Abstract
Charcot arthropathy is a type of destructive osteoarthropathy characterized by neurotrophic and sensory disorders. The condition is relatively rare, with an insidious onset, and it is easily misdiagnosed. Total knee arthroplasty (TKA) can cause excessive joint wear, continuous inflammatory stimulation of the prosthesis, postoperative residual cavity, prosthesis loosening and subsidence, peripheral fracture, infection, and other complications. Furthermore, these complications are more likely to occur in patients with Charcot arthropathy because of disease-specific pathological characteristics, when TKA is performed. Therefore, Charcot arthropathy was once a contraindication to TKA. Recently, with the optimization of joint prostheses and the maturity of surgical techniques, more studies have reported successful cases of TKA in patients with Charcot arthropathy. We report a case of Charcot arthropathy in our hospital, and describe the patient’s medical history, clinical symptoms, signs, imaging findings, diagnosis, and the entire TKA process, to explore the TKA strategy and prosthesis selection in a patient with Charcot arthropathy.
Introduction
Charcot arthropathy, also known as neurogenic arthropathy, is a destructive osteoarthrosis characterized by neurotrophic and sensory disorders.1,2 Total knee arthroplasty (TKA) causes excessive wear, inflammatory stimulation, and a postoperative residual cavity. The common complications after TKA are infection, hematocele, periprosthetic fracture, and prosthesis loosening and subsidence. Charcot arthropathy is associated with problems such as poor articular bone, impaired blood supply, and the inability to effectively avoid trauma, which not only reduces the effect of TKA, but also greatly increases the incidence of its complications. As a result, Charcot arthropathy was once considered an absolute contraindication for TKA.3–6 Recently, with the continuous optimization of joint prostheses and gradual improvement of TKA technology, more studies have reported the successful application of TKA in the treatment of Charcot joint disease (Table 1). Nevertheless, the treatment remains challenging and controversial. Therefore, the authors retrospectively analyzed one case of TKA for Charcot knee joint, and compared the case with similar reports. The follow-up time in our case was longer, and relevant examination and data were more comprehensive. The authors hope to provide a reference for selecting TKA in the clinical treatment of Charcot arthropathy.
Reports of rotating-hinged knee prosthesis replacement for Charcot arthropathy in the last 5 years.

Case presentation
The patient’s private information and medical records were obtained with written informed consent and with the approval of the institutional ethics committee of The Affiliated Jiangnan Hospital of Zhejiang Chinese Medical University (Approval No. XSZYY202010016). The reporting of this study conforms to the CARE guidelines. 7
A 54-year-old man was admitted to our Orthopedics Department with a complaint of left knee swelling and deformity for more than 20 days. There was no obvious trauma history before the onset of the symptoms. The left knee was swollen, deformed, and mildly painful, and the patient was able to bend and stretch but was unable to bear weight when walking. Fifteen days earlier, he was admitted to a local hospital, and X-rays showed fracture and displacement in the left tibial plateau. He was transferred to our hospital for further treatment.
The patient was previously healthy, but routine examination at the local hospital found positive serum Treponema pallidus antibody (Tp-Ab). Otherwise, there was no major internal or surgical disease.
Physical examination findings revealed stable vital signs, no spinal deformity or limited movement, no somatic sensation, no obvious abnormalities in muscle strength and sensation in both upper limbs, and bilateral Hoffmann sign (−). The following were also identified: swelling of the left knee joint, floating patellar test (+), knee varus deformity, mild tenderness, positive percussion pain, and range of motion (ROM): 120° to 20°. Muscle strength was grade 4 in the left lower extremity and grade 5 in the right lower extremity, the American Knee Society clinical score (KSS) was 45 points, and the KSS functional score was 30 points. Left lower extremity pigmentation was present, with plantar lateral shallow hypoesthesia, absence of the left big toe, bilateral Babinski sign (–), and good blood circulation and movement of the extremities (Figure 1a, b).

Preoperative lower extremity images showing: (a) left knee swelling; (b) left lower extremity varus deformity.
Laboratory examination revealed no obvious abnormalities in liver and kidney function, electrolytes, blood glucose, glycosylated hemoglobin, and tumor indicators; high blood platelet count; slightly high hypersensitive C-reactive protein concentration; and high erythrocyte sedimentation rate.
Preoperative imaging examination: X-rays, and computed tomography (CT) coronal, sagittal, and three-dimensional reconstruction images of the left knee showed a comminuted fracture on the medial side of the left tibia, articular surface fragmentation and collapse, displacement with bone resorption, and peripheral soft tissue calcification (Figure 2a–e). Magnetic resonance imaging (MRI) of the left knee showed that the medial and lateral patellar retinaculae, anterior and posterior cruciate ligaments, and medial collateral ligaments were torn. In addition, there were medial meniscus injuries and bone contusions (Figure 3a–d). MRI of the cervical and lumbar vertebrae showed no syringomyelia (Figure 4a, b). Left lower extremity CT venography (CTV) showed obvious stenosis of the lumen of the left popliteal vein at the level of the fractured end of the tibial plateau but no obvious luminal filling defect, with open circulation in the distal collateral veins. The local femoral vein had a narrow filling defect and slight luminal stenosis (Figure 4c).

Preoperative imaging: (a) Anteroposterior X-ray of the left knee. (b) Lateral X-ray of the left knee. (c) Coronal computed tomography (CT) of the left knee. (d) Sagittal CT of the left knee (e) Three-dimensional CT reconstruction of the left knee showing a comminuted fracture of the left medial tibia, fragmentation and collapse of the articular surface, displacement with bone resorption, and surrounding soft tissue calcification.

Preoperative magnetic resonance imaging (MRI): (a and b) Sagittal MRI of the left knee joint. (c and d) Coronal MRI of the left knee joint showing injuries and tears in the internal and external patellar retinaculae, anterior and posterior cruciate ligaments, and medial collateral ligaments; medial meniscus injury; and bone contusions on the patella, lower femur, and upper tibia and fibula.

Preoperative magnetic resonance (MRI) and computed tomography (CT) imaging: (a) Cervical spine MRI. (b) Lumbar MRI showing no syringomyelia. (c) CT venography (CTV) of the left lower extremity showing obvious stenosis of the lumen of the left popliteal vein at the fracture end of the tibial plateau and mild stenosis of the local femoral vein lumen.
Electromyography showed that the left tibial nerve and sural nerve were severely damaged, while the right sural nerve had prolonged latency and was slightly damaged.
Surgical Technique: Routine disinfection, and towel and tourniquet pressure were performed. The articular cavity was exposed via an anterior midline incision over the left knee using a medial and parapatellar approach. Intraoperative findings revealed a large amount of synovial hyperplasia of the knee joint, distal femoral condylar wear, medial tibial collapse, displacement of the fracture block, bone absorption, large amount of hyperplastic tissue and bone fragments, and absent medial collateral ligament and anterior cruciate ligament (Figure 5a). The soft tissue was released and cleaned, and several osteophytes were removed. An opening of approximately 10 mm was made in front of the posterior cruciate ligament on the femur, and the marrow cavity was gradually expanded along this opening. The size of the distal medial device was determined, the femoral condyle was grooved, and the osteotomy module was placed, for testing. The distal femoral osteotomy was guided according to the module, the tibial plateau was dislocated, and the anterior cruciate ligament was positioned and inserted on the tibia. The size of the middle device was determined according to the osteotomy of the lateral platform and the expansion of the proximal tibia. A medial bone defect on the tibial plateau was found at this point. The residual bone block was repaired to a suitable size, and was then reset and fixed with two screws. Then, the joint space was balanced (Figure 5b, c). The upper and lower medullary plugs were placed, and the joint cavity was lavaged, then antibiotic bone cement and the tibia−femoral rotation-hinged knee prosthesis was placed (Figure 5d). Finally, the joint edges were cleaned routinely, a local analgesic cocktail (ropivacaine and compound betamethasone) was placed in the joint cavity, and the incision was closed in layers. A sample of the synovial membrane was collected for pathological examination, and the results showed chronic synovitis with degeneration and necrosis (Figure 5e).
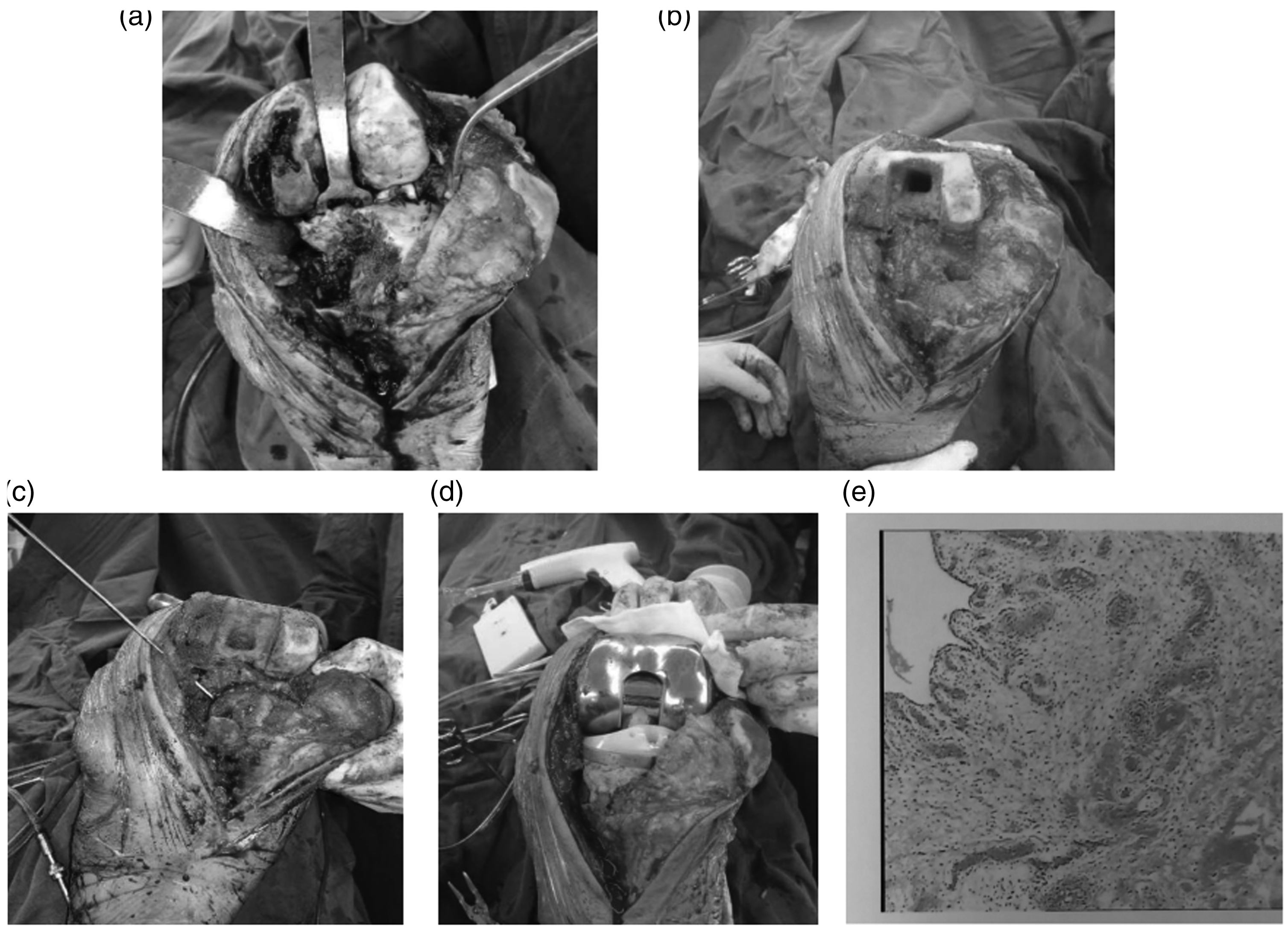
Intraoperative findings: a. Large amount of synovial hyperplasia of the knee joint, distal femoral condylar wear, medial tibial collapse, fracture block displacement, bone absorption, large amount of hyperplastic tissue and numerous bone fragments, and absence of the medial collateral ligament and anterior cruciate ligament. b–d. Intraoperative osteotomy and model placement. e. Intraoperatively, inflamed synovial tissue was excised, and pathological examination revealed chronic synovitis with degeneration and necrosis (hematoxylin and eosin staining; ×100).
Postoperative treatment comprised multimodal analgesia, routine anticoagulation, infection prevention, and anti-inflammatory and anti-swelling treatment. The surgical drain was removed 24 hours after the operation, at which time, left knee X-rays (Figure 6a, b) were reviewed, and guided functional exercise was initiated. A knee brace was worn for 6 weeks to avoid excessive joint motion. Within 2 weeks after the operation, when active straight-leg raises were started, the patient could walk with the assistance of walking aids (Figure 6c).

The patient’s postoperative status: a and b. Left knee joint X-ray showing a normal left knee joint space and that the lower extremity force line was restored. c. Good walking activity.
Follow-up: Outpatient reexamination was performed 1, 3, 6, 9, 12, and 18 months after surgery. Anteroposterior and lateral radiographs of the left knee were taken, and full-length anteroposterior radiographs of both lower extremities were reviewed if necessary to evaluate the limb alignment, and the position and stability of the prosthesis. The KSS score was used to evaluate pain, ROM, and knee joint stability.
At the last follow-up (outpatient reexamination 18 months after surgery), X-rays of the left knee joint showed that the prosthesis was in a good position, and no loosening, displacement, fracture, or periprosthetic fractures were observed. The KSS clinical score was 95 points, which was significantly improved compared with the preoperative score of 45 points. The KSS functional score of 90 was also significantly improved compared with the preoperative score of 30. The ROM of 125° to 5° was also improved compared with the preoperative ROM of 120° to 20°, and the left lower limb muscle strength of grade 5 was also improved compared with preoperative grade 4. The knee deformity was corrected, and the mobility and stability of the knee joint were improved. No complications, such as infection, prosthesis loosening, and periprosthetic fracture were found (Figure 7a–d). The patient was satisfied with the treatment process and results, and felt that the affected knee had been well treated. The diagnosis and treatment procedures are summarized in Table 2.

The patient’s postoperative status 18 months postoperatively: a and b. X-ray of left knee showing a normal joint space, normal lower limb force line, stable internal fixation, good bone union, and a small amount of ectopic ossification. c and d. Good knee range of motion.
Diagnosis and treatment procedures in the current case.

CT, computed tomography; MRI, magnetic resonance imaging; TKA, total knee arthroplasty.
Discussion
Current research status of the pathological mechanism of Charcot arthropathy
Charcot arthropathy is a type of neurogenic arthropathy and is a relatively rare disease. Its etiology is unclear, and a variety of central or peripheral nerve damaging diseases can cause similar damage, namely syringomyelia, spinal cord tumors, myeloschisis, spinal cord injury, congenital insensitivity to pain with anhidrosis (CIPA), diabetes, syphilis, Yaws disease, Guillain–Barré syndrome, chronic alcoholism, advanced renal failure, family amyloidosis, and intraarticular steroid injection.8–11 Joint lesions in Charcot arthropathy are characterized by painless bone destruction in which bone resorption and proliferation coexist. Charcot arthropathy is generally accompanied by joint swelling, osteoporosis, limited activity, and in severe cases, joint deformity. The clinical symptoms are no or mild pain, which is inconsistent with the severe joint damage shown by imaging.
There are many theories regarding the pathological process of Charcot arthropathy, namely: 1. Autonomic nerve dysfunction leads to peripheral vessel diastolic reflex, resulting in pathological hyperemia of joint vessels, which leads to excessive activation of osteoclasts that eventually disrupts the osteoclast balance. 12 2. Damage to the sensory nerves, such as those responsible for proprioception, leads to the decrease or even disappearance of the protective joint reflex, which then makes the joint unable to effectively avoid the effects of external trauma. In addition, the joint is damaged further by excessive joint activity, and the repeated action of minor trauma on the pathological joint with the loss of sensory nerves leads to joint disintegration and fracture. Repeated microtrauma causes joint disintegration and fracture in diseased joints with sensory nerve deficiency. 13 3. Lack of nutrition to the nerves and abnormal bone metabolism changes caused by vascular injury lead to joint nutrition disorders. 4. Owing to the joint destruction, inflammatory factors continue to act on joints with neuropathy, generating an inflammatory response and promoting the proliferation and differentiation of osteoclasts, finally creating a vicious cycle.14,15
There is currently no unified conclusion regarding the pathological process in Charcot arthropathy, but a relatively mature theory divides the condition into three stages. 1. Damage stage: Vascular reflex disorder, joint protective reflex decline, lack of neurotrophic function, inflammatory factor infiltration and other factors lead to joint osteogenesis–osteoclast imbalance. Then, the bone begins to dissolve and break down, causing swelling and joint damage.16,17 2. Repair stage: The body’s compensatory repair mechanism creates dense fibrous tissue and uses the remaining osteogenic function to reassemble the debris after joint destruction. 18 3. Static stage: In the late stage of disease progression, the joints are in a state of devascularization, and bone destruction and repair reach a new balance. The final state has been classified as destructive, proliferative, and mixed types. 19
Diagnosis and differential diagnosis of Charcot arthropathy
The clinical diagnosis of Charcot arthropathy is generally based on the inconsistent features of mild clinical symptoms and severe joint destruction shown by imaging. Patients generally complain of joint swelling, instability, and deformity without an obvious orthopedic trauma history. The primary disease is insidious, according to the imaging results, and is often misdiagnosed as fracture or osteoarthritis; thus, affecting the treatment. When a patient has no joint pain or when the pain is mild, and the degree of pain is significantly lower than the degree of joint destruction on imaging, doctors should be alert, pay attention to the relevant medical history, determine whether there is a primary nerve damaging disease, and perform a neurological examination. Generally, patients have signs of muscle atrophy, muscle strength loss, and pain and warmth sensation loss. If necessary, MRI of the cervical and thoracolumbar spine, electromyography, and other auxiliary examinations can be combined to help diagnose the disease. CT and MRI of the knee joint can help to assess the extent of joint destruction to guide further treatment. Currently, the most common clinical staging method is the modified Eichenholtz staging method. Eichenholtz conducted a continuous cohort study of 68 patients with Charcot joint disease, then proposed three lesion stages. First stage: development. Second stage: healing. Third stage: reconstruction. According to this staging, some scholars add stage 0, the precursor stage, and this staging is called the modified Eichenholtz stage I. 20
Charcot arthropathy often lacks specific symptoms and imaging findings; therefore, other diseases must be excluded during the diagnosis. The differential diagnoses are thrombosis, spontaneous osteolysis, degenerative osteoarthropathy, rheumatoid arthritis, gouty arthritis, suppurative arthritis, traumatic arthritis, hemophilic arthropathy, psoriatic arthritis, pigmented villonodular synovitis, joint tuberculosis, tumor, ankylosing spondylitis, calcium dihydrogen pyrophosphate deposition, and other diseases. These differential diseases are mainly identified by serological examination (blood routine examination, C-reactive protein, procalcitonin, coagulation function, immunological indicators); imaging examination, such as X-ray, CT, and MRI; pathological examination; and electromyography.21–24 The diagnosis of Charcot arthropathy should also include the diagnosis of the primary disease.
In the current case, radiographs taken at a local hospital indicated a fracture of the left tibial plateau. However, the patient had no obvious history of trauma; therefore, simple fracture of the left tibial plateau was not considered initially. Physical examination showed knee varus, and radiographs showed soft tissue calcification and bone destruction, which should be differentiated from knee osteoarthritis. However, the degree of destruction in knee osteoarthritis should induce severe pain, which was significantly inconsistent with our patient’s mild pain symptoms. This mismatch between pain and symptoms suggested that Charcot arthropathy should be considered. There were no differences in MRI findings between the cervical and lumbar vertebrae, while Tp-Ab (+) status was found when the medical history was examined. Combined with the results of electromyography, indicating nerve damage, Charcot arthropathy caused by syphilis was considered.
Research progress in the treatment of Charcot arthropathy
The treatment of Charcot arthropathy should first control the primary disease, such as incision and drainage of syringomyelia, blood glucose control in diabetes, and antibiotic control for syphilis, and patients should be transferred to the corresponding specialist for these procedures. Control of the primary disease can effectively delay the progression of disease and joint destruction, relieve pain and swelling, and improve muscle strength and muscle reflexes.25–27
To control the primary disease, according to the modified Eichenholtz stage, conservative treatment is adopted for patients at stage 0 and stage I. Bracing and weight-bearing restriction are preferred, including rest, orthosis fixation, and plaster fixation, combined with muscle strength recovery training and physical therapy. Patients with pain can take non-steroidal anti-inflammatory drugs, and those with unresolving swelling can undergo arthrocentesis. Calcitonin and bisphosphonates can be added to inhibit bone resorption and slow osteolysis. During this period, patients should avoid knee sprains or falls because trauma can aggravate the primary lesions and accelerate joint destruction.
After 3 months of conservative treatment, if the disease still cannot be controlled, and sustained joint destruction and instability persist, surgical treatment can be considered for joints in stage II and III. The same is true for joints with deformity, infection, and other complications.28,29 However, the ideal procedure remains controversial. The main methods are debridement, internal/external fixation with bone grafting and plating, TKA, arthrodesis, and amputation. Debridement is performed in infected joints. Arthrodesis leads to complete loss of knee function and should be considered carefully. Arthrodesis is suitable for patients with extremely loose ligaments, extremely unstable joints, serious bone destruction, or initial TKA failure. Amputation is more difficult for patients to accept, and is performed only as a salvage operation.
In the current case, the patient had severe bone destruction and obvious joint deformity, and conservative treatment was obviously difficult to ensure his performance of daily activities. Currently, only TKA can restore the damaged shape of the knee and, thus, restore joint function. The patient had no obvious signs of infection, and no debridement was required. Arthrodesis and amputation should be considered after TKA has failed; therefore, TKA was the preferred treatment for this patient.
Current status of TKA and rotation-hinged knee prosthesis in the treatment of Charcot knee joint
Because of the presence of osteoporosis, sclerosis, poor bone quality, nutritional disorders, inadequate blood flow, and unavoidable micro-trauma in patients with Charcot arthropathy, in the past, complications occurred easily, such as loosening and subsidence of the artificial joint, fractures around the artificial joint, infection, and hematocele. In addition, primary diseases causing nerve damage, such as diabetes, are associated with a high risk of infection. Patients with Charcot arthropathy with these diseases were not previously considered candidates for TKA surgery.3–6 However, with developments in prosthetic design and manipulation techniques, recent studies have shown a significant increase in the likelihood of successful TKA for Charcot arthropathy.11,28,30–33 Deng et al. reviewed and analyzed 12 patients (12 knees) with Charcot arthropathy who underwent TKA. The patients were followed-up for an average of 6.5 years. One patient developed an infection and prosthesis loosening 2 years postoperatively and underwent staged surgery, and the others had no postoperative pain. In these successful cases, the knee deformity was corrected, and the stability and function were excellent. In addition, there were no complications, such as infection, prosthesis loosening, and deep venous thrombosis. Currently, because TKA can quickly relieve symptoms, improve joint function, correct deformity, rebuild joint stability, and improve quality of life, greater numbers of patients with Charcot arthropathy undergo TKA. However, arthrodesis is still a remedy, if TKA fails.
To treat Charcot arthropathy using TKA, the appropriate operative timing should be selected, as follows: 1. The primary disease has been controlled. When the primary disease is unknown and contraindications are excluded, surgery to improve joint function can be considered. 2. Charcot arthropathy is in the “reconstruction stage” in the Eichenholtz staging, and imaging suggests that the bone destruction is static. Patients in the development and healing stages should be treated conservatively and await the reconstruction stage. If blind surgery is performed in these two stages, early prosthesis loosening may be caused by further bone destruction after surgery.32,34 Surgical indications: Eichenholtz stage II and III patients with uncontrolled conditions or sustained joint destruction and instability after 3 months of conservative treatment. 34 Surgical contraindications: active infection around the knee or systemic lesions; limb muscle strength grade ≤2; local severe osteoporosis; completely fused knee joint; severe underlying diseases; and other states of illness that may lead to surgical risk and a poor prognosis. 35
The primary consideration with surgical treatment of Charcot arthropathy is joint stability, followed by activity and function. Therefore, non-restricted and light-restricted prostheses are generally not choices in TKA. Specific prosthesis selection depends on joint stability and bone defects. Serious bone defects require bone grafting; however, poor bone quality in Charcot arthropathy results in poor bone healing of the graft and the host bone. Furthermore, severe joint deformity is often accompanied by joint ligament and capsule laxity and serious joint instability, which require a more stable knee joint after surgery. Therefore, in the current case, the authors’ medical group chose the rotation-hinged knee prosthesis. The rotating shaft protects the tibial stem from anchoring to the platform to prevent postoperative dislocation due to soft tissue laxity, while the rotation-hinged stem reduces the load on the grafted bone and reduces the risk of later collapse or fragmentation of grafted bone. Bae et al. followed nine patients (11 knees) for 12 years, agreeing with the use of a rotation-hinged knee prosthesis for severe knee instability. 29 Kim et al. reported the only case in which a rotation-hinged knee prosthesis developed a fracture 1 year after surgery. The authors suggested that using a rotation-hinged knee prosthesis increased the surface stress on the bone cement, leading to subsequent aseptic loosening of the prosthesis. However, after reading this article, we concluded that the reason for the failure was that the surgeon did not perform bone cement fixation of the prosthesis. 6 The concern with a rotating-hinged knee prosthesis is that its high stability results in biomechanical changes in knee movement, which will undoubtedly lead to associated complications. Although there is currently no relevant report confirming this concern, we believe that a decision to choose a high-restrictive prosthesis should be made carefully. For patients with relatively mild joint instability, surface prostheses and semi-restrictive prostheses should be selected, if possible, to maintain the option for future revision surgery.30,36
In the current case, the patient suffered injuries and tears in the internal and external patellar retinaculae, anterior and posterior cruciate ligaments, and medial collateral ligaments, with large bone defects. These findings suggest poor soft tissue and bone support of the knee joint. Thus, using the semi-restricted and surface prostheses, it is difficult to achieve good fixation, and a fully-restricted prosthesis should be used. The rotating-hinged knee prosthesis improves knee stability and retains the rotation between the femur and tibial prosthesis, which reduces the prosthesis-cement-bone interface shear force to avoid premature prosthesis loosening.
Summary experience
Finally, the author's experience in this case and the literature review of TKA in the treatment of Charcot arthropathy (summary, Table 3) indicate the following: 1. The timing of surgery, and the indications and contraindications must be appropriate. 2. Prosthesis selection should be done carefully. 3. Charcot arthropathy is often accompanied by large bone defects, which require proper bone grafting. Autologous bone grafts can be used for small lacunar bone defects, and allogeneic bone grafts or a gasket can be considered for large defects. 4. Bone cement. In addition to the fixation, bone cement can also be used to reshape the bone structure, and the cement can be mixed with antibiotics to prevent infection. 5. Osteophytes, free fracture fragments, and synovial tissues in the articular cavity should be completely removed to avoid affecting postoperative flexion and extension activities and bone metabolism. 6. Perioperative management. In addition to conventional medications, more attention should be paid to muscle training and braces to stabilize the joint. For patients with intraoperative periprosthetic fractures, external fixation should be performed for at least 3 months, and the amount of activity in rehabilitation training should be reduced as appropriate. 7. In accordance with the literature review, no patellar displacement was found, possibly because of joint stability; therefore, patellar replacement was not performed in this case.
Summary experience.

Supplemental Material
sj-pdf-1-imr-10.1177_03000605211058871 - Supplemental material for Rotation-hinged knee prosthesis for the treatment of Charcot arthropathy: a case report and literature review
Supplemental material, sj-pdf-1-imr-10.1177_03000605211058871 for Rotation-hinged knee prosthesis for the treatment of Charcot arthropathy: a case report and literature review by Weibin Du, Rongdan Dai, Rongliang Chen and Fuxiang Shen in Journal of International Medical Research
Footnotes
Ethics statement
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of competing interests
All the authors declare that they have no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work was supported by the Medical and Health Science and Technology Project of Zhejiang Province (NO. 2020KY797) and the Major Science and Technology Program for Social Development of Xiaoshan District, Hangzhou (NO. 11216 and 2019318).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
