Abstract
Hemophilia is a congenital coagulopathy characterized by a deficiency of coagulation factors and the development of haematomas and haemarthrosis, either spontaneously or after minor trauma. Recurrent joint hemorrhage in hemophilia patients leads to progressive and degenerative arthropathy, which affects around 90% of patients with severe disease and contributes significantly to disease morbidity. Positive diagnosis is based on biology. Imaging, particularly MRI, plays an essential role in assessing the evolution and complications, especially osteoarticular complications. We report 2 cases of severe hemophilia A, who presented with almost identical clinical and radiological symptoms. The patients developed severe arthropathy with a course marked by recurrences of haemarthrosis.
Keywords
Introduction
Hemophilia is an X-linked recessive inherited disorder affecting almost exclusively males. 1 It is a rare but serious pathology characterized by a deficiency in coagulation factor. There are 2 forms : hemophilia “A,” characterized by a factor VIII deficiency with a frequency of 1 in 10,000, and hemophilia “B,” characterized by a factor IX deficiency, which is 5 times less frequent. 2 It manifests itself through bleeding, the severity of which correlates with that of the coagulation factor deficiency, and the haemorrhagic manifestations of hemophilia predominate in the joints, with haemarthrosis in particular. Repeated haemorrhagic episodes can lead to degenerative arthropathy, which is the most frequent complication in patients with severe and moderate hemophilia, and a single episode of haemarthrosis is enough to alter joint tissue. 3 People with hemophilia A and B have similar imaging findings, and can only be distinguished by the results of the laboratory tests that provide the positive diagnosis. 4 Imaging is useful for detecting and monitoring complications, particularly osteoarticular. 5
Case Report
Case 1
A 13-year-old boy with hemophilia A discovered at the age of 11 months. He was admitted for haemarthrosis following a fall from a height with a point of impact in the mouth; On somatic examination, he presented with a swollen, painful, and warm knee with extension and flexion deficits, all evolving in an apyretic context; the patient had a history of frequent episodes of haemarthrosis leading to haemophilic arthropathy; biologically, coagulation factor VIII level was 0.9%; anti-FVIII inhibition rate = 97% and anti-FVIII assay = 32 BU/ml, fibrinogen level was 4 g/l; aPTT was 3.8.
An MRI was ordered for evaluation and revealed irregularity of the femorotibial plateaus with subchondral geodes with fibrinous signal (T1 and T2 hyposignal) associated with osteophytic beaks of the femoral and tibial condyles and patellar base, and erosion of the articular cartilage in the medial and lateral compartments; an abundant liquid effusion in the quadricipital bursa, the intercondylar fossa and the popliteal fossa with T1 hyposignal, T2 hypersignal, containing a liquid-liquid level and a few spots with T1 hypersignal and T2* GRE asignal, associated with active bleeding (Figure 1); we also noted thickening of the femoral synovial membrane, with intermediate T2 signal and hemosiderin deposits in T1 and T2 hyposignal and asignal on GRE sequences (Figure 2) and pinching of the femorotibial joint space in the medial compartment (Figure 3).

Severe hemophilic arthropathy of the knee: T1 sagittal (a) and T2 gradient echo (b) images. Heterogeneous signal hemarthrosis, with liquid-liquid level (star) and hemosiderin deposits in T2 hyposignal (red arrow) with a few spots in T1 hypersignal and GRE asignal associated with active bleeding (white arrow). Severe erosion and irregularity of femoral and tibial articular cartilage and osteophytic beaks and subchondral geodes (blue arrowhead).

Sagittal DP TSE FAT SAT image: abundant sub-quadricipital hemarthrosis (star) and enlarged synovial recesses (white arrow).
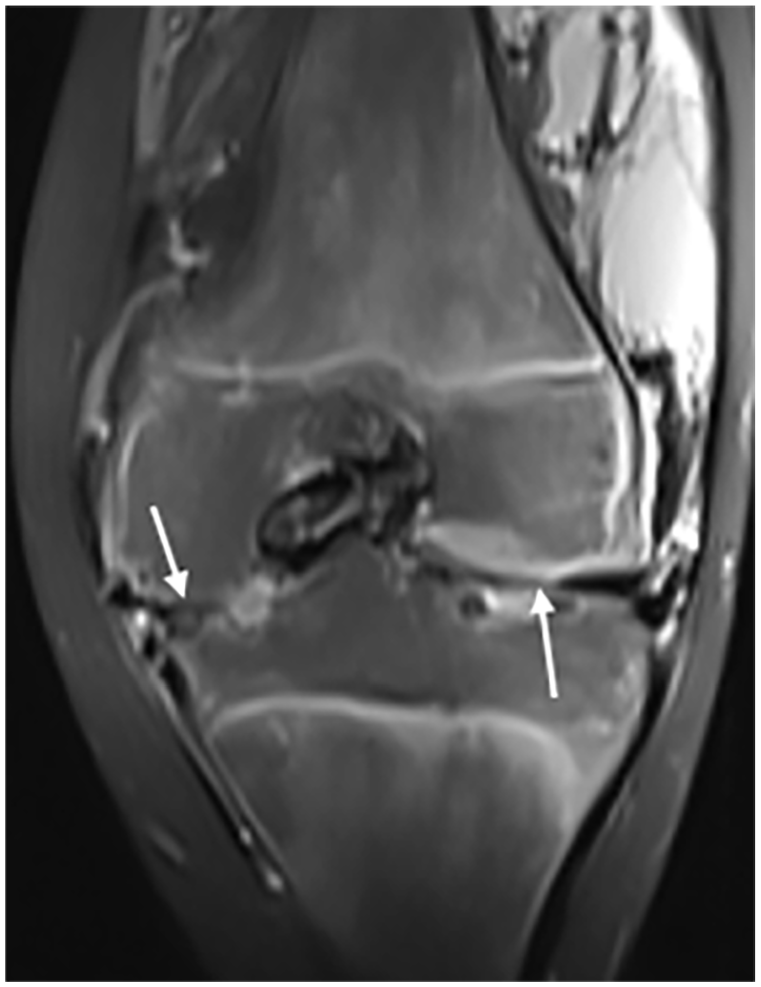
Coronal T1 FAT SAT image after gadolinium injection : pinching of the femorotibial joint spaces (arrows) and widening of the intercondylar fossa.
The patient was treated with factor VIII with a splint and monitored for 6 months, and the bleeding stopped. A knee prosthesis was proposed and will be fitted at the age of 15.
Case 2
Adolescent aged 15, followed for severe hemophilia A inhibitor +; he was seen for a haemorrhagic syndrome, with an enlarged, painful knee, and chronic synovitis with no notion of fever. The coagulation factor VIII level was 0.2%; the anti-FVIII inhibition level was 99.56% and the anti-FVIII level was 23.68 BU/ml; the fibrinogen level was 3.2 g/l; the aPTT was 3.8 seconds.
Radiological findings included pinching of the tibiofemoral joint space and irregularity of the tibiofemoral and patellofemoral plateaus with cartilage erosions; an exposure of the bone in places, as well as subchondral geodes and osteophytic beaks (Figure 4); abundant fluid effusion in the quadricipital bursa, intercondylar fossa, and lateral recesses, with intermediate T1 and T2 hypersignals suggesting bleeding and thickening of the femoral synovial membrane, with intermediate T2 signal and T1 and T2 hyposignal and asignal signal abnormalities on GRE sequences associated with hemosiderin deposits (Figure 5).

Coupe coronal (a) et sagittal (b) T1 FSE section: intermediate signal hemarthrosis (star) and pinching of the medial tibiofemoral joint space associated with irregularity of the tibiofemoral and patellofemoral plateaus, with cartilage erosion and local exposure of bone and osteophytic spurs (red arrow) and subchondral geodes (white arrow).

Advanced hemarthrosis: T2 gradient-echo weighted sagittal (a) and DP TSE sag FS (b) image. Joint fluid effusion (orange star) with T2 hyposignal and asignal signal abnormalities on GRE sequences related to hemosiderin deposits (red arrow).
Discussion
Hemophilia is a rare disease caused by a genetic coagulation disorder, resulting from the absence or dysfunction of a coagulation factor. 6 Depending on the coagulation factor deficiency, hemophilia can be classified as hemophilia A (factor VIII) or hemophilia B (factor IX). The 2 types of hemophilia can be classified according to the plasma level of the coagulation factor present; it is said to be severe when the percentage of coagulant activity is less than 1% of normal, moderate between 1% and 5% and minor between 6% and 40%.7,8 The severity of bleeding in hemophilia patients is directly related to the amount of residual coagulation factor activity in the blood. In patients with severe hemophilia, bleeding may occur spontaneously or following minor trauma, with 70-90% of bleeding episodes occurring mainly in muscles and joints, particularly synovial joints. The severity of hemophilia is not proportional to osteoarticular involvement, and even in the absence of clinical haemarthrosis, many of these patients develop arthropathy. 5 Patients with moderate or mild hemophilia experience bleeding episodes even after minor surgical procedures, which require immediate treatment to prevent complications. 9 Several studies have shown that the severity of hemophilia has no impact on the presence of haematomas or external hemorrhages, but does have a significant influence on the frequency of haemarthrosis.5,7 The aspects of osteoarticular damage in hemophiliacs are strongly related to the number of episodes of haemarthrosis, since it is repeated haemarthrosis that causes osteoarticular lesions. 10 The same types of lesions may therefore be present in all 3 severity categories, and a minor hemophilia may therefore be responsible for hemarthrosis. 11 The loss of mobility resulting from cartilage and bone damage has an impact on these patients’ quality of life. Osteoarticular disorders are the most common, and the knee joint is the most affected. 12
The development of haemarthrosis is the most important clinical manifestation; the clinical symptoms of acute haemarthrosis manifest as rapid swelling, with patients usually experiencing the first signs of stiffness, tingling, and pain. 6
In the same joint, repeated bleeding leads to synovial hypertrophy, haemosiderin deposits, cartilage destruction, and changes in the structure of the subchondral bone. These haemosiderin deposits may also further promote synovial hypertrophy and swelling. The onset of haemarthrosis induces chondrocyte apoptosis, which disrupts the renewal of the cartilage matrix. 13 Synovial tissue undergoes hypervascularization, initially synoviocyte hypertrophy, which increases the risk of new haemorrhagic events and inflammatory changes. These changes cause damage to cartilage by producing enzymes and cytokines that destroy tissue, 14 leading to joint destruction. In a recent study, Zheng et al demonstrated the role played by iron in cartilage damage by regulating the expression of FGF23 and SOX9 in chondrocytes from hemophiliac patients with haemarthrosis. 15 Patients with hemophilia A showed elevated levels of COL-18N, which could serve as a potential marker for monitoring the progression of arthropathy and thus adapting optimal treatment to prevent further joint damage. 16
For several years, the World Federation of Haemophilia has recommended the assessment of joint damage using radiological scoring systems and orthopedic exploration. 17 Diagnostic imaging methods can be used to divide the degenerative joint process into different clinical and pathological stages. The aim is to identify joint changes, measure their severity and evaluate the follow-up of therapeutic effects.
The most frequently used are ultrasound, plain radiography, and magnetic resonance imaging (MRI).
Ultrasound is an inexpensive, readily available, and excellent technique for determining the presence of soft tissue inflammation. At the initial stage, there is an effusion of variable echostructure, hypervascularization of the synovium. Its use makes it easy to distinguish between an effusion and synovial hypertrophy, and to monitor progress after an acute episode, making it easier to adjust the duration of treatment. In advanced forms, thinned or irregular cartilage, subchondral erosions, and fibrous synovial hypertrophy may be observed. 13
Despite its limitations, radiography remains one of the most commonly used methods for assessing haemophilic arthropathies, MRI is the reference tool for studying joint changes in hemophilia patients.
MRI has proved to be the most useful tool for analyzing soft tissue and osteochondral changes present in the early stages of haemophilic arthropathy. 4 It reveals joint damage at an early stage and can therefore be used to advantage in the first episodes of haemarthrosis, when X-rays are still normal.18,19 The signal of hemarthroses varies with time, but in the acute stage it is usually heterogeneous, with areas that are strongly hyperintense in T1 and hypointense in T2. In the subacute stage, the presence of methemoglobin may be responsible for a T1 and T2 hypersignal. Declivity levels better visible on T2 are sometimes objectified. Hemarthrosis may also present as an aspecific fluid effusion when MRI is performed remote from an acute or subacute episode, presenting as hypointense in T1 and hyperintense in T2, as was the case with our patients. In T1 and T2 weighted sequences, the synovium is hypo or isointense in muscles due to hemosiderin deposits and fibrosis, especially when gradient echo sequences are used (ferromagnetic susceptibility). Subchondral lesions may contain fluid (hyposignal T1 and hypersignal T2), fibrin (hyposignal T1 and T2) or haemorrhagic material (hypersignal T1 and T2), but usually combined in varying proportions. Gadolinium injection can differentiate synovitis from intra-articular effusion.3,13
Associated lesions include soft tissue ossifications, particularly periarticular, and fractures. They are caused by bone insufficiency, chondrocalcinosis, superinfections, particularly of the joints. 13
Differential diagnosis includes juvenile idiopathic arthritis, which may give radiographic signs similar to those of hemophilic arthropathy. MRI shows synovitis but no hemosiderin deposits.
Pigmented villonodular synovitis, whose radiographic and MRI findings are very similar to those of hemophilia, but only 1 joint is affected. 4
Intravenous administration of the missing coagulation factor is the gold standard for preventing hemorrhage and haemophilic arthropathy. 6
Recurrent haemarthrosis leads to the progression of a degenerative and irreversible process.
Limitations of Our Study
Several radiological scores have been established as part of monitoring and evaluation in haemophilic arthropathy and MRI remains the most accurate diagnostic standard for assessing soft tissue changes at an early stage. We were unable to evaluate the radiological score in our study.
Conclusion
Hemophilia is a rare disease. It can give rise to serious osteoarticular complications secondary to repeated hemarthrosis linked to coagulation factor deficiency. These lesions are manifested by synovial, cartilaginous, and bony changes. Numerous studies have shown the predominance of ultrasonography, but MRI is the standard reference modality for assessing arthropathy and diagnosing the musculoskeletal complications of hemophilia. The knee is the joint most frequently affected by hemophilic arthropathy, and represents a major morbidity factor in these patients. Repeated joint bleeding causes damage to bone, cartilage and synovium, inducing inflammatory and degenerative processes.
Footnotes
Acknowledgements
The authors thank his professors and colleagues who helped in the completion of this work.
Author Contributions
All authors contributed to the implementation and realization of this work. All authors declare that they have read and approved the final version of this manuscript
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from a legally authorized representative for anonymized patient information to be published in this article.
