Abstract
Objectives. Atypical hemolytic uremic syndrome (aHUS) is a rare complement-mediated kidney disease with genetic predisposition and represents up to 10% of pediatric hemolytic uremic syndrome (HUS) cases. Few studies have evaluated aHUS in Latin American population. We studied a Colombian pediatric cohort to delineate disease presentation and outcomes. Methods. A multicenter cohort of 27 Colombian children with aHUS were included. Patients were grouped by age at onset. Clinical features were compared using analysis of variance (ANOVA) and Fisher exact tests. Renal biopsy was performed on 6 patients who were suspected of having other renal diseases before aHUS diagnosis. Results. Most patients were male (70%). The onset of aHUS occurred frequently before age 4 years (60%) and followed gastroenteritis as the main triggering event (52%). Age groups showed comparable clinical presentation, disease severity, treatment, and outcomes. Pulmonary involvement (67%) was the main extrarenal manifestation, particularly in the 1 to 7 age group (P = .01). Renal biopsies were as follows: 3 had membranoproliferative glomerulonephritis (MPGN) type I, one MPGN type III, one C3-glomerulonephritis, and one rapidly progressive GN. Genetic screening was available in 6 patients and identified 2xCFHR5, 2xMCP, 1xADAMTS13/THBD, and 1xDGKE mutations. A total of 15 relapses were seen, of which 8 (72%) occurred in the 1 to 7 age group. The renal outcome was not significantly different regardless of age group. Conclusion. In our cohort, we observed a relatively high frequency of extrarenal involvement at first presentation represented by pulmonary manifestations. The renal prognosis at initial presentation was worse than in previous reports.
Keywords
Background
Atypical hemolytic uremic syndrome (aHUS) is a rare, genetically-mediated systemic disease characterized by the triad of microangiopathic hemolytic anemia, thrombocytopenia, and acute kidney injury (AKI).1,2 Chronic and uncontrolled activation of the complement system leads to the activation of endothelial cells, recruitment of platelets, and thrombotic microangiopathy (TMA). 3 Due to the rarity of the disease, the global epidemiology of aHUS is not well known. Some population-based estimates suggest a prevalence in the age group of 20 years or younger ranging from 2.2 to 9.4 per million population. 4 Among all cases of typical hemolytic uremic syndrome (HUS) in children, up to 10% are recurrent cases of aHUS. 2
According to the etiology, aHUS can be divided into subgroups: complement alternative pathway dysregulation,3,5 anti-complement factor H autoantibodies (anti-CFH), 6 mutations in the coagulation pathway gene, 7 and also in combination of the above.7,8 Regardless of the etiology, patients with aHUS may have extra-renal manifestations (ie, neurological, cardiovascular, pulmonary, and gastrointestinal), and poor outcomes: up to 25% of mortality rate, and about 50% of disease progression to end-stage kidney disease (ESKD)2,5,9
Various hereditary or acquired deficiencies in the complement alternative pathway proteins have been identified in up to 60% of patients with aHUS, 9 and about 67% of cases with genetic mutations are affected during childhood. 2 Most mutations are seen in complement factor H (CFH)10,11 while membrane cofactor protein (MCP or CD46) 5 and complement factor I (CFI) 12 mutations are known to predispose the development of aHUS. Gain-of-function mutations in complement factor B (CFB) 13 and complement 3 (C3) 14 genes have been reported in a few cases. Additionally, mutations in the thrombomodulin gene (THBD), encoding membrane-bound thrombomodulin 15 and diacylglycerol kinase epsilon (DKGE) gene have been also related to aHUS. 16
The genetic composition of Latin American populations is a heterogeneous result of admixture mainly between European, Native American, and African populations.17,18 Colombia has a strong background of African and indigenous populations which has differentially shaped the genetic population structure when compared with regions of other countries.19,20 Patient registries are required to characterize the diseases natural history and to evaluate clinical therapies. Since genetic backgrounds largely underlie the pathogenesis of aHUS, investigation of the clinical and genetic characteristics in patients with different ethnic backgrounds may be of clinical value. However, our knowledge of the pediatric aHUS clinical profile in our region is limited. This study aims to describe a series of pediatric cases with aHUS in a nationwide Colombian cohort.
Methods
Study Population and Design
From 2010 to 2019, unrelated Colombian children with aHUS were prospectively collected from different medical centers throughout Colombia (Figure 1). Diagnosis of aHUS was based on clinical criteria: hemolytic anemia characterized by a hemoglobin (Hb) level <10 g/dl, thrombocytopenia with a platelet count <150 × 109/l, elevated levels of lactate dehydrogenase (LDH), negative Coombs test, and neurological/pulmonary/gastrointestinal organ involvement. All are in association with acute kidney injury (AKI) (ie, serum creatinine levels greater than age-related norms).21,22

Distribution of pediatric aHUS cases in Colombia.
Patients included in the sample were not required to have an identified complement gene mutation or factor H autoantibody, nor were they required to have previous or ongoing treatment with eculizumab. Genetic analyses (if available) were obtained retrospectively. We excluded patients presenting with HUS secondary to drugs, autoimmune diseases, infections (caused by Shiga toxin-producing Escherichia coli, Streptococcus pneumoniae, or HIV), bone marrow, or solid organ transplantation.
Three groups of patients were individualized according to their age at onset: 0 to 1, 1 to 7, and ≥7 years. We compared the clinical findings including clinical manifestations, disease severity, extrarenal involvement, treatment, outcomes, and overall renal survival between the age groups.
Renal biopsy was performed on 6 patients who were suspected of having another renal disease before aHUS diagnosis. The presence of proteinuria was defined as >0.2 g/24 h or a positive dipstick for proteins. Hypertension was defined as a consistent blood pressure >p95 for age and length.
Eculizumab Treatment
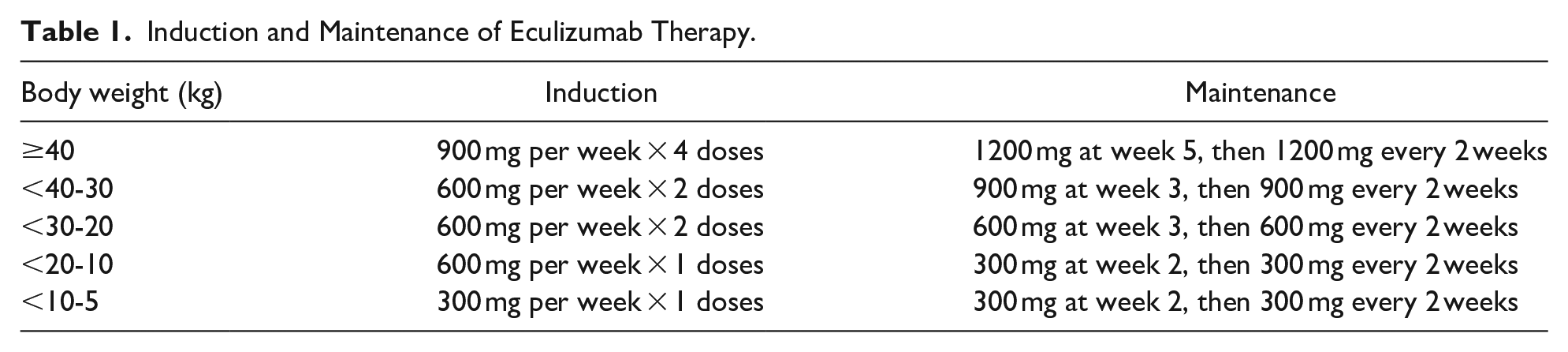
Induction and maintenance of Eculizumab therapy were based on the Colombian aHUS clinical consensus criteria. 22 Eculizumab was administered according to the patient body weight (Table 1). Patients ever-treated received >1 dose of eculizumab prior to, during, or after enrollment. Patients treated were vaccinated against Neisseria meningitides prior to eculizumab initiation and received prophylactic antibiotics up to 2 weeks post-eculizumab.22,23 On the other hand, patients never-treated had no history of eculizumab being used at any time. Reasons for not receiving eculizumab were difficulties in acquiring the drug due to cost and availability in the region. 22 Remission was defined by a normal platelet count, the absence of hemolysis, and (1) a glomerular filtration rate of >60 mL per minute per 1.73 mm2 using the Schwartz formula or (2) stable kidney function for at least 6 months.22,24
Induction and Maintenance of Eculizumab Therapy.
Serum Complements Measurements
Serum concentrations of complement 3 (C3) and complement 4 (C4) were measured by nephelometry at each hospital.
Complement Genetics
All coding sequences of the CFH, CFI, MCP, C3, CFB, DGKE, ADAMTS13, and THBD genes were sequenced with Illumina technology (MiSeq), with a minimum average coverage of 500× and a minimum coverage of 20×. Screening for complex genetic disorders affecting CFH, CFHR1, CFHR3, and CFHR5 was undertaken using multiplex ligation–dependent probe amplification from MRC Holland.
Statistical Analysis
Numerical variables were summarized by means and ranges. Binary and categorical variables were summarized by frequency (in percent). To test for important differences in either age groups or eculizumab treatment groups, the analysis of variance (ANOVA) was performed for numeric variables and Fisher exact test for categorical variables. The Shapiro-Wilk statistical test confirmed the normal distribution of the quantitative data for further statistical evaluations (P > .05). As such the above-mentioned so parametric statistical tests were used for the data analysis in this study. P-values <.05 were considered statistically significant. Statistical computations were conducted using R-CRAN version 4.2 software. 25
Results
Patients
During the study period, we identified 27 children who met the diagnostic criteria for aHUS. Figure 1 shows the distribution of this multicenter study in Colombia. Pediatric aHUS patients’ characteristics are summarized in Table 2. Most patients were male (70%) with a male-to-female ratio of 2. The mean age was 5.1 (interquartile range: 0.2-16 years) and 37% of cases were presented in the 1 to 7 year age group.
Characteristics of Pediatric aHUS Patients According to the Age of Onset.

Ranges in the square brackets.
Abbreviations: yr, years; Hb, hemoglobin; PLT, platelet; LDH, lactate dehydrogenase; Cr, serum creatinine; PU, proteinuria; ESKD, end-stage kidney disease.
Based on analysis of variance (ANOVA) or Fisher exact test.
Clinical Findings and Extrarenal Manifestations
Age groups were comparable in clinical manifestations, and disease severity (Table 2). Most patients (60%) experienced disease onset before age 4 years. Hypertension (blood pressure > 95th percentile for age) was present at onset in 24/27 (88%) patients. Prespecified triggers for the development of aHUS were as follows: gastroenteritis (52%), upper respiratory infection (19%), and urinary infection (15%). A combination of triggering events was seen as well; for instance, diarrhea and fever were seen in 9/27 patients, 6 of whom also had bloody diarrhea.
All patients had extrarenal involvement. Nineteen cases (70%) had ≥2 extrarenal manifestations and 5/27 (18%) patients had ≥4 extrarenal manifestations. Pulmonary involvement (18/27, 67%) was the main extrarenal manifestation. Other pulmonary failures (21%), pulmonary edema (10%), diffuse alveolar hemorrhage (8%), and pleural effusion (7%) were recorded. A higher pulmonary frequency involvement was observed in the 1 to 7 age group (P = .01). Twelve cases (44%) presented with neurologic manifestations (altered consciousness or coma, seizures, and focal neurologic deficit), and 3/12 cases had cerebral infarction and edema during their first aHUS flare. Cardiovascular involvement was observed in 9/27 (33%) cases, and mainly manifested as pulmonary hypertension (14%), hypertrophic cardiomyopathy (11%), and a combination of cardiac failure with dilated cardiac chambers and decreased ejection fraction (7%). Renal involvement was more severe in the 1 to 7 year age group, all requiring hemodialysis at the first aHUS episode.
Renal Biopsy
Clinical data of 6 patients with biopsy-proven glomerulopathy before aHUS diagnosis are summarized in Table 3. Three of 6 patients had membranoproliferative glomerulonephritis (MPGN) type I, one MPGN type III, one C3 glomerulonephritis (C3GN), and one rapidly progressive glomerulonephritis (Figure 2).
Clinical Data of 6 Pediatric aHUS Patients With Biopsy-Proven Glomerulopathy.

Abbreviations: CKD, chronic kidney disease; ESKD, end-stage kidney disease; FSGS, focal and segmental glomerulosclerosis; C3GN, C3 glomerulonephritis; IF, immunofluorescence staining; MPGN, membranoproliferative glomerulonephritis; RPGN: Rapidly progressive glomerulonephritis; PE, plasma exchange; PU, proteinuria.
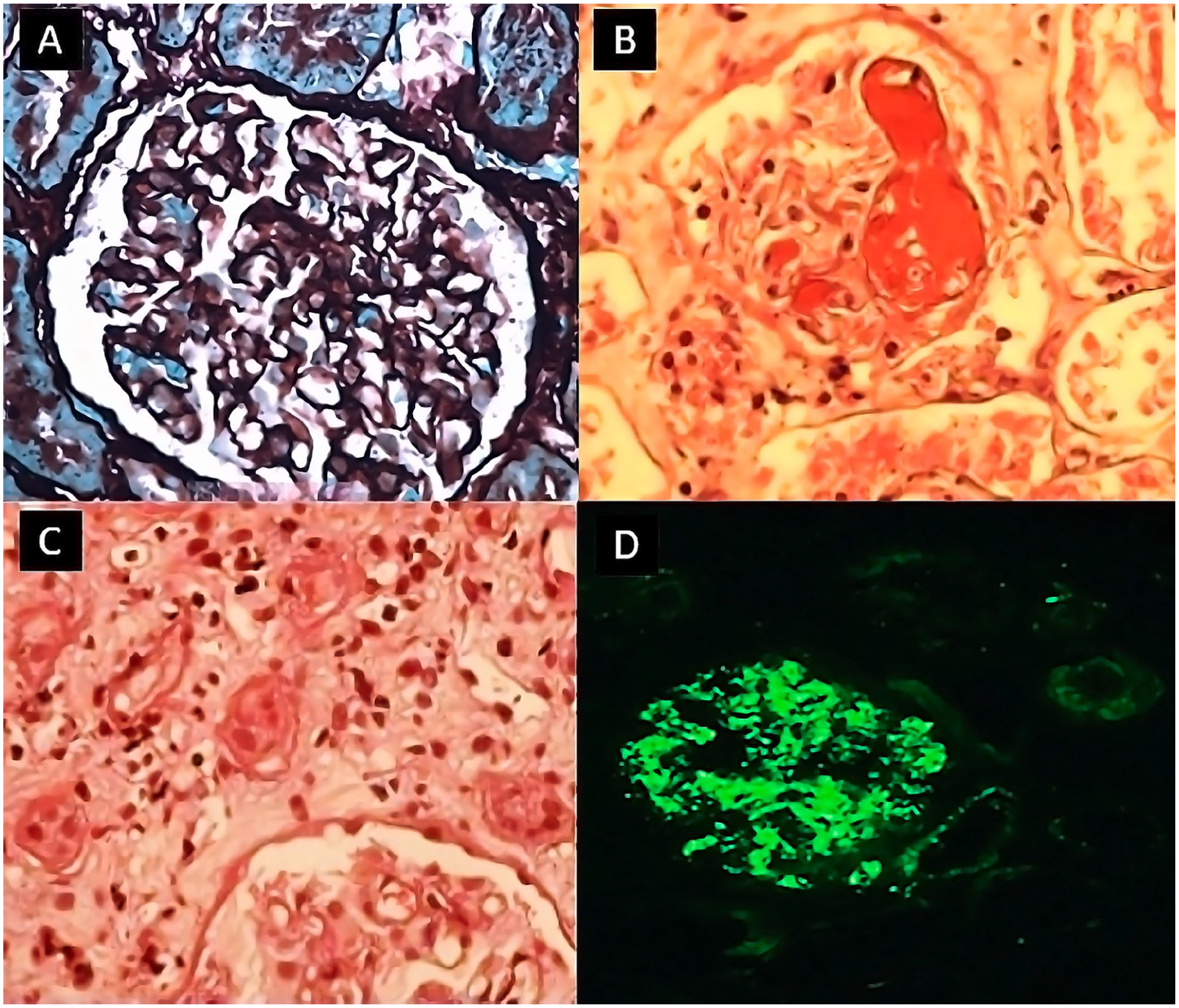
Light microscopy findings in children who developed glomerulopathy associated with aHUS. (A) Membranoproliferative glomerulonephritis—silver stain. (B and C) Rapidly progressive glomerulonephritis. (B) Glomerulus with sclerotic changes, endothelial swelling, and focal mesangiogliosis secondary to thrombotic microangiopathy. (C) Endothelial proliferation and fibrinoid microthrombi. (D) C3 Glomerulopathy (with anti-C3c antibody 400×) diffuse granular deposition.
Genetic Background
Genetic screening to assess alterations in complement-regulating proteins was available in 6/27 (22%) patients. Two patients were identified with homozygous complement factor H related 5 (CFHR5) mutation (c.3019G > T, p.V1007L; c.3644G > A, p.R1215Q), 2 patients with heterozygous MCP mutation (c.565T > G, p.Y189D; c.565T > G, p.Y189D), one case with homozygous ADAMTS13 and heterozygous THBD disease-associated variants (c.3287G > A, p.Arg1096His; c.1456G > T, p.Asp486Tyr) respectively, and one case with heterozygous DGKE variant of uncertain significance (c.994G > C, p.Val332Leu). We were not able to assess the clinical significance of the mutation types in this study due to a limited number of cases per mutation.
Treatment
Small volume infusion of fresh-frozen plasma, steroids, plasmapheresis, and eculizumab was comparable used in age groups (Table 2). Plasmapheresis was used in 15 (56%) patients, 9/15 (60%) required 1 to 5 sessions, 4/15 (27%) 6 to 10 sessions, and 2/15 (13%) more than 15 sessions. The goal was to change 1 to 1.5 times blood volume with fresh plasma until platelets >100 000 × 109/l. 16 Eleven cases (40%) were on concomitant antiproteinuric agents, such as angiotensin-converting enzyme inhibitor (ACEI) or angiotensin II receptor blocker (ARA). A total of 22 patients (81%) underwent hemodialysis, of which 11/22 (50%) of the cases required hemodialysis for 1 to 7 days, and 7/22 (32%) for more than 20 days.
There were 20/27 (74%) and 7/27 (26) patients ever-treated and never-treated with eculizumab, respectively. Baseline clinical characteristics, disease severity, cardiovascular, central nervous system, and pulmonary involvement were comparable between patients who were ever-treated and never-treated with eculizumab (P > .05). A higher frequency of gastrointestinal system involvement was observed in ever-treated patients (P = .008). Complete remission was observed in 14/20 (70%) ever-treated patients. The proportions of patients receiving hemodialysis and plasma exchange/plasma infusion did not differ between ever-treated and never-treated patients (P > .05). No side effects were observed with eculizumab treatment in this study.
Outcome and Renal Survival
Relapses were observed in 8/27 cases. A total of 15 relapses were seen, of which 8 (72%) occurred in the 1 to 7 age group. Most relapses occurred after a viral infection, usually of the upper respiratory tract. The time between aHUS onset and the first relapse varied widely, from 1 month to a maximum of 5.5 years. In 42% of patients, chronic hypertension was seen after the onset; proteinuria was sustained in 20% of cases, and most of these patients had to be treated for several months with antihypertensive agents.
Two patients received renal transplants without disease recurrence. Despite an initial decrease in the severity of renal and extrarenal involvement after induction therapy, 5/20 (35%) ever-treated patients remained with slight proteinuria (>500 mg/day) and 3/5 progressed to ESRD, of which 1/3 case was on chronic dialysis, received 1 dose of eculizumab after a TMA manifestation, and died the next day with serious, severe acute respiratory failure and acute cardiac failure. In these 5 cases, maintenance therapy could not be complete due to difficulties in drug acquisition and availability.
Overall Kaplan-Meier estimate of the 5-year survival without ESKD was 65% in the aHUS children. The Kaplan-Meier estimates for ESKD-free survival probability were not different between patients when analyzed by aHUS age groups (log-rank P = .73) (Figure 3). Analyses of ESRD-free survival in Eculizumab treatment groups showed longer ESRD survival in ever-treated patients (log-rank P = .016) (Figure 4).

Cumulative Kaplan-Meier estimates for end-stage renal disease survival probability analyzed according to pediatric atypical hemolytic uremic syndrome (aHUS) age groups at initial presentation. Number of patients at risk shown for months after initial aHUS presentation.

Cumulative Kaplan-Meier estimates for end-stage renal disease survival probability analyzed according to Eculizumab treatment group (b). Number of patients at risk shown for months after initial aHUS presentation.
Discussion
We have defined the clinical data and outcomes of aHUS in a nationwide Colombian series of 27 children according to age groups. Six patients with aHUS underlying glomerulopathies were also described. This study confirms previous registry findings 11 and provides new insights regarding these phenotypic variables on the clinical manifestation and progression of aHUS in this population.
In this study, the majority of registry participants are male, supporting previous French aHUS pediatric registry data. 11 In contrast, Noris et al, 5 reported a majority of female patients (47%), suggesting variance in aHUS characteristics by region and patients enrollment differences. Most of the patients (53%) presented between the age of 1 and 7 years, which is also the most common age for children with typical HUS, which suggests age at onset cannot always help distinguish between typical and aHUS. 1 Cases presenting with LN were excluded since an association of this condition with microangiopathic hemolytic anemia is well established and up to 10% of LN patients have this complication. Complement dysregulation and CFH dysfunction in the susceptibility to SLE appears to emerge from genetic studies.26,27
Although the renal microvasculature is a common site of TMA, aHUS can manifest in many organ systems (ie, gastrointestinal, respiratory, cardiovascular, nervous) and can mimic many conditions. 28 Extrarenal involvement in aHUS has been mainly reported at initial presentation in both adults and children. 29 Case series or literature reviews have reported extrarenal involvement rates up to 46% in recently diagnosed children depending on the organ system.5,21,29,30 The Global aHUS Registry (n = 1321 patients; adult, n = 842; pediatric, n = 479) is the largest aHUS cohort to systematically record extrarenal manifestations associated with aHUS. The main extrarenal manifestation associated with aHUS was gastrointestinal involvement in both adults and children at the initial presentation phase (47% vs 33%, respectively). 29 Similarly, an adult aHUS series of cases published previously 31 in this population showed a high frequency of gastrointestinal involvement (75%).
One of the most important observations in this study is the relatively high frequency of extrarenal involvement in pediatric patients at initial presentation mainly represented by pulmonary manifestations (67%) compared to the pediatric pulmonary involvement rate reported in the Global aHUS Registry (12%). 29 In this study, a greater proportion of pediatric patients experienced Gastrointestinal (59% vs 47%) and central nervous system (44% vs 25%) involvement than Global aHUs Registry. 29 Complement abnormalities and extrarenal manifestations correlation associated with aHUS were not assessed due to insufficient genetic information.
Pulmonary complications of aHUS are typically seen in cases of multiple organ dysfunction and pulmonary edema associated with cardiac dysfunction and/or systemic volume overload. 28 Careful monitoring of fluid and respiratory status is required to reduce the risk of pulmonary compromise. 28 As further data are collected it may become possible to predict patients at risk of specific pulmonary complications, providing useful information to guide monitoring health strategies.
In our study, up to 15% of cases were diagnosed with other biopsy-proven renal diseases before aHUS diagnosis. We observed 3 cases of MPGN type I, one MPGN type III, and one of C3GN. A close link between aHUS and MPGN has been reported in previous case reports.32-34 Cases have been described where renal biopsies first suggested MPGN, and then aHUS or vice versa, 35 which recognize similar aetiopathogenetic mechanisms involving complement dysregulation. Mutations in CFH, CFI, and MCP have been reported in patients with type I and type II MPGN which suggest a genetically determined dysregulation of the alternative pathway of complement in MPGN. 36 Loss of regulation of complement alternative pathway activity leads to deposition of complement factors and glomerular inflammation, resulting in proliferative glomerulonephritis.37,38 Different phenotypical expressions of renal disease ranging from aHUS to C3GN have been reported.27,32,36 In our MGPN/C3GN cases, renal biopsy showed C3 mesangial deposition. Serum C3 levels were persistently low. These findings support the alternative pathway hyperactivation mechanism and both C3GN and aHUS.
CFH is a cell surface regulatory protein that protects from uncontrolled complement activation by serving as a cofactor in the cleavage of C3b and accelerating the decay of C3 convertase. 39 The CFH family is completed by a total of 6 CFH-related proteins (CFHRs) that are encoded by genes present as a cluster located on 3′ of CFH (CFHR1, CFHR2, CFHR3, CFHR4, and CFHR5). 39 In this study, homozygous CFHR5 mutation was found in 2 patients. Genomic deletions of these genes lead to an undesirable effect on CFH thereby impairing its functions.39,40 Genetic screening in healthcare centers should be recommended to increase understanding of complement mutations influence on aHUS phenotypic variables and progression. This may involve developing either local or regional testing protocols to ensure appropriate testing of samples as our understanding and technology advance.5,29
Plasma exchange/plasma infusions (PE/PI) have been the standard of care for aHUS in less developed countries and health institutions where the anti-C5 inhibitor Eculizumab is not readily available. 9 Eculizumab cost must be considered, as it is very expensive with >300 000 USD/year of treatment per patient and many insurance companies have budget restrictions. 41 In our country, it may be a challenging therapeutic strategy given the difficulty of costs and availability. 22
Eculizumab, a humanized monoclonal IgG antibody, was the first successful terminal complement inhibitor available to pediatric aHUS patients. It binds to complement protein C5, preventing cleavage into C5a and C5b. Blocking the formation of C5b prevents the continuation of the complement cascade and blocks the formation of C5b-9 membrane attack complex (MAC).9,23,24 In our study, eculizumab therapy allowed to achieve complete remission in 70% of cases ever-treated, a similar rate was reported by Greenbaum et al 42 in a study that evaluated the efficacy and safety of weight-based dosing of eculizumab in 22 children with aHUS and reported a remission rate up to 73% by week 24.
aHUS is known for its poorer prognosis than typical HUS, requiring dialysis in half of the children and reaching ESKD up to 48% after 5 years of follow up.9,11,29 In our study, more than half of children required hemodialysis at initial presentation with relapses mainly in the 1 to 7 age group. This may be attributable to the relatively high frequency of triggering events, the unknown mutational profile effect in our population, and the delay in the initiation of complement inhibitor therapy. Patients who required hemodialysis were the ones whose clinical presentation was a severe acute kidney injury.
To our limitation, firstly, this is a retrospective study with small sample size. Second, genetic screening was available in a few children, thus, we could not analyze if the mutational profiles significantly differed in this ethnic group. Second, a retrospective study may have biased the data regarding treatment outcomes.
Conclusions
In this study, we provided an assessment of clinicopathological features and outcomes of aHUS in Colombian children. The clinical profile was characterized by a relatively high frequency of extrarenal involvement at first presentation represented by pulmonary manifestations. The renal prognosis at initial presentation was worse than in previous reports. More extensive genetic screening is required for this etiologically unknown population, including genetic studies of the complement terminal pathway factors and coagulation pathway factors, and possibly, autoantibodies against CFH.
Footnotes
Acknowledgements
Not applicable.
Author Contributions
ZE and AD conceptualized and designed the study. MC, GG, RB, CP, LG, LR, PA, MCa, YA, LA, WM, OL, RG, AQ, GA, and CM carried out the clinical data collection. ZE and AD analyzed and interpreted the clinical data. ZE, AD, and JV wrote the first draft of this manuscript. MC, GG, DS, AQ, EE, GA and CM reviewed and revised the manuscript. All authors read and approved the final manuscript.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The study was approved by the Ethics Committee of the Clínica de la Costa, Barranquilla, Atlántico, Colombia, recognized by all participating hospitals. All methods were performed in accordance with the ethical standards as laid down in the Declaration of Helsinki and its later amendments or comparable ethical standards. Written informed consent was obtained for the collection of data from all legal representatives of participants.
Consent for Publication
Not applicable.
