Abstract
Introduction. Osteoarticular infections (OI) in children cause considerable morbidity with associated long-lasting sequelae. Comprehensive
Introduction
Osteoarticular infections (OI) require hospitalization and can be associated with significant morbidities and long-lasting sequelae in children. OI may manifest as osteomyelitis (OM), septic arthritis (SA) or a combination of OM and SA depending on the anatomic localization of the infection and age. The majority of SA and OM are pyogenic infections. They may also be related to atypical pathogens. Children below the age of 6 years are more commonly affected. 1 In older children, clinical symptoms are helpful for the diagnosis however in neonates and infants the diagnosis may be challenging as clinical presentation is often non-specific. Early recognition of the disease is particularly important as missed or delayed diagnosis is associated with dissemination and multifocal osteomyelitis, joint destruction, and long-term sequelae.2,3 In addition, bacteriological confirmation including antibiotic sensitivity testing is critical for the choice of the optimal antibiotic treatment regimen and duration. Incidence rates of SA or OM are so far best studied in high-resource settings ranging from 1 to 15 per 100,000. 4 Studies looking at SA in low resource settings are scarce but suggest potentially higher incidence rates of up to 20 per 100 000 in Africa. 5 Data on incidence rates of OI in central and East Asia are very limited and the disease is likely underdiagnosed and underreported because of limited available microbiology facilities and management resources. 6 The Ministry of Health of Mongolia has been collaborating with the Swiss Mongolian Pediatric Project (SMOPP) 7 and implementing state-of-the-art hip sonography screening and preventive treatment for developmental dysplasia of the hip in neonates since 2009. 8 Experience from this project show a high number of pathological hip joints unlikely to be related to primary developmental dysplasia. It was therefore hypothesized that these may be sequelae of former inadequately diagnosed and treated OI. Furthermore, specific data on pediatric OI from Mongolia, are not available and pathogen detection was limited. The aim of this study was therefore to investigate the uptake and outcomes of the use of a standardized process for pathogen detection, sensitivity testing (disk diffusion) and antimicrobial management in children with OI in Mongolia.
Methods
Study Settings
The National Centre for Maternal and Child Health (NCMCH) is a governmental, tertiary care children’s hospital in Ulaanbaatar, Mongolia. The hospital has 424 budgeted beds and is responsible for pediatric and pediatric surgical services in the country with 28 subspecialty departments. It provides treatment for approximately 40 000 inpatients and an average 175 000 outpatients per year. Departments recruiting and managing OI patients are emergency, general pediatric, surgery, pediatric and neonatal intensive care, and the outpatient departments. The hospital has a radiology department and a central laboratory that includes hematology, biochemistry, and microbiology. The latter is equipped with a Bactec® blood culture analysis instrument, which was however not routinely used for pathogen detection of OI prior to this study.
Ethical Approval
The study protocol was reviewed and approved by the Institutional Review Board at the National Center for Maternal and Child Health (04/2020) and the Ethical Review Committee of Ministry of Health, Mongolia (152/2020). Written informed consent was obtained from the parents of each child prior to participation.
Study Design and Participants
This was a prospective intervention study using a standardized operating procedure (SOP) with OI diagnostic and treatment algorithms that was developed together with the local team (Supplement 1). The study recruited patients treated for OI at NCMCH between 1st September 2019 and 1st September 2022.
Based on prior in-house statistical analyses for the management of children with OI, an unfavorable outcome rate was 20%. With the proposed intervention, we anticipated a 10% reduction in this unfavorable outcome rate. Power calculations (Stata: power oneproportion) determined that at least 86 subjects were required to detect a change in the proportion from 0.2 to 0.1, with 80% power, using a 5% significance level and a two-sided test.
Inclusion criteria for children aged 4 months – 18 years were: clinical suspicion of OI based on two or more of the following signs and symptoms: (a) fever (≥38.0 °C), (b) soft tissue redness or swelling of joint or limb, (c) pain on movement or limited range or loss of movement, and (d) limp or non-weight bearing or refusal to walk.
Inclusion criteria for neonates/infants aged 0 to 3 months were: clinical suspicion based on one or more of the following signs and symptoms: (a) soft tissue redness or swelling of joint or limb, (b) diminished spontaneous movement of extremity and (c) handling discomfort/ irritability when touched, and (d) joint effusion on sonography. Predefined exclusion criteria for study participation were: children with known immunodeficiency or oncological diagnosis, unwilling to give informed consent or absence of a guardian for children under 14 years to give informed consent.
Diagnostic and Treatment Protocol
The standardized operating procedure for diagnosis, treatment and follow-up of OI was available for trained research and clinical staff involved in the study. Parent information hand-out (Mongolian language) was given to the screened study participants.
All children had an inclusion visit at baseline and four predefined follow-up data collection visits at days 4, 14, 28 and 90 (Supplement 2). At baseline the patient’s history, signs and symptoms were recorded on the clinical report form. Routine laboratory investigations included hemoglobin, white blood cell count (WBC), erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) and blood culture using Bactec® blood culture bottles (BD Bactec™ Peds Plus™ Medium) and conventional agar plate method. Plain radiography of the affected area was performed if OM was suspected. Sonography was performed to identify arthritis signs and effusion in affected joints. Moreover, in order to detect a subluxation/dislocation of the hip due to a large effusion the Graf’s method of hip ultrasound for <24 months old children was performed.
Microbiological Diagnosis
If effusion was detected (defined as the distance femoral cortex to hip capsule >5 mm or >2 mm difference of distance compared to unaffected side), sonography-guided aspiration of joint fluid was performed. If OM was suspected bone aspiration was performed by a pediatric surgeon. Samples (joint fluid and/or bone aspirate) if possible and depending amount were aliquotted in the following priority (1) 1 ml into Bactec® blood culture bottles and (2) 1 ml into sterile containers for conventional agar plate culture methods and transferred to the microbiology laboratory at NCMCH. The Bactec® bottles containing the aspirated synovial fluid or aspirated bone fluid were incubated in the Bactec 9240 system for up to 5 days. Sensitivity testing disk diffusion method of Clinical Laboratory Standards Institute (CLSI) was done with 15 antibacterials (amoxicillin, ampicillin - sulbactam, ceftriaxone, clindamycin, azithromycin, cefotaxime, vancomycin, ceftazidime, erythromycin, ciprofloxacin, chloramphenicol, cefazolin, clarithromycin, lincomycin, and meropenem).
Antibiotic Treatment
The empirical treatment regimen for infants 0 to 3 months of ages was intravenous amoxicillin plus gentamicin; and for children 4 months – 18 years a first-generation cephalosporin (cefazolin) plus gentamicin. Second line antibiotics were used if the following criteria were met: (a) adjustment according to antibiotic susceptibility testing, (b) no improvement of symptoms (inflammation and pain, range of movement), (c) referral cases from rural hospitals where first line antibiotics had already been started. Second line parenteral antibiotic regimen included: third generation cephalosporin (cefotaxime, ceftriaxone or ceftazidime). The empiric antibiotic treatment was adjusted according to pathogen and its antibiotic susceptibility profile. Depending on antibiotic susceptibility testing the following further antibiotics were available for treatment: vancomycin, lincomycin, meropenem and ampicillin – sulbactam etc.
Children 0 to 3 months were treated intravenously without switching to an oral treatment. Children ≥4 months were switched to an oral antibiotic according to SOP (Supplement 1) if the following criteria were met: (a) improvement of symptoms, with decreased inflammation and pain, (b) afebrile or decreased body temperature for 24-48 hours, (c) decrease in CRP by at least 30% from maximum value, (d) no signs of complications, such as endocarditis, pneumonia, septic thrombosis etc. or (e) absence of rare pathogens (eg, Salmonella sp) or resistant pathogens, methicillin resistant S. aureus (MRSA).
Data Collection and Definitions
A paper-based case report form was used to collect data which was then entered by the local team into a central trial database (secuTrial® interActive Systems GmbH, Berlin, Germany) hosted by the clinical trials unit in Lucerne, Switzerland.
Statistical Analysis
Continuous variables are expressed as means and standard deviations or as medians with interquartile ranges as appropriate and categorical variables are expressed as percentages. Categorical data were analyzed using the chi-square test or nonparametric test (Fisher’s Exact test). Comparative analyses of descriptive and inferential data were performed using Student’s t-test or Mann-Whitney U-test.
Results
Patients’ Characteristics
All OI (OM and or SA) patients presented during the study period were screened for potential inclusion of which all (n = 71) agreed to take part in the study. The median (Interquartile range- IQR) age at diagnosis in was 26.3 (0.9-85.0) months and 36 children (50.7%) were female. A total of 42 (59.1%) children presented with septic arthritis (SA), 6 (8.5%) with osteomyelitis (OM) and 23 (32.4%) with SA associated with OM (SA + OM). A complete set of follow-up data (at 90 days follow-up visit) were available in 70 (98.6%) children (further baseline characteristics are shown in Table 1.
Baseline Characteristics of Included Children.
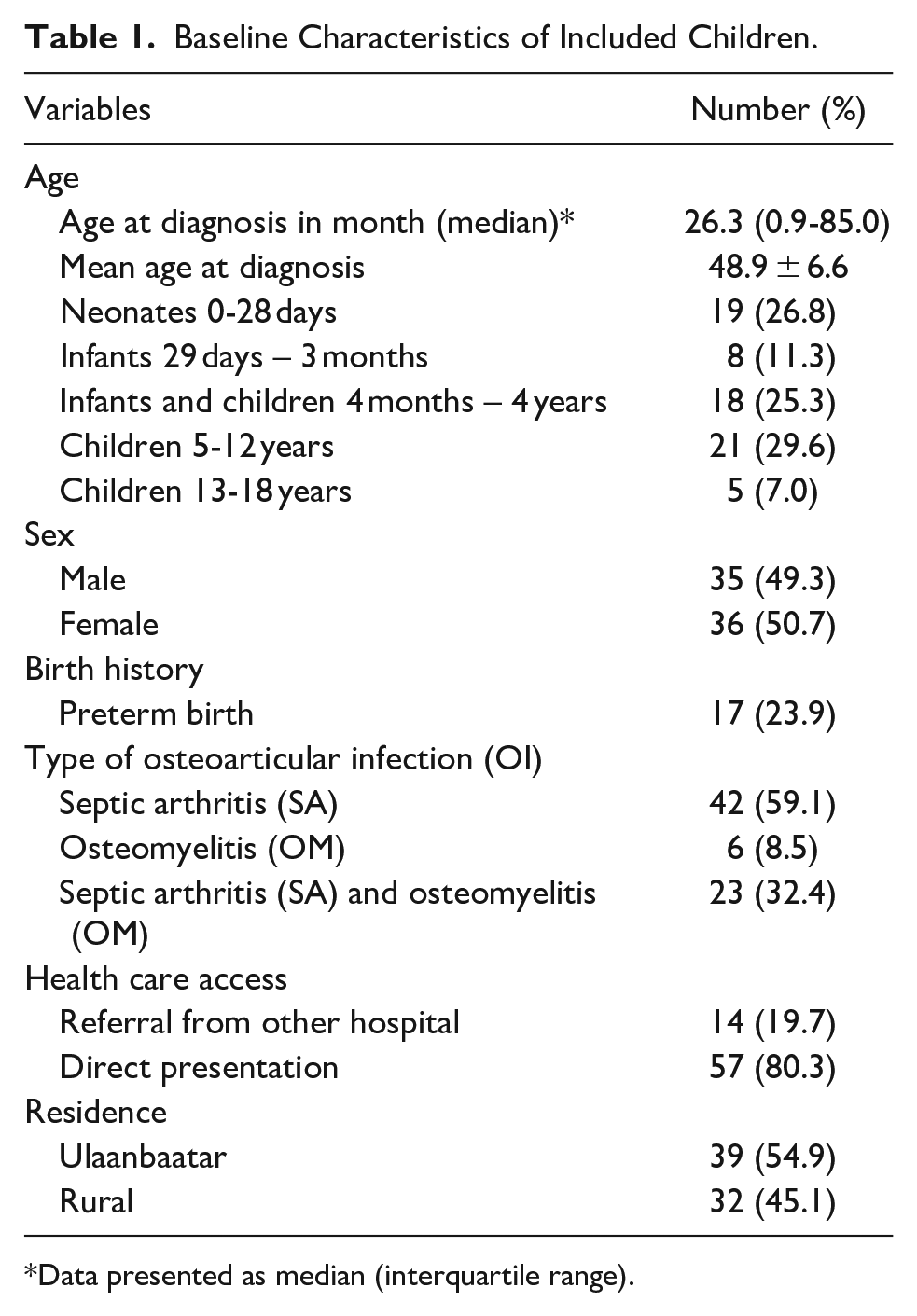
Data presented as median (interquartile range).
Underlying Conditions and Potential Risk Factors
The most commonly noted underlying conditions and potential risk factors for the infants aged 0 to 3 months were intravenous access prior to admission (23/27; 85.2%) and preterm birth (13/27; 48.2). For infants and children aged 4 months to 4 years the majority had dental caries (13/18; 72.2%) and respiratory tract infections (6/18; 33.3%). For patients aged ≥5 years dental caries (16/26; 61.5%) and minor trauma (9/26, 34.6%) were the most prevalent conditions and potential risk factors. Further details can are summarized in Table 2.
Underlying Conditions and Potential Risk Factors.

Data presented as median (interquartile range).
Clinical Manifestations
Of all patients 15.5% (11/71) had symptoms for more than 7 days before admission. The most common symptoms were movement restriction/reduced function/mobility (92.9%), fever (71.8%), joint swelling (70.4), pain (46.5%). For OM the femur was the most affected bone (16/29; 55.2%). Multifocal OM sites were identified in 3 (3/29; 10.3%) children. Children with SA predominantly showed infections of the hip (29/65; 44.6%) and the knees (16/65; 24.6%). Multifocal SA sites were identified in 5 children (7.7%). The clinical manifestation in the different age groups is summarized in Table 3. Osteomyelitis mainly affected the femur in all age groups. Patients with SA predominantly showed infections of the hip in the neonates and the 1 to 3 months age groups while infections of knee was prevalent in older age groups (Supplement 3).
Clinical/Laboratory Data.

Data presented as median (interquartile range).
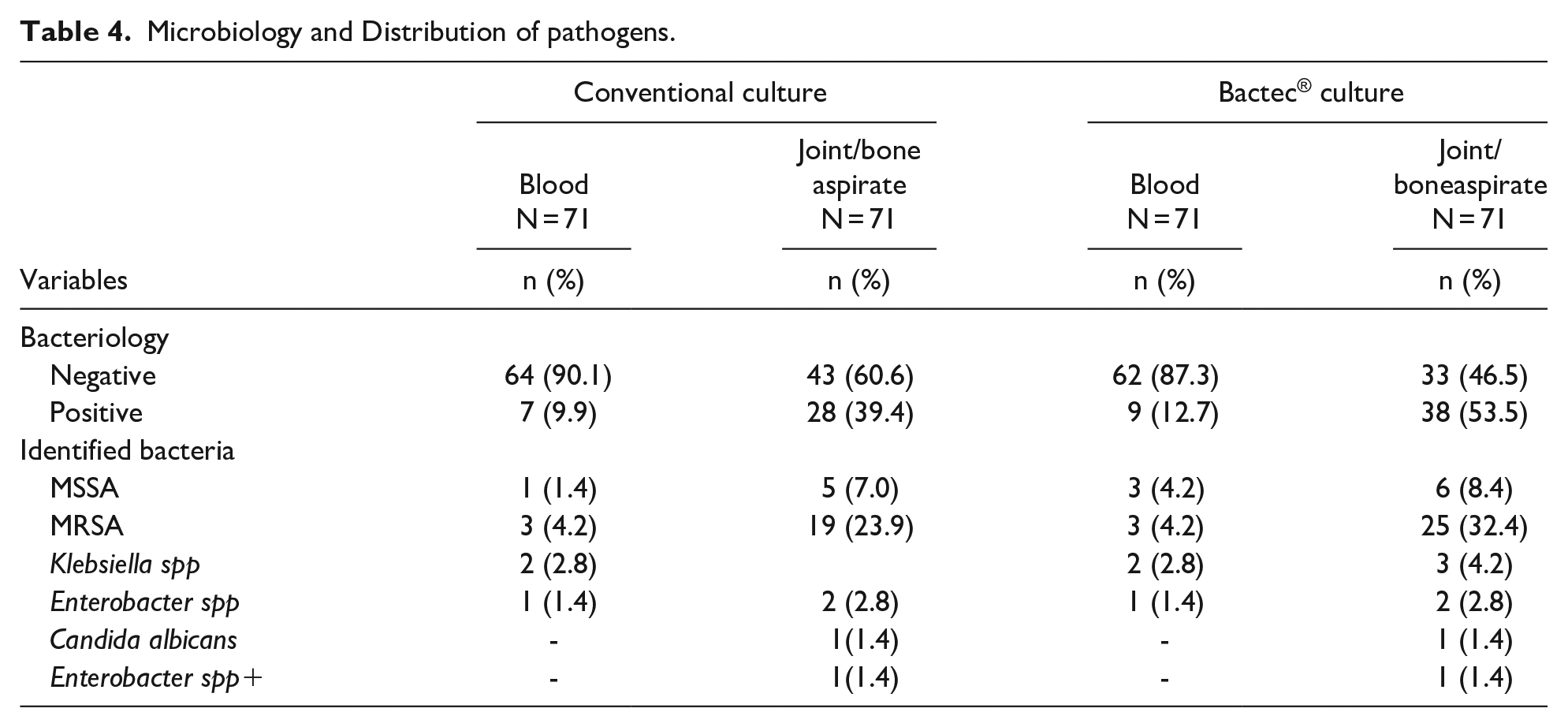
Microbiology Data
A causative pathogen from blood culture was identified in 7/71 (9.9%) by the conventional agar method and in 9/71 (12.7%) using Bactec® culture methods. From the joint or bone aspirates conventional methods detected a pathogen in 28/71 (39.4%)) and via Bactec® culture in 38/71 (53.5%). The most common identified pathogen was Staphylococcus aureus 31/38 (81.6%). Out of these MSSA were detected in 6/31 (19.4%) while MRSA were detected in 25/31 (80.6%) and multi-drug resistance in 4/71(5.6%; Table 4). Of the three cases with multifocal OM sites, one case was caused by MRSA and two cases caused by Klebsiella spp.
Microbiology and Distribution of pathogens.
Radiological Findings
Sonography was abnormal in 90% of all cases: edema of soft tissue and synovial thickening 64/71 (90.1%), joint effusion 63/71 (88.7%) and hyperechogenicity of the surrounding soft tissues 47/71 (66.2%). At the admission, plain radiographs were performed in 33 OM suspected cases and the most common findings were swelling soft tissue (100%), enlargement of joint cavity (46.5%) Osteolytic lesions in bones and bone destruction (40.8%) and periosteal reaction/apposition (40.8%). Plain radiographs were abnormal in 37/71 (52.1%) children at the day 14 follow-up examination (Table 5). The findings were consistent with osteomyelitis and the osteolytic lesions and periosteal apposition were the most common signs in OM cases. At day 90 follow-up control, 5 children had sequelae (Supplement 4).
In 34 cases (0-24 month old children) a hip sonography by Graf method was done. A total of 9/34 (26.5%) children had an abnormal a hip sonography (Type C and Type D) and were treated with a hip flexion and abduction orthosis (Tubingen); but in two cases developed femoral head necrosis due to OI.
Radiological Findings in OI.

Management
Compared to older patients, neonates and young infant children (1-3 months) had longer hospital stays. The median (IQR) duration of intravenous antibiotic therapy was 28 (26-28) days in neonates and 28 (28-28) in 1-3 month age group as indicated in the SOP (Supplement 1). In older age groups, the median duration of intravenous antibiotic therapy was 9 to 12 days, followed by 14 day-oral antibiotics for SA and 21 day-oral antibiotics for OM or SA + OM. Clindamycin was the most frequently (27/71; 38.0%) administered empiric antibiotic followed by Cephazolin + Gentamicin (21/71, 29.6%) and Amoxicillin + Gentamicin (12/71; 16.9%). Meropenem (2/71, 2.8%) and others (ampicillin + sulbactam; 2/71, 2.8%) were also administered in those who did not improve despite empirical antibiotic therapy. Needle aspiration only and joint drainage were performed in 27/71 (38.0%) and 14/71 (19.7%) patients, respectively. Open surgery was performed in 37/71 (52.1%) patients (6 children with OM and 17 with SA, 14 with SA and OM association
OI Management.

Data presented as median (interquartile range).
Discussion
This is the first study to prospectively assess the management of OI in Mongolian children. It includes diagnostic, microbiologic, treatment and outcome data following the implementation of a standardized management protocol, as well as the introduction an automated pathogen detection system (Bactec®). The adoption of the standardized diagnostic and treatment approach was excellent among healthcare providers and patients and their families. Newborns and infants ≤ 3 months represented the largest proportion of patients in our study cohort. This differs from reports in studies conducted in European countries, where this age group accounts for only 3% to 9% of all cases.9,10 Similarly, the mean age at diagnosis of OI of our cohort was 4.1 years (median age 26.3 months), which is lower than that reported in other cohorts: 6.6 in Brasil, 11 12.8 in India 12 and 4.2 years in Belgium. 13 The lower median age and higher proportion of newborns and infants in our study may be due to increased exposure to pathogens, likely resulting from limited hygiene, poor nutritional conditions, and suboptimal healthcare quality. In Mongolia, neonatal sepsis is common, and deaths due to neonatal sepsis increased from 3.2% (21/647) in 2017% to 4.6% (27/591) in 2021. 14 This increase may be a result of prolonged intravenous access, raising the risk of bacterial dissemination, and increasing the likelihood of improper handling of intravenous catheters during both placement and use.
More than half of the patients were diagnosed with SA, the hip being the most affected joint.
The predominance of septic arthritis (59.1%) observed in our cohort aligns with findings from similar-sized tertiary care referral hospitals, where septic arthritis accounts for 45% to 66% of all osteoarticular infections (OI).11,13 In these settings, 45% to 66% of OI cases are classified as septic arthritis. However, other studies from India, 12 Cambodia 6 and Europe 10 have shown a higher prevalence of osteomyelitis. In younger children, concomitant infections of joints and bones are common due to transphyseal blood vessels. 10 In our cohort, 51.8% (14/27) of children aged 0 to 3 months had concurrent infections of both joints and bones. SA, especially in young children, may be associated with a large effusion which may predispose to subluxation and dislocation of the hip. If it is not recognized and treated immediately, it may lead to destruction of the femoral head and subluxation or dislocation of the hip.15,16 In our cohort, 9 cases involved children under 24 months who developed hip subluxation due to significant hip joint effusion. A retrospective study of 14 septic joints in children under 3 months of age reported hip subluxation or dislocation in 2 out of 3 affected hips. 17
Reviewing microbiology of our cohort, almost half of the cases were culture negative. This is comparable to other studies where the culture negative rate is around 30% to 50% with conventional methods.18 -20 To increase the yield of pathogen detection, we used haemoculture flasks (Bactec®) to culture joint and bone aspirates. The added value of Bactec® culture system was shown by 3% to 15% more pathogens being detected in blood and from joint/bone specimens respectively, compared to conventional culture. This increase is also demonstrated in other studies showing the superiorty of the Bactec® system for the detection of clinically significant microorganisms in synovial fluid specimens.21,22 Staphylococcus aureus was the most found pathogen in our cohort with MRSA accounting for 80.6% (25/31). This is in line with other studies in different countries showing consistently S. aureus being the most common causative bacterium of OI including Japan 23 and China. 24 Moreover, in our study, MRSA was the predominant agent in older children like other studies.25,26 In OI studies in children, K. kingae plays an important role as causative pathogen.10,25,27 In our cohort K. kingae was not detected, likely as a result of the difficulty of culturing this fastidious pathogen and as no PCR was available for detection. With this diagnostic innovation future studies will be able to report the frequency of K. kingae associated OI also in Mongolian children.
In Mongolia, data on antibiotic susceptibility is limited. A retrospective study including of 11 889 gram-positive isolates of specimens submitted to the microbiology laboratory at NCMCH in 2019 concluded that compared with published studies from neighboring nations, the rates of antimicrobial resistance among gram positive isolates at NCMCH, particularly with respect to S. aureus and S. pneumoniae, were much higher and multidrug-resistance among E. coli, Enterobacter spp and Klebsiella spp increased.28,29 These results could be explained by the high consumption of antibiotics in the country. In a community-based survey, over 70% of children had received antibiotics in the preceding 6 months, more than half of which had not been prescribed by a healthcare professional. 30 In 2018, the WHO released its first global report on the consumption and use of antibiotics, and Mongolia was the country with the highest consumption among the six Western Pacific Region countries that submitted the data. 31
Imaging techniques performed to diagnose OM in other studies were mainly MRI and bone scan, with a high yield for both diagnostic tools (around 94%). 32 Although arthrocentesis has a therapeutic aim in SA, the need for a bone aspiration for a suspected uncomplicated OM is more controversial because this procedure does not seem to affect the outcome. In our study cohort, more than half of patients received orthopedic operations: in children suffering from OM, operations were performed in almost two-thirds of cases and in SA in 40.5%. Studies on pediatric OI reported that SA and OM mortality had decreased significantly due to improved diagnosis and treatment in the last two decades. However, despite this long-term consequences are still present. 32 The rate of sequelae in developed countries is low: for example, in Spain, with early diagnosis, it ranges between 5% and 10%. 33 A cross sectional study in Switzerland revealed objective sequelae of OI in 12%. 2 In countries with limited resources and delayed treatment, sequelae may be much more frequent. In some studies, the incidence of permanent sequelae was reported in up to 50% of cases.34 -36 In our study cohort at the time of last follow-up at 90 days after admission, the rate was however low.
This study had some limitations, including the limited number of participants, the absence of a control group of patients who received conventional services of OI management.
Conclusions
A standardized protocol of diagnosis and treatment was taken up well by the patients and treating staff and led to improved pathogen detection. In the Mongolian setting children with OI are younger compared to other cohorts and this may be linked to higher prevalence of neonatal sepsis and management thereof. Having faster and more sensitive methods of pathogen detection (for eg, PCR detection) may facilitate further optimized antimicrobial management and reduce the burden of prolonged broad antimicrobial treatment, leading to increase of resistant rates.
Supplemental Material
sj-docx-1-gph-10.1177_2333794X241298801 – Supplemental material for Microbiological Spectrum of Osteoarticular Infections and Their Management in Mongolian Children
Supplemental material, sj-docx-1-gph-10.1177_2333794X241298801 for Microbiological Spectrum of Osteoarticular Infections and Their Management in Mongolian Children by Munkhtulga Ulziibat, Michael Buettcher, Uuganbayar Altankhuyag, Battulga Chuluunbaatar, Zorigtbaatar Mikhlay, Chimgee Sharav, Sarantsetseg Bira, Stefan Essig, Nicole Ritz and Bayalag Munkhuu in Global Pediatric Health
Supplemental Material
sj-docx-2-gph-10.1177_2333794X241298801 – Supplemental material for Microbiological Spectrum of Osteoarticular Infections and Their Management in Mongolian Children
Supplemental material, sj-docx-2-gph-10.1177_2333794X241298801 for Microbiological Spectrum of Osteoarticular Infections and Their Management in Mongolian Children by Munkhtulga Ulziibat, Michael Buettcher, Uuganbayar Altankhuyag, Battulga Chuluunbaatar, Zorigtbaatar Mikhlay, Chimgee Sharav, Sarantsetseg Bira, Stefan Essig, Nicole Ritz and Bayalag Munkhuu in Global Pediatric Health
Supplemental Material
sj-docx-3-gph-10.1177_2333794X241298801 – Supplemental material for Microbiological Spectrum of Osteoarticular Infections and Their Management in Mongolian Children
Supplemental material, sj-docx-3-gph-10.1177_2333794X241298801 for Microbiological Spectrum of Osteoarticular Infections and Their Management in Mongolian Children by Munkhtulga Ulziibat, Michael Buettcher, Uuganbayar Altankhuyag, Battulga Chuluunbaatar, Zorigtbaatar Mikhlay, Chimgee Sharav, Sarantsetseg Bira, Stefan Essig, Nicole Ritz and Bayalag Munkhuu in Global Pediatric Health
Supplemental Material
sj-docx-4-gph-10.1177_2333794X241298801 – Supplemental material for Microbiological Spectrum of Osteoarticular Infections and Their Management in Mongolian Children
Supplemental material, sj-docx-4-gph-10.1177_2333794X241298801 for Microbiological Spectrum of Osteoarticular Infections and Their Management in Mongolian Children by Munkhtulga Ulziibat, Michael Buettcher, Uuganbayar Altankhuyag, Battulga Chuluunbaatar, Zorigtbaatar Mikhlay, Chimgee Sharav, Sarantsetseg Bira, Stefan Essig, Nicole Ritz and Bayalag Munkhuu in Global Pediatric Health
Footnotes
Acknowledgements
We thank the members of the study group for their contributions to the study and all the participating families. We would like to acknowledge the Swiss Mongolian Pediatric Project (SMOPP) team, in particular Thomas Baumann and Raoul Schmidt who enabled the research collaboration, fostered the idea, enlarged the SMOPP team with pediatric infectious diseases specialists.
Contributors Statement
Michael Buettcher, Nicole Ritz and Bayalag Munkhuu designed the study. Michael Buettcher wrote the study protocol. The final study protocol was reviewed by Nicole Ritz, Bayalag Munkhuu and Stefan Essig. Munkhtulga Ulziibat, Uuganbayar Altankhuyag, Battulga Chuluunbaatar, Zorigtbaatar Mikhlay, Chimgee Sharav, Sarantsetseg Bira conducted and supervised the study. Munkhtulga Ulziibat and Stefan Essig conducted statistical analyses. Michael Buettcher, Bayalag Munkhuu, Nicole Ritz, Stefan Essig commented on analysis and drafting of the manuscript, which was prepared by Munkhtulga Ulziibat. All authors critically reviewed the various versions of the manuscripts. All authors approved content and form of the final manuscript and supplementary data files.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Swiss - Mongolian Pediatric Project (SMOPP) supported the study with a small funding grant.
Ethical Approval
The study protocol was reviewed and approved by the Institutional Review Board at the National Center for Maternal and Child Health (04/2020) and the Ethical Review Committee of Ministry of Health, Mongolia (152/2020). Written informed consent was obtained from the parents of each child prior to participation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
