Abstract
Context
Several phase III randomized controlled trials (RCTs) have shown the importance of perioperative systemic therapy, especially for the efficacy of immune checkpoint inhibitors (ICIs) in both neoadjuvant and adjuvant settings for muscle-invasive bladder cancer (MIBC).
Objective
To synthesize the growing evidence on the efficacy and safety of systemic therapies for MIBC utilizing the data from RCTs.
Evidence acquisition
Three databases and ClinicalTrials.gov were searched in October 2024 for eligible RCTs evaluating oncologic outcomes in MIBC patients treated with systemic therapy. We evaluated pathological complete response (pCR), disease-free survival (DFS), progression-free survival (PFS), event-free survival (EFS), overall survival (OS), and adverse events (AEs).
Evidence synthesis
Thirty-three RCTs (including 14 ongoing trials) were included in this systematic review. Neoadjuvant chemotherapy improved OS compared to radical cystectomy alone. Particularly, the VESPER trial demonstrated that dd-MVAC provided oncological benefits over GC alone in terms of pCR rates, OS (HR: 0.71), and PFS (HR: 0.70). Recently, the NIAGARA trial showed that perioperative durvalumab plus GC outperformed GC alone in terms of pCR rates, OS (HR: 0.75), and EFS (HR: 0.68). Despite the lack of data on overall AE rates in the VESPER trial, differential safety profiles in hematologic toxicity were reported between dd-MVAC and durvalumab plus GC regimens. In the adjuvant setting, no study provided the OS benefit from adjuvant chemotherapy. However, only adjuvant nivolumab had significant DFS and OS benefits compared to placebo.
Conclusions
Neoadjuvant chemotherapy remains the current standard of care for MIBC. Durvalumab shed light on the promising impact of ICIs added to neoadjuvant chemotherapy. Nivolumab is the only ICI recommended as adjuvant therapy in patients who harbored adverse pathologic outcomes. Ongoing trials will provide further information on the impact of combination therapy, including chemotherapy, ICIs, and enfortumab vedotin, in both neoadjuvant and adjuvant settings.
Keywords
Introduction
Radical cystectomy (RC) is the definitive local surgical treatment for muscle-invasive bladder cancer (MIBC). However, despite an appropriate radical surgical approach, there remains a substantial risk of recurrence, which increases in the presence of extravesical tumor extension or node-positive disease.1,2 Since deaths are usually caused by distant metastases, not local recurrence, 3 neoadjuvant and/or adjuvant systemic therapy is a reasonable treatment strategy to eradicate possible micro-metastases, potentially improving oncologic outcomes.2,4–7 Following the oncological benefits demonstrated in randomized controlled trials (RCTs), neoadjuvant cisplatin-based chemotherapy followed by RC is the guideline-endorsement treatment for MIBC. 7 Additionally, a meta-analysis of individual participant data (IPD) from 10 RCTs demonstrated that adjuvant cisplatin-based chemotherapy is also a valid treatment option for improving outcomes in MIBC. 8
Immune checkpoint inhibitors (ICIs) have dramatically changed the treatment landscape for metastatic urothelial carcinoma.9–11 Several randomized controlled trials (RCTs) have assessed the impact of ICI monotherapy in adjuvant settings.12–14 In addition, several phase I/II trials demonstrated the promising role of ICIs and/or combination with chemotherapy in the neoadjuvant setting.15–17 However, a large amount of emerging evidence inhibits comparing and clarifying which regimens are optimal for which patients. Furthermore, a recent trial was designed to consistently use of ICIs for both neoadjuvant and adjuvant settings, inhibiting a robust meta-analysis with fair comparison between novel combination therapy versus conventional neoadjuvant GC alone.18,19 Therefore, we conducted the systematic review exclusively including RCTs to comprehensively synthesize the historical and current evidence of the impact of systemic therapy for MIBC, including both neoadjuvant and adjuvant therapies.
Evidence acquisition
The protocol of this study has been registered in the International Prospective Register of Systematic Reviews database (PROSPERO: CRD42024618884).
Search strategy
This systematic review was conducted based on the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (Supplementary Table 1).20,21 Three databases (PubMed®, Web of Science™, and Embase®) and ClinicalTrials.gov were searched in October 2024 to identify trials assessing oncologic outcomes in MIBC patients treated with systemic therapy. The detailed search words were (muscle-invasive bladder cancer OR MIBC) AND (neoadjuvant OR adjuvant OR chemotherapy OR immune checkpoint inhibitor) AND (randomized controlled OR RCT), detailed in Supplementary Appendix 1. Subsequently, abstracts presented at major conferences (i.e., the American Society of Clinical Oncology and the European Society for Medical Oncology) were searched to include trial updates. Additionally, manual literature searches were conducted by reviewing the reference lists of relevant studies and performing citation tracking to identify additional pertinent articles. The primary outcomes of interest were oncologic outcomes, including pathological complete response (pCR), event-free survival (EFS), and progression-free survival (PFS) in the neoadjuvant setting, disease-free-survival (DFS) in the adjuvant setting, and cancer-specific survival (CSS) and overall survival (OS) in both settings. Additional outcomes of interest were rates of adverse events (AEs). Two investigators independently screened the titles and abstracts and thereby performed the full-text review for all potentially relevant trials. Disagreements were resolved by obtaining consensus among co-authors.
Inclusion and exclusion criteria
Studies that included patients with MIBC (Participants) and assessed the efficacy of neoadjuvant/adjuvant systemic therapy (Interventions) compared to definitive local therapy alone or another systemic therapy regimen (Comparisons) assessing their differential effects on pCR, EFS, PFS, DFS, OS, and/or rates of AEs (Outcomes) in randomized controlled trials (RCTs) (Study design) were included. We excluded studies lacking original patient data, reviews, letters, editorial comments, replies from authors, case reports, and articles not written in English. Relevant references of eligible trials were scanned for additional studies of interest.
Data extraction
The trial design and demographic data on eligible trials were extracted by two authors independently: the first author's name, publication year, inclusion criteria, endpoints of studies, agents and dosage of the intervention and control arms, age, the number of patients on each demographic (i.e., sex, TN stage, primary tumor location, neoadjuvant chemotherapy [NAC]), follow-up periods, pCR, EFS, PFS, CSS, OS, and the number of AEs (any and severe defined as greater than CTCAE grade 3).
Risk of bias assessment
We assessed the quality and risk of bias of published RCTs following the Cochrane Handbook for Systematic Reviews of Interventions risk-of-bias tool (RoB version 2) (Supplementary Figure 1). 22 The certainty of evidence for each RCT was evaluated using the GRADE system by two authors. (Supplementary Table 2). 22 Two authors independently performed the risk-of-bias and certainty of evidence assessment of each study.
Evidence synthesis
Study selection and characteristics
The PRISMA flow chart details our article selection process (Figure 1). We identified 599 publications as an initial literature search. After removing duplicates, 493 publications remained for titles and abstract screening. Based on our inclusion criteria, 438 publications were excluded, leaving 55 publications for full-text review. Additionally, we found six further abstracts via citation searching. As a result, 33 RCTs (13 for neoadjuvant setting, 9 for adjuvant setting, and 11 for ongoing trials) were included in the systematic review.4,14,18,23–49 The trial design, patient demographics, oncologic outcomes, and safety profiles are summarized in Tables 1 and 2 and Supplementary Table 3–5.
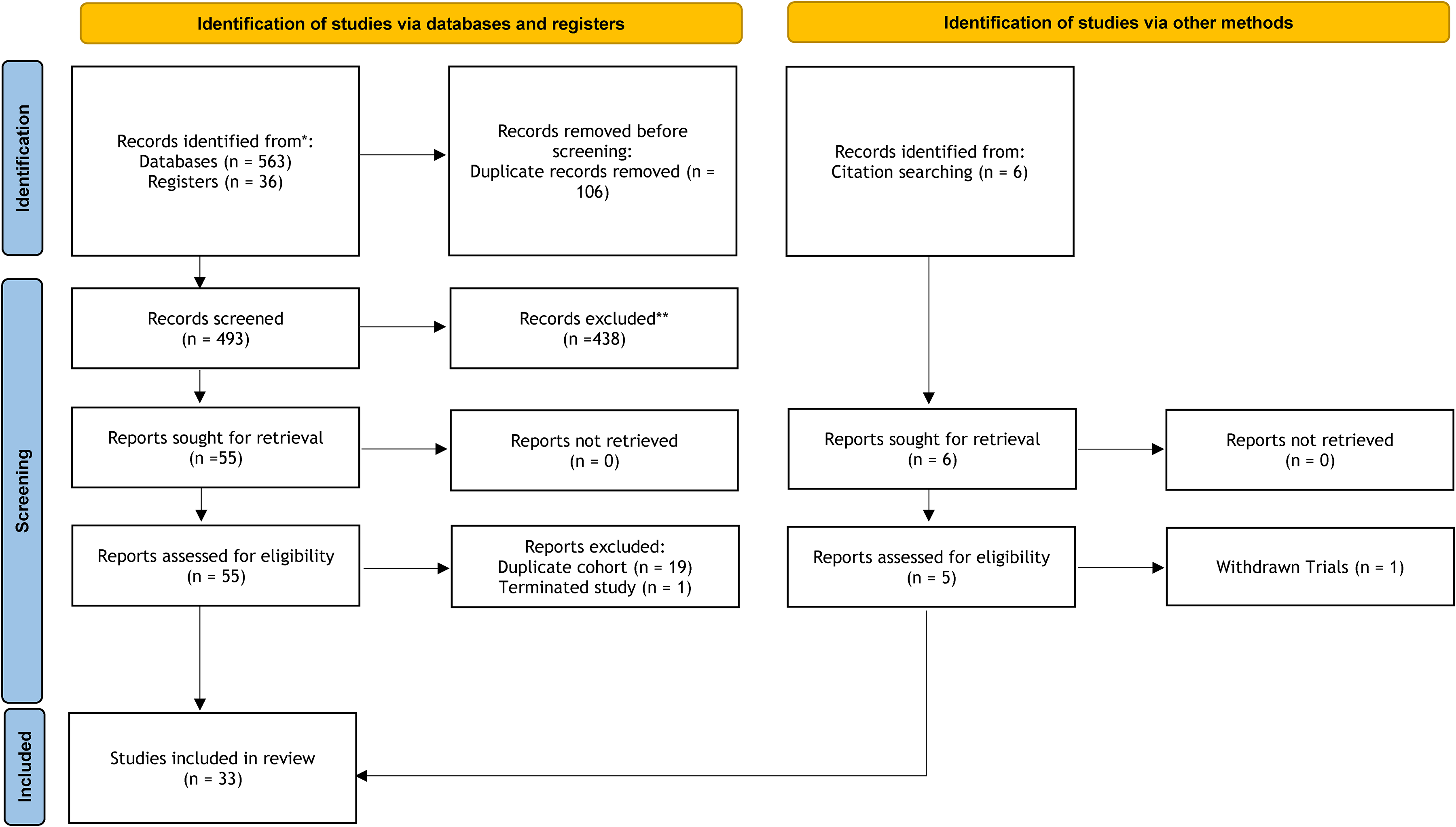
PRISMA 2020 flow diagram for systematic reviews which included searches of databases, registers and other sources
Trial design and oncologic outcomes of included RCTs comparing neoadjuvant combination therapy with conventional chemotherapy.
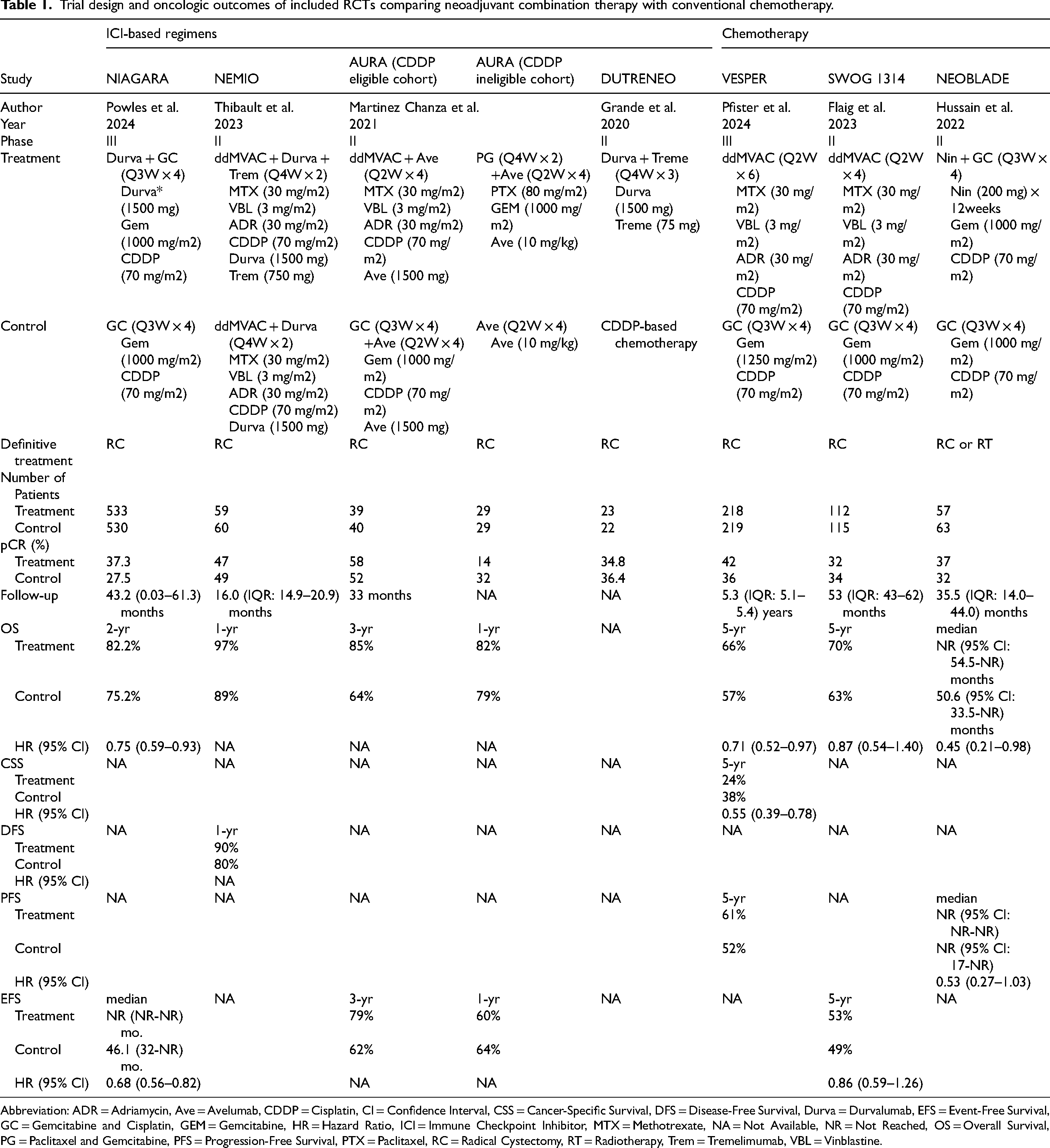
Abbreviation: ADR = Adriamycin, Ave = Avelumab, CDDP = Cisplatin, CI = Confidence Interval, CSS = Cancer-Specific Survival, DFS = Disease-Free Survival, Durva = Durvalumab, EFS = Event-Free Survival, GC = Gemcitabine and Cisplatin, GEM = Gemcitabine, HR = Hazard Ratio, ICI = Immune Checkpoint Inhibitor, MTX = Methotrexate, NA = Not Available, NR = Not Reached, OS = Overall Survival, PG = Paclitaxel and Gemcitabine, PFS = Progression-Free Survival, PTX = Paclitaxel, RC = Radical Cystectomy, RT = Radiotherapy, Trem = Tremelimumab, VBL = Vinblastine.
Trial design and oncologic outcomes of included RCTs comparing adjuvant ICI therapy with placebo/observation
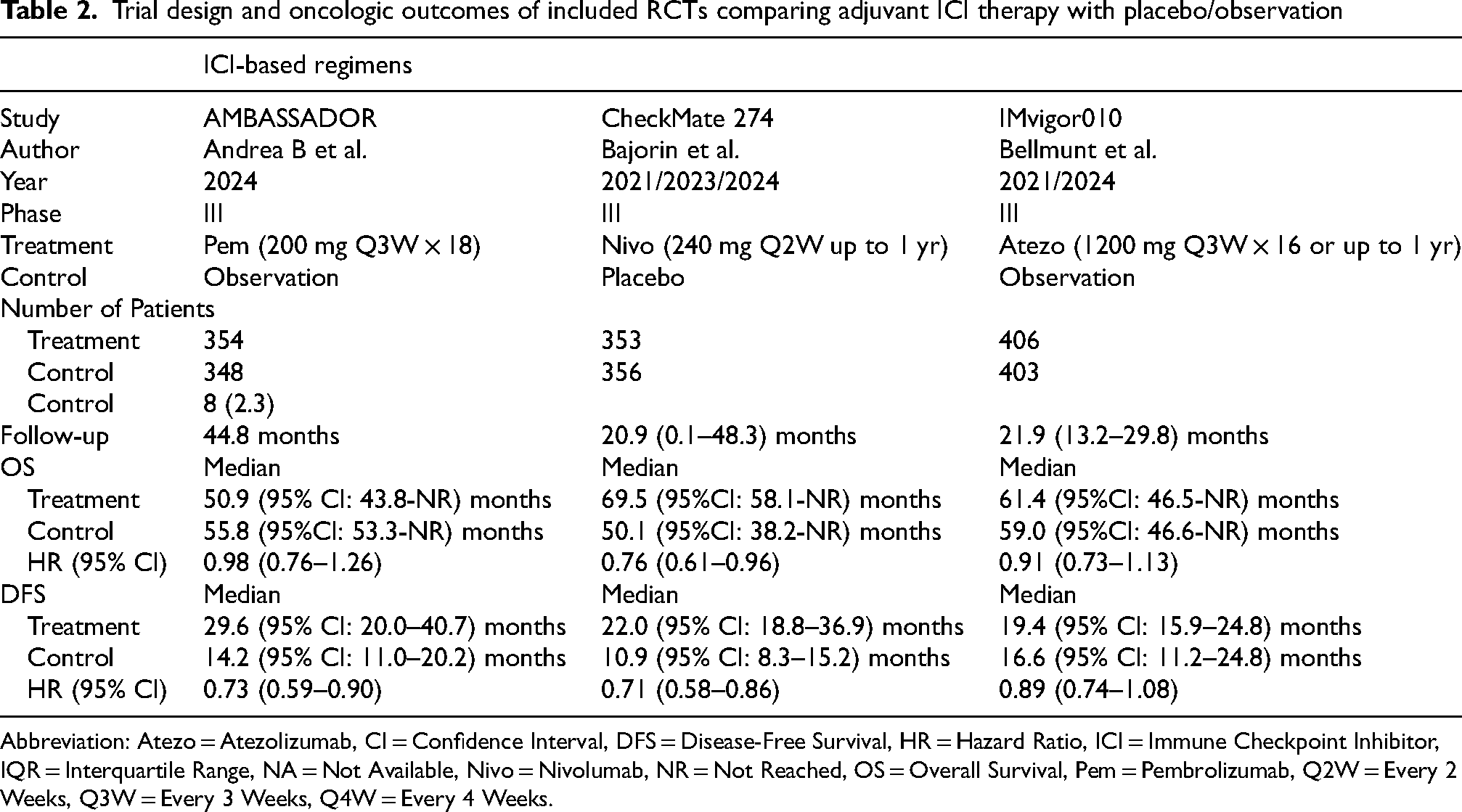
Abbreviation: Atezo = Atezolizumab, CI = Confidence Interval, DFS = Disease-Free Survival, HR = Hazard Ratio, ICI = Immune Checkpoint Inhibitor, IQR = Interquartile Range, NA = Not Available, Nivo = Nivolumab, NR = Not Reached, OS = Overall Survival, Pem = Pembrolizumab, Q2W = Every 2 Weeks, Q3W = Every 3 Weeks, Q4W = Every 4 Weeks.
Neoadjuvant therapy prior to radical cystectomy
A total of 13 RCTs were included in the systematic review (Table 1 and Supplementary Table 3). Six trials compared neoadjuvant chemotherapy plus definitive local therapy with definitive local therapy alone.4,25,27,29,31,32 Three trials compared a novel chemotherapy regimen with a conventional gemcitabine plus cisplatin (GC) regimen followed by definitive local therapy.23,26,30 Four trials assessed the efficacy of ICI monotherapy or in combination with chemotherapy.18,24,28,33
Neoadjuvant chemotherapy
History of neoadjuvant chemotherapy
From the 1990s to 2010s, six RCTs were conducted to assess the oncologic impact of neoadjuvant cisplatin-based chemotherapy in addition to definitive local treatment (Supplementary Table 3).4,25,27,29,31,32 In the Nordic Cystectomy Trial (NCT) 1 in 1993, Rintala et al. firstly demonstrated that neoadjuvant cisplatin plus doxorubicin followed by radiotherapy (RT) and RC significantly improved pCR rates and 5-year OS estimates compared to RT plus RT alone. 31 However, a study by Martinez-Pineiro et al. in 1995 found no survival benefit for neoadjuvant cisplatin monotherapy, though responders to chemotherapy (defined as downstaging to pT0, pTis, pTa, or pT1) had significantly better OS. 29 Then, pCR, as well as pathological downstaging after neoadjuvant chemotherapy, have been recognized as important predictors for survival in patients who underwent neoadjuvant chemotherapy and RC.
In 2003, Grossman et al. published the results of the SWOG-8710 trial, evaluating the impact of neoadjuvant MVAC (three 28-day cycles: methotrexate [30 mg/m2] on days 1, 15, and 22; vinblastine [3 mg /m2] on days 2, 15, and 22; and doxorubicin [30 mg/m2] and cisplatin [70 mg/m2] on day 2) versus RC alone in 317 patients with cT2-4a bladder cancer. 4 This trial demonstrated that neoadjuvant MVAC significantly reduced the risk of death by 33% (hazard ratio [HR]: 1.33; 95% confidence interval [CI]: 1.00-1.76). In addition, improved survival was associated with the absence of residual cancer in the cystectomy specimen, consistent with previous findings.4,29
In 2004, a combined analysis of the Nordic Cystectomy Trials (NCT1 and NCT2, with 325 and 317 patients, respectively) assessed the efficacy of cisplatin-based NAC versus RC alone.31,32 Despite variations in regimens and dosages of cisplatin, the analysis showed a significant OS benefit with cisplatin-based NAC (HR: 0.8; 95% CI: 0.64-0.99). 50 Since then, based on robust evidence of the utility of NAC, cisplatin-based NAC has been recognized as a standard treatment strategy for cisplatin-fit patients with MIBC. However, the optimal chemotherapy regimen in the neoadjuvant setting other than MVAC was unclear. Based on the safety and noninferiority of GC compared to MVAC in the advanced/metastatic setting, 51 neoadjuvant use of GC had been adopted in clinical practice.
Optimal regimens for neoadjuvant chemotherapy
Since then, three RCTs have assessed the optimal regimen for NAC compared to neoadjuvant GC.23,26,30 The NEOBLADE phase II trial evaluated the addition of nintedanib, a tyrosine kinase inhibitor, to GC in the neoadjuvant setting. 26 Though effective in lung cancer, nintedanib failed to improve pCR rates (37% for nintedanib group and 32% for placebo group) and was associated with increased hematologic AEs. 26
The VESPER trial has assessed the impact of perioperative dose dense-MVAC (dd-MVAC: six cycles of dose-dense methotrexate, vinblastine, doxorubicin, and cisplatin once every 2 weeks) compared to GC. 30 The trial design was unique owing to including both neoadjuvant (88%) and adjuvant (12%) settings. This trial provided several insights into the field of NAC. Compared to GC regimen, dd-MVAC resulted in better organ-confined response (ypT3N0) (77% vs. 63%, p < 0.001) and 3-year PFS (66% vs. 56%, HR: 0.70 [95% CI, 0.51–0.96], p = 0.025) in the neoadjuvant setting. 6 In addition, extended follow-up data showed that 5-year OS was significantly favor in the dd-MVAC group (66% vs. 57%, HR: 0.71 [95% CI, 0.52–0.97]) in the neoadjuvant setting. 30 However, dd-MVAC potentially has increased risk of myelosuppression, such as anemia (22% vs. 7.8%, p < 0.001) and febrile neutropenia (6.5% vs. 2.4%, p = 0.053), leading to lower achievement rates of preplanned six cycles (60% of patients received the planned six cycles in the dd-MVAC arm and 84% received four cycles in the GC arm). 6 Balancing the harm and benefit is important, especially for neoadjuvant settings prior to invasive radical surgery. In addition, optimal candidate selection, which is more likely to benefit from dd-MVAC is another clinical unmet need.
The SWOG S1314 trial aimed to test the utility of coexpression extrapolation gene expression models as potential predictive biomarkers for pathologic response to GC and dd-MVAC at the time of surgery. This trial suggests the prognostic value of scoring system based on coexpression extrapolation gene expression models for pathological downstaging (odds ratio [OR]: 2.33, 95%CI: 1.11–4.89, p = 0.02) as well as OS (HR: 0.45, 95%CI: 0.20–0.99; p = 0.047). 23 Further biomarker assessment, including molecular subtype, RNA, microRNA, DNA, and circulating tumor cells/DNA, is needed to optimize patient selection in the neoadjuvant setting.
Neoadjuvant immune checkpoint inhibitors and/or in combination with chemotherapy
Based on promising anti-tumor activity from neoadjuvant studies investigating ICI (29–46% of pCR rates for ICI monotherapy and 33–43% of pCR rates for ICI plus chemotherapy),16,52–55 there are four comparative trials assessing the efficacy of ICI monotherapy or in combination with chemotherapy and three of them are phase II trials.18,24,28,33 The DUTRENEO trial, a prospective, randomized, and phase II trial, assessed the impact of ICI-based combination, durvalumab (anti-PD-L1 antibody) and tremelimumab (anti-CTLA-4 antibody) combination in patients with “hot tumor” evaluated by tumor inflammation score compared to cisplatin-based chemotherapy. 24 Unexpectedly, durvalumab plus tremelimumab failed to show an improvement in the rates of pCR (34.8% for durvalumab + tremelimumab and 36.4% for cisplatin-based chemotherapy) and pathological downstaging (56.5% for durvalumab + tremelimumab and 58.5% for cisplatin-based chemotherapy). 24
The AURA trial is a uniquely designed, prospective, multicenter, randomized, phase II trial for patients with MIBC. 28 Cisplatin-eligible patients were randomized to GC plus avelumab (n = 40) or dd-MVAC plus avelumab (n = 39), and cisplatin-ineligible patients were randomized to gemcitabine plus paclitaxel (GP) chemotherapy plus avelumab (n = 28) or avelumab alone (n = 28). 28 The pCR rates (ypT0/is/a), the primary endpoints were 52% for GC plus avelumab, 58% for dd-MVAC plus avelumab, 14% for GP plus avelumab, and 32% for avelumab alone.56,57 Additionally, avelumab plus cisplatin-based chemotherapy provided substantial survival benefits with 64% and 85% of preliminary 3-year OS for GC plus avelumab and dd-MVAC plus avelumab. 56 These are lessons from this trial; 1) Avelumab in addition to cisplatin-based chemotherapy has robust pCR rates; 2) Avelumab in addition to dd-MVAC achieved the highest OS benefit; 3) Avelumab monotherapy had promising pCR rates, but it was reduced when in combination with GP.56,57
The NEMIO, open-label, randomized, non-comparative phase I/II trial, has also assessed the impact of ICIs in addition to dd-MVAC. 33 Patients with cT2-4N0-1 MIBC eligible for RC were randomized to dd-MVAC plus durvalumab plus tremelimumab or dd-MVAC plus durvalumab. 33 This trial showed one of the highest pCR (ypT0) rates in the neoadjuvant setting using dd-MVAC plus durvalumab (49%). Nonetheless, the benefit of adding tremelimumab on pathological response appears limited (47%). 33
Last but not least, the NIAGARA, randomized, phase III trial assessing the efficacy of perioperative durvalumab (4 cycles for neoadjuvant and 8 cycles for adjuvant therapies) in addition to GC chemotherapy has recently been published. 18 This trial consisted of 533 patients treated with perioperative durvalumab plus GC and 535 treated with GC alone. 18 Durvalumab plus GC significantly improved EFS (HR: 0.68, 95% CI: 0.56–0.82) and 2-year OS estimate (82.2% vs. 75.2%, p = 0.01) compared to GC alone. 18 Furthermore, there were no differences in the rates of severe treatment-related AEs (40.6% vs. 40.9%) and patients who underwent RC (88.0% vs. 83.2%). 18 This trial first demonstrated the survival benefit of adding neoadjuvant ICI to chemotherapy. Durvalumab plus GC combination will become another standard of care treatment strategy in this setting.
Adjuvant therapy after radical cystectomy
We found nine RCTs assessing the effectiveness of adjuvant therapy, including six RCTs for adjuvant chemotherapy and three for adjuvant ICI monotherapy (Table 2, Supplementary Table 5).14,34–40 Seven of nine RCTs set the observation/placebo as a comparator, but two of them set the delayed chemotherapy as a comparator.
Adjuvant chemotherapy
In the 1990s, three RCTs were performed to assess the efficacy of adjuvant cisplatin monotherapy or cisplatin-based combination therapy against observation.36,38,39 In 1995, Stöckle et al. showed that adjuvant MVAC or methotrexate, vinblastine, cisplatin, and epirubicin (MVEC) (n = 26) significantly improved PFS in patients with pT3b, pT4a, and/or pN1, pN2 compared to observation (n = 23, p = 0.005). In 1996, an RCT by Freiha et al. comprising 50 patients with pT3b or pT4 bladder cancer also demonstrated that adjuvant cisplatin, methotrexate, and vinblastine (CMV) combination significantly improved PFS compared to observation (median PFS: 37 vs. 12 months, p = 0.01), while there was no OS benefit (Median OS: 63 vs. 36 months, p = 0.32). 36 These trials suffered from limited number of patients and the possible impact of savage chemotherapy on survival outcomes. Considering this, the EORTC 30994 trial compared the impact of adjuvant chemotherapy (four cycles of GC or MVAC) with deferred chemotherapy in patients with pT3-T4 or pN + bladder cancer. 37 However, this trial closed earlier than preplanned date due to poor accrual: 91% of adjuvant chemotherapy arm received planned chemotherapy but only 47% of deferred chemotherapy arm. 37 This trial revealed that adjuvant chemotherapy significantly prolonged PFS (HR: 0.54, 95% CI: 0.40–0.73), while it did not improve OS (HR: 0.78, 95% CI: 0.56–1.08). Despite statistically significant recurrence and metastasis-free survival benefits from IPD meta-analysis synthesizing these RCTs, 8 previous RCTs consistently demonstrated the PFS benefit but no OS benefit for adjuvant chemotherapy. Furthermore, setting an optimal trial design was found to be another essential aspect of establishing a robust conclusion in this setting.
Adjuvant immunotherapy
There are three phase III RCTs assessing the impact of adjuvant ICI monotherapy in patients with high-risk urothelial carcinoma who received NAC with ypT2-T4 or N + or those who did not receive NAC and were pT3-T4 or N + at radical surgery (Table 2).13,14,34 These trials included patients with upper tract urothelial carcinoma (UTUC) (ranged 7 to 22%) and those who underwent NAC (ranged 45 to 64%). The IMvigor010 trial compared the efficacy of adjuvant atezolizumab with observation. 14 Unexpectedly, this trial failed to show DFS (HR: 0.89, 95% CI: 0.74–1.08) and OS (HR: 0.91, 95% CI: 0.73–1.13) benefits in overall population and bladder cancer cohort. 14 The AMBASSADOR trial assessing the impact of adjuvant pembrolizumab showed the DFS benefit in the overall cohort (HR 0.69, 95% CI: 0.54–0.87) as well as bladder cancer cohort (HR: 0.63: 95% CI: 0.50–0.80). However, OS was similar between the adjuvant pembrolizumab and observation groups (HR: 0.98, 95% CI: 0.76–1.26). 34 The CheckMate 274 trial compared the efficacy of adjuvant nivolumab with placebo. 13 This trial showed the DFS (HR: 0.71, 95% CI: 0.58–0.86) and OS (HR: 0.76, 95% CI: 0.61–0.96) benefits for adjuvant nivolumab in the overall cohort. 13 In addition, similar to the AMBASSADOR trial, DFS benefit was more prominent in patients with bladder cancer (HR: 0.63, 95% CI: 0.51–0.78). 13 Furthermore, DFS benefit was also better in patients who received NAC (HR: 0.53, 95% CI: 0.39–0.72). 13 Therefore, adjuvant nivolumab is currently the standard of care for patients with high-risk muscle-invasive urothelial carcinoma, especially considered for those with MIBC and who received NAC.
Ongoing clinical trials
Table 3 summarizes the phase II/III comparative trials regarding perioperative systemic treatment for MIBC. There are six phase III trials; their primary endpoint is mostly EFS. The KEYNOTE-866 (NCT03924856) is a phase III RCT aiming to clarify the impact of adding pembrolizumab to GC in the neoadjuvant setting. The VOLGA (NCT04960709) is a phase III, multicenter, international RCT that will enroll 830 patients with MIBC. This trial aims to assess the efficacy of neoadjuvant enfortumab vedotin (EV) plus perioperative durvalumab ± tremelimumab. The KEYNOTE-905/EV-303 (NCT03924895) is a phase III RCT assesses the efficacy of perioperative EV plus pembrolizumab. In bladder preservation settings, the INTACT (NCT03775265) and KEYNOTE-992 (NCT04241185) trials aim to clarify the impact of adding ICIs to chemotherapy. These ongoing trials will clarify the role of adding ICIs to chemotherapy and shed light on the impact of EV in the neoadjuvant setting. In addition, the impact of ICIs as a part of trimodal therapy has been assessed in several ongoing trials. These ongoing trials will change the clinical practice in the near future.
Details on ongoing phase 2/3 RCTs assessing the impact of systemic combination therapy for MIBC
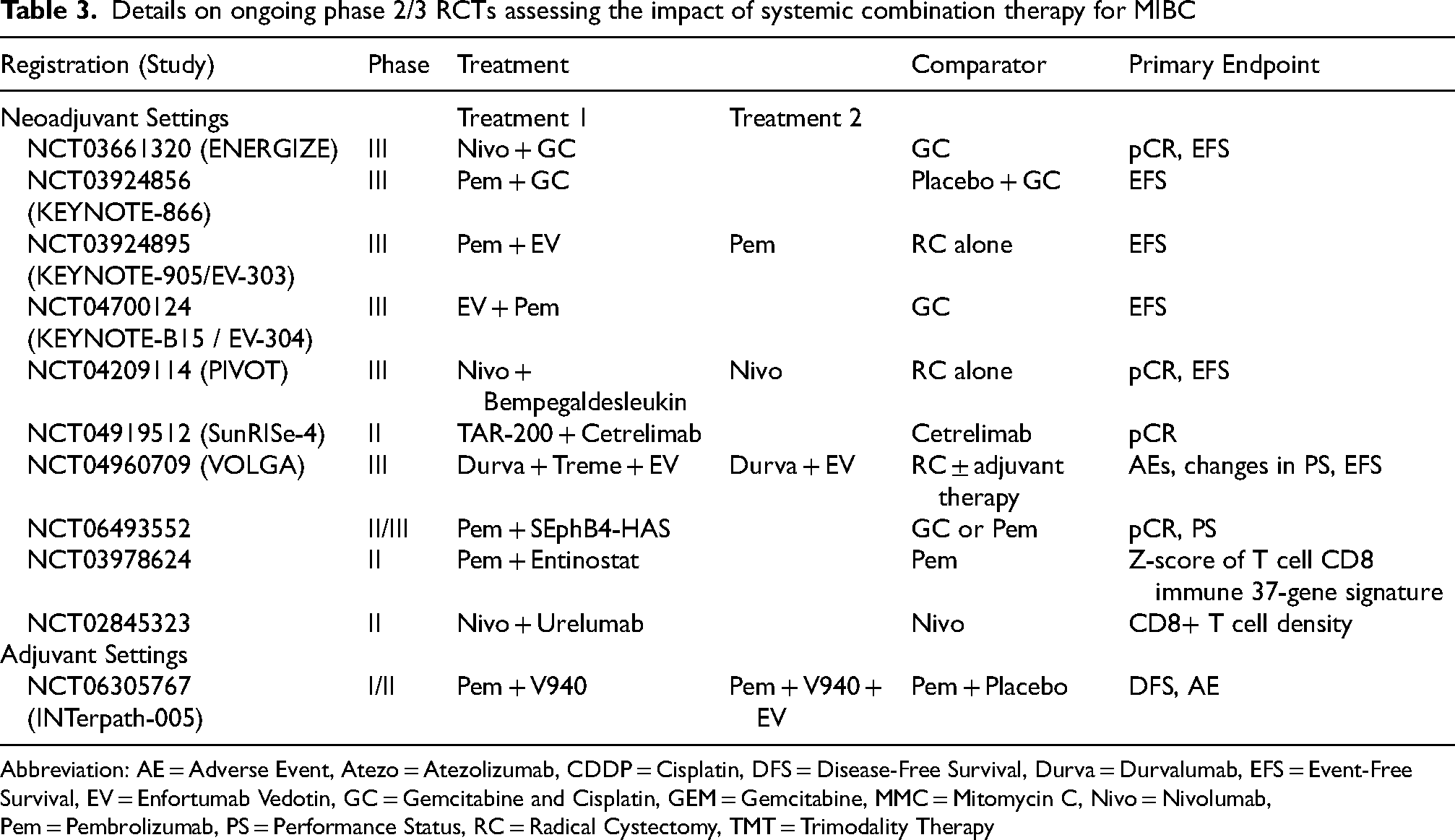
Abbreviation: AE = Adverse Event, Atezo = Atezolizumab, CDDP = Cisplatin, DFS = Disease-Free Survival, Durva = Durvalumab, EFS = Event-Free Survival, EV = Enfortumab Vedotin, GC = Gemcitabine and Cisplatin, GEM = Gemcitabine, MMC = Mitomycin C, Nivo = Nivolumab, Pem = Pembrolizumab, PS = Performance Status, RC = Radical Cystectomy, TMT = Trimodality Therapy
Discussion
This systematic review comprehensively summarizes the historical and current status of the efficacy of perioperative systemic therapy in the management of MIBC. There were four key takeaways from this review. First, neoadjuvant dd-MVAC and perioperative durvalumab plus GC had significant OS benefits compared to neoadjuvant GC followed by RC. Second, despite promising pCR rates, there are still no robust survival data on adding ICI to dd-MVAC. Third, patients who achieved pCR after neoadjuvant therapy were significantly associated with better OS compared to those who did not. Fourth, adjuvant ICI monotherapy, such as nivolumab, established a standard of care, especially for patients who progressed despite neoadjuvant chemotherapy.
Based on the positive results from the VESPER and NIAGARA trials evaluating novel neoadjuvant combinations versus GC chemotherapy, there is growing interest which regimen- dd-MVAC or durvalumab plus GC- is more effective.18,30 Neoadjuvant dd-MVAC (HR for OS: 0.71 [95% CI, 0.52–0.97]) in the VESPER trial and perioperative durvalumab plus GC (HR for OS: 0.75 [95% CI, 0.59–0.93]) in the NIAGARA trial had significant OS benefits compared to neoadjuvant GC followed by RC.18,30 Based on similar HRs for OS, indirect comparison via network meta-analysis will not demonstrate the superiority of either dd-MVAC or durvalumab plus GC. In the two trials, despite using marginally different endpoints such as EFS and PFS, these outcomes were also similar between the two regimens compared to GC alone.18,30 Additionally, the NIAGARA trial set the consistent durvalumab use in both neoadjuvant and adjuvant settings, inhibiting a fair comparison between the two trials. 18 However, with the assumption that there are similar oncologic benefits between the two regimens, differential safety profiles should be considered in adoption for daily clinical practice. Given concerns about the potential for intensive myelosuppression with dd-MVAC, durvalumab plus GC may be a suitable option for elderly and comorbid patients with MIBC(Figure 2). Conversely, neoadjuvant dd-MVAC can select the patients who should be offered adjuvant nivolumab depending on their pathologic outcomes on RC specimen. This would minimize the treatment burden for selected patients, thereby resulting in cost-effectiveness. Balancing the risks and benefits is, indeed, important for shared-decision making especially for neoadjuvant settings. To seek optimal regimens for neoadjuvant therapy, considering promising pCR rates, ICI plus dd-MVAC will theoretically outperform ICI plus GC in terms of OS benefit. Further clinical trials and extended follow-up data from trials above are awaited.
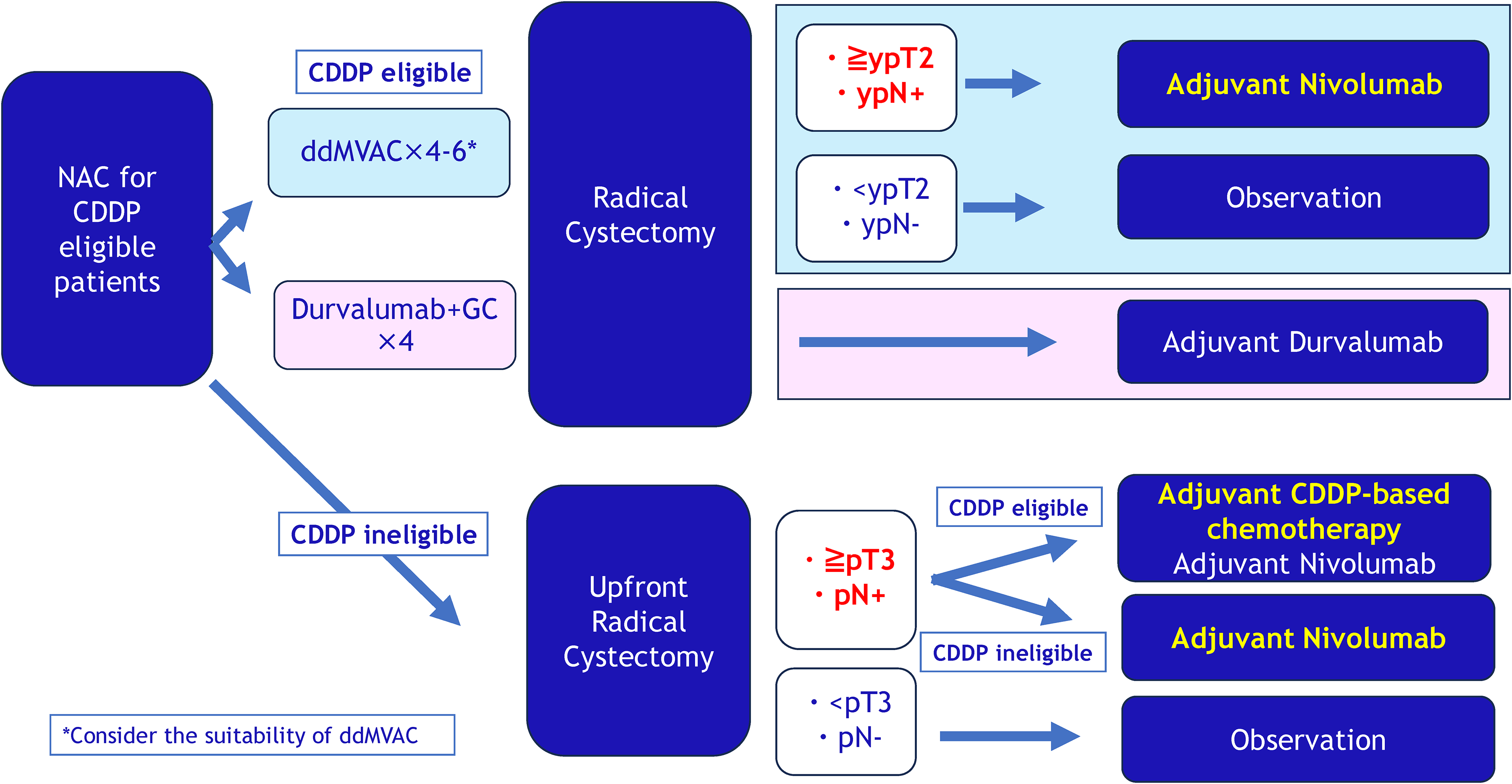
Current treatment strategy for MIBC
Neoadjuvant trials consistently demonstrated the substantial survival benefit for patients who achieved pCR compared to those who did not.4,27,28,51 Even in the era of ICI-based combination therapy, pCR rates seem to be optimal surrogate endpoints, which are not needed for long-term follow-up duration for phase I/II trials. 58 In support of this, several ongoing trials have been set up to make pCR a primary endpoint (Table 3). Like coexpression extrapolation gene expression models, more robust, reliable, and generalizable predictive biomarker for pCR is awaited in the era of intensified ICI-combination regimens. 23
Only adjuvant nivolumab monotherapy resulted in OS benefit for patients with adverse pathologic findings on RC specimens. 13 The subgroup analysis of the CheckMate274 trial suggested the efficacy of adjuvant nivolumab would be more prominent in patients with MIBC and those who received NAC. Despite pembrolizumab having DFS benefit but not OS benefit, a recently published meta-analysis demonstrated the statistically significant OS benefit for adjuvant ICIs when synthesizing nivolumab, pembrolizumab, and atezolizumab. 59 A key to the success of the adjuvant therapy is optimal patient selection. In the post-hoc analysis of the IMvigor010 trial, postoperative circulating tumor DNA (ctDNA) status is a promising predictive factor for the efficacy of adjuvant atezolizumab. 60 Combining ypTstage and ctDNA status can be utilized to select the optimal candidates for adjuvant ICIs.
Despite several important findings in this systematic review, most neoadjuvant trials assessing the impact of adding ICIs to chemotherapy are preliminary. In addition, limited numbers of trials comparing the same treatment arms with control arms inhibited performing a robust meta-analysis, especially for the neoadjuvant setting. Further well-designed phase III RCTs, survival data with extended follow-up from included trials, and individual patient data meta-analysis will clarify the optimal regimen for ICI-based neoadjuvant therapy for MIBC. Moreover, advancements and improved generalizability in molecular and genetic analyses, as well as liquid biopsy, are urgently needed to optimize patient selection for intensified perioperative systemic treatment, which may carry a certain level of toxicity.
Conclusions
Based on OS benefits from recent trial updates, neoadjuvant dd-MVAC and perioperative durvalumab plus GC are recommended for cisplatin-eligible patients who planned RC instead of neoadjuvant GC alone. Treatment decisions should be performed using shared-decision making process, including differential safety profiles. Despite promising pCR rates for adding ICI to dd-MVAC, there is still no data showing any survival benefit. The achievement of pCR after neoadjuvant therapy was significantly associated with better OS than those who did not. Adjuvant nivolumab is currently a standard of care, especially for patients who progressed despite NAC. Ongoing trials assessing the impact of perioperative novel combinations, including EV and ICIs will change the treatment landscape of MIBC in the coming future.
Supplemental Material
sj-docx-1-blc-10.1177_23523735251335122 - Supplemental material for Advancements in systemic therapy for muscle-invasive bladder cancer: A systematic review from the beginning to the latest updates
Supplemental material, sj-docx-1-blc-10.1177_23523735251335122 for Advancements in systemic therapy for muscle-invasive bladder cancer: A systematic review from the beginning to the latest updates by Takafumi Yanagisawa, Akihiro Matsukawa, Jeremy Yuen-Chun Teoh, Keiichiro Mori, Tatsushi Kawada, Satoshi Katayama, Paweł Rajwa, Fahad Quhal, Benjamin Pradere, Marco Moschini, Shahrokh F. Shariat, Jun Miki1 and Takahiro Kimura in Bladder Cancer
Footnotes
Abbreviations and Acronyms
Acknowledgment
None.
ORCID iDs
Authors’ contributions/CRediT
TY and AM contributed to protocol/project development, data collection and management, data analysis, and manuscript writing/editing. JYCT, KM, TK (Tatsushi Kawada), and SK contributed to data analysis and manuscript writing/editing. PR, FQ, BP, and MM contributed to manuscript writing/editing. SFS, JM, and TK (Takahiro Kimura) contributed to manuscript editing and supervision.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
EUSP Scholarship of the European Association of Urology (PR).
Conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Takahiro Kimura is a paid consultant/advisor of Astellas, Bayer, Janssen and Sanofi. Shahrokh F. Shariat received follows: Honoraria: Astellas, AstraZeneca, BMS, Ferring, Ipsen, Janssen, MSD, Olympus, Pfizer, Roche, Takeda. Consulting or Advisory Role: Astellas, AstraZeneca, BMS, Ferring, Ipsen, Janssen, MSD, Olympus, Pfizer, Pierre Fabre, Roche, Takeda. Speakers Bureau: Astellas, Astra Zeneca, Bayer, BMS, Ferring, Ipsen, Janssen, MSD, Olympus, Pfizer, Richard Wolf, Roche, Takeda. The other authors declare no conflicts of interest associated with this manuscript.
Information of co-authors
Takafumi Yanagisawa (E-mail: t.yanagisawa.jikei@gmail.com)
Akihiro Matsukawa (E-mail: a.matsu.audi1055g@gmail.com)
Jeremy Yuen-Chun Teoh (E-mail: jeremyteoh@surgery.cuhk.edu.hk)
Keiichiro Mori (E-mail: morikeiichiro29@gmail.com)
Tatsushi Kawada (E-mail: tktk5524@yahoo.co.jp)
Satoshi Katayama (E-mail: skatayama18@gmail.com)
Pawel Rajwa (E-mail: pawelgrajwa@gmail.com)
Fahad Quhal (E-mail: F.Quhal@hotmail.com)
Benjamin Pradere (E-mail: benjaminpradere@gmail.com)
Marco Moschini (E-mail: marco.moschini87@gmail.com)
Shahrokh F. Shariat (E-mail: shahrokh.shariat@meduniwien.ac.at)
Jun Miki (E-mail: junmiki.jikei@gmail.com)
Takahiro Kimura (E-mail: tkimura0809@gmail.com)
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
