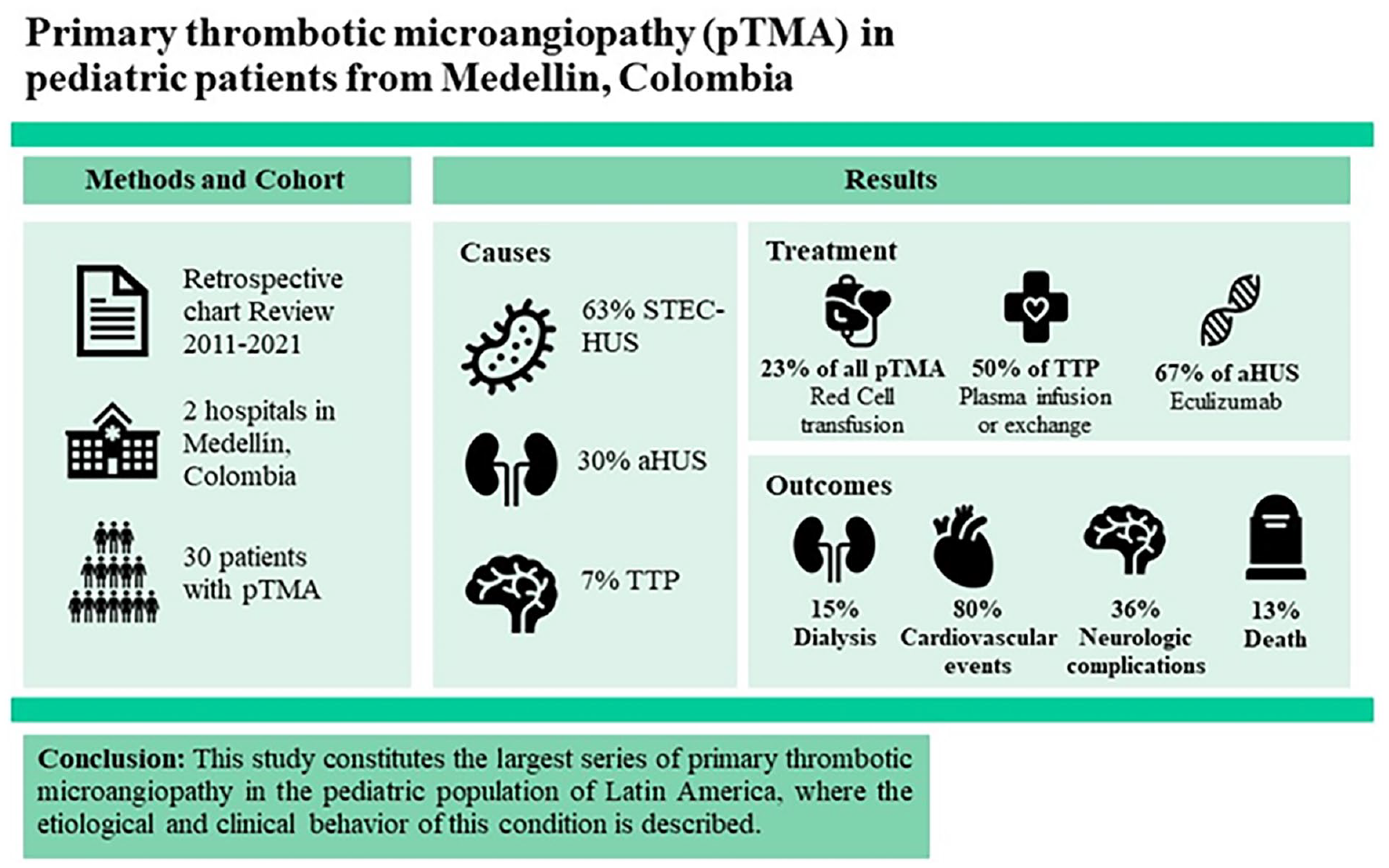
Abstract
Background. Primary thrombotic microangiopathy includes hemolytic uremic syndrome caused by Shiga toxin-producing Escherichia coli, atypical hemolytic uremic syndrome, and thrombotic thrombocytopenic purpura. Methodology. A retrospective study that included patients younger than 18 years diagnosed with primary thrombotic microangiopathy between 2011 and 2021. Results. Thirty patients were included, of which 63% corresponded to a hemolytic uremic syndrome caused by Shiga toxin-producing Escherichia coli, and 30% to the atypical hemolytic uremic syndrome. The median age was 2.8 years and female sex predominated at 57%. On admission to the emergency room, fever and fatigue were the most frequent symptoms (93%), followed by oliguria and anuria (80%). 48% of patients received hemodialysis during their care. Mortality was estimated at 13%. Conclusion. This study constitutes the largest series of primary thrombotic microangiopathy in the pediatric population of Latin America, where the etiological and clinical behavior of this condition is described.
Background
Thrombotic microangiopathy (TMA) is a syndrome in which a series of pathophysiological phenomena are present, including consumption thrombocytopenia, microangiopathic hemolytic anemia, and organ dysfunction resulting from endothelial damage and microvascular thrombosis. 1
Thrombotic microangiopathy can be divided into primary or secondary, depending on the underlying etiologic mechanism. Primary causes include hemolytic uremic syndrome caused by Shiga toxin-producing Escherichia coli (STEC-HUS) usually serotype O157:H7; atypical hemolytic uremic syndrome (aHUS), in which there is a genetic or acquired dysregulation in the alternate pathway of the complement system, and thrombotic thrombocytopenic purpura (TTP), caused by severely reduced activity (≤5%-10% of normal) of ADAMTS13 (A Disintegrin-like and Metalloprotease with Thrombospondin Type 1 Motif No. 13). Secondary causes may occur as a complication of various diseases and conditions such as autoimmune diseases, pregnancy, medications, cancer, hematopoietic cell transplantation or solid organ transplantation.1,2
pTMA (primary thrombotic microangiopathy) represents a diagnostic and therapeutic challenge, due to the infrequency of presentation, broad differential diagnoses, and delays in the detection and confirmation of patients suspected of this entity. 3 In addition, it has been estimated that 9% to 15% of patients die within a year after the first manifestation.4,5
There are few studies worldwide that define the epidemiology of pTMA in the pediatric population. In a series of 258 pediatric patients in Japan with pTMA, 64.3% had STEC-HUS, 15.5% atypical HUS, and 5.8% TTP. This study reported an incidence of 0.37 to 0.47 cases per 100,000 children under 15 years of age. 6 Another study in Oklahoma estimated the incidence of HUS and TTP at 3.3 and 4 to 11 per million population, respectively.3,7 Clinical practice confirms that pTMA is one of the causes to be considered when acute kidney injury occurs in infants and children.8-10
Although in recent decades there has been an increase in the knowledge of pathophysiology, genetics, diagnosis, and treatment, there are still areas with information gaps in our environment. Therefore, this study aims to clarify epidemiological, sociodemographic, and clinical details of a population of pediatric patients with pTMA in two high-complexity hospitals in Medellin, Colombia between 2011 and 2021.
Methodology
This is a retrospective observational study which included pediatric patients with suspected first episode of primary thrombotic microangiopathy from two high complexity level hospital institutions in the city of Medellín, Colombia between January 1, 2011 and December 31, 2021.
Because this is a rare disease, no sample calculation was performed and all patients under 18 years of age who met the three criteria for primary thrombotic microangiopathy were included: thrombocytopenia (less than 150,000/mm3 or 25% decrease from baseline), microangiopathic hemolytic anemia (age-defined anemia, elevated schistocytes and/or LDH, and/or decreased haptoglobin), and evidence of target organ injury, manifested as one or more of the following: evidence of neurological, renal, gastrointestinal, cardiovascular, pulmonary, and visual involvement. Patients with a history of autoimmune disease, cancer, transplantation, or predisposing conditions for secondary TMA were not included.
The cause of pTMA was determined by clinical variables and the result of Shiga toxin detection (polymerase chain reaction to detect the ST1-ST2 gene or stool culture to detect the O157:H7 strain of Escherichia coli) and ADAMTS13 or PLASMIC score 11 for differentiation between atypical hemolytic uremic syndrome and thrombotic thrombocytopenic purpura in the histories in which the result was available.
Data Collection
For data collection, the medical records of patients who met the eligibility criteria were used as a source. A form including all the sociodemographic and clinical variables necessary to fulfill the objectives was designed. The information collected was stored in a database in Excel 2010.
Clinical Variables
We included sociodemographic variables (age, race, sex, origin), clinical variables (symptoms on admission, systems involved), laboratory variables (hemogram, peripheral blood smear, lactate dehydrogenase, Shiga toxin, ADAMTS13, complement levels, creatinine, urea nitrogen, urinalysis, PLASMIC score), treatment (plasmapheresis, eculizumab, antihypertensives, transfusions), and outcomes (death, cardiovascular, renal, neurological, psychological complications).
Statistical Analysis
Quantitative variables were analyzed according to the normal distribution fit, as mean and standard deviation or median and range. For qualitative variables, absolute and relative frequencies were used. Data analysis was performed using SPSS version 24.
This study was approved by the Health Research Ethics Committee of the Universidad Pontificia Bolivariana, Hospital Pablo Tobon Uribe, and Hospital San Vicente Fundación, Medellín, Colombia.
Results
Sixty-eight patients were identified from 2011 to 2021. After analyzing patient histories individually, pTMA was diagnosed in 30 patients (see Figure 1).

Flowchart of patients with primary TMA.
The 30 patients included were distributed as follows: 63% STEC-HUS, 30% aHUS and 7% TTP. The median age was 2.8 years and female sex predominated with 57% (see Table 1). Fever and fatigue were the most frequent manifestations on admission to the hospital (93%), followed by oliguria or anuria (80%), asthenia (67%), abdominal pain (67%), nausea, and diarrhea (63%). Diarrhea and dysentery occurred more frequently in aHUS (56% and 44%) than in STEC-HUS (32% and 26%) respectively. Focal neurological deficits occurred in 1 of the 2 TTP patients (see supplemental material).
Sociodemographic Characteristics of Patients with pTMA at Presentation.

Abbreviations: aHUS, atypical hemolytic uremic syndrome; STEC-HUS, hemolytic uremic syndrome caused by Shiga toxin-producing Escherichia coli; TTP, thrombotic thrombocytopenic purpura.
Regarding laboratory findings at the time of admission, the mean hemoglobin in patients with pTMA was 8.6 ± 1.9 g/dL, with a mean platelet count of 63,000 ± 41,000/μL, highlighting a lower platelet count of 9000 ± 0.7/μL, hemoglobin 7.6 ± 0.7 g/dL, and hematocrit 22.1 ± 2.6% in the cases of TTP. Mean creatinine was 3.2 ± 1.8 with no significant differences between groups. Schistocytes were present in 90% of patients in the peripheral blood and evidence of proteinuria in the urinalysis was present in 89% of patients, with a distribution of 100% and 94% TTP and STEC-HUS, respectively. The PLASMIC score was higher in TTP, with a mean of 5.5 ± .1 (see Table 2).
Laboratory Findings at Admission in Patients with pTMA.

Abbreviations: aHUS, atypical hemolytic uremic syndrome; BUN, blood urea nitrogen; LDH, lactate dehydrogenase; SD, standard deviation; STEC-HUS, hemolytic uremic syndrome caused by Shiga toxin-producing Escherichia coli; TTP, thrombotic thrombocytopenic purpura.
Regarding the treatment received by patients with pTMA, plasma exchange was performed in 50% and 44% of patients with TTP and aHUS, respectively. Eculizumab was applied to 67% of aHUS patients. All patients with aHUS and TTP were given antihypertensive drugs during treatment. Transfusion therapy was required during treatment. 37% received red blood cells. Renal replacement therapy (RRT) was required as therapeutic medication, with hemodialysis being the most common modality (48%) in pTMA, but the subgroup of patients with TTP where all received this intervention stands out (see Table 3).
Treatment, Stay in Critical Care During the Follow-Up of Patients with pTMA.

Abbreviations: aHUS, atypical hemolytic uremic syndrome; PICU, Pediatric Intensive Care Unit; RRT, renal replacement therapy; STEC-HUS, hemolytic uremic syndrome caused by Shiga toxin-producing Escherichia coli; TTP, thrombotic thrombocytopenic purpura.
The median stay in the pediatric intensive care unit was 10 days (range 0-51), with a longer stay in patients with aHUS (18 days, range 0-51). Overall mortality reported in the study was 13%, which varied according to etiology, 15.7% in patients with STEC-HUS and 50% in TTP, and 0% in aHUS. Cardiovascular complications occurred in 100% and 89.4% of patients with TTP and STEC-HUS, respectively. Renal complications occurred in 66.6% and 63.1% of patients with STEC-HUS and aHUS. Half of the patients with TTP presented with neurological complications (see Figure 2).

Complications in patients with pTMA after the acute phase.
Discussion
pTMA is an uncommon medical condition caused by diverse etiologies with similar signs but with a series of characteristics that make them heterogeneous. This is the first study that evaluates the etiology, demographic and clinical characteristics, and outcomes of STEC-HUS, aHUS, and TTP in the pediatric population in Colombia. STEC-HUS was the most frequent diagnosis in patients with suspected pTMA at 63%, followed by aHUS at 30%, like what was found by Schönermarck et al 9 in a multicenter study in Germany, where STEC-HUS was found in 69% and aHUS in 30% of cases.
The median age in this study was 2.8 years, but varied according to etiology; 2.1, 4.6, and 5.3 years for STEC-HUS, aHUS, and TTP, respectively. 6 In 258 Japanese pediatric patients with TMA, the median age at diagnosis was 4.3 years with data comparable to our study. Regarding sex, this study showed a female predominance at 54%, like Epperla et al 10 with 59% and Bendapudi et al 11 with 60%.
Previous studies used the presence of diarrhea as a discriminating factor between aHUS and STEC-HUS.12,13 However, it has been clarified that diarrhea is not unique to STEC-HUS and patients with aHUS may also develop gastrointestinal symptoms. 14 In our study, 56% of patients with aHUS had diarrhea, which was higher than in patients with STEC-HUS with 32%. Schönermarck et al 9 found that 100% of pediatric patients with aHUS and 89% of patients with STEC-HUS showed gastrointestinal symptoms, illustrating the limitations of the differential diagnosis. In the Colombian context, gastrointestinal infections are common due to sociodemographic and sanitation conditions in our population, especially in rural areas, so considering diarrhea and pTMA criteria, STEC-HUS should always be suspected as the first cause.
Initial laboratory studies showed a mean hemoglobin 8.6 ± 1.9 g/dL, with a significant impact in TTP with 7.6 ± 0.7 g/dL. In the study by Froissart et al, 15 the mean hemoglobin was 8.0 ± 2.2 g/dL. Regarding other paraclinical variables such as platelet count in this study, the mean was 63 103 ± 41/μL, while Dahlan et al 16 reported a mean of 60.5 103 ± 65/μL for this same variable. The mean platelet count in patients with TTP was 9 103 ± 0.7/μL, which is in accordance with reports in the world literature and is relevant as it is a criterion for predictive scores for the diagnosis of early TTP.9,11,17
The presence of schistocytes was reported in 90% of patients in this study, higher than the 79% reported by Bayer et al. 17 This finding is relevant because, although they may be present while the sample is being taken, in some laboratories they are not routinely reported. This leads to possible confusion at the time of diagnosis. Additionally, this study showed a high mean in serum creatinine compared to other pediatric and adult series,9,11,17 which is relevant because it may correspond to late diagnoses in the course of the disease.
This study found a strong overlap in clinical presentation with respect to general, gastrointestinal, renal, cardiovascular, and neurological symptoms in each of the pTMA etiologies. These signs, symptoms, and laboratory variables are in most cases not enough when analyzed in isolation to diagnose each clinical entity. This reinforces the importance of additional tests such as polymerase chain reaction (PCR) for STEC-HUS and ADAMTS13 activity to assess the correct diagnosis.
As for severe deficiency of ADAMTS13 activity (≤5%-10%) which is a marker of TTP, its assessment takes considerable time to confirm the diagnosis and establish a treatment, which must be urgent; therefore, clinical and laboratory measures have been used to predict its activity.11,18 One of them is the PLASMIC score, a validated tool to predict severe deficiency of ADAMTS13 based on the evaluation of seven variables, where a score ≥6, would represent a 72% risk of severe deficiency of ADAMTS13.11,19
In this study, the mean PLASMIC score for patients with TTP (7% of pTMA) was 5.5 ± 2.1, with ADAMTS13 levels of 5%. Although there were only two patients with TTP, the mean creatinine was 3.1 ± 0.1, a rare finding in this entity that could have affected the result and is possibly explained by late diagnosis. For this reason, it is evident that further validation studies are needed regarding the usefulness of the PLASMIC score as a predictive measure and for rapid decision-making in the context of urgent plasma exchange. Adjustments to prediction are currently being proposed to improve its performance.20-22
Plasma exchange was performed in 23% of patients with pTMA, like the 29.1% and 13.9% reported by Ashida et al 6 and Henry et al 23 50% and 44% of patients with TTP and aHUS in the present study received plasma exchange. This is a validated measure in these two conditions and potentially lifesaving if instituted early. Eculizumab, an anti-C5 monoclonal antibody, is an approved treatment measure for aHUS with growing evidence.24,25 In this study, 67% of patients with aHUS received eculizumab, a finding of interest due to the availability and increase in its use in our environment, which is strikingly equal to or higher than that reported in studies conducted in developed countries.6,26,27
Supportive treatment during the acute phase is essential in pTMA treatment. Studies by Bayer et al 17 and Henry et al 23 reported that 40% and 43.5% of patients, respectively, received a red blood cell transfusion, similar to the 37% found in this study. The use of antihypertensive drugs was reported in 100% of patients with aHUS and TTP, related to the greater renal and cardiovascular involvement estimated in these two entities. Hemodialysis was required in 48% of patients with pTMA, like that found in the study by Ashida et al 6 (39%), but distant from Dahlan et al 16 (11%). The difference between the studies lies in the late diagnosis in the present study, which contributes to greater renal involvement and the need for this therapy.
Our study estimated mortality at 13%, which is consistent with Minoia et al 27 and Dahlan et al. 16 There was a higher mortality rate among patients with TTP than in the other entities, which is consistent with other studies6,16 as well. The rate of complications in this study is high compared with other pediatric patient series. The majority, with no difference between the entities studied, experienced cardiovascular complications, the most common being arterial hypertension. 61% and 66% of patients with STEC-HUS and aHUS developed some degree of chronic kidney disease. A relevant fact in this study is that all TTP patients required renal replacement therapy in the acute phase. However, they did not have any degree of chronic kidney disease.
This study has some limitations. pTMA is a disease that occurs infrequently. The present study has a small sample, so the comparative analysis between the different entities has some limitations. The retrospective nature of the study did not allow follow-up of variables of interest in the diagnostic setting (PCR for STEC or ADAMTS13) and in some patients, results were not collected. In addition, long-term outcomes were measured according to patient visits during outpatient follow-up, but some patients did not continue follow-up in the hospitals included in the study. It is noteworthy that in the present series, two patients had TTP, which did not allow a direct comparison with the other entities, but this finding is consistent with other studies due to the rarity of this condition.6,9
The study was conducted in two highly complex medical centers in a major Colombian city. This may overestimate the true behavior and availability of resources in other cities or municipalities with a lower level of healthcare complexity than in our country.
Conclusion
This study constitutes the largest series of pTMA in the pediatric population in Latin America. This study examines the etiological, clinical, laboratory, and result behavior of STEC-HUS, aHUS, and TTP in this region of the world, which is not significantly different from studies conducted in Japan, Germany, or the United States. This was a retrospective study which provides important information about the behavior of this rare disease. Further prospective studies in Latin America may provide additional insight.
Supplemental Material
sj-docx-1-gph-10.1177_2333794X241307535 – Supplemental material for Primary Thrombotic Microangiopathy in Pediatric Patients
Supplemental material, sj-docx-1-gph-10.1177_2333794X241307535 for Primary Thrombotic Microangiopathy in Pediatric Patients by Andrés David Aranzazu Ceballos, Lina María Martínez Sánchez, Ana Paulina Pamplona Sierra, Daniela Vergara Yánez, Liliana Franco Hincapié and Richard Baquero Rodriguez in Global Pediatric Health
Footnotes
Acknowledgements
We would like to acknowledge to the designer AA for her contribution in the graphical abstracts.
Author Contributions
ADAC, LMM, DVY, RBR, APP, and LFH contributed to the design and supervised data collection. ADAC, LMM, RBR and LFH contributed to data analysis.All of the authors contributed to the preparation of the manuscript. All authors have read and agreed to the published version of the manuscript.
Data Availability
All data are fully available without restriction by the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
The study was approved by the institutional review board and the Ethics Committee of the Universidad Pontificia Bolivariana [Ref. No. 2019-23], Hospital Pablo Tobon Uribe [Ref. No. 2020-005], and Hospital San Vicente Fundación [Ref. No. 26-2020]. The research was conducted in accordance with the ethical guidelines of Helsinki’s Declaration (2013). Data sheets were coded with numbers to maintain the anonymity and confidentiality of patient’s data. This article does not contain any studies with animals performed by any of the authors.
Informed Consent
The ethical committee has approved this form of consent.
Consent for Publication
All authors approved the manuscript for publication.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
