Abstract
Nephrogenic rests (NRs) are foci of embryonic nephrogenic cells that persist beyond the 36th week of gestation. They are precursor lesions of Wilms tumor and are found incidentally in approximately 1% of infants. The term nephroblastomatosis (NBS) is utilized when nephrogenic rests extensively or multifocally affect the kidneys. We report a case of a left nephroblastoma with bilateral nephroblastomatosis, treated with neoadjuvant chemotherapy followed by a left nephrectomy, highlighting the imaging features of nephrogenic rests on ultrasound, CT, and MRI, and the potential possibilities of distinguishing nephroblastomas from nephrogenic rests in one hand, and the possibilities of distinguishing active nephrogenic rests from inactive ones in the other hand. We furthermore discuss the role of histology in making those distinctions, and the role of chemotherapy in reducing nephrogenic rests’ size and cellularity.
Introduction
Nephrogenic rests (NRs) are foci of embryonic nephrogenic cells, that persist beyond the 36th week of gestation. They have the potential to transform to Wilms tumor (WT), 1 and they can be a fortuitous finding in 1% of infants. 2
Nephroblastomatosis (NBS) is the term used when there are diffuse or multiple NRs. 1 It’s important to note that almost 28 to 40% of unilateral WTs and 90 to 100% of bilateral WTs are accompanied with NRs. 3
Our case draws attention to the significance of imaging and histology in the distinction between a nephrogenic rest and a nephroblastoma, as well as between a hyperplastic (active) nephrogenic rest and a non-hyperplastic one. Additionally, the role of chemotherapy in reducing the size nephrogenic rests is highlighted.
Case Report
A 3-year-old male child with unremarkable personal and family history and no anomalies during pregnancy, presented for abdominal distension. The rest of physical examination showed no other signs nor malformations.
The biological work-up showed no abnormality, and no elevated infectious markers.
An abdominal ultrasound was performed, showing a voluminous heterogeneous left renal mass, associated with multiple small and bilateral renal nodules, which exhibited an oval-shaped, hypoechoic and isoechoic homogeneous appearance, with no flow on color Doppler (Figure 1). A chest-abdomen-pelvis computed tomography (CT) was subsequently performed, showing the left voluminous renal mass, which exhibited a heterogeneous enhancement after contrast, in addition to the small bilateral oval-shaped intra and perilobar renal nodules, which displayed a hypodense appearance with a discrete homogeneous enhancement after contrast (Figure 2).
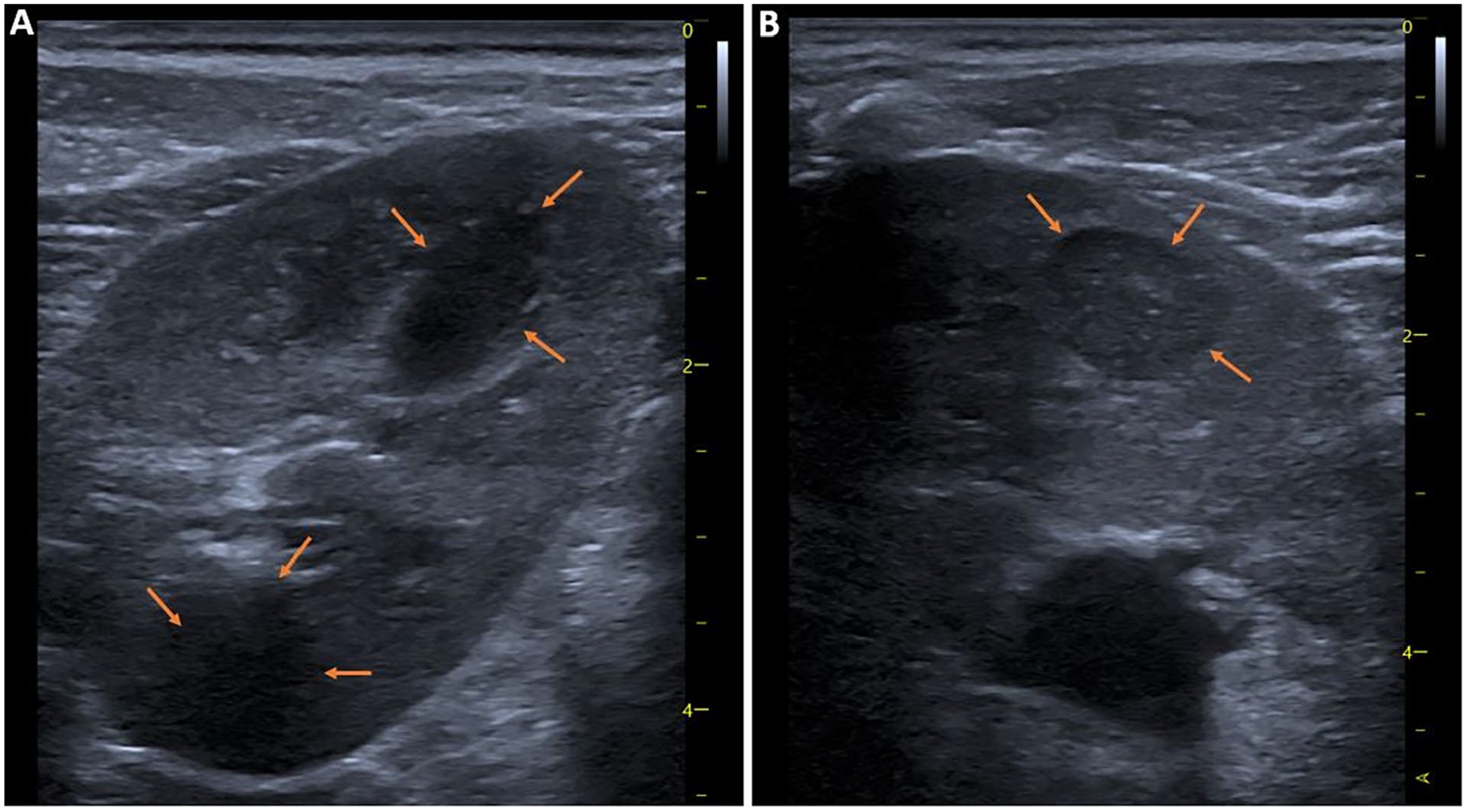
Ultrasound images of the left kidney using the superficial probe, showing nephrogenic rests manifesting as well-defined hypoechoic (A) and isoechoic (B) nodules.

Axial CT scan image of the abdomen, showing the nephrogenic rests manifesting as small bilateral oval-shaped nodules, which are homogeneous with almost no enhancement (orange arrows), associated with a left nephroblastoma manifesting as a voluminous, round-shaped and heterogeneously enhanced mass of the lower pole of the left kidney (white arrows).
Neither invasion of adjacent tissues nor lymphadenopathies nor distal lesions were found.
Magnetic resonance imaging (MRI) showed the voluminous left renal mass, manifesting a heterogeneous iso-intense signal on T1 and T2, with diffusion restriction and heterogeneous enhancement after contrast. In association with the small bilateral oval-shaped nodules, which manifested a hypointense T1 signal, and a variable T2 signal in comparison to the renal cortex. All of them exhibited diffusion restriction, and either a discrete or no enhancement after contrast, with a maximal diameter not exceeding 25 mm (Figure 3).

Axial MRI images, in T2-weighted sequence (A and B), ADC map (C), and post-contrast (D), showing the bilateral nephrogenic rests presenting as oval-shaped nodules, T2 hypointense (A, orange arrows) and T2 hyperintense (B, orange arrow), with low ADC (C, orange arrows) and no post-contrast enhancement (D, orange arrows). Associated with a left nephroblastoma presenting as a voluminous round mass, T2 hyperintense (B, blue arrows) with low ADC (C, blue arrows), and heterogeneous post-contrast enhancement (D, blue arrows).
The suggested diagnosis on imaging was nephroblastoma with bilateral nephroblastomatosis, due to age and imaging characteristics.
The patient underwent neoadjuvant chemotherapy, receiving four courses of Vincristine at a weekly dose of 1.5 mg/m2 for four weeks, in conjunction with two courses of Actinomycin D at a dose of 45 μg/kg, one administered in the first week and another in the third week.
Next, a control MRI was performed 2 months after the initial one, showing a 69% decrease in size of the nephroblastoma, and a decrease in size of the nephroblastomatosis (Figure 4), in addition to a decrease in apparent diffusion coefficient (ADC) for both the nephroblastoma (mean pre-chemotherapy ADC was measured at 0.51 × 10−3 mm2/seconds vs mean post-chemotherapy ADC at 0.65 × 10−3 mm2/seconds), and the nephroblastomatosis (mean pre-chemotherapy ADC was measured at 0.52 × 10−3 mm2/seconds vs mean post-chemotherapy ADC at 0.68 × 10−3 mm2/seconds).

MRI images showing decrease in size of the nephrogenic rests: Pre-chemotherapy ADC map (A) versus post-chemotherapy (B), and pre-chemotherapy coronal T2 (C) versus post-chemotherapy (D). Also note a decrease in size of the nephroblastoma (blue arrows).
The patient benefited from a left nephrectomy, with a histological examination of the specimen which confirmed the diagnosis of Wilms tumor with nephroblastomatosis. The WT exhibited a predominant blastemal component, while the NRs, mainly hyperplastic, were observed in both intralobar and periloar regions, demonstrating a direct interface with normal renal parenchyma, devoid of a pseudocapsule (Figure 5).

Histopathologic examination images with routine stain, showing: Hyperplastic perilobar nephroblastomatosis manifesting a direct interface with normal renal parenchyma with no pseudocapsule (A, ×10). Hyperplastic intralobar nephroblastomatosis with a direct interface with normal renal parenchyma (B, ×10). Post-chemotherapy Wilms tumor with chemotherapy-induced changes with foamy macrophages (C, ×4). Post-chemotherapy Wilms tumor of blastemal type (D, ×4).
Discussion
Nephrogenic rests can be categorized depending on their location, either as perilobar (found in the cortex peripherally or in the columns of Bertin) or intralobar. Intralobar NRs are more strongly associated with the development of Wilms tumor.
NRs exhibit varying proportions of blastema, stromal, and epithelial components. 1 However, the natural progression of nephrogenic rests remains unpredictable. Some of these cells may halt their differentiation process or eventually regress and turn into sclerotic NRs. On the other hand, others may advance to develop into hyperplastic NRs. 4 As a result, they are histologically categorized to four types: the dormant and the sclerosing rests which are typically microscopic with no ability to turn cancerous. And the hyperplastic and the neoplastic rests which are more sizable, have a high mitotic rate, and can exhibit cellular atypia. 1
Histological criteria that support a diagnosis of hyperplastic rests consist of an oval shape, absence of pseudoencapsulation, and presence of sclerosing tissue amidst hyperplastic tissue.3,5 To the contrary of tiny nephroblastomas, which are typically expansive, displaying a spherical shape, with the presence of a pseudocapsule. Nephroblastomas can be accompanied with hyperplastic or sclerosing nephrogenic tissue.3,5 The absence or presence of a pseudocapsule is a useful distinctive criterion before chemotherapy. However, post-chemotherapy nephrogenic nodules can also develop a fibrous capsule, making it challenging to distinguish them from nephroblastomas. 6 In our experience, WTs tend to manifest foamy macrophages, necrosis and fibrosis as part of their chemotherapy-induced change whereas in NRs we usually only see fibrosis.
On ultrasound, the perception of NRs is difficult especially the smaller ones. They appear as hypoechoic or isoechoic nodules.7,8
On CT, NRs are typically isodense (or slightly hyperdense). And exhibit either a homogeneous and discrete enhancement or no enhancement post-contrast injection.7,8
The distinction between a WT and a NR can be difficult on imaging, nevertheless, a diagnosis of WT should be considered more likely when a renal mass appears spherical, exophytic, and has a size exceeding 17.5 mm in maximal diameter. In our case, however, three NRs exceeded 17.5 mm in maximal diameter, measuring 18, 19, and 25 mm respectively). The signal intensity on MRI T1, T2 and diffusion sequences does not provide useful distinctions between WTs and NRs, as both exhibit restricted diffusion.3,9 Our case shows that diffusion restriction tends to decrease after chemotherapy (as indicated by elevation of ADC values), which is probably a translation of fibrotic changes and a decreased cellularity. To note that signal homogeneity is associated more often with perilobar nephrogenic rests, whereas intralobar rests and Wilms tumors are more often heterogeneous. Moreover, Imaging techniques lack the capability to reliably detect a pseudocapsule, which serves as a crucial pathological differentiator between a nephrogenic rest and a Wilms tumor in pre-chemotherapy specimens. 3
NRs on MRI can be iso or hypointense on T1 with generally no contrast uptake. Their signal is variable on T2, but more essentially, they exhibit a homogeneous signal. 10
It is believed that the distinction between hyperplastic NRs and non-hyperplastic ones on MRI can be oriented by signal intensity on T2 sequences, since hyperplastic NRs (with malignant potential) tend to be hyperintense similarly to WTs, while non-hyperplastic ones tend to be hypointense. 11 Some studies, however, have shown that the imaging features of hyperplastic and non-hyperplastic NRs (including density, signal, and shape) do not allow for their differentiation, 3 which is consistent with our case’s findings.
Moreover, a malignant transformation of a NR can be suggested on follow-up based on the persistence or the increase in size of nodules after chemotherapy 12 ; an area of nephroblastomatosis that becomes more large, more round or more heterogeneous should raise concern for malignant transformation. 10
Chemotherapy can be prescribed for NRs with the aim of reducing their size and preventing potential malignant degeneration. However, some authors argue that chemotherapy does not effectively prevent malignant transformation. As an alternative, these authors rather propose close imaging surveillance.7,13 For children with a high risk of Wilms tumor (due to genetic mutations, and other syndromes and predisposing conditions), screening at 3-month intervals is recommended to detect precursor lesions early. 7 For children with nephroblastomatosis, closer surveillance intervals are advised due to their higher risk of Wilms tumor. MRI is the preferred method for follow-up; however, a cost-effective approach could involve alternating between MRI and ultrasound, which would also reduce the need for sedation associated with MRI usage. 7
Conclusion
The characteristics on imaging that are more suggestive of nephroblastoma than nephrogenic rests are sphericity in shape, heterogeneity, and a maximum diameter usually less than 17.5 mm but can reach up to 25 mm. Additionally, nodules become highly suspicious during follow-up when they grow larger and rounder. Differentiating hyperplastic (active) nephrogenic rests and inactive ones seems to be not possible on imaging. Histology can distinguish nephroblastomas from a NR, as the absence of a pseudocapsule supports the diagnosis of a NR before administering chemotherapy, however, in post-chemotherapy specimens, even NRs can develop a pseudocapsule, making them difficult to differentiate from nephroblastomas. Furthermore, our case shows that chemotherapy is effective in reducing the size and the cellularity of nephrogenic rests and nephroblastoma.
Footnotes
Author Contributions
All authors contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
No ethical approval is required for de-identified single case reports based on our institutional policies.
Informed Consent
Written informed consent was obtained from a legally authorized representative.
