Abstract
INTRODUCTION:
Antibody drug conjugates represent a promising class of antineoplastic agents comprised of a monoclonal antibody linked to a potent cytotoxic payload for targeted delivery of chemotherapy to tumors. Various antibody drug conjugates have demonstrated impressive efficacy in patients with metastatic urothelial carcinoma in clinical trials, leading to two FDA approved therapies and several other agents and combinations in clinical development.
MATERIALS AND METHODS:
A comprehensive systematic review was undertaken utilizing the principles of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. Queried databases included Ovid MEDLINE, Ovid Embase, Web of Science Core Collection and Cochrane CENTRAL Trials. The search sought to identify prospective therapeutic clinical trials in humans with metastatic urothelial carcinoma with a single-arm or randomized controlled trial design investigating antibody drug conjugate-containing regimens.
RESULTS:
The literature search yielded 4,929 non-duplicated articles, of which 30 manuscripts and conference abstracts were included, which derived from 15 clinical trials including 19 separate cohorts with efficacy outcome results. Eleven trials investigated ADC monotherapy, while two investigated combination regimens, and the remaining two studies were mixed. Five unique ADC targets were represented including Nectin-4, Trop-2, HER2, Tissue Factor, and SLITRK6. Twelve clinical trial cohorts required prior treatment (63%). Objective response rate was reported for all studies and ranged from 27–52% for ADC monotherapies and 34–75% for ADC plus anti-PD-1 agents. Time to event outcome reporting was highly variable.
CONCLUSION:
In addition to enfortumab vedotin and sacituzumab govitecan, various HER2-targeted antibody drug conjugates and ADC-anti-PD-1 combination regimens have demonstrated efficacy in clinical trials and are poised for clinical advancement.
Keywords
INTRODUCTION
Approximately 5% of patients with urothelial cancer (UC) present with de novo metastatic disease [1]. However, a proportion of patients initially diagnosed with localized disease, especially muscle invasive UC, will develop distant recurrence despite adequate initial therapy [2, 3]. The 5-year survival for cisplatin-eligible patients treated with first-line cisplatin-based combination regimens is approximately 13–15%, and these data predate the dawn of immunotherapy, antibody drug conjugates, and effective targeted therapy in this disease [4]. Unfortunately, up to 50% of patients are cisplatin-ineligible at diagnosis leading to a tempered five-year survival of 6% for all-comers with de novo metastatic UC in the United States [1, 5].
Despite these poor historical outcomes, treatment advances have ushered in a new era for patients with metastatic UC. Maintenance avelumab (anti-PD-L1) in patients with at least stable disease after first-line chemotherapy, represents the current first-line standard for all platinum- and IO-eligible patients with metastatic UC [6]. Pembrolizumab is approved for treatment-naïve, platinum-ineligible patients with metastatic UC, and PD-1 axis inhibitors may also be used in the second line for patients with progressive disease after initial chemotherapy [8–12]. Finally, erdafitinib, an FGFR inhibitor, has been FDA approved for patients with previously-treated metastatic UC harboring specific oncogenic FGFR2/3 alterations [13].
Novel immuno-oncology (IO) and targeted therapies have clearly revamped the metastatic UC treatment paradigm. However, one can argue that the most interesting therapeutic class in this space in recent years has been the antibody drug conjugates (ADC) for which there are two currently approved agents, enfortumab vedotin and sacituzumab govitecan. In addition, there is a myriad of promising agents and combinations on the horizon. ADCs are multifaceted molecules comprising a molecularly targeted monoclonal antibody portion and a potent cytotoxic payload joined by a chemical linker. The linker can be either indefinitely fixed between the antibody and payload (non-cleavable) or cleavable under predictable conditions allowing for controlled dissociation of the ADC constituents. ADCs are designed for targeted delivery of potent cytotoxic compounds to cancer cells while minimizing off-tumor toxicity. In this systematic review, we will analyze the developmental landscape of ADCs in metastatic urothelial carcinoma including an appraisal of clinical trial data for FDA approved ADCs, investigational compounds, and novel ADC-based combinations. As a detailed understanding of this therapeutic class is integral to the modern management of patients with metastatic UC, in addition to focusing on efficacy outcomes, we will also review pertinent concepts of target selection, drug sequencing, and unique pharmacologic properties and toxicities of select ADCs in metastatic UC.
METHODS
Systematic review
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines [14]. The protocol has been registered in the PROSPERO international prospective register of systematic reviews under the ID CRD42022367738 a priori.
Search strategy
A clinical librarian designed the systematic search strategy following a consultation with the research team using the research team’s protocol as a framework. The librarian also performed a Medical Subject Heading (MeSH) analysis of pre-identified reference articles provided by the research team. These articles were later used to validate search concepts. The search strategy then was peer reviewed by a senior medical librarian. Ovid MEDLINE, Ovid Embase, Web of Science Core Collection, and Cochrane Central Trials databases were searched to find relevant articles published from the inception of each database to November 4, 2022. Final searches were performed in all databases on this date. Databases were searched using a combination of controlled vocabulary and free text terms for urinary bladder neoplasms, antineoplastic agents, antibody drug conjugate, and randomized controlled trials or controlled clinical trials. The search was not limited by language or year. The final search found a total of 5,705 studies with 4,929 original articles. These results were exported into EndNote 20 (Clarivate Analytics, Philadelphia, PA), where they were de-duplicated, and then uploaded to Covidence Systematic Review software (Melbourne, Australia) for screening.
Select conferences, including American Society of Clinical Oncology (ASCO) Annual Meeting, ASCO Genitourinary Cancers Symposium, American Association for Cancer Research Annual Meeting, and the European Society of Medical Oncology Congress were reviewed for abstracts meeting inclusion criteria from 2015–2022, to find additional relevant studies not retrieved by the database search.
Inclusion criteria
Prospective therapeutic clinical trials in humans (phase I-IV) with a single-arm or randomized controlled trial design investigating an ADC monotherapy or combination therapy including an ADC were included. Trials were included if they enrolled patients with locally advanced unresectable or metastatic urothelial carcinoma, either in dedicated cohorts or, in the case of advanced solid tumor studies, if there were at least 10 patients with locally advanced unresectable or metastatic urothelial carcinoma treated with the ADC-containing regimen. Trials including patients with treatment-naïve or previously treated disease were included. Only trials with clinical efficacy outcome results were included.
Exclusion criteria
Case reports, case series, systematic reviews, meta-analyses, retrospective analyses, preclinical studies, or animal studies were excluded. Studies including similar therapeutic classes, such as bispecific antibodies or monoclonal antibodies were excluded.
Selection and data extraction
After the search was performed, a web-based software platform, Covidence, was used to streamline the systematic review. References were imported and two reviewers (authors MJG and PS) screened citations imported into the Covidence platform based on the title and abstract, with full-text interrogation against the inclusion/exclusion criteria as necessary. Conflicts were reconciled via discussion between the co-authors. Publications containing repeat analyses of the same trial population with longer follow-up were included, but Table 1 synthesizes this information by trial cohort. Data extraction was conducted using full text articles into a Microsoft Excel 2020 (Microsoft Office, Microsoft Corporation, Redmond, WA, USA) document by co-authors MJG and PS.
Clinical trials of antibody drug conjugates in metastatic urothelial carcinoma
Abbreviations: Cis = cisplatin; RCT = randomized controlled trial; n = number of patients per cohort; Tx = treatment; ORR = objective response rate; DoR = duration of response; PFS = progression free survival; OS = overall survival; TRAE = treatment related adverse event. *Based on entire study population for NCT01631552 not solely patients with metastatic urothelial carcinoma.
RESULTS
The systematic review identified 30 unique conference abstracts and manuscripts representing analyses of prospective clinical trials with efficacy outcome data investigating ADC-containing regimens for the treatment of patients with locally advanced unresectable or metastatic urothelial carcinoma (Fig. 1) [15–44]. These 30 records comprised 15 different clinical trials with 19 distinct cohorts which are detailed in Table 1. Of the 15 trials included, 93% were phase I and/or phase II studies, while one trial (EV-301) had a phase III design. Eleven (73%) of trials investigated ADC monotherapy, while two (13%) investigated ADC combinations, and the remaining two studies had multiple treatment arms with monotherapy and combination regimens (EV-103 and TROPHY-U-01). All combination studies investigated regimens consisting of an ADC and a PD-1 inhibitor. There were 5 unique ADC targets- Nectin-4, Trop-2, HER2, Tissue Factor, and SLITRK6 across trials. Biomarker-selected cohorts were almost solely limited to HER2 IHC, although one study required Nectin-4 expression (EV-101). Prior treatment was heterogeneous as were cisplatin eligibility requirements. Twelve clinical trial cohorts required prior treatment (63%), specifying at least “standard chemotherapy” or “platinum-chemotherapy” in all but two of these (83%). These two cohorts specified prior treatment with anti-PD-1 or ant-PD-L1 agents only (EV-201 cohort 2, TROPHY-U-01 cohort 2). There were three cohorts from one study (EV-103) which strictly enrolled treatment-naïve patients.
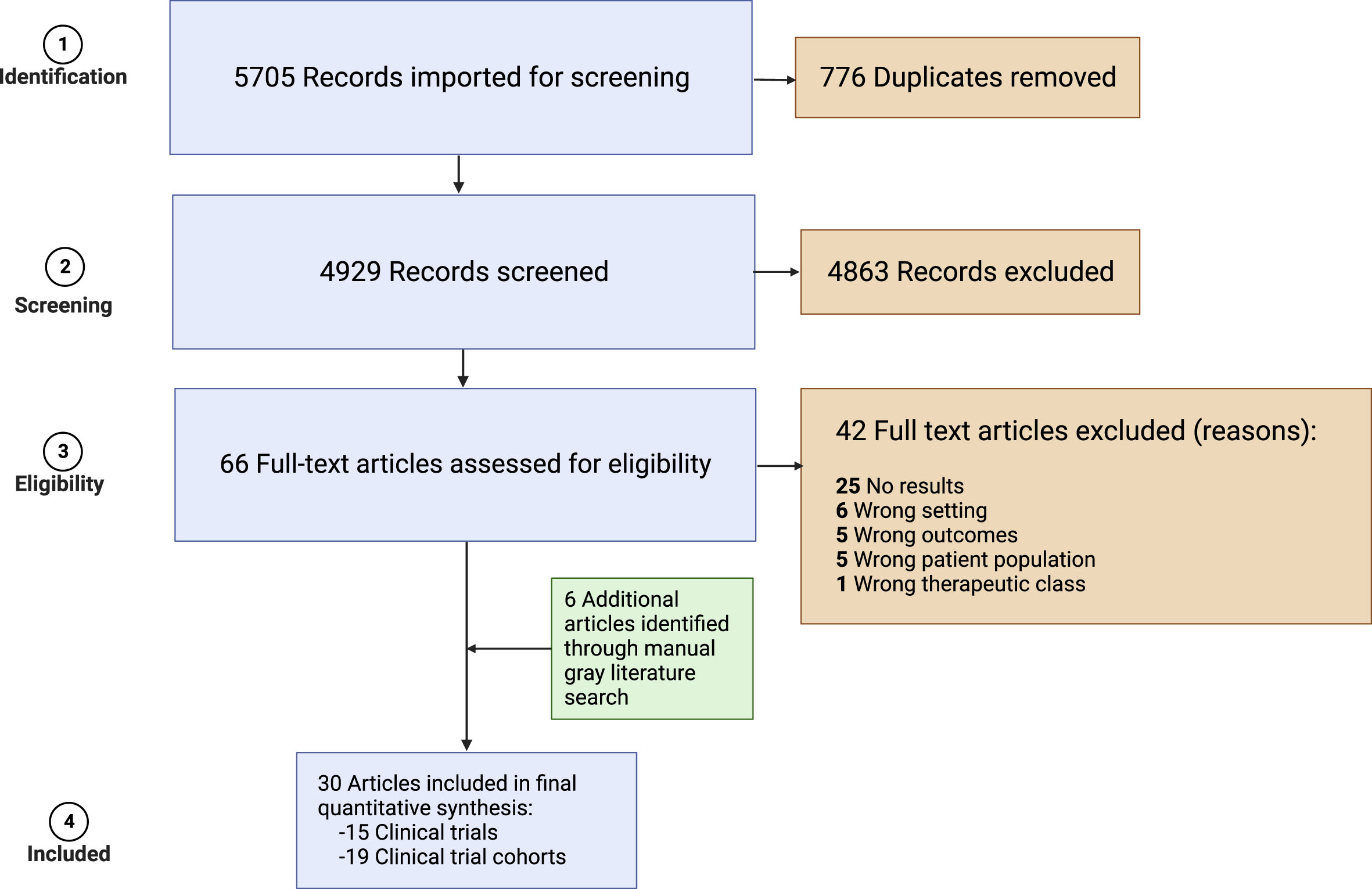
PRISMA 2009 flow diagram.
Duration of follow-up was reported for 14/19 cohorts presenting efficacy data (74%). The longest follow-up was 24.9 months for enfortumab vedotin plus pembrolizumab (EV-103). Four cohorts had a duration of follow-up≤6 months. All included studies presented data on ORR which ranged from 27–52% for ADC monotherapies and 34–75% for ADC plus anti-PD-1 agents (note that this is a range of ORRs from individual trials and not generated by formal meta-analytical techniques). For ADC monotherapy trials in strictly cisplatin eligible populations, ORR ranged from 25–51%. Sacituzumab govitecan (TROPHY-U-01 Cohort 2) and enfortumab vedotin (EV-201 Cohort 2) were evaluated for cisplatin ineligible patients with ORR of 28% and 52%, respectively. Two trials evaluated ADC plus anti-PD-1 agents in previously treated cohorts with ORR 34% for sacituzumab govitecan plus pembrolizumab and 37% for trastuzumab deruxtecan plus nivolumab. Finally, enfortumab vedotin plus pembrolizumab was the only combination regimen evaluated for cisplatin ineligible patients in two separate cohorts with ORR 65% in EV-301 Cohort K and EV-103.
Time to event outcome (DOR, PFS, OS) reporting was highly variable. PFS was the most reported time to event endpoint in 12/19 cohorts (63%), followed by duration of response in 11/19 cohorts (58%), and overall survival in 9/19 cohorts (47%). Detailed data on these outcomes for each ADC or ADC combination arm are presented in Table 1. Toxicity outcome reporting was also highly variable; the percentage of Grade≥3 TRAEs was reported in 11/19 cohorts (58%).
DISCUSSION
ADCs represent a unique therapeutic class with targeted cytotoxic delivery which has transformed the management of metastatic urothelial carcinoma. The developmental landscape for metastatic UC is advancing as these agents progress from early to late phase clinical trials, move into earlier treatment settings for certain patients, and are incorporated into combination strategies with other therapeutic classes. The targets that have been most fruitful have been Nectin-4 (enfortumab vedotin) and Trop-2 (sacituzumab govitecan), although the HER2-targeted ADCs have also demonstrated promising early results. Furthermore, recently presented results from ADC-IO combination trials are generating a great deal of excitement and are worthy of discussion as well.
Enfortumab vedotin- Nectin-4 ADC
The development of enfortumab vedotin, an ADC targeting Nectin-4, has been an outstanding triumph in the modern era of metastatic UC. Nectin-4 is a cell-surface protein with physiologic roles in maintaining adherens junctions, regulating JAK-STAT signaling, and serving as an entry point receptor for the measles virus [45–48]. Although several normal tissues display weak to moderate Nectin-4 expression, the protein is overexpressed on virtually all malignant urothelial cells [31, 49]. Though Nectin-4 is not definitively an oncogenic driver, downstream signaling coincides with activation of the PI3K-AKT pathway [48]. Collectively, these properties make Nectin-4 a promising ADC target in bladder cancer.
Enfortumab vedotin is comprised of a high affinity human IgG4 anti-Nectin-4 monoclonal antibody conjugated to MMAE, a microtubule destabilizer, via a cathepsin B–cleavable peptide linker (Fig. 2) [49, 50]. While unconjugated anti-nectin monoclonal antibodies lack cytotoxic activity in Nectin-4 overexpressed tumors, preclinical models demonstrating efficacy for the ADC supported moving this agent into clinical studies [49]. The phase I dose escalation/ dose expansion study, EV-101, enrolled patients with previously treated solid tumors with Nectin-4 overexpression by immunohistochemistry [31]. Ultimately, this study demonstrated that Nectin-4 was ubiquitously overexpressed on metastatic UC tumors, established 1.25 mg/kg on days 1, 8, and 15 of a 28-day cycle as the recommended phase II dose, and characterized the predominantly on-target and MMAE-related toxicity profile of enfortumab vedotin at this dose level. Commonly occurring treatment related adverse events (TRAE) included rash, peripheral sensory neuropathy, fatigue, alopecia, nausea, and diarrhea. Mostly, these were grade 1 and 2, but 10% of the population discontinued therapy and 4% suffered death attributable to TRAEs. In line with preclinical studies, impressive clinical efficacy punctuated these phase I results. Most patients with metastatic UC experienced some degree of tumor regression, with 43% meeting criteria for objective response (38% PR, 5% CR) and a median duration of response (DOR) of 7.3 months.
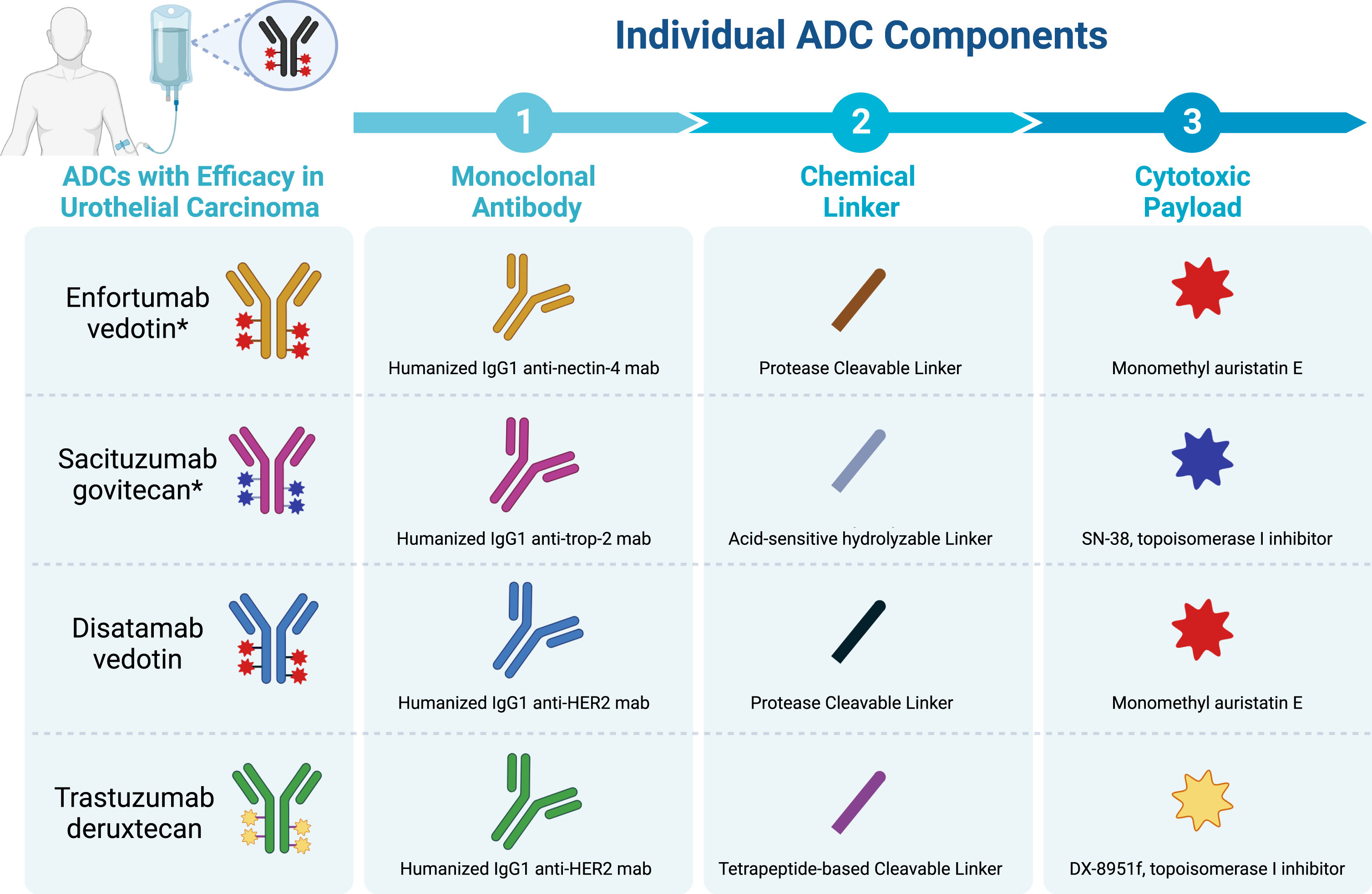
Select Antibody Drug Conjugates with efficacy in metastatic urothelial carcinoma and their constituent monoclonal antibodies, linkers, and payloads. Abbreviations: ADC = antibody drug conjugate; mab = monoclonal antibody. *US FDA approved.
The overall tolerability and striking phase I efficacy of enfortumab vedotin among an historically challenging population in EV-101 set the stage for ensuing phase II and III clinical trials. EV-201 (NCT03219333) was a single arm phase II study investigating enfortumab vedotin in two separate cohorts with locally advanced or metastatic urothelial carcinoma [29]. Cohort 1 included patients previously treated with platinum-based regimens and PD-1 axis inhibitors, and further demonstrated efficacy of enfortumab vedotin in this setting with an objective response rate (ORR) of 44% (32% PR, 12% CR), median DOR 7.6 months, and median overall survival of 12.4 months [29]. Cohort 2 included patients previously treated with PD-1 axis inhibitor therapy who were cisplatin-ineligible and platinum-naïve; in this population ORR was 52% (31% CR, 20% PR) with a median DOR of 10.9 months [38]. The toxicity profile mirrored that of enfortumab vedotin in EV-101.
Finally, EV-301 was a large phase III randomized controlled trial comparing enfortumab vedotin monotherapy against investigator choice chemotherapy for patients with locally advanced or metastatic UC previously treated with platinum regimens and PD-1 axis inhibitor therapy. This study demonstrated an improvement in median OS of 4 months and a 30% reduction in the risk of death favoring the enfortumab vedotin arm and sustained at a median follow-up of 24 months [25, 30]. Notably, subgroup analyses have demonstrated that enfortumab vedotin affords clinical benefit over chemotherapy in clinically challenging subgroups (eg. Patients with age≥65, liver metastases, upper tract disease, and primary IO resistance) and is similarly efficacious in patients with and without FGFR 2/3 alterations [25, 51].
Based on the results of EV-201 and EV-301, the US FDA granted accelerated approval and subsequently regular approval for the use of enfortumab vedotin in previously treated patients with locally advanced or metastatic UC who received at least PD-1 axis inhibitor therapy and who may or may not have received platinum chemotherapy based on cisplatin eligibility [52, 53]. Enfortumab vedotin is also under investigation in previously untreated, cisplatin-ineligible patients with metastatic UC. In this setting, the ORR was 45% with a duration of response of 13.2 months [28].
Sacituzumab govitecan- TROP2 ADC
Trophoblast cell surface antigen-2 (TROP-2) is a cell-surface protein highly expressed on various epithelial carcinomas including UC; TROP-2 overexpression is associated with activation of the ERK-MAPK pathway leading to cell cycle progression, proliferation, and cell survival [54]. Various ADCs targeting TROP-2 are being evaluated in clinical trials, sacituzumab govitecan being furthest along in development at the time of preparation of this manuscript. This agent has received US FDA approval for previously treated patients with metastatic triple negative breast cancer as well as for patients with platinum- and PD-1 axis inhibitor relapsed/refractory metastatic UC, and it is being investigated in various other solid tumors including small cell and non-small cell lung cancer and hormone receptor positive breast cancer among others [55–57].
Whereas enfortumab vedotin contains a protease-cleavable linker, sacituzumab govitecan’s is a CL2A pH sensitive linker characterized by intermediate stability in the serum [58, 59]. This leads to release of payload within TROP2-positive cells as well as in the tumor microenvironment to enhance the bystander effect on neighboring tumor cells which may not be bound by the humanized RS7 monoclonal antibody portion of the drug. The payload for sacituzumab is SN-38, the active form of topoisomerase I inhibitor prodrug, irinotecan. As ADC payloads go, SN-38 is less potently cytotoxic allowing for its coupling to a more readily cleavable linker and allowing for tolerance of a higher drug-antibody ratio (DAR) [58].
After demonstrating preclinical efficacy against a range of solid tumors and tolerance of sacituzumab govitecan in animal models, this agent was investigated in a first-in-human phase I study which enrolled 25 patients with treatment refractory advanced solid tumors. Adverse events were characterized by familiar SN-38-related toxicity such as neutropenia, nausea, vomiting, fatigue, alopecia, and diarrhea and evidence of cytoreduction was demonstrated across diverse tumor types, although the sole patient with heavily pretreated metastatic UC experienced primary progression after 4 cycles at a starting dose of 12 mg/kg. Ultimately the RP2D of 10 mg/kg on days 1 and 8 of 21 was selected and used as the starting dose for the ensuing multi-tumor phase I/II study including patients with metastatic UC who had progressed on at least one line of therapy [60]. Nearly 1/3 of patients with mUC achieved an objective response to sacituzumab govitecan with comparable response rates in subgroups with visceral metastases (ORR 27%) and patients previously treated with PD-1 axis inhibitors (ORR 23%). These promising results prompted further investigation in a global phase II study, TROPHY-U-01. While this was a multicohort phase II trial investigating sacituzumab-based regimens in various clinical settings for patients with metastatic UC, cohort 1 included 113 patients treated with sacituzumab govitecan monotherapy who were previously treated with platinum chemotherapy and a PD-1 axis inhibitor. The ORR was 27% among this cohort with a duration of response of 7.2 months and a median overall survival of 10.9 months. Again, neutropenia and diarrhea were among the most encountered grade≥3 adverse event rates of 35% and 10% respectively.
Based on the distinct targets, linker characteristics, and payloads of enfortumab vedotin and sacituzumab vedotin, one would expect that patients treated with these agents should not be susceptible to cross-resistance. In fact, this is substantiated by preclinical findings demonstrating that enfortumab vedotin-resistant cells maintain expression of TROP2 despite diminished Nectin-4 expression and remain sensitive to sacituzumab govitecan treatment [61]. Finally, thorough investigation into clinical response and resistance dynamics in sacituzumab govitecan-treated patients with triple negative breast cancer have yielded molecular findings that may ultimately lead to strategies to prevent, delay, or circumvent drug resistance [62]. Genomic alterations leading to qualitative or quantitative changes in TROP2 as well as alterations affecting the target of SN-38, Topoisomerase I, were shown to mediate clinical acquired resistance based on pre-treatment and post-progression comparisons, and this may happen in a polyclonal fashion [62]. More work is needed to establish the frequency of such alterations to determine the wide-scale clinical implications and develop novel management approaches.
HER2 ADCs in bladder cancer
HER2 (erbB-2) is a receptor tyrosine kinase most notably overexpressed on hormone-receptor negative breast cancer cells, leading to heterodimerization with other epidermal growth factor family receptors and ultimately proliferative signaling and oncogenesis [63, 64]. As the arsenal of HER2-directed therapies has expanded beyond the monoclonal antibodies, so too has the role for this therapeutic class in the solid tumor treatment landscape, namely for gastroesophageal tumors with HER2 overexpression, non-small cell lung cancers with HER2 mutations, and breast cancers with low HER2 expression [65–69].
HER2 is overexpressed in 12.4% of UC [65, 70]. Moreover, potentially targetable HER2 mutations in UC may be underrecognized with a prevalence of 11% and are enriched in tumors with micropapillary histology [71–73]. Despite considerable interest in HER2 as a target in UC, trials involving kinase inhibitors, monoclonal antibodies, and even the ADC trastuzumab emtansine (T-DM1) have generated underwhelming efficacy results or concerning toxicity profiles [74, 75].
Disatamab vedotin (RC-48) is an ADC with a humanized anti-HER2 monoclonal antibody joined via a cleavable linker to MMAE. In a phase I study of disatamab vedotin in patients with HER2 overexpressing tumors, this agent showed an acceptable safety profile and led to the selection of 2.5 mg/kg every 2 weeks as the recommended phase 2 dose [76]. There were 57 patients enrolled, the majority of whom had gastric adenocarcinoma (n = 47), while 4 patients with UC were included. However, the disease control rate among the UC patients was 100% with all patients experiencing cytoreduction and 2 patients achieving partial responses to therapy. A pooled analysis of two phase II trials investigating disatamab vedotin in patients with HER2-positive metastatic UC previously treated with at least chemotherapy confirmed efficacy in this population [41]. In this analysis, 107 patients with HER2 immunohistochemistry (IHC) 2 + or 3 + demonstrated an ORR of 50.5% (95% CI, 40.6–60.3%). In patients with IHC 2 + with HER2 amplification by FISH or HER2 IHC 3+, the ORR was 62.2% whereas for patients with HER2 IHC 2+, the ORR was 39.6%. The median PFS for the entire cohort was 5.9 months (95% CI, 4.2–7.2) with median OS 14.2 months (95% CI, 9.7–18.8). Grade≥3 neutropenia occurred in 12.1% of patients. Promising combinations with HER2 ADCs and anti-PD-1 agents are also in clinical trials for HER2 overexpressing metastatic UC.
Additional ADC targets
In 2016, results from a phase I dose escalation study of AGS-15E in patients with metastatic UC were presented [42]. This ADC targets SLITRK6, a transmembrane protein expressed at high levels in UC [77]. AGS-15E is composed of a SLITRK6-specific human monoclonal IgG conjugated with MMAE via a protease-cleavable dipeptide linker [77]. Overall, the phase I study demonstrated a modest ORR of 30% with 51% grade≥3 adverse events in previously treated patients with metastatic UC. There has not been any further study of this agent beyond this phase I trial. Another agent, tisotumab vedotin, has been investigated in patients with metastatic UC [18]. This ADC targets tissue factor, a transmembrane glycoprotein whose role has been primarily established in the coagulation pathway, although it is also aberrantly expressed and involved in proliferative cell signaling in various malignancies including bladder cancer. The protease cleavable linker of tisotumab vedotin facilitates delivery of MMAE after lysosomal trafficking, ultimately leading to disruption of microtubule polymerization. In the phase I/II InnovaTV-201 trial, 15 patients with metastatic UC were included among the larger cohort of patients with advanced solid tumors. The ORR for the metastatic UC cohort was 27% with median follow-up of just 5.8 months for the entire study population. Grade≥3 TRAEs were experienced by 41%, mostly consistent with payload toxicity; coagulation parameters were unaffected by this agent. Ongoing investigation with tisotumab vedotin is focused primarily on patients with advanced cervical cancer (NCT03786081, NCT04697628).
ADC combination strategies
For metastatic UC, other therapeutic classes such as PD-1 axis inhibitors and FGFR TKIs have demonstrated single-agent activity in certain populations as well. These agents are ripe for combination approaches with ADCs, with the potential for synergistic activity.
Certain properties of ADCs make them optimal agents to pair with immune checkpoint inhibitors, specifically those targeting the PD-1 pathway. ADCs lead to direct activation and maturation of dendritic cells as well as immunogenic cell death, triggering an anti-tumor immune response that may be enhanced by inhibition of PD-1 [78]. Certain ADCs have been shown to stimulate intratumoral migration of CD8 + T-cells as well [79, 80]. Although immune checkpoint inhibitors can lead to long-term responses, a minority of patients with metastatic UC achieve durable disease control with these agents; therefore, combination regimens which may expand this population of long-term responders are highly sought after [81–84]. The combination of pembrolizumab (anti-PD-1) and enfortumab vedotin has been evaluated in a phase Ib/II study for patients with previously untreated advanced UC who were ineligible for cisplatin chemotherapy [85]. The ORR with this combination was an astounding 73.3% (95% CI, 58.1–85.4%) with a 15.6% complete response rate. Median PFS and OS were 12.3 months and 26.1 months, respectively. Responses were demonstrated irrespective of PD-L1 or Nectin-4 expression levels. Grade≥3 treatment-related adverse events occurred in 64.4% of patients and were immune-mediated in 26.7%, most of which were cutaneous reactions. The US FDA granted accelerated approval for enfortumab vedotin with pembrolizumab for patients with locally advanced or metastatic urothelial carcinoma who are ineligible for cisplatin-based chemotherapy [86]. Finally, during preparation of this manuscript, results were reported for the randomized phase III study, EV-302 (NCT04223856), evaluating enfortumab vedotin plus pembrolizumab vs. gemcitabine plus cisplatin or carboplatin in treatment-naïve patients with advanced UC. In this study, enfortumab vedotin plus pembrolizumab improved progression free survival relative to chemotherapy (12.3 vs. 6.3 months, HR, 0.45 [95% CI, 0.38–0.54; P < 0.00001]) as well as overall survival (31.5 vs. 16.1 months, HR, 0.47 [95% CI, 0.38–0.58; P < 0.00001]). This regimen has been FDA approved in this setting [87]. Given the marked improvement in overall survival compared to frontline chemotherapy, this regimen is expected to replace platinum-based chemotherapy as the standard of care for patients with treatment-naive metastatic UC.
Several other trials with ADC and PD-1 axis inhibitor combinations have been reported. In cohort 3 of TROPHY-U-01 (phase II), investigation of sacituzumab govitecan plus pembrolizumab is underway for patients with metastatic UC previously treated with platinum chemotherapy [20]. With median follow-up of 5.8 months, the ORR was 34% (95% CI, 20.1–50.6%) but with grade≥3 TRAEs occurring in 59% of patients- mostly diarrhea (24%), anemia (20%), and febrile neutropenia (10%). Early data from HER2-ADC and PD-1 axis inhibitor combinations has been reported as well. In a phase Ib study, trastuzumab deruxtecan (T-DXd), an ADC comprised of the HER2 monoclonal antibody trastuzumab covalently linked to a topoisomerase I inhibitor payload, was combined with nivolumab in patients with platinum-relapsed/refractory metastatic UC with either HER2 IHC 2 + /3+(cohort 3) or IHC1+(cohort 4) [44]. The ORR for cohort 3 (n = 30) was 36.7% (95% CI, 19.9–56.1%) with a 13.3% complete response rate. The median PFS was 6.9 months with median OS of 11.0 months. Grade≥3 TRAEs occurred in 73.5% of patients with a TRAE-related discontinuation rate of 32.4%. Notably, treatment-related interstitial lung disease or pneumonitis, which has been reported across other studies involving T-DXd, occurred in 23.5% of patients with one death attributed to this toxicity [44, 88]. In another phase Ib/II study, disatimab vedotin was combined with the PD-1 inhibitor toripalimab in a mixed cohort of treatment naïve (57%) and previously treated patients with locally advanced or metastatic UC with variable HER2 and PD-L1 expression [43, 89]. The confirmed ORR was 76.7% (95% CI, 57.7–90.1%) in the 36 patients with at least one tumor assessment, with ORR 100% (n = 5/5) among treatment-naïve patients with HER2 IHC 2 + /3 + and PD-L1 positivity (combined proportion score≥10%). Given the short follow-up, time to event analyses were immature, but the estimated median PFS was 9.2 months for the cohort. The cumulative grade≥3 toxicity rate was not reported, but immune-related grade≥3 AEs occurred in 7.3% of patients.
Several other novel ADC-based combination strategies are being investigated in clinical trials including in combination with chemotherapy (NCT03547973; cisplatin plus sacituzumab govitecan), FGFR inhibitors (NCT04963153; erdafitinib plus enfortumab vedotin), PARP inhibitors (NCT04039230; sacituzumab govitecan plus talazoparib), and dual ADC therapy (NCT04724081, sacituzumab govitecan plus enfortumab vedotin). These combinations have yet to report data but are conceptually promising.
Conclusion
ADCs represent a versatile class of medications with roles in treating patients with various tumor types and likely many more indications on the horizon [90]. These agents are designed to deliver potent cytotoxic compounds in a targeted fashion, with the potential to optimize delivery, for instance, by altering the drug-antibody ratio or modifying the linker technology. Optimal combinations of antibody, payload, and linker may be specific to tumor types, yet some agents (e.g., trastuzumab deruxtecan and sacituzumab govitecan) have demonstrated activity against a variety of cancers. Patients with metastatic urothelial carcinoma have benefitted tremendously from ADCs, namely enfortumab vedotin and sacituzumab govitecan, which are both FDA approved in previously treated patients. In addition to these agents moving into earlier lines of therapy and being utilized in earlier stages of disease, we are likely to see ADC-based combination regimens enter the clinic as well. HER2 targeted ADCs alone and in combination with immune checkpoint inhibitors have also demonstrated impressive response rates in early phase clinical trials. Further development will hinge on uncovering mechanisms of resistance to specific ADCs in urothelial carcinoma. The findings of this systematic review highlight both the importance of ADCs in the current management of patients with urothelial carcinoma and the great promise of these agents for future development.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments.
FUNDING
The authors report no funding.
AUTHOR CONTRIBUTIONS
Conception: MG, DP; Performance: MG, PS, ZN, MA; Interpretation of data: MG, DP, PS, MA, ZN
ETHICAL CONSIDERATIONS
As a review of the literature, and as no animal or human research was involved, our study is exempt from any requirement for Institutional Review Board approval.
CONFLICTS OF INTEREST
Michael J Grant reports honoraria from AstraZeneca and Daiichi Sankyo. Paul Stockhammer, Matthew R. Austin, Zsuzsanna Nameth have no conflicts of interest to report. Daniel P. Petrylak is an Editorial Board member of this journal, but was not involved in the peer-review process nor had access to any information regarding its peer-review. He reports grants or contracts from Ada Cap (Advanced Accelerator Applications), Agensys Inc, Arvinas, Astellas, AstraZeneca, Bayer, BioXcel Therapeutics, Bristol Myers Squibb, Clovis Oncology, Daiichi Sankyo Company Limited, Eisai, Eli Lilly, Endocyte, Ferring, Genentech, Gilead Sciences, Innocrin, MedImmune, Medivation, Merck, Mirati, Novartis, Pfizer, Progenics, Replimune, Roche, Sanofi Aventis, Seattle Genetics; Daniel P. Petrylak also reports consulting fees from Ada Cap (Advanced Accelerator Applications), Amgen, Astellas, AstraZeneca, Bayer, Bicycle Therapeutics, Boehringer Ingelheim, Bristol Myers Squibb, Clovis Oncology, Eli Lilly, Exelixis, Gilead Sciences, Incyte, Infinity Pharmaceuticals, Ipsen, Janssen, Merck & Company Inc, Mirati, Monopteros, Pfizer, Pharmacyclics, Regeneron, Roche, Sanofi Aventis Pharmaceuticals, Seattle Genetics, Urogen.
DATA AVAILABILITY
The data supporting the findings of this study are available within the article and/or its referenced manuscripts.
