Abstract
Bladder preservation remains a highly desirable therapeutic goal in urothelial carcinoma, particularly when durable survival can be achieved. Radical cystectomy is the standard treatment for muscle-invasive disease; however, it results in irreversible bladder loss and significant morbidity. Among patients with locally advanced or metastatic urothelial carcinoma, recurrence rates after standard therapy remain high. Consolidation therapy with enfortumab vedotin and pembrolizumab following cisplatin-based induction chemotherapy has demonstrated promising efficacy, with substantial complete response rates and improved bladder preservation. This single-center retrospective analysis evaluated the efficacy and safety of enfortumab vedotin plus pembrolizumab as consolidation therapy in 11 patients with metastatic urothelial carcinoma who completed cisplatin-based induction chemotherapy. More than 90% of patients achieved a radiographic or clinical complete response, and several maintained prolonged treatment-free remission. Treatment-related adverse events, including dermatologic toxicity, reversible peripheral neuropathy, and hyperglycemia, were observed but were predominantly manageable, allowing treatment continuation in most cases. Notably, the regimen demonstrated clinical activity in cisplatin-intolerant patients and in those who did not achieve an initial complete response after cisplatin therapy. Compared with conventional cisplatin-based therapy alone, consolidation with enfortumab vedotin plus pembrolizumab was associated with favorable tolerability, durable responses, and a high rate of bladder preservation. These findings support further investigation of enfortumab vedotin and pembrolizumab as a potential bladder-sparing strategy in advanced urothelial carcinoma.
Introduction
Bladder preservation is highly desired by patients with urothelial cancer if long-term survival can be achieved. However, radical cystectomy remains a primary treatment modality for localized bladder cancer with muscle invasion. Despite the use of perioperative platinum-based chemotherapy and adjuvant nivolumab, patients with T3–T4 or node-positive (N+) disease face a high risk of distant recurrence, along with permanent bladder loss following cystectomy, with a reported disease-free survival (DFS) of 21 months. 1 In the phase III NIAGARA trial, patients with muscle-invasive bladder cancer (MIBC) who were eligible for cisplatin were randomly assigned to receive neoadjuvant durvalumab in combination with gemcitabine and cisplatin every 3 weeks for four cycles, followed by radical cystectomy and adjuvant durvalumab every 4 weeks for eight cycles. The addition of perioperative durvalumab improved event-free survival (EFS), with a 2-year EFS of 68% compared with 60% in the control group without durvalumab. The frequency of radical cystectomy was similar between the two arms (88% vs. 83%). Pathologic CR (pCR) rates at the time of cystectomy were 34% in the durvalumab plus chemotherapy group compared with 26% in the chemotherapy-alone group. 2
In the KEYNOTE-905/EV-303 phase III trial, patients with MIBC who were ineligible for cisplatin-based chemotherapy received three cycles of preoperative enfortumab vedotin (EV) plus pembrolizumab followed by radical cystectomy. Postoperatively, patients received six cycles of EV plus pembrolizumab, followed by eight cycles of pembrolizumab alone. This regimen demonstrated an improved 2-year EFS of 74% compared with 39% in the surgery-alone group. pCR rates at the time of cystectomy were 57% in the EV plus pembrolizumab group compared with 8% in the surgery-alone group.3,4
For patients with unresectable or metastatic urothelial carcinoma, complete response (CR) rates are substantially lower than those observed in earlier-stage MIBC. First-line standard treatment includes nivolumab plus gemcitabine–cisplatin (CheckMate 901), 5 which demonstrated a CR rate of 16% and a median overall survival of 22 months, or enfortumab vedotin plus pembrolizumab (EV-302), 6 which demonstrated a CR rate of 30% and a median overall survival of 31 months. Lymph node (LN)–only metastatic disease is a favorable prognostic factor in patients with mUC, and a subset of these patients achieve durable DFS with cisplatin-based chemotherapy. 5 In the CheckMate 901 trial of 608 randomized patients, 54 treated with nivolumab plus gemcitabine–cisplatin and 56 treated with gemcitabine–cisplatin had LN-only mUC. In that subgroup, the CR rate was 63% with nivolumab plus gemcitabine–cisplatin vs. 34% with gemcitabine–cisplatin. 5
Due to the dose-limiting toxicities of cisplatin (e.g. renal failure, hearing loss, and neuropathy), prolonged administration is challenging, limiting its use in routine practice. Dose limitations and suboptimal outcomes in patients with intratumoral clonal resistance to platinum-based chemotherapy highlight the need for alternative treatment strategies, particularly among patients unable to complete four full cycles and those who do not achieve a CR with cisplatin.
EV is an antibody–drug conjugate (ADC) composed of a fully human monoclonal antibody targeting nectin-4 (a cell-adhesion protein expressed in >95% of urothelial carcinoma cells) linked to the cytotoxic agent monomethyl auristatin E (MMAE), a microtubule-disrupting agent. After EV binds to nectin-4 on the tumor cell surface, MMAE is released intracellularly through proteolytic cleavage, leading to cell-cycle arrest and apoptosis. 7 EV can generally be administered without the same cumulative dose limitations as cisplatin until CR is achieved. The sequential use of cisplatin followed by EV may potentially eradicate resistant clones and increase CR rates.
Historical trials have shown that a subset of patients who achieve a pCR following cisplatin-based neoadjuvant chemotherapy experience lower relapse rates and improved long-term survival. 1 These favorable outcomes among complete responders have generated interest in organ-sparing approaches for patients who wish to preserve their bladder. We aimed to design a regimen capable of increasing CR rates through extended consolidation therapy, thereby allowing a greater proportion of patients to safely preserve their bladder under close surveillance. Achieving high CR rates with organ preservation in patients with locally advanced unresectable mUC may also inform future strategies in earlier-stage MIBC.
Here, we describe the outcomes of 11 patients with mUC treated with EV consolidation plus pembrolizumab following cisplatin-based induction chemotherapy. This novel approach was designed to improve bladder preservation rates, extend treatment-free remission, and enhance survival outcomes within the evolving therapeutic landscape of advanced urothelial carcinoma.
Methodology
Study design
Patient selection
Patients included in the study were diagnosed with locally advanced or mUC, defined as muscle-invasive disease (T3–T4), lymph node involvement, or stage IV disease with metastases to the lungs or distant lymph nodes (e.g. para-aortic or pelvic nodes). All patients were treated at The University of Texas Health Science Center at Houston–affiliated Memorial Hermann Cancer Center between 2017 and 2023. Each patient had undergone induction cisplatin-based chemotherapy and elected to forgo cystectomy. Patient selection criteria are outlined in Figure 1.
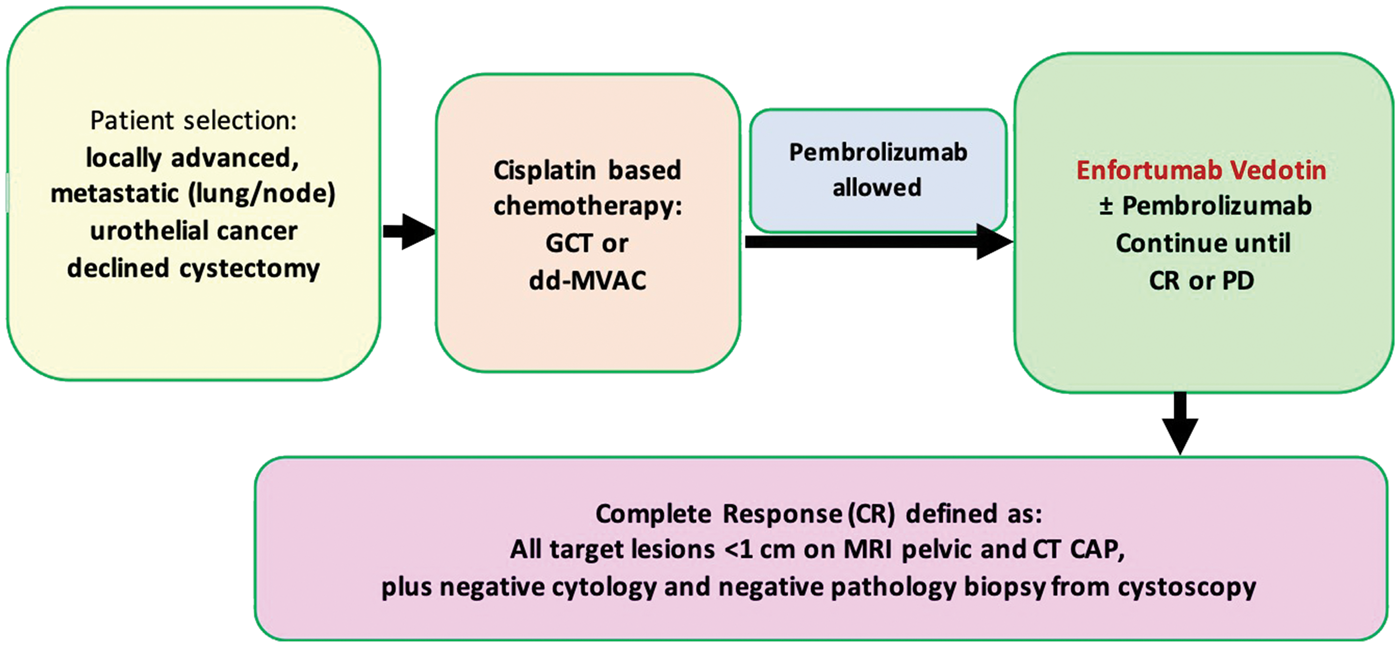
Flowchart of the study paradigm.
Ethical issues and informed consent
The study protocol was approved by the Institutional Review Board of The University of Texas Health Science Center at Houston. The research was conducted in accordance with the ethical standards of The University of Texas Health Science Center at Houston and the Declaration of Helsinki for research involving human participants. A waiver of HIPAA Privacy Authorization was obtained from the Institutional Review Board. All patients provided consent for publication.
Hypothesis
The purpose of this study was to determine whether EV plus immunotherapy following cisplatin-based chemotherapy could prolong and deepen treatment responses, increase subsequent treatment-free remission rates, and facilitate bladder preservation in patients who achieved a CR.
Pathological evaluation
Biopsy samples were obtained during initial cystoscopy prior to chemotherapy, at surveillance cystoscopy following chemotherapy for restaging, and from cystectomy specimens when applicable. Germline and somatic mutations were identified using the Tempus® (Chicago, IL) next-generation sequencing assay.
Study procedures
First-line cisplatin-based chemotherapy
Patients received one of two cisplatin-based regimens as initial therapy. The gemcitabine, cisplatin, and paclitaxel (GCT) regimen consisted of split-dose cisplatin 35 mg/m2 on days 1 and 8 and gemcitabine 300–500 mg/m2 on days 1 and 8, with or without paclitaxel 70 mg/m2 on days 1 and 8. Granulocyte colony-stimulating factor (G-CSF) was administered on days 2 and 9 of each 3-week cycle.
The dd-MVAC regimen consisted of methotrexate 30 mg/m2, vinblastine 3 mg/m2, and doxorubicin 30 mg/m2 on day 1, plus cisplatin 35 mg/m2 on days 1 and 8. G-CSF was administered on days 2 and 9 of each 2-week cycle.
Cisplatin-based chemotherapy was administered for 1–6 cycles, as tolerated. Following completion of cisplatin-based therapy, all patients received pembrolizumab immunotherapy in one of two approaches. Some patients received maintenance pembrolizumab and were subsequently switched to EV upon disease progression during pembrolizumab treatment. Others initiated combination therapy with EV plus pembrolizumab from the outset. EV was administered at 1.25 mg/kg on days 1, 8, and 15 of each 4-week cycle as monotherapy, or on days 1 and 8 of each 3-week cycle when combined with pembrolizumab. Pembrolizumab was administered as a 400 mg flat dose every 6 weeks.
MRI-defined responses
Clinical CR (cCR) on MRI T2-weighted imaging (MRI-T2W) was defined as a dark T2 signal without intermediate T2 signal intensity and no visible lymph nodes. On diffusion-weighted imaging (MRI-DWI), cCR was defined as no visible tumor on the b1000 image and/or low signal intensity on the ADC map; a uniform, linear signal in the bladder wall at the prior tumor site was considered acceptable.
Residual disease was defined as MRI-T2W demonstrating an intermediate T2 signal without evidence of a T2 scar or without regression of lymph nodes, or MRI-DWI demonstrating minimal regression of signal intensity on b1000 images and/or persistent low signal on the ADC map.
CR
CR was defined as the absence of viable tumor cells in the resection specimen from cystoscopy and no evidence of disease on pelvic MRI and CT restaging scans, with target lymph nodes measuring <1 cm.
Results
Between 2017 and 2023, a total of 11 patients were included in this study (Tables 1–3). Of these, four patients had stage IV disease: 3 presented with metastases to the lungs and distant lymph nodes, and one presented with metastasis to the para-aortic lymph nodes. Eight patients initially presented with obstructive hydronephrosis requiring placement of external nephrostomy tubes and ureteral stents. Following chemotherapy, all eight patients experienced resolution of obstruction within 6 months of treatment, allowing for removal of the nephrostomy tubes.
Frequency distribution of patients’ demographic and clinical characteristics.
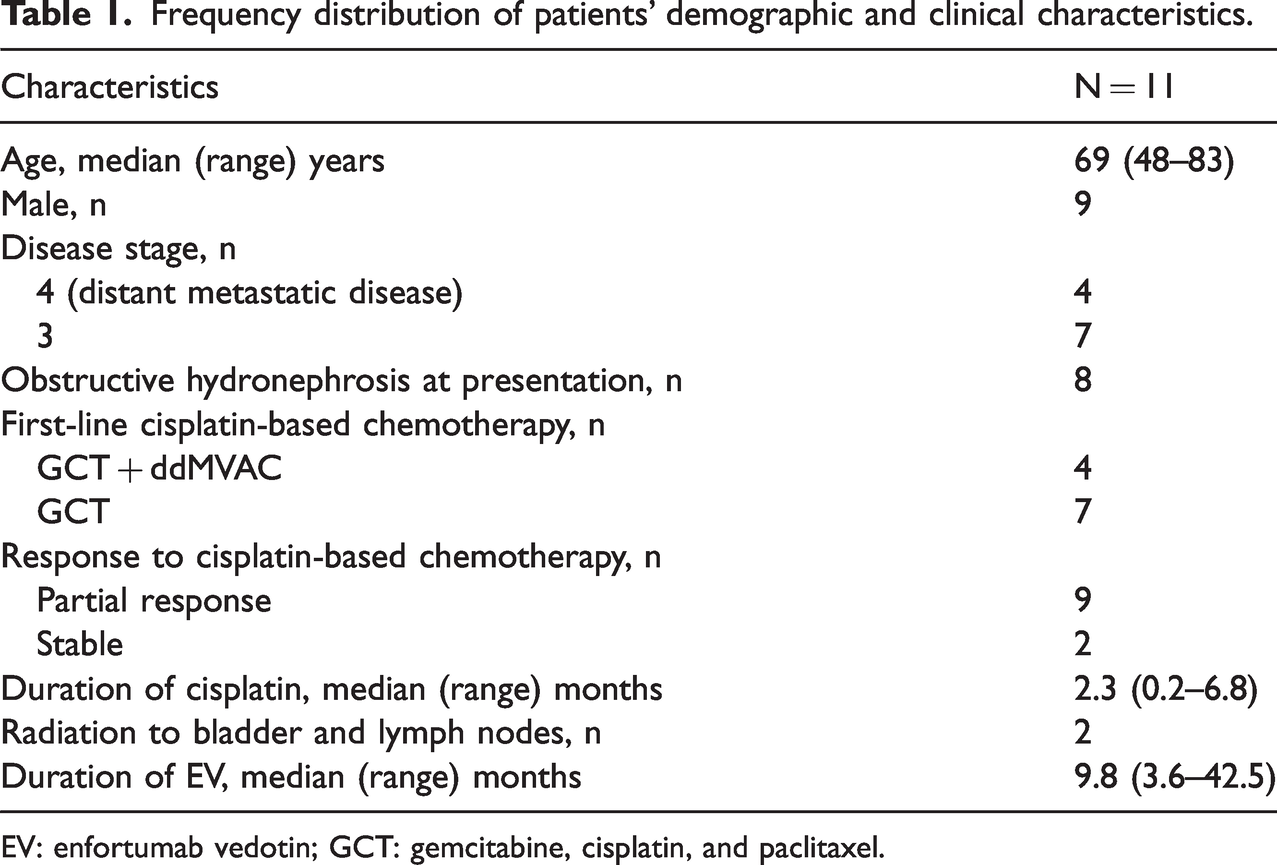
EV: enfortumab vedotin; GCT: gemcitabine, cisplatin, and paclitaxel.
Details of each patient’s treatment duration and treatment outcome.

CIS: carcinoma in situ; CR: complete response; EV: enfortumab vedotin; F: female; IDDM: insulin dependent diabetes mellitus; M: male; Pembro: pembrolizumab; PR: partial response; RP: retroperitoneal; XRT: radiation; ATM: ataxia-telangiectasia mutated; BCG: Bacille Calmette-Guérin.
Outcomes among patients given EV-pembrolizumab for mUC.

CR: complete response; EV: enfortumab vedotin; mUC: metastatic urothelial carcinoma.
Radiation therapy and radiosensitizers
Two patients received pelvic radiation to the bladder and regional lymph nodes. Both achieved CR; however, both subsequently developed radiation-induced complications. One patient achieved a CR with concurrent EV and radiotherapy, with EV used as a radiosensitizer. 2 years later, the patient developed radiation-induced hemorrhagic cystitis and subsequently elected to undergo cystectomy. The other patient received pembrolizumab with concurrent radiation therapy for synchronous bladder and prostate cancers and achieved a CR; however, 1 year later, the patient developed radiation-induced ischemic hemorrhagic colitis requiring sigmoidectomy.
EV side effects
We observed frequent adverse effects during treatment with EV. Ten of the 11 patients treated with EV developed grades 1–2 maculopapular rash, primarily involving the torso, arms, and thighs, while sparing the face, palms, and soles. The rash was typically managed with topical steroids. Ten patients developed new or worsening neuropathy during EV treatment, including three who experienced grade 3 neuropathy requiring assistance with a walker or wheelchair. One patient developed lower-extremity muscle weakness and was diagnosed with demyelinating neuropathy requiring treatment with rituximab and intravenous immunoglobulin (IVIG). This patient had a germline mutation associated with neurofibromatosis type 1. Neuropathy symptoms improved to near-baseline levels in all 10 affected patients after discontinuation of treatment (including EV). All patients required dose reduction of EV (to 0.75 mg/kg) and decreased dosing frequency (to every 2 weeks) due to rash and neuropathy after 3–6 months of therapy.
In addition to dermatologic and neurologic toxicities, two patients developed hyperglycemia. One patient had blood glucose levels reaching the 400s mg/dL, which returned to baseline (nondiabetic range) after discontinuation of EV while maintaining a CR of mUC. The other patient developed type 1 insulin-dependent diabetes mellitus attributed to pembrolizumab treatment.
CR, bladder preservation, and recurrence
Ten patients achieved a CR. The swimmer plot illustrating CR duration (in months), duration of EV treatment, and patient outcomes is shown in Figure 2. Among these 10 patients, 4 experienced recurrence during surveillance. One patient with carcinoma in situ (CIS) elected to undergo cystectomy; surgical pathology confirmed TisN0 disease. Another patient with CIS achieved a subsequent CR after intravesical Bacille Calmette-Guérin (BCG) therapy and has had no further recurrence. One patient with a germline pathogenic ataxia-telangiectasia mutated (ATM) mutation developed a second primary T2 lesion in the prostatic urethra 1 year into surveillance and achieved a second CR after resuming EV plus pembrolizumab for 6 months (Figure 3). Another patient was found to have nodal recurrence in the retroperitoneum and mediastinum, requiring resumption of EV plus pembrolizumab. One patient achieved a partial response to cisplatin but subsequently experienced disease progression during EV plus pembrolizumab therapy and later died from liver metastases.
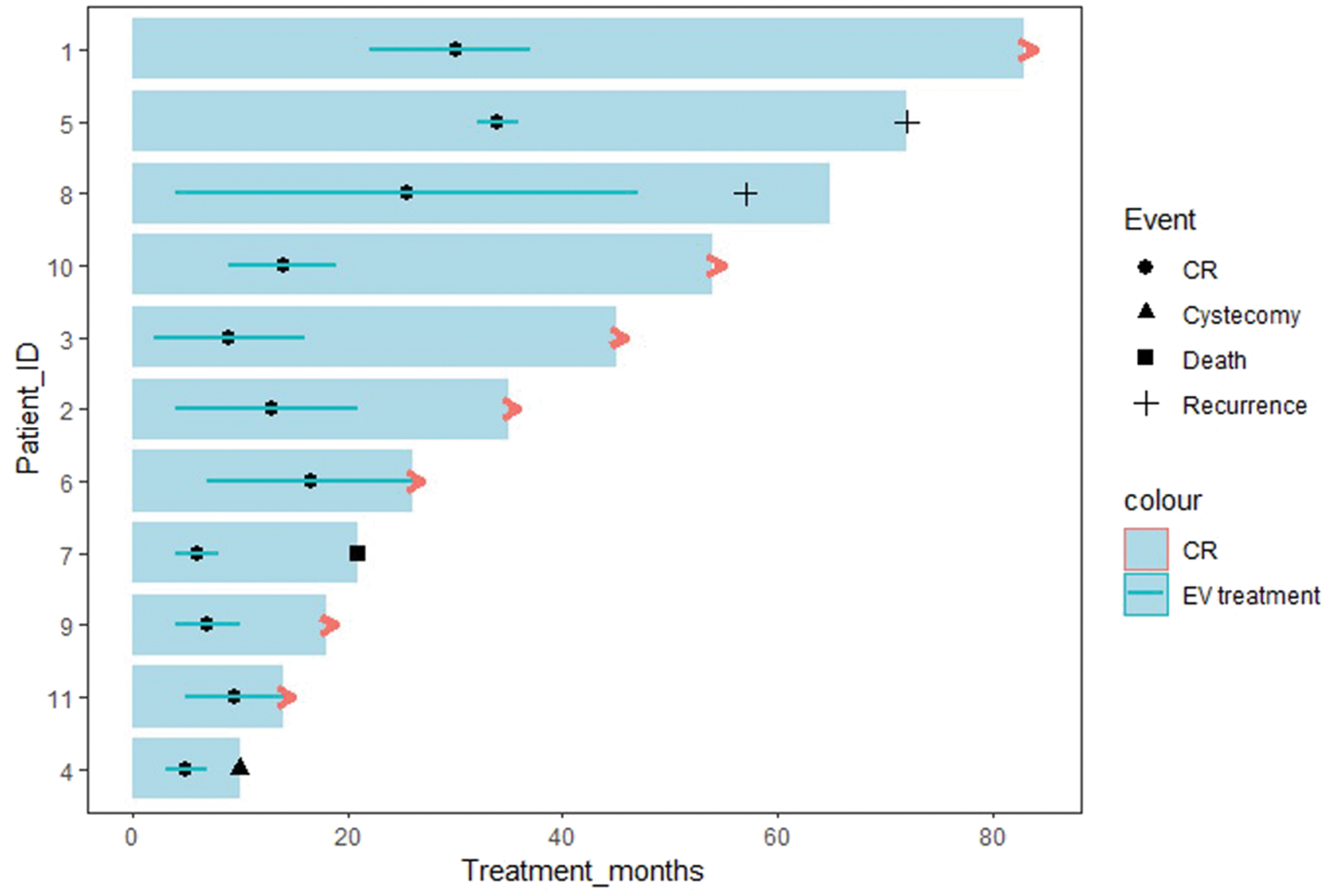
Swimmer plot of complete response duration (months), EV treatment, and patient outcome.
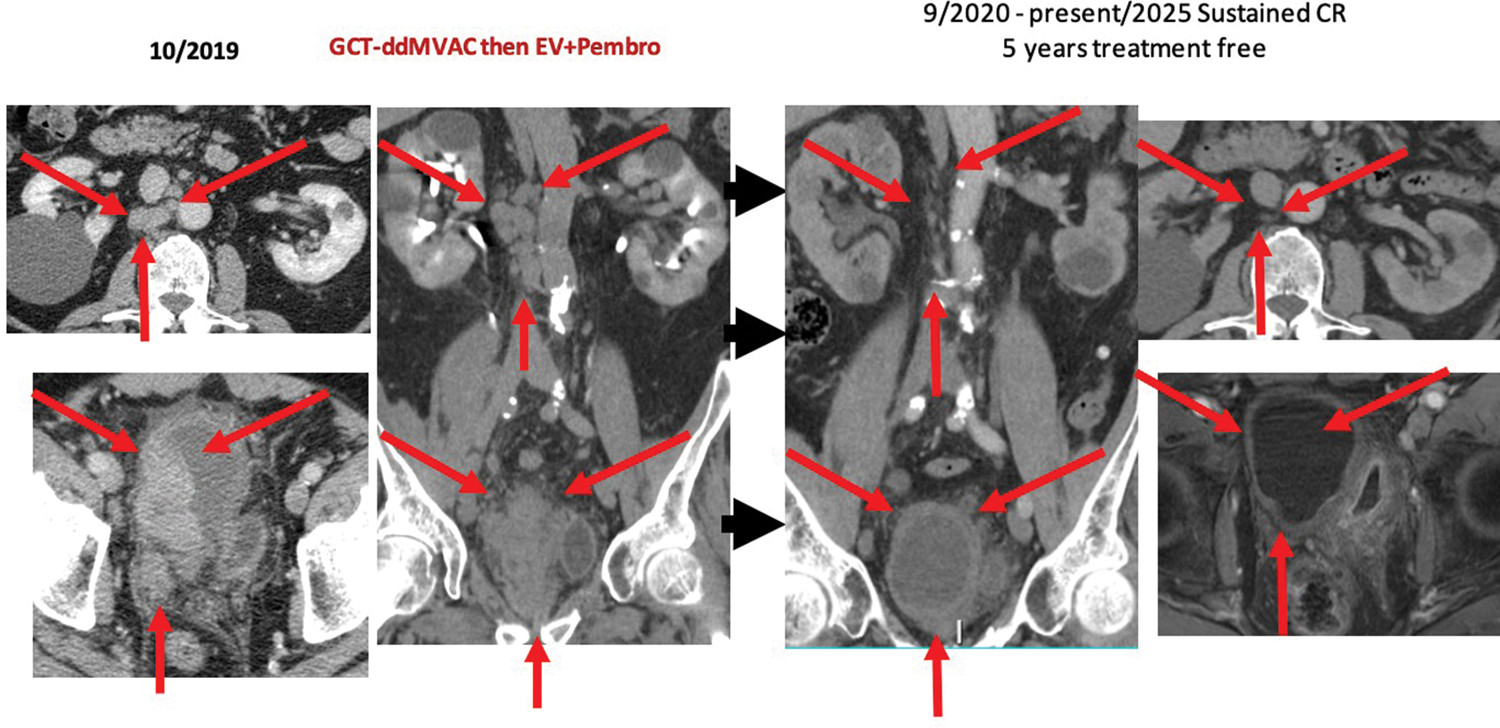
A 75-years-old male with a 7 cm bladder UC of the right lateral bladder wall, metastatic to the lungs and associated with painful, bulky para-aortic lymphadenopathy, requiring placement of a nephrostomy tube. The patient was treated with GCT followed by ddMVAC and achieved a partial response, then received EV + pembrolizumab and achieved CR. Genetic testing revealed a germline pathogenic ATM mutation. One year after achieving CR, the patient developed a new second primary T2 lesion in the prostatic urethra and resumed treatment with EV plus pembrolizumab. CR: complete response; EV: enfortumab vedotin; UC: urothelial carcinoma; GCT: gemcitabine, cisplatin, and paclitaxel; ATM: ataxia-telangiectasia mutated.
Discussion
The current study demonstrated that cisplatin-based chemotherapy followed by consolidation with EV plus immunotherapy achieved a >90% CR rate in predominantly node-metastatic, locally advanced bladder cancer. This approach resulted in prolonged treatment-free remission and enabled bladder preservation in the majority of patients, who will require lifelong close surveillance. Based on our experience, we identified two clinical scenarios in which cisplatin-eligible patients with locally advanced urothelial carcinoma may benefit from this treatment strategy: Patients without a CR after 4–6 cycles of cisplatin-based chemotherapy: For these patients, switching to an alternative effective regimen—specifically EV combined with pembrolizumab—may potentially increase the overall CR rate. In our cohort, a small number of patients achieved CR with cisplatin-based chemotherapy alone and were therefore not offered EV plus pembrolizumab; these patients were excluded from the present analysis. Patients who are cisplatin-intolerant or borderline eligible: Some patients who received only 1–2 cycles of cisplatin-based chemotherapy developed significant toxicities, including renal failure, heart failure, arrhythmia, fluid overload, severe infections, or general clinical decline, preventing completion of treatment. These patients are unlikely to derive the full therapeutic benefit of cisplatin because of early discontinuation or dose reduction. However, their tumors may remain chemotherapy-sensitive. In such cases, transitioning to EV plus pembrolizumab may offer a higher likelihood of CR compared with immunotherapy alone or switching to a less effective carboplatin-based regimen.
Historical data have shown that carboplatin does not provide the same long-term survival benefit as cisplatin in the neoadjuvant or node-metastatic setting.8,9 Many of our initially cisplatin-eligible patients were unable to tolerate the third or fourth cycle of cisplatin and had bulky residual disease following treatment. Although all patients in our study received cisplatin, 3 received only one cycle because of intolerance, and 2 did not complete 4 cycles of 70 mg/m2 cisplatin as first-line therapy. Nonetheless, these patients achieved favorable responses and ultimately attained CR after EV treatment. Unlike cisplatin-based chemotherapy, EV was generally well tolerated and could be administered for an extended duration without causing irreversible grades 3–4 adverse events. Our study did not include patients with primary cisplatin-refractory disease but did include patients who either progressed after cisplatin or were unable to tolerate full-dose cisplatin. One patient who progressed on pembrolizumab monotherapy after cisplatin subsequently achieved a CR with EV plus pembrolizumab. Avelumab maintenance therapy, which has a reported response rate of 9.7% and a progression rate of 37%, is limited to patients who have not progressed on first-line platinum chemotherapy. 10 Our strategy of administering EV in combination with pembrolizumab following cisplatin-based therapy represents a promising alternative, capable of achieving deeper responses and durable remissions in responders. Additionally, it may serve as an effective salvage option for patients who progress after platinum-based chemotherapy. Ideal candidates for this approach include patients initially eligible for cisplatin who present with stage IV urothelial carcinoma involving distant nodal metastases, limited pulmonary metastases, or muscle-invasive, locally advanced disease seeking bladder preservation after successful downstaging with chemotherapy.
The CheckMate 274 phase III trial demonstrated that, among patients with high-risk muscle-invasive urothelial carcinoma who underwent radical cystectomy, adjuvant nivolumab improved DFS from 11 to 21 months compared with placebo, although recurrence remained common. 1 finding indicates that approximately half of patients with T2 muscle-invasive urothelial carcinoma (UC) experienced recurrence or death within 1–2 years despite undergoing radical cystectomy and perioperative systemic therapy (chemotherapy and/or immunotherapy). Therefore, if patients do not achieve significant downstaging to clinical stage yT0–T1 after neoadjuvant chemotherapy, they may benefit more from a novel combined approach incorporating cisplatin-based and EV-based chemoimmunotherapy, as used in this study, rather than proceeding directly to cystectomy.
In the phase III NIAGARA trial, neoadjuvant durvalumab combined with gemcitabine and cisplatin for four cycles in patients with MIBC (T2–T4N0–N1) achieved a pCR rate of 34%. 2 In the KEYNOTE-905/EV-303 phase III trial, the pCR rate was 57% in the neoadjuvant group receiving EV plus pembrolizumab for three cycles. 4 In the phase II HCRN GU 16-257 trial, neoadjuvant gemcitabine, cisplatin, and nivolumab administered for four cycles in patients with T2–T4N0 MIBC resulted in a pCR rate of 43%. 11 In comparison, patients in our study had more advanced stage III–IV disease and required additional treatment with EV to eradicate residual disease following cisplatin. Our CR rate of >90% exceeds those reported in the above neoadjuvant trials, likely reflecting the addition of EV after platinum-based chemotherapy in a consolidation setting.
Similarly, in the bladder-sparing ReTAIN trial, patients with T2–T3N0 disease (79% with T2 disease) received three cycles of neoadjuvant ddMVAC. In the subsequent ReTAIN-2 trial, patients with T2–T3N0 disease were treated with three cycles of neoadjuvant DDMVAC + NIVO. 12 Patients who achieved a CR and harbored mutations in ATM, RB1, FANCC, or ERCC2 were offered active surveillance without cystectomy or radiation. However, metastatic recurrence rates were high in both groups (32% in the non-mutation plus cystectomy group vs. 38% in the active surveillance group) during a median follow-up of 41 months. 13 In contrast, the metastatic recurrence rate in our study was low, occurring in only 2 of 11 patients. One patient developed metastatic disease in mediastinal lymph nodes, while another initially demonstrated a partial response to EV but subsequently progressed. Among the eight patients who achieved CR and retained their bladders, three developed local recurrence with Tis–T2 lesions considered second primary tumors. These were detected through routine cystoscopic surveillance and were successfully managed with resumption of therapy, resulting in a second CR. The remaining five patients remained disease-free at the end of follow-up.
One patient with a germline ATM mutation developed a second primary T2 recurrence. Based on our limited experience and extrapolation from breast cancer management, patients with homologous recombination deficiency germline mutations may not be ideal candidates for organ preservation because of a potentially higher risk of developing second primary bladder tumors or, in men, prostate cancer.
Although trimodality therapy for MIBC commonly incorporates cisplatin, 5-FU, mitomycin, or a taxane as radiosensitizers, there are currently no published prospective trials evaluating bladder preservation in advanced mUC using neoadjuvant EV or EV administered concurrently with trimodality chemoradiation (CRT). In our study, EV appeared to be safely administered concurrently with pelvic and para-aortic lymph node radiation, and one patient achieved a durable CR lasting 2 years. However, radiation therapy was associated with significant complications. One patient developed radiation-induced cystitis and another developed radiation-induced colitis; both required surgical resection of the affected organs (bladder and sigmoid colon, respectively). Therefore, if a patient achieves a CR with systemic therapy alone, we advocate avoiding pelvic radiation to minimize the risk of long-term radiation-related complications. In our cohort, it remains uncertain whether one patient might have achieved a CR with extended EV therapy alone had radiation been omitted. Based on this experience, we favor extending EV-based immunotherapy in place of radiation, when feasible, to reduce the long-term toxicity associated with pelvic irradiation.
Longoni et al. 12 analyzed surveillance, epidemiology, and end results (SEER) data and reported several findings comparing bladder preservation with radical cystectomy. First, partial cystectomy was associated with a 5-year cancer-specific mortality (CSM) of 26%, similar to 28% observed with radical cystectomy in highly selected tumors (solitary, <3 cm, localized to the bladder dome or trigone, and not involving the bladder neck or ureteral orifices). Second, bimodal therapy (BMT: TURBT + either radiation or chemotherapy) or trimodal therapy (TMT: TURBT + ChemoRadiation) was associated with nearly double the 5-year CSM compared with radical cystectomy (RC) + perioperative chemotherapy among patients with nonorgan-confined (NOC; T3–T4 and/or N1–N3) UC of the bladder, with findings consistent across age groups, including those >80 years.14–16 In our cohort, 8 of 11 patients initially presented with obstructive hydronephrosis requiring external nephrostomy tubes and ureteral stents, and 3 presented with distant nodal (retroperitoneal) and lung metastases. Therefore, our patients would not have met the selection criteria for partial cystectomy or trimodality therapy as described in the Longoni SEER analyses.
Trials such as KEYNOTE-B15/EV-304, KEYNOTE-905/EV-303, and the VOLGA trial (NCT04960709 is a Phase 3, open-label study investigating neoadjuvant EV plus immunotherapy (durvalumab ± tremelimumab) in cisplatin-ineligible patients with MIBC; VolGA) are currently evaluating neoadjuvant EV plus immunotherapy administered for 3–4 cycles in patients with MIBC, followed by cystectomy in all participants. However, these trials exclude patients with clinically node-positive disease. In our study, we did not restrict the number of EV cycles, as we observed that 3–4 cycles of EV plus immunotherapy were insufficient to achieve CR in the majority of patients with node-positive, locally advanced UC. Some patients exhibited delayed responses, requiring close to 1 year of EV-based immunotherapy to achieve CR.
The two patients in our cohort who developed recurrence with CIS elected intravesical therapy rather than cystectomy. Intravesical salvage options include gemcitabine, docetaxel, and repeat BCG. TAR200 and adstiladrin (nadofaragene firadenovec-vncg) for BCG-unresponsive non-MIBC were not yet available at the time of our study. 17
Dermatologic reactions associated with EV typically resolved within a few days after infusion, leading many patients to extend dosing intervals to every 2–3 weeks. However, extended dosing intervals were associated with local bladder recurrence in two patients. Based on this experience, we found that EV was most effective when administered on the standard days 1 and 8 schedule until a cCR was achieved. Additionally, a retrospective study by Molina et al. 18 reported that patients who received EV after prior immunotherapy (18 patients) had a higher incidence of severe skin toxicity (including two deaths, 11%) compared with patients who had not received prior immunotherapy before EV (7 patients). Patients who developed dermatologic rash during EV treatment demonstrated higher response rates (57% vs. 24%). 19 Neuropathy was also observed but was generally reversible over several months after treatment discontinuation.
This study has several limitations. As a single-center retrospective analysis with a small sample size, the findings are subject to potential selection bias and limited generalizability. In addition, patients with liver or bone metastases were excluded, as our clinical experience suggests that metastatic involvement at these sites rarely results in durable complete remission after treatment discontinuation. Despite these limitations, our results indicate that combined EV + pembrolizumab following cisplatin-based chemotherapy may represent a viable treatment option for patients who are initially eligible for cisplatin but are unable to complete four cycles of cisplatin-based therapy as well as for those with residual disease after completing cisplatin-based regimens. Prospective studies are warranted to further evaluate this treatment paradigm.
We are actively assessing the role of genomic profiling and post-treatment ctDNA monitoring (both performed by Tempus) to facilitate early detection and surveillance of recurrence in conjunction with MRI, CT, and cystoscopy. We recently published a case report describing a complete pathologic response after pembrolizumab and EV in a patient with upper tract UC. 20 In a similar ongoing study, we are recruiting elderly patients with upper tract UC to receive EV + immunotherapy consolidation with the goal of kidney preservation. The results of the present study, together with our ongoing investigations, suggest a promising strategy—EV-based immunotherapy consolidation following cisplatin-based induction chemotherapy—in the management of UC, with the potential to improve both oncologic outcomes and patient quality of life.
Footnotes
Acknowledgments
We express our sincere gratitude and appreciation to Ms. Virginia Mohlere, ELS, who helped revise the grammar of our manuscript.
Author contributions
Chan and Cen conceptualized the study and drafted the manuscript. Chan, Cen, Shu, Canfield, Gonzalez, Rowe, Blanco, and Amsbaugh treated patients and collected data on treatment response. Tammisetti and Ayoub reviewed imaging studies to assess treatment response. Zhao and Tannir performed the statistical analyses. All authors reviewed and approved the final manuscript.
Data availability statement
Data are available from the corresponding author upon request.
Declaration of conflicting interests
Putao Cen, MD, serves on advisory boards and on speaker bureau for Eisai, Merck, Taiho, Pfizer, Astellas, and AstraZeneca.
Funding
Not applicable
