Abstract
Total elbow arthroplasty is effective for pain relief and the functional improvement of severe symptomatic hemophilic osteoproliferative arthropathy. Nevertheless, high complication rates have been reported. This report describes clinical results obtained at 30-month follow-up of total elbow arthroplasty in a hemophilic patient with severe flexion contracture. A 50-year-old patient with advanced left elbow hemophilic arthropathy underwent unlinked total elbow arthroplasty. He had sustained an intraoperative fracture of the medial part of a supracondylar humerus complication. To control perioperative bleeding, strict factor VIII replacement therapy was performed under the guidance of hematologists. The total elbow range of motion was 35° preoperatively, but it had improved to 110° postoperatively. The Patient-Rated Elbow Evaluation Japanese version score, which was 53.7 preoperatively, improved to 10.7 postoperatively. During the 30 months after operation, no complication occurred. Good clinical results have been obtained under close collaboration with hematologists and close patient adherence following treatment.
Introduction
With hemophilia, a recessive X-linked inherited coagulopathy caused by a deficit of clotting factors VIII and IX, 1 spontaneous and repeated intra-articular bleeding can engender progressive joint destruction in the ankle, knee, elbow, and hip joints. Articular problems of hemophiliac patients begin in infancy. 2 Following the knee, the elbow is commonly reported as the second most common target joint. 3 Total elbow arthroplasty (TEA) is selected for patients with marked elbow pain and severe limitation of the range of elbow movement.4–6 A recent review article described postoperative outcomes of 51 TEAs in 38 patients with hemophilic arthropathy across 7 publications. 7 For hemophilic elbow arthropathy, TEA provides good improvement of elbow pain and elbow function postoperatively. 7 However, repeated bleeding and inflammation in the elbow joints can lead to the early development of skeletal deformities and abnormal and difficult anatomy, which might complicate surgery.4,6 Moreover, the rate of complication from infection and the revision rate are higher than those for TEA performed for other indications such as inflammatory arthropathy, osteoarthritis, and traumatic elbow injuries. 7 This report describes a case of a 50-year-old hemophilia patient diagnosed as having hypertrophic elbow osteoarthropathy with severe flexion contracture, who underwent unconstrained TEA, complicated by intraoperative medial humeral supracondylar fracture.
Case report
A 50-year-old man, a desk worker, was referred to our hospital with left elbow pain, his nondominant side, and limited active range of left elbow motion. The patient had been diagnosed as having hemophilia shortly after birth. Since the age of 13, he had been treated intermittently with factor VIII. He had received bilateral ankle synovectomy and left total hip arthroplasty and bilateral total knee arthroplasty for multiple hemophilic arthritis. His left elbow joint, with repeated intra-articular bleeding from an early age, was one target joint. Physical examination revealed considerable left elbow pain when he moved actively. Passive elbow extension was as great as −60°; flexion was 95° with no instability (Figure 1(a) and (b)). The range of forearm motion was 30° for pronation and 30° for supination. The grip and key pinch strengths of the affected hand were, 27.5 kg and 6.0 kg, respectively, whereas those of the unaffected hand were, 45.0 kg and 10.0 kg, respectively. The preoperative Patient-Rated Elbow Evaluation Japanese version (PREE-J) 8 scores were 53.7. Radiographs of the left elbow showed narrowing joint space and bone hyperplasia (Figure 2(a) and (b)). Computed tomography (CT) showed some loose bodies in the olecranon fossa, subchondral bone irregularities of the trochlea of humerus (Figure 2(c)), and sclerosis with occlusion of the humerus and medullary cavity and a few hypertrophic loose bodies around the radial head (Figure 2(d) and (e)). Clinical and image findings led to our diagnosis of hemophilic arthropathy of the left elbow. After consultation with a hematologist, the decision was made to proceed with total elbow joint replacement surgery. Factor VIII replacement therapy was done to maintain the target concentration of coagulation factors during the perioperative period under the guidance of hematologists. Before the day of surgery, 5000 U/24 h in continuous infusion (58 kg patient body weight) factor VIII (Advate®; Takeda Pharmaceutical Co. Ltd., Tokyo, Japan) and 3000 U in bolus infusion were administered intravenously. We performed left TEA with an unconstrained prosthesis (Kudo elbow type 6; Zimmer-Biomet Inc., Warsaw, IN, USA) under general anesthesia. A triceps fascial tongue exposure approach 9 was used. Cartilage defect and eburnation in the joint surface of the head of the radius were found and subsequently excised (Figure 3(a)). The joint loose body (3 cm long axis, 1.5 cm short axis) in the front of the head of the radius was removed (Figure 3(b)). The medial and lateral collateral ligaments, which were intact, were dissected to allow dislocation of the elbow joint. The articular surface of the capitellum of the humerus and the ulnar trochlear notch showed a cartilage defect (Figure 3(c)). We selected the medium size among the prostheses to stabilize the humerus component. The space in the marrow was too narrow to insert the component. The humeral component was cementless; the all-polyethylene ulnar component was cemented. Postoperative X-ray showed a fracture line on the medial part of the supracondylar humerus (Figure 4(a)). His upper left limb was swollen and blistered because of cutaneous circulatory disturbance (Figure 4(b)). Therefore the elbow was immobilized to have 60°–90° flexion in a posterior splint for 14 days. The patient started active assisted elbow exercises under the supervision of a physiotherapist 2 weeks after the operation. The patient continued to wear a night splint set at the same position as postoperatively for an additional 3 weeks. He was instructed to wear a sling during the day. Mild grip strength training was started 4 weeks postoperatively. As postoperative factor VIII replacement therapy, 5000 U/24 h in continuous infusion was administered on postoperative days 1–4 (POD 1–4). Then, on POD 5–12, a dose of 2000 U twice daily in bolus infusion was administered, followed by 2000 U once daily in bolus infusion on POD 13–21. Factor VIII inhibitor remained negative in perioperative tests. Radiographic images taken 3 months after surgery showed the bone union of a fracture line on the medial part of the supracondylar humerus. We strictly instructed this patient not to overload his left elbow joint by performing activities in daily life such as carrying heavy objects or supporting weight with the upper limb. At the 30-month follow-up visit, the patient was found to have no neurovascular deficit or implant difficulty visible by X-ray (Figure 5(a) and (b)). He was able to achieve extension to −30° (Figure 5(c)), flexion to 140° (Figure 5(d)), supination to 90° (Figure 5(e)), and pronation to 80° (Figure 5(f)). Moreover, the grip and key pinch strengths of the affected side were restored respectively to 46 kg and 12 kg, respectively. The PREE-J score was 10.7 compared to 53.7 before the operation. No elbow joint pain was experienced during daily activities; elbow joint function improved.

The patient’s elbow extended −60° (a) and flexed 95° (b) before the operation.

Radiographs of the left elbow showed narrowing joint space and bone hyperplasia before the operation: (a) anteroposterior view; (b) lateral view. (c) Computed tomography (CT) coronal view showed some loose bodies in the olecranon fossa (arrowhead) and subchondral bone irregularities of the trochlea of humerus (arrow). (d) CT axial view showed sclerosis with occlusion of the ulnar medullary cavity (arrowhead) and a few hypertrophic loose bodies around radial head (arrow) at the proximal radioulnar joint. (e) CT coronal view showed sclerosis with occlusion of the humerus medullary cavity at the proximal supracondylar humerus (arrowhead).

(a) Cartilage defect and eburnation were found in the joint surface of the head of the radius. (b) Extracted joint free body. (c) Cartilage defects were found in the articular surface of the capitellum of the humerus (arrow) and ulnar trochlear notch (arrowhead).
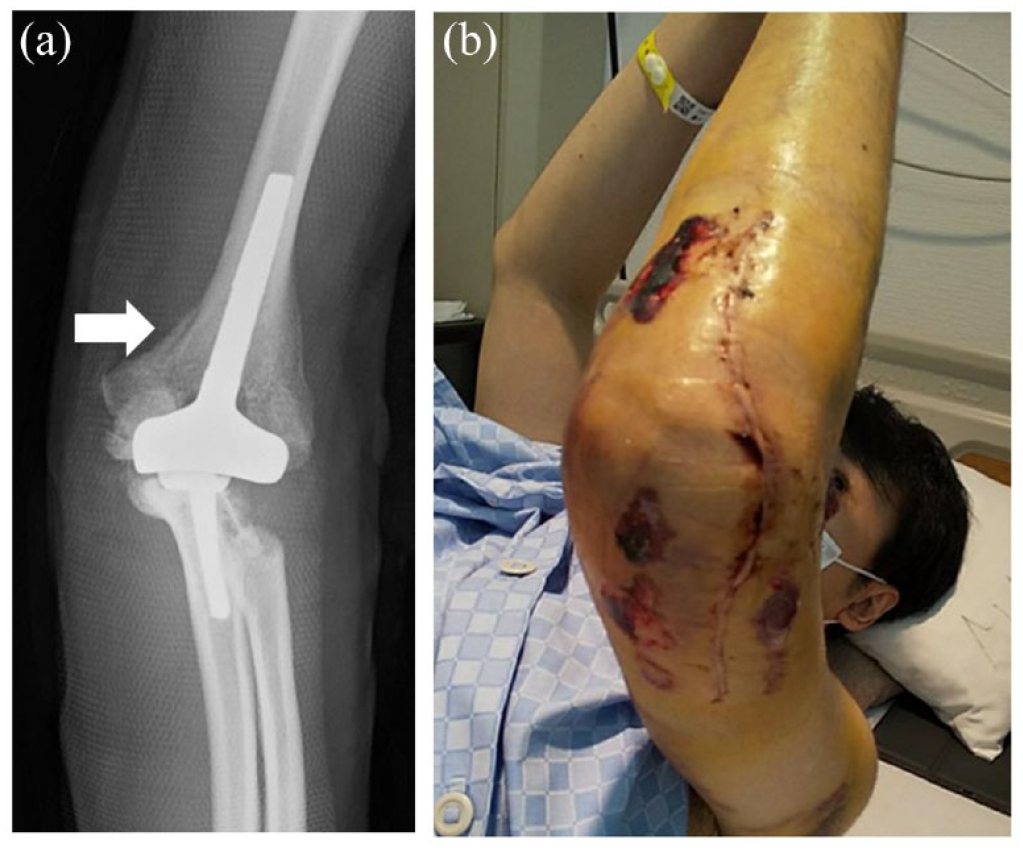
(a) Postoperative X-ray showed a fracture line on the medial part of the supracondylar humerus (arrow). (b) Swollen upper left extremity with blisters caused by skin circulation difficulties.

Radiographs at 30-month follow-up: (a) anteroposterior view; (b) lateral view. The patient’s elbow flexed 140° (c) and extended −30° (d) and pronated 80° (e) and supinated 90° (f) at the 30-month follow-up.
Discussion
Our case was characterized by manifestations of hemophilic hypertrophic osteoarthropathy of the elbow with severe flexion contracture. For this case, we selected an unlinked prosthesis that provided stability, acting as a good spacer for restoring tension to the soft tissues, based on the preoperative image findings indicating it as osteoproliferative, rather than osteolytic, which we judged as free of bilateral collateral ligamentous structural destruction. In fact, intraoperative findings showed that their ligaments were intact. Another reason that we used an unconstrained prosthesis is that it entailed less sacrifice of bone from the lower humerus than that associated with unlinked types. 10 If, in the future, a revision surgery is unfortunately required because of aseptic loosening, then the possibility of choosing a semi-constrained type as a salvage operation must be considered.
In patients with hemophilia, bleeding and inflammation in the joints from an early age engender the development of skeletal deformities and anatomical variation, 2 which might then engender difficulties with surgery, especially in recognizing an anatomical landmark.11,12 Our patient was found to have sclerosis with occlusion of the humerus and ulnar medullary cavity and some hypertrophic loose bodies during operation. The narrow medullary cavity was reamed to insert medium-sized cementless humerus components suitable for the patient’s physique. The reaming caused thinning of the bone cortex of the medial and lateral epicondyles of the humerus. This thinning in turn caused a medial supracondylar fracture of the humerus when the component was inserted into the humerus. For such patients, it might be difficult to prevent fracture complications related to the placement of appropriately sized prostheses. An alternative might be to require an extra-small humeral and ulnar component and appropriately small reamers. 4 According to the latest systematic review, postoperative complication rate, including periprosthetic joint infection, TEA with aseptic loosening, ulnar nerve neuropathy, and significant postoperative hematoma was 49%. 7 Revision rates were found to be 29% in patients with hemophilia. 7 Furthermore, the revision rate for TEA in hemophilic elbow arthropathy is somewhat higher, when compared to other joint arthroplasties such as hip and knee replacement. 7 Joint instability after arthroplasty might lead to wear or breakage of the polyethylene bushing and subsequent failure. Great care was taken to select an appropriate prosthesis for the pathology of the patient’s elbow joint.
The replacement therapy of coagulation factors during the perioperative period is crucially important to perform surgery safely and to prevent complications such as excessive bleeding, deep infection, prosthesis loosening, and ulnar nerve neuropathy caused by adhesion with surrounding tissues. Regarding factor VIII replacement therapy, bolus injection and continuous infusion are two common methods. 1 To control perioperative bleeding, we performed the combination therapy of bolus injection and continuous infusion under the guidance of hematologists. Close collaboration with hematologists is important to ensure safe surgery.
According to an earlier study, the implementation of patient education for orthopedic surgery patients has positive effects of clarifying the physical condition and requirements for self-care. 12 Compared to survival rates of hip and knee arthroplasties, those of TEA are low.13,14 Daily work on the elbow depending on the loads being lifted can cumulatively become as destructive as high loads and stresses. 15 Under our medical instruction, the patient might have been very cautious in daily activities to avoid the imposition of a heavy loading on his elbow.
Conclusion
This case study presented good results obtained for this hemophilic patient with hypertrophic elbow osteoarthropathy undergoing unlinked TEA and strict factor VIII replacement therapy during the perioperative period. Our follow-up time was short. Follow-up must be continued for further evaluation of the efficacy of these methods.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information and accompanying images to be published in this article.
