Abstract
Background
Enfortumab vedotin (EV) and Sacituzumab govitecan (SG) are antibody-drug conjugates (ADCs) with demonstrated activity in advanced bladder cancer. A subset of bladder tumors harbors a DNA repair deficiency in either the homologous recombination (HR) or nucleotide excision repair (NER) pathway that has the potential to impact sensitivity to specific classes of therapeutics.
Objective
Define the impact of HR or NER deficiency on sensitivity to ADC payloads alone or in combination with DNA repair targeted agents in bladder cancer.
Methods
Isogenic cell pairs with versus without HR or NER deficiency were profiled using DNA repair and drug sensitivity assays. Sensitivity to the ADC payloads monomethyl auristatin E (MMAE) and SN-38 alone or in combination with small molecule inhibitors of poly(ADP-ribose) polymerase (PARP), ATR, or USP1 were measured using cell viability assays.
Results
BRCA2 loss was sufficient to confer an HR deficient phenotype and increase sensitivity to cisplatin and PARP inhibition in bladder cancer cell lines. HR deficiency, but not NER deficiency, increased sensitivity to MMAE and SN-38 in bladder cancer cells. The combination of SN-38 and PARP inhibition displayed synergistic cell killing independent of HR or NER status.
Conclusion
HR and NER deficiency have distinct impacts on sensitivity to cisplatin and ADC payloads in bladder cancer preclinical models.
Keywords
Introduction
DNA repair deficiency is a common feature of tumors and can drive tumor evolution and therapy response. In bladder cancer, alterations in several DNA repair pathway have been characterized. 1 A subset of bladder tumors harbor germline or somatic alterations in a mismatch repair (MMR) gene, resulting in MMR deficiency (MMRd) and a distinctive microsatellite instability (MSI) phenotype characterized by high mutation burden. 2 MMRd/MSI tumors often have a rich immune infiltrate, and the anti-PD1 agent pembrolizumab is approved for MMRd/MSI tumors independent of tumor subtype. 3
Approximately 10% of bladder tumors harbor a somatic missense mutation in the nucleotide excision repair (NER) gene ERCC2. 4 The NER pathway repairs bulky adducts created by genotoxic agents such as ultraviolet radiation and platinum chemotherapies, and several studies have identified an association between somatic ERCC2 mutations and increased response rates to cisplatin-based chemotherapy in muscle-invasive bladder cancer (MIBC).5–8 Functional studies have demonstrated that these clinically observed ERCC2 mutations are sufficient to confer loss of normal NER function and drive cisplatin sensitivity in preclinical bladder cancer models. 6 Together, these data suggest that ERCC2 mutations are one determinant of clinical response to cisplatin-based chemotherapy in MIBC. Mutations in other NER genes (eg ERCC3-6) are less common in bladder cancer, but do occur in a small percentage of cases and may be associated with a similar cisplatin sensitivity phenotype. 9
Homologous recombination (HR) deficiency, often conferred by loss-of-function alterations in BRCA1 or BRCA2, has been extensively studied in certain tumor types including breast, ovarian, pancreatic, and prostate cancers. In bladder cancer, germline or somatic alterations in BRCA1 or BRCA2 are uncommon, occurring in <5% of cases. 4 However, a mutational scar-based signature of HR deficiency is present in ∼20% of bladder tumors, suggesting that HR deficiency may be present in a substantial subset of cases. 10 The presence of a predicted deleterious BRCA1/2 mutation is associated with improved response to cisplatin-based chemotherapy regimens in a bladder cancer clinical cohort, 11 but functional validation of the relationship between HR deficiency and cisplatin sensitivity in bladder cancer systems has not been reported.
An alternative approach to target tumor HR deficiency is through synthetic lethal approaches using DNA repair targeted agents such as poly(ADP-ribose) polymerase (PARP) inhibitors. Several PARP inhibitors are now approved for use in specific clinical settings for HR-deficient breast, ovarian, pancreatic, and prostate tumors. In bladder cancer, initial clinical trials of PARP inhibition did not show activity12,13; however, case series and two recently published clinical trials suggest a potential benefit in subsets of biomarker-selected patients with tumors that harbor a mutation in one or more DNA repair genes.14–16 Therefore, the current role of PARP inhibitors in bladder cancer is uncertain, and the functional relationship among BRCA1/2 alterations, HR deficiency, and PARP inhibitor sensitivity in bladder cancer cells has not been defined.
Beyond PARP inhibition, other DNA repair targeted therapeutic strategies have also been investigated in bladder cancer. ATR (ataxia telangiectasia and Rad3-related) is a serine/threonine kinase that plays a central role in cellular response to DNA damage and replication stress, and ATR inhibitors have been investigated as monotherapy and in combination approaches across a variety of disease contexts. 17 In bladder cancer, the addition of the ATR inhibitor berzosertib to cisplatin and gemcitabine did not improve outcomes in patients with advanced disease. 18
In the past several years, two antibody-drug conjugates (ADCs) have been approved for use in metastatic urothelial cancer. 19 Enfortumab vedotin (EV) combines a nectin-4 targeting antibody with the microtubule destabilizer monomethyl auristatin E (MMAE) while sacituzumab govitecan (SG) combines a trop-2 targeting antibody with the Type 1 topoisomerase inhibitor SN-38. Both EV and SG were approved for use in the post-cisplatin, post-anti-PD1/PD-L1 setting, although the indication for SG was recently withdrawn. EV combined with pembrolizumab is also approved for use in the first-line metastatic setting.20–22
Given the uncertainty regarding the impact of HR or NER deficiency on therapeutic vulnerabilities in bladder cancer, as well as expanding role for ADCs such as EV and SG in bladder cancer, we hypothesized that defining the impact of HR or NER loss on sensitivity to established and emerging bladder cancer therapeutics would provide mechanistic insights that could inform clinical efforts to identify predictive biomarkers and rationale therapeutic combinations. Therefore, we tested the impact of HR and NER deficiency on sensitivity to ADC payloads alone and in combination with PARP inhibitors and other DNA repair targeted agents.
Materials and methods
Cell lines
Human bladder cancer cell lines J82 and KU19-19 were purchased from ATCC and DSMZ, respectively. KE-182 is an ERCC2-mutant derivative of KU19-19 created by CRISPR/Cas9-mediated gene editing. 6 The H460 human lung adenocarcinoma cell line with ERCC4 knockout has also been previously described. 23 The CAPAN-1 (BRCA2-mutant) and CAPAN-1-CR (CAPAN-1 cells with stable expression of wild-type BRCA2) human pancreatic adenocarcinoma cell lines were kind gifts from the Chris Lord laboratory. All cell lines were cultured in RPMI media supplemented with 10% fetal bovine serum, 1% L-glutamine, and 1% penicillin-streptomycin at 37 °C in a 5% CO2 incubator.
siRNA-mediated BRCA2 depletion
Two individual siRNAs targeting the BRCA2 gene (siBRCA2-1, siBRCA2-2) as well as a non-targeting control (siNTC) were purchased from Integrated DNA Technologies Inc (Supplementary Table 1). Cells were seeded at 50% confluency in 6-well plates and transfected with 30 nM siRNA in Opti-MEM media (Gibco) with Lipofectamine 3000 (Life Technologies). Transfected cells were incubated for 48 h, trypsinized, re-plated into 24-well plates at 20,000 cells per well, re-transfected with 15 nM siRNA, incubated for 24 h, then treated with drugs as outlined below.
In vitro drug sensitivity assays
Cells were seeded at 20,000 cells per well in 24-well plates as above. A complete list of drug names and sources are listed in Supplementary Table 1. All drugs were dissolved in DMSO except cisplatin which was dissolved in 0.9% normal saline. Stock solutions of each drug were diluted in a stepwise manner in RPMI media and added to cells in quadruplicate. After 96 h, media was removed and CellTiter-Glo 2.0 Reagent (Promega) was added to each well. The plates were read using a Luminescence Microplate Reader (BioTek). Cell viability was calculated as the ratio of the average luminescence signal at each drug concentration divided by the average luminescence signal of untreated cells. GraphPad PRISM 9.5.1 software was used to plot survival curves and calculate IC50 values using a non-linear regression model. A significant difference in drug sensitivity between two cell lines was defined as non-overlapping 99% confidence intervals for the IC50 values (p < 0.01). For combination treatment experiments, the degree of synergy was calculated using the Combination Index (CI) formula, based on IC50 values for each drug component as a single agent and as part of the combination using CompuSyn software, v1.0 (ComboSyn, Inc.; Paramus, NJ). A CI value less than one indicates synergy whereas a CI value greater than one indicates antagonism. 24 Comparisons between CI values was performed using unpaired 2-sided t-test, with p < 0.01 considered to be statistically significant.
Immunoblotting
Cells were lysed using ice-cold RIPA buffer (Pierce) supplemented with protease and phosphatase inhibitors (Roche). Lysates were sonicated and protein concentrations were measured using the Bradford assay. After adding Sample Buffer (Bio-Rad), the samples were heated to 55-95 °C for 10 min then loaded in 3–8% NuPAGE Bis-Tris gradient gels (Thermo Fisher Scientific) and electrophoresed at 120 V for 3 h. Proteins were then transferred to a 0.45-micron PVDF membrane using a 30 V overnight transfer process. The membrane was then blocked with 5% skim milk in TBST buffer for one hour and probed with BRCA2 (1:500 dilution) and Vinculin (1:1000 dilution) primary antibodies overnight at 4 °C. Antibody details are listed in Supplementary Table 1. Detection of the bound primary antibodies was performed using IRDYE-conjugated secondary antibodies (LI-COR Biosciences) and imaged using a Li-COR Odyssey Infrared Imaging System.
Immunofluorescence
Glass coverslips were inserted into the wells of 12-well plates followed by cell seeding. The following day, cells were irradiated using a cell irradiator (Rad Source), incubated for 45 min, then fixed with 4% paraformaldehyde for 20 min. Fixed cells were then permeabilized on ice for 5 min using 0.2% Triton X-100 in 1% BSA/PBS, blocked with 1% BSA/PBS for one hour at room temperature, then incubated overnight at 4 °C with anti-Rad51 primary antibody (Supplementary Table 1) at a dilution of 1:200. The next morning, a fluorescence-linked secondary anti-Rabbit antibody (Alexa-Fluor 488) was applied at a 1:1000 dilution and the slides were incubated in the dark at room temperature for one hour. The coverslips were then mounted on slides using ProLong Gold Antifade reagent containing DAPI (Invitrogen). Fluorescence microscopy was performed with a Zeiss microscope, and the images were analyzed and quantified with ImageJ software by selecting individual nucleus. R version 4.2.2 was used to create bar graphs and perform Student's t-test to compare results across samples.
Results
Homologous recombination (HR) repair deficiency sensitizes bladder cancer cells to cisplatin and PARP inhibition
Given that the functional impact of HR loss on bladder cancer cell properties has not been directly tested, we performed siRNA-mediated BRCA2 depletion in the KU19-19 and J82 bladder cancer cell lines (Figure 1(a)) and measured the impact on HR function as well as sensitivity to cisplatin and PARP inhibition. BRCA2 depletion resulted in loss of HR function as evidenced by lack of radiation-induced Rad51 foci formation (Figure 1(b)) and significant increase in sensitivity to cisplatin as well as the PARP inhibitors olaparib and talazoparib (Figure 1(c) and (d)). These results provide functional evidence that HR deficiency confers sensitivity to cisplatin and PARP inhibitor therapy in bladder cancer cells.
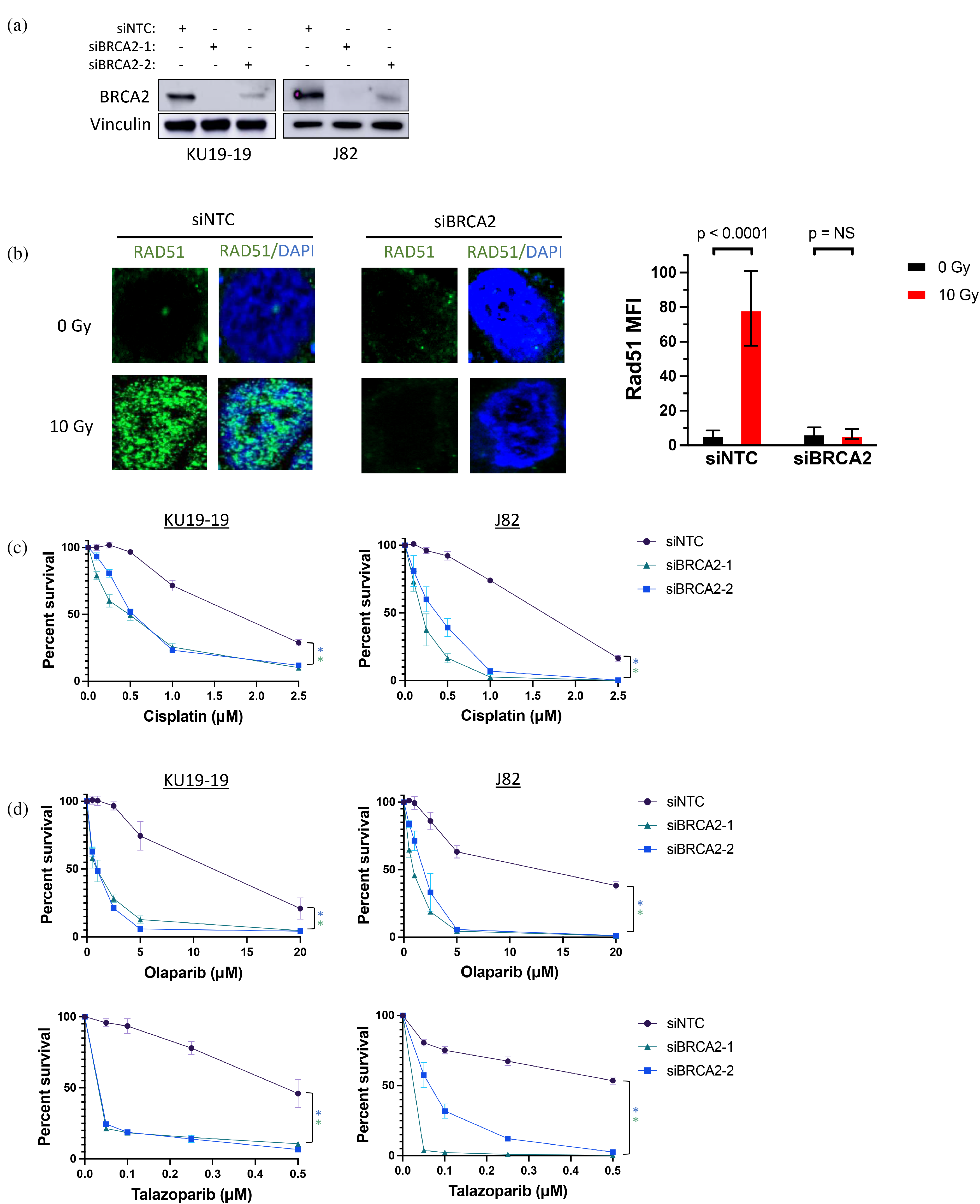
Homologous recombination (HR) deficiency confers cisplatin and PARP inhibitor sensitivity in bladder cancer cells. (a) Immunoblot showing siRNA-mediated depletion of BRCA2 protein in KU19-19 and J82 bladder cancer cell lines. Vinculin is shown as a loading control. (b) Immunofluorescence (IF) microscopy shows loss of radiation-induced Rad51 foci formation in BRCA2-depleted compared to BRCA2-intact bladder cancer cells. (c) Cell viability assays demonstrate increased sensitivity to cisplatin following BRCA2 depletion in bladder cancer cell lines. (d) Cell viability assays demonstrate increased sensitivity to PARP inhibitors olaparib and talazoparib in bladder cancer cell lines. kDa, kilodalton. NTC, non-targeting control. Gy, Gray. MFI, mean fluorescence intensity. *p < 0.01 (IC50 values). Error bars represent standard deviation of data collected from assays performed in triplicate (panel B) or quadruplicate (panels C, D).
HR deficiency sensitizes cells to SN-38 and MMAE
We next wished to test the impact of HR deficiency on sensitivity to MMAE and SN-38, the payloads of EV and SG, respectively. In KU19-19 and J82 bladder cancer cell lines, there was a significant increase in sensitivity to both MMAE and SN-38 in BRCA2-depleted compared to mock-depleted cells (Figure 2(a) and (b)). In addition, we leveraged a HR-deficient BRCA2-mutant pancreatic cancer cell line (CAPAN-1) as well as a derivative of this line in which HR function has been restored by stable expression of wild-type (WT) BRCA2 (CAPAN-1-CR). Similar to the bladder cancer cell lines with engineered HR deficiency, the HR-deficient CAPAN-1 cell line was significantly more sensitive to both MMAE and SN-38 than its engineered HR-proficient CAPAN-1-CR counterpart (Figure 2(c)). Together these results show that cellular HR deficiency increases sensitivity to both MMAE and SN-38.
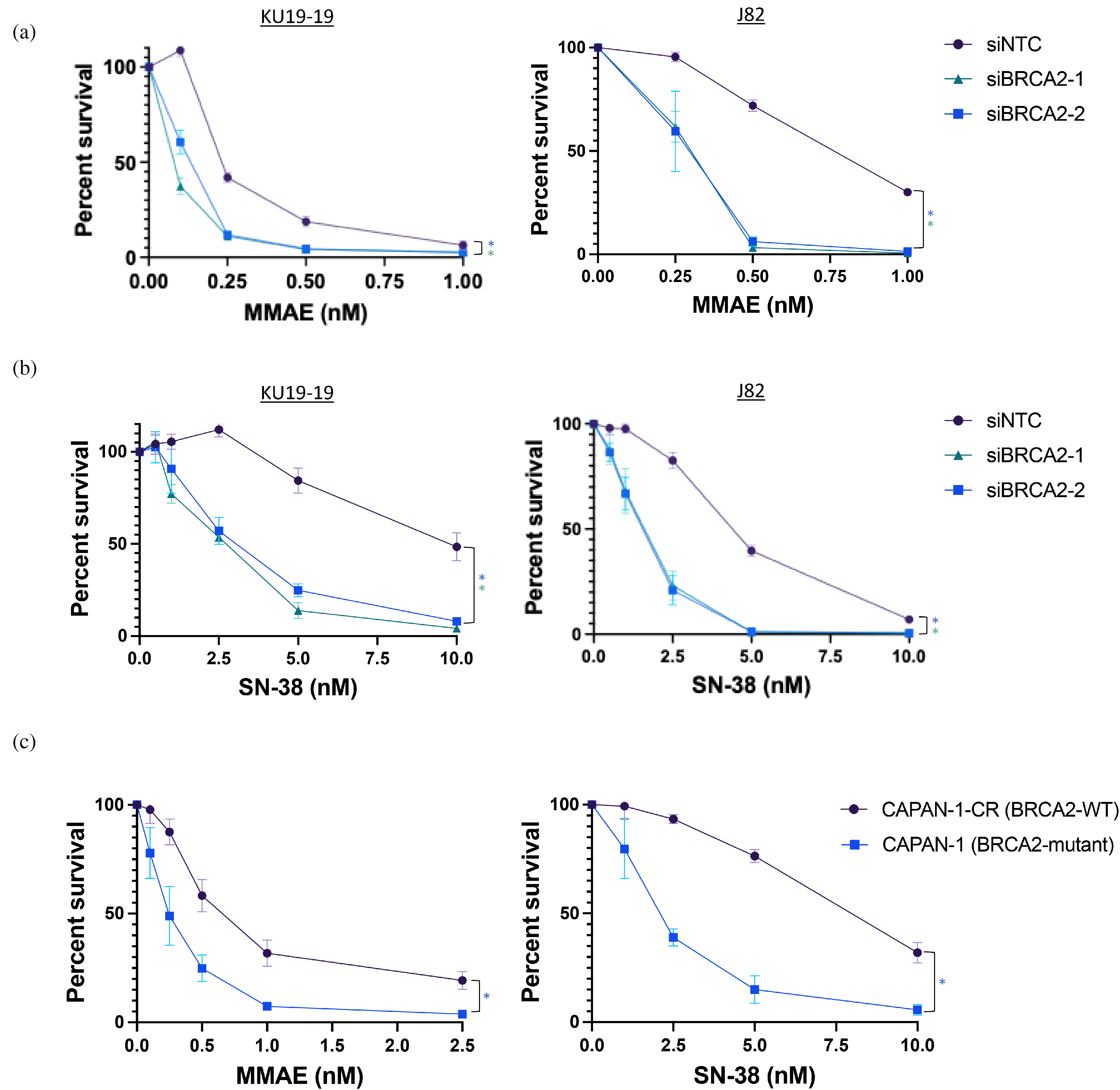
Homologous recombination (HR) deficiency confers increased sensitivity to MMAE and SN-38 in bladder cancer cells. (a) Cell viability assays demonstrate increased sensitivity to MMAE in HR-deficient compared to HR-proficient bladder cancer cell lines. (b) Cell viability assays demonstrate increased sensitivity to SN-38 in HR-deficient compared to HR-proficient bladder cancer cell lines. (c) Cell viability assays demonstrate increased sensitivity to MMAE and SN-38 in HR-deficient CAPAN-1 (BRCA2-mutant) compared to CAPAN-1-CR (BRCA2-WT) pancreatic cancer cell lines. *p < 0.01 (IC50 values). Error bars represent standard deviation of data collected from assays performed in quadruplicate.
NER deficiency does not sensitize cells to SN-38 or MMAE
We next tested the impact of NER deficiency on sensitivity to MMAE and SN-38 in the NER-proficient KU19-19 bladder cancer cell line as well as KE-182, a derivative of KU19-19 with an engineered ERCC2 mutation introduced by CRISPR/Cas9 gene editing. 6 KE-182 is NER-deficient and displays dramatic increase in cisplatin sensitivity (Figure 3(a)). However, unlike bladder cancer cell lines with HR deficiency, the NER-deficient KE-182 cell line did not display increased sensitivity to either MMAE or SN-38 compared to its NER-proficient KU19-19 counterpart (Figure 3(b)). In order to determine the impact of NER deficiency conferred by loss-of-function of NER genes beyond ERCC2, we used an H460 cell line in which ERCC4 had been deleted. 23 Similar to the ERCC2-mutant setting, loss of ERCC4 was sufficient to confer a dramatic increase in cisplatin sensitivity (Figure 3(c)) but did not impact sensitivity to either MMAE or SN-38 (Figure 3(d)). These data demonstrate that NER deficiency drives sensitivity to cisplatin but not MMAE or SN-38 in bladder cancer cells.
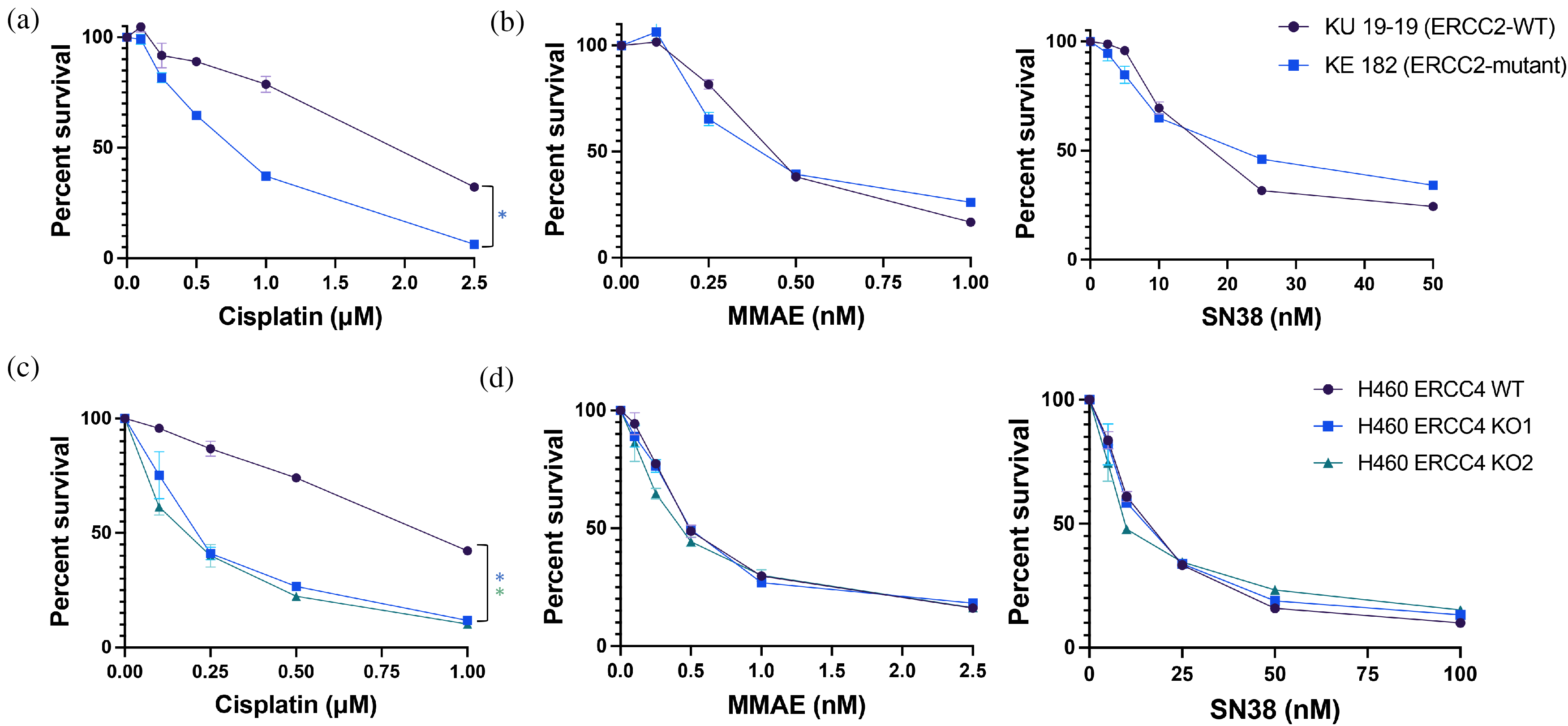
Nucleotide excision repair (NER) deficiency does not increase sensitivity to MMAE or SN-38. (a)-(b) Cell viability assays demonstrate increased sensitivity to cisplatin, but not MMAE or SN-38 in ERCC2-mutant KE182 cells compared to the parental ERCC2-wildtype (WT) KU19-19 cells. (c)-(d) Cell viability assays demonstrate increased sensitivity to cisplatin, but not MMAE or SN-38 in ERCC4-deleted H460 cells compared to parental ERCC4-wildtype H460 cells. *p < 0.01 (IC50 values). Error bars represent standard deviation of data collected from assays performed in quadruplicate.
SN-38 and PARP inhibition are synergistic irrespective of HR or NER status
Given the potential for synergy when combining a cytotoxic agent such as MMAE or SN-38 with a DNA repair targeted agent such as a PARP inhibitor, we next wished to systematically test these drug combinations in the presence or absence of underlying HR or NER deficiency. In addition to PARP inhibition, we also tested combinations with berzosertib, an ATR inhibitor, and ML323, a USP1 inhibitor. ATR is a central DNA damage response (DDR) kinase and ATR inhibition alone or in combination with other DNA repair targeted or cytotoxic agents is being tested in a variety of clinical contexts. 17 USP1 is a de-ubiquitinating enzyme that plays an important role in the DDR and is being investigated as a synthetic lethal target in the HR-deficient tumor setting. 25
We first tested the combination of MMAE or SN-38 with PARP inhibition (olaparib, talazoparib), ATR inhibition (berzosertib), or USP1 inhibition (ML323) in HR-proficient (WT BRCA2) and HR-deficient (BRCA2-depleted) KU19-19 and J82 bladder cancer cell lines (Figure 4(a)). For MMAE combinations, additivity or mild antagonism was observed across drug classes (log10 combination index [CI] 0-1; see Methods). Combining either berzosertib or ML323 with SN-38 was additive; however, synergy was observed when SN-38 was combined with PARP inhibition in the presence or absence of HR deficiency, particularly in the J82 cell line.
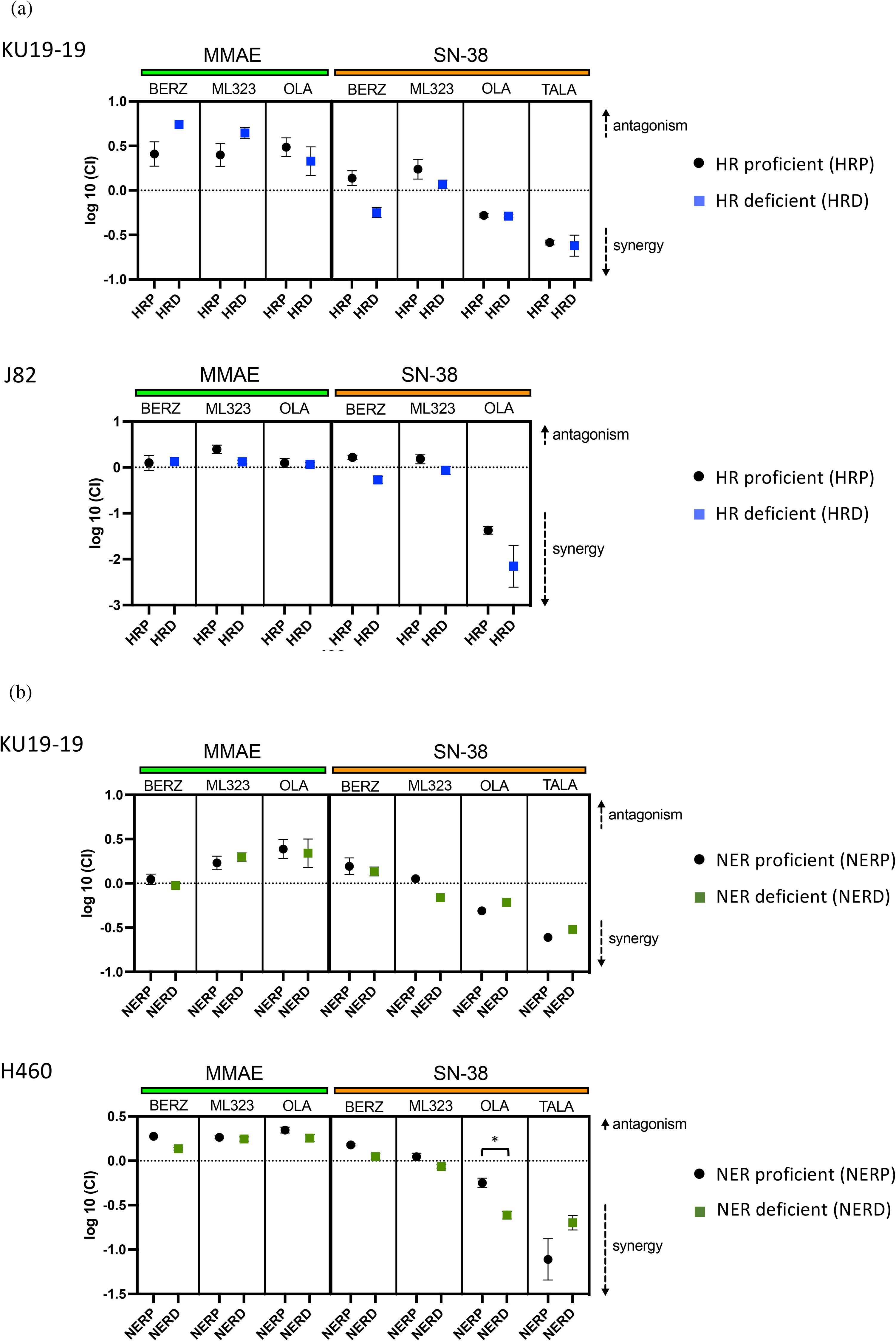
Combined activity of MMAE and SN-38 with DNA repair inhibitors. (a) Cytotoxic activity of combining MMAE or SN-38 with ATR inhibition (berzosertib, BERZ), USP1 inhibition (ML323), or PARP inhibition (Olaparib, OLA; talazoparib, TALA) in KU19-19 (top graph) and J82 (bottom graph) bladder cancer cell lines with versus without HR deficiency conferred by BRCA2 depletion. (b) Cytotoxic activity of combining MMAE or SN-38 with ATR inhibition (berzosertib, BERZ), USP1 inhibition (ML323), or PARP inhibition (Olaparib, OLA; talazoparib, TALA) in NER-proficient (ERCC2 WT) KU19-19 cells versus NER-deficient (ERCC2-mutant) KE182 cells (top graph) or NER-proficient (ERCC4 WT) H460 cells versus NER-deficient (ERCC4-deleted) H460 cells (bottom graph). Combination activity is quantified by the combination index (CI, see Methods) with positive log10(CI) values indicative of antagonism and negative log10(CI) values indicative of synergism. HRP, homologous recombination proficient (WT BRCA2); HRD, homologous recombination deficient (BRCA2 depleted); NERP, nucleotide excision repair proficient; NERD, nucleotide excision repair deficient. Error bars represent standard deviation of data collected from assays performed in quadruplicate.
We next tested the same drug combinations in NER-proficient and NER-deficient cell lines. In both KU19-19/KE182 and H460 ERCC4 WT/KO cell pairs, and similar to HR-proficient/deficient cell pairs, we observed additivity or mild antagonism when combining MMAE with ATR, USP1 or PARP inhibition. Combining SN-38 with ATR inhibition or USP1 inhibition showed additivity. However, similar to the HR-proficient and HR-deficient lines, we observed substantial synergy when combining SN-38 with PARP inhibition. The magnitude of synergy in NER-proficient and NER-deficient cell lines was similar in KU19-19, but significantly more synergy was observed in the NER-deficient H460 cell line than its NER-proficient counterpart (p < 0.01; Figure 3(b)). Across HR and NER cell pairs, talazoparib generally demonstrated greater synergy than olaparib when each was combined with SN-38, possibly due to talazoparib's increased PARP trapping activity. Together, these data provide functional evidence for synergy between PARP inhibition and ADCs possessing the Type 1 topoisomerase inhibitor SN-38.
Discussion
ADCs have transformed the management of metastatic bladder cancer, and both the number and clinical scope of ADCs are likely to expand. 19 Understanding the molecular drivers of ADC response - as well as identifying mechanisms of ADC resistance – are active areas of investigation. ADCs are unique in that tumor response to an ADC can be influenced by the expression level of the target molecule (e.g. nectin-4, trop-2) on the tumor cell surface as well as by the intrinsic sensitivity of the tumor cell to the cytotoxic payload (e.g. MMAE, SN-38). Expression of nectin-4 and trop-2 vary across bladder cancer preclinical models and in clinical cohorts.26–28 Supporting the importance of target expression as a mediator of ADC response, a recent study showed nectin-4 gene amplification was associated with improved EV response in metastatic bladder cancer. 29
There is limited data correlating specific tumor molecular features with sensitivity to MMAE or SN-38. DNA repair pathway deficiency is a common tumor feature and can be therapeutically targeted in several contexts such as use of PARP inhibitors for homologous recombination (HR)-deficient tumors and use of pembrolizumab (anti-PD1) for mismatch repair (MMR)-deficient tumors. Several recent studies have identified subsets of bladder tumors with specific DNA repair pathway deficiencies. 1 Although BRCA1/2 mutations are rare in bladder tumors (∼1–3%), approximately 20% of bladder tumors harbor mutational signatures suggestive of HR deficiency.4,10 Recent studies of PARP inhibitors in advanced bladder cancer suggest activity in patients with tumor features of HR deficiency.14,15 Our functional data demonstrate that BRCA2 loss is sufficient to drive an HR-deficient phenotype and increase sensitivity to both cisplatin and PARP inhibition in bladder cancer preclinical models. To our knowledge, this is the first direct experimental evidence that HR deficiency is sufficient to drive PARP inhibitor sensitivity in a bladder cancer-specific setting and supports further clinical investigation of PARP inhibitor use in bladder cancer cases with known or suspected HR deficiency.
We also observed increased sensitivity to the SG payload SN-38 in the HR-deficient bladder cancer models. Type I topoisomerases create single-strand DNA (ssDNA) breaks to regulate DNA topology, and SN-38 and other type I topoisomerase inhibitors act as poisons by trapping bound topoisomerase at ssDNA breaks. 30 As DNA replication forks encounter the trapped topoisomerase complexes, ssDNA are converted into double-strand DNA (dsDNA) breaks. In HR-proficient cells, these dsDNA breaks are efficiently repaired, whereas in HR-deficient cells, the dsDNA breaks accumulate and lead to cell death.31,32 Given that type I topoisomerases inhibitors such as SN-38 and deruxtecan are payloads in several approved and investigational ADCs, 33 the potential to target HR-deficient tumors using these ADCs is particularly attractive.
Unlike SN-38 and other topoisomerase inhibitors, microtubule destabilizers such as MMAE do not cause direct DNA damage. Rather, MMAE and related compounds destabilize microtubule formation leading to inability to assemble and segregate chromosomes during mitosis, ultimately resulting in mitotic collapse and cell death. 34 Despite having a mechanism of action that does not involve direct DNA damage, we also observed increased sensitivity to MMAE in HR-deficient bladder cancer models; however, the difference in MMAE sensitivity between HR-proficient and HR-deficient models was less dramatic than with either PARP inhibition or SN-38 treatment. It is possible that the increase in dsDNA breaks resulting from HR deficiency may lead to concomitant increase in baseline DNA damage-induced apoptotic signaling, thereby potentiating the effect of shared downstream apoptotic signaling induced by MMAE. Correlative studies investigating the genomic correlates of EV response in completed and on-going clinical studies will likely help clarify the relationship between HR deficiency and EV response in bladder cancer. 35
Approximately 10% of bladder tumors exhibit nucleotide excision repair (NER) deficiency conferred by somatic mutations in the NER gene ERCC2, and ERCC2-mutant bladder tumors display increased sensitivity to platinum-based chemotherapy in clinical cohorts and preclinical models.6–8 However, we did not observe increased sensitivity to either MMAE or SN-38 in our NER-deficient preclinical models. This is consistent with other mechanistic studies showing that the NER pathway is specific for bulky intrastrand lesions and is not the primary pathway responsible for repairing DNA-protein crosslinks as induced by SN-38 or mitotic catastrophe as induced by MMAE.6,36 These results suggest that despite the expanding role for ADCs, cisplatin-based regimens may still be useful in patients with ERCC2-mutant bladder tumors, such as the combination of gemcitabine-cisplatin plus nivolumab, which was recently FDA-approved in the first-line setting for advanced bladder cancer patients. 37 The potential to use mutations in ERCC2 and other DNA repair genes to guide therapy in the MIBC setting has also been investigated in several trials, including the RETAIN trial 38 as well as the on-going Alliance A0 trial. 39
Given the potential for synergistic interactions, we also tested the activity of SN-38 and MMAE in combination with multiple DNA repair inhibitors in both HR- and NER-proficient /deficient cell pairs. One of the notable findings from these combination studies was the synergy observed between SN-38 and PARP inhibition across cell models and irrespective of HR or NER status. The PARP1 protein plays a crucial and multifaceted role in response to ssDNA breaks, including those resulting from topoisomerase I inhibitors, 40 providing mechanistic rationale for the observed synergism in both HR-proficient and HR-deficient contexts. Synergistic cell toxicity has also been previously reported in non-bladder cancer systems 41 and together these data provide preclinical evidence to support clinical efforts to combine topoisomerase I inhibitor-based ADCs with PARP inhibition.
Unlike SN-38, combining MMAE with PARP inhibition did not demonstrate synergism in any cell line context; instead, modest antagonism was observed in some cell lines as well as with ATR and USP1 inhibitor combinations. Further work will be required to validate these findings and characterize potential mechanisms of antagonism. Currently, EV is approved for use in combination with pembrolizumab (anti-PD1) in the first-line setting for advanced bladder cancer, 42 and future studies will need to involve approaches that attempt to build upon the success of EV plus pembrolizumab.
Our study is limited by the use of a limited number of preclinical models that do not fully recapitulate the clinical or molecular heterogeneity of bladder cancer. Furthermore, although we chose to model HR deficiency by using models with BRCA2 depletion or mutation, multiple mechanisms of HR deficiency exist across tumors and may be associated with distinct patterns of response to PARP inhibitors or other agents. Despite these caveats, our preclinical findings provide a basis for prioritizing additional in vitro and in vivo preclinical studies as well as clinical efforts to identify reliable predictive biomarkers and rational combination approaches to personalize treatment approaches for bladder cancer.
Supplemental Material
sj-pdf-1-blc-10.1177_23523735251317865 - Supplemental material for Impact of DNA repair deficiency on sensitivity to antibody-drug conjugate (ADC) payloads in bladder cancer
Supplemental material, sj-pdf-1-blc-10.1177_23523735251317865 for Impact of DNA repair deficiency on sensitivity to antibody-drug conjugate (ADC) payloads in bladder cancer by Surish P Shanmugam, Yuzhen Zhou, Isabella Stelter, Timothy Hanlon, Raie T Bekele, Joaquim Bellmunt, Zoltan Szallasi and Kent W Mouw in Bladder Cancer
Footnotes
Acknowledgements
The authors have no acknowledgments.
Author contributions
Conceptualization: SPS, ZS, KWM. Methodology: SPS, YZ, JB, ZS, KWM. Resources: SPS, YZ, RTB, KWM. Investigation: SPS, YZ, TH, IS. Supervision: JB, ZS, KWM. Writing: SPS, KWM. Editing: SPS, YZ, TH, IS, RTB, JB, ZS, KWM.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by NCI R21CA288234 (to KWM).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mouw:
Advisory/Consulting: EMD Serono, Pfizer, UroGen, Riva Therapeutics, Natera
Research Support: Pfizer, Novo Ventures
Equity: Riva Therapeutics, Illudent Therapeutics
Writing/editor fees: UpToDate
Speaking fees: OncLive
Patents: Institutional patent filed on mutational signatures of DNA repair deficiency
Editorial boards: Science Advances
Bellmunt:
Stock and Other Ownership Interests: Rainier
Honoraria: UpToDate
Consulting or Advisory Roles: Pierre Fabre, Pfizer, Merck, Novartis, AstraZeneca/MedImmune, Bristol-Myers Squibb, EMD Serono/Merck
Research Funding: Pfizer/Gilead, Pfizer/EMD Serono
Patents, Royalties, Other Intellectual Property: UpToDate Bladder Cancer
Travel, Accommodations, Expenses: Ipsen, Genentech/Roche
Editorial board: Editorial Board member of this journal, but was not involved in the peer-review process nor had access to any information regarding its peer-review.
Szallasi: Z. Szallasi is an inventor on a patent used in the myChoiceHRD assay issued and with royalties paid.
All other authors declare no conflicts of interest.
Data availability
The data supporting the findings of this study are available within the article and/or its supplementary material.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
