test.
aAbsolute values in liter, and 6MWT values in meter.
Complications
Initial pleural aspiration and biopsy were uncomplicated in all study patients. Reexpansion pulmonary edema and/or patient discomfort resulted in the premature termination of 6 of 10 attempts at complete pleural drainage. No pneumothorax or major hemorrhage was caused.
Discussion
We found that patients with confirmed tuberculous pleural effusions randomized to therapeutic pleural drainage showed significantly superior improvements in several lung function parameters at 3-month follow-up, including change in FVC, FEV1, TLC, and DLCO, despite the fact that complete drainage per protocol was achieved in less than half of all patients randomized to undergo the intervention.
In 1996, Wyser et al investigated the influence of corticosteroids on TB pleural effusions and concluded that standard anti-TB therapy and early complete drainage are adequate for the treatment of TB pleurisy. 14 Their study did not include a control group. A subsequent randomized controlled trial by Lai et al found that the addition of pleural space drainage to anti-TB drug treatment had neither a beneficial effect on RPT development nor shortened the duration of fever or other clinical symptoms. 16 Lai et al failed to show significant improvement in FVC (treatment group 85.5% vs control group 88%; P = .568), and TLC and FEV1 were not measured and efficacy of drainage was not evaluated. Dyspnea was the only proven benefit and showed faster improvement in the drained group (median 4 days vs 8 days, P < .001). Contrary to this, a recent study by Bhuniya et al where they performed pleural drainage using pleural manometry showed significant differences after 6 months in regard to mean percentage predicted of FEV1 (drainage group 87.62 vs control group 84.92, P = .02) and FVC (84.46 vs 83.31, P = .00). 18 They reported a lower appearance of RPT in drained patients and also commented that patients with therapeutic thoracentesis experienced immediate relief from dyspnea after drainage but did not substantiate this finding with any objective tool. Previous studies report immediate improvement in FVC and FEV1, both showing an increase in excess of 10% after thoracentesis of large pleural effusions. 31,32 We could not find any immediate improvement in FVC or FEV1 after the procedure, which might be due to pain and coughing caused by the draining process, but TLC showed a significant immediate improvement (3.00 L predrainage, 95% CI 2.49-3.49 vs 3.40 L postdrainage, 95% CI 2.48-4.00, P = .047). Additionally, complete drainage did not lead to any differences in walking distances measured by Borg between both groups at any time during the follow-up, and overall improvement after 3 months also was not significant. On the other hand, most patients experienced clinical improvement in chest pain and relief of dyspnea after drainage of effusion. The highest dyspnea relief was achieved immediately after drainage, which confirms findings of former studies. 14,18,33
Although not significantly, it appears that complete drainage seemed to reduce RPT. Earlier studies reported that RPT ≥10 mm causes significant clinical symptoms in patients with pleural TB, and incidences vary from 26% to 50.4%. 12,16,18 In our recent study, the control group presented double (60%) as much cases with RPT ≥10 mm than the intervention group (30%) which matches the numbers of current studies. 18
Compared to former studies, 14,17,18 we were particularly interested in the immediate and short-term influence of complete drainage of TB pleural effusions. This study is a randomized controlled trial and as such we focused on FVC improvement and investigated the influence of drainage on all lung function parameters including the TLC and the DLCO. In contrast to previous studies, 16,17 we decided to use a single once-off drainage for achieving dryness of pleural effusion, not a pigtail drainage over several days. The reason for this was that once-off drainage, if proven as effective, would be an available treatment procedure simply performable at day care clinics in rural areas. As far as we know, this is the first study that evaluated efficacy of drainage after the procedure to ensure complete dryness.
One limitation of our study is that, despite randomizing patients, both groups differed with regard to their baseline characteristics. From the outset, the intervention group presented a larger effusion size, higher dyspnea grade, and more restriction in lung function parameters. Simple randomization schedules in a small number of patients can cause an unequal allocation of data. Nevertheless, the intervention group achieved significant improvements in primary and key secondary outcome parameters. Further limitations are the small number of patients, a short follow-up period, and the inability to perform complete dryness of effusion continuously as defined in the protocol. Dyspnea associated with pulmonary reexpansion is known to limit the maximum volume drained. 33 -35
In conclusion, we believe that therapeutic drainage may offer additional short-term functional benefits to patients with large tuberculous pleural effusions. Larger scale prospective studies with more realistic pleural drainage protocols, a longer follow-up period, and the use of pleural manometry (to decrease the risk of reexpansion pulmonary edema) are needed to define the role of this intervention in reducing long-term restrictive ventilatory impairment and the need for surgical decortication.
Footnotes
Authors’ Note
Prof Coenraad Koegelenberg conceived the study and supervised the study performance together with Prof Tobias Welte. Hannah Fengels drafted the first manuscript version together with Dr Johannes Bruwer, recruited patients, performed 6MWT and data collection. Spirometry data were obtained by Francois Swart and David Maree. Dr Elisma Wilken assessed the recruitment of patients. Dr Enas Batubara, Dr Johannes Bruwer, and Prof Coenraad Koegelenberg performed pleural procedures (tapping, biopsy, and drainage) according to the manuscript. Analysis and interpretation of data were performed by Andrea Gonnermann and Hannah Fengels. Clinical trial registration was obtained by the Pan African Clinical Trials Registry (identification number: PACTR201411000938131).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by the Susan De Kock Bursary 2013 of the University of Stellenbosch (15,000.00 ZAR).
