Abstract
Objective
This study investigated the efficacy and safety of superselective transcatheter arterial embolization for angiomyolipoma at the renal hilum.
Methods
Between August 2012 and January 2015, 13 patients with 16 angiomyolipomas at the renal hilum underwent initial, prophylactic, superselective transcatheter arterial embolization. The patients were followed by computed tomography or magnetic resonance imaging, and volume-reduction ratios after embolization were measured.
Results
The mean or median post-embolization volume reduction ratios were 23% (follow-up duration, 1–2 months), 55% (3–6 months), 55% (7–12 months), 66% (1–2 years), 67% (2–3 years), and 54% (>3 years). After initial embolization, none of the 16 tumors bled or required surgery; two (13%) tumors recurred; and three (19%) tumors received repeat embolization. Estimated glomerular filtration rates were not decreased at medians of 7 days (near the time of discharge) and 39 days (first clinical follow-up) post-procedure, compared with baseline. Except for post-embolization syndrome, no procedure-related complications occurred.
Conclusions
Superselective embolization for renal hilar angiomyolipoma is safe and kidney-preserving, with good tumor volume reduction and bleeding prevention.
Introduction
Renal angiomyolipomas (AMLs) contain abnormal vessels, fat tissue, and smooth muscle and occur at a frequency of 1% to 3% in the general population. 1 Renal AMLs can occur sporadically or in association with tuberous sclerosis complex (TSC) or lymphangioleiomyomatosis (LAM), or both. Although benign, renal AMLs can grow large enough to be symptomatic; they may bleed, which leads to hematuria and retroperitoneal hemorrhage; and they may cause severe renal dysfunction or even death due to blood loss. Increasing aneurysm size is associated with rupture, and an aneurysm size of ≥5 mm is a significant predictor of rupture. 2 , 3 The criteria for prophylactic treatment of unruptured renal AMLs are the presence of symptoms and an aneurysm that is at least 5 mm in diameter. 2 , 4
The prophylactic treatment goal is symptom relief or prevention of severe bleeding as well as preservation of normal renal parenchyma; 5 treatment can be accomplished with nephron-sparing surgery or superselective transcatheter arterial embolization (TAE). Superselective TAE is a minimally invasive and safe endovascular procedure and is favored for treating unruptured renal AMLs. This technique superselectively occludes the feeding arteries and aneurysms of tumors, thus sparing normal renal parenchyma. 6 , 7 However, regrowth and repeated hemorrhage after embolization remain concerns and necessitate repeat treatment. 8 Compared with embolization, nephron-sparing surgery provides a “permanent cure” for resectable lesions, 9 , 10 with the clear advantage of eliminating the potential for recurrent treatment. 4 , 11 However, nephron-sparing surgery has increased procedure-associated adverse sequelae, namely urinary leakage (5%), urinary fistulas (7%), hemorrhage (3%), and ileus (3%). 3 ,11–13 In addition, the tumor location and extension (central and extending to the renal hilum) can make it impossible to avoid renal pelvic injury, which can preclude nephron-sparing surgery and necessitate complete (total) nephrectomy. 5 , 6 , 14 , 15 Therefore, although nephron-sparing surgery is a reliable alternative to embolization, 16 , 17 nephrectomy or superselective embolization is more appropriate for hilar AMLs not amenable to nephron-sparing surgery. Superselective embolization is superior to nephrectomy regarding preserving renal function. However, superselective embolization for hilar AMLs can be particularly difficult to perform when feeding arteries arise from proximal renal arteries, such as the renal main trunk, anterior segmental artery, and posterior segmental artery. In these situations, particular care during embolization should be exercised to avoid reflux through the normal renal artery, which causes non-target embolization of normal renal parenchyma. Severe non-target embolization can induce renal infarction followed by renal dysfunction.
Superselective TAE is an important but technique-challenging treatment for renal hilar AMLs. However, although several studies have assessed the post-embolization regression of renal AMLs in general,18–24 no studies have evaluated the effectiveness of superselective TAE for treating renal hilar AMLs. Therefore, we investigated the efficacy of prophylactic, superselective TAE for renal hilar AMLs for reducing tumor volume, preventing bleeding, and protecting renal function.
Methods
Study design
This retrospective study was approved by our institutional review board (approval number: 16-168); the need for informed consent was waived because of the study’s retrospective design.
From our radiologic information system, we retrieved a list of all patients who underwent prophylactic, superselective TAE for unruptured renal AMLs in our department between August 2012 and January 2015. AMLs that invaded or occupied the renal pelvis on computed tomography (CT) were defined as renal hilar tumors (Figure 1a).
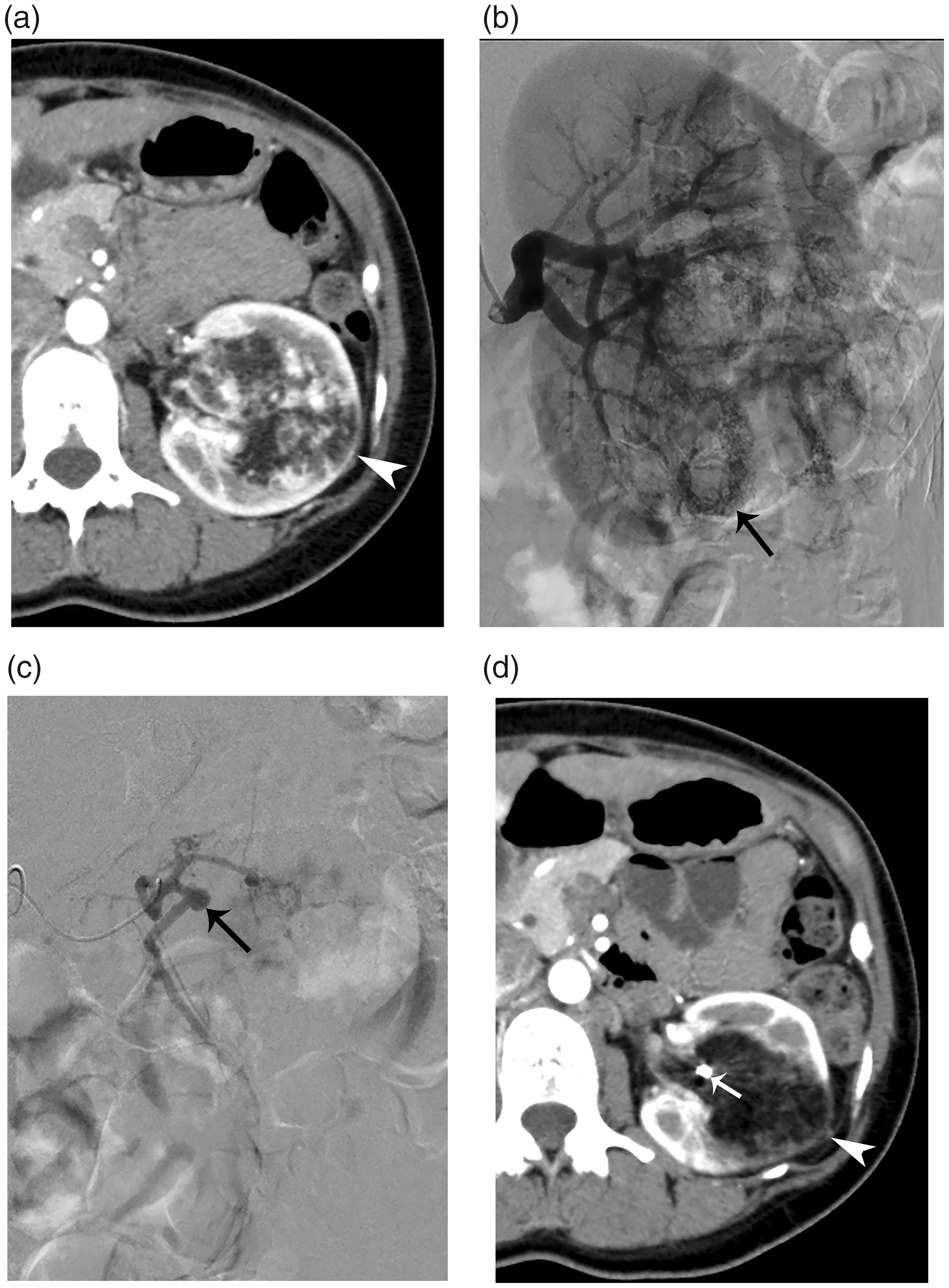
A 39-year-old woman with sporadic renal angiomyolipoma. (a) Axial contrast-enhanced computed tomography before embolization showing a fat-containing angiomyolipoma (arrowhead) at the left renal hilum. (b) Pre-embolization renal arteriography showing tortuous tumoral vessels and tumor staining (arrow). (c) Superselective arteriography of a tumor-feeding artery showing a single large aneurysm (arrow).
Superselective embolization
Before embolization, axial dynamic contrast-enhanced CT and coronal multiplanar reconstruction images in the arterial phase were used to detect the location and number of renal arteries and potential extrarenal feeders. In addition, renal arteriography (Figure 1b) was performed using a 4-Fr J-shaped catheter to identify abnormal tortuous vessels of the target AML, as well as any intratumoral aneurysms. A superselective microcatheter (inner diameter, 1.9 Fr; distal outside diameter, 1.98 Fr; Masters Parkway Soft; Asahi Intecc Co., Ltd., Nagoya, Japan) was advanced into the feeding artery or aneurysm (Figure 1c) through a 4-Fr catheter, and embolic agents were injected. Specifically, 1-mm multiporous gelatin sponge particles (GSPs) dissolved in contrast agent (Gelpart; Nippon Kayaku Co., Ltd., Tokyo, Japan), which could easily change shape and pass through the 1.98-Fr catheter without resistance, were tightly injected into tumor-feeding arteries for embolization. After these particles had totally occluded tumor-feeding arteries, metallic coils were placed in the proximal portion of the tumor-feeding arteries and in intratumoral aneurysms to avoid recanalization of the tumor-feeding arteries. Four of the tumors were embolized using 1-mm multiporous GSPs only, owing to difficulty placing coils inside the tumor-feeding arteries. The remaining 12 tumors were embolized using a combination of 1-mm GSPs and microcoils (Tornado [Cook Medical LLC, Bloomington, IN, USA; n = 6]; Hilal [Cook Medical LLC; n = 2]; Target [Stryker Corp., Kalamazoo, MI, USA; n = 1]; GDC [Stryker Corp.; n = 1]; or Trufill DCS Orbit [Codman & Shurtleff Inc., Raynham, MA, USA; n = 2]). After embolization, renal arteriography was performed to evaluate the embolization percentage (i.e., the percentage difference between the tumor staining during pre-embolization renal arteriography and that during renal arteriography after embolization) of the tumors and the presence of intratumoral aneurysms. For each tumor, the embolization percentage was assessed by two radiologists who worked together to reach a consensus.
Follow-up
For each of the 13 patients, we evaluated the following parameters: presence of concomitant disease (TSC or LAM); need for retreatment during follow-up; complications during or after TAE; and renal function and tumor volume before and after embolization.
Post-embolization syndrome (PES) constitutes fever, nausea, vomiting, and pain, and lasts for 3 to 7 days after TAE. These symptoms are attributable to inflammatory mediators and typically, are easily managed by conservative treatment. 7 , 20 , 25 Renal function evaluated by serum creatinine concentration and estimated glomerular filtration rate (eGFR) was evaluated before the procedure, during hospitalization, and at each clinical follow-up.
After embolization, imaging follow-up comprised contrast-enhanced CT (Figure 1d) or magnetic resonance imaging (MRI) with non-contrast MR angiography (MRA). Generally, MRI with non-contrast MRA is the first choice to avoid irradiation and contrast injection. Routine time points for imaging follow-up were 1 month, 6 months, and then annually after embolization; however, these times varied among the patients, for practical reasons. All follow-up images were reviewed, and the final available imaging time point for each patient was considered the observation endpoint. During follow-up after initial embolization, tumor regrowth to the original volume or greater, regrowth of intratumoral aneurysms to larger than 5 mm, recurrent bleeding, or the presence of associated symptoms was considered regrowth of the target tumor and was followed by re-embolization.
For each tumor, pre- and post-embolization CT or MR images in the axial plane were loaded into Volume Analyzer Synapse Vincent Software (Fujifilm, Tokyo, Japan). The tumor margins in each slice were delineated, and tumor volume was calculated automatically by the software.
Statistical analysis
Normally-distributed continuous data are described as means ± SD, and non-normally-distributed continuous data are presented as medians (interquartile range). Paired t-tests were used to compare the eGFRs after procedures with those before procedures and to compare the serum creatinine concentrations at the observation endpoint with those before the initial embolization. P values less than 0.05 (two-sided) were considered statistically significant, and the statistical analyses were performed using PASW Statistics for Windows, version 18.0 (SPSS Inc., Chicago, IL, USA).
Results
Patients
Of 26 patients who underwent prophylactic, superselective TAE for unruptured renal AMLs, 13 patients (12 women and 1 man) with 16 renal hilar AMLs were included in this study. Thirteen patients were excluded owing to the absence of renal hilar AML.
The patients’ and tumor characteristics are shown in Table 1. Of the 13 patients, 3 women had TSC, 6 women had sporadic LAM, and 4 patients (3 women and 1 man) had no concomitant disease. The patients’ age at the initial TAE was 39.6 ± 10 years (mean ± standard deviation [SD]; range, 28–64 years). Of the 13 patients, 9 (69%) had bilateral and multiple AMLs, and 4 (31%) had a solitary AML. Of the 16 tumors, 9 (56%) were located in the left renal hilum and 7 (44%) in the right renal hilum. Before TAE, the greatest tumor diameter (mean ± SD) was 8.2 ± 1.9 cm (range, 5.4–13.2 cm), and the median volume was 123 mL (interquartile range, 85–216 mL; range, 44–553 mL). Eight tumors (50%) had intratumoral aneurysms that were at least 5 mm in diameter on dynamic contrast-enhanced CT.
Patient and tumor characteristics before embolization and at the last follow-up.

1Tumors 3 and 5 were in the same patient; 2Tumors 12, 15, and 16 were in the same patient.
F, female; M, male; LK, left kidney; RK, right kidney; TSC, tuberous sclerosis complex; LAM, lymphangioleiomyomatosis; sLAM, sporadic LAM; AML, angiomyolipoma; N, none; P, present; TAE, transcatheter arterial embolization.
Initial embolization
The 13 patients underwent 15 initial TAE procedures for 16 renal hilar AMLs; two tumors (nos. 12 and 15) were embolized during the same session (procedure 12, Table 2). Renal arteriography after initial embolization showed that no aneurysm in the target tumor was ≥5 mm in diameter. In 12 of the 16 target tumors, the initial procedure achieved an embolization percentage of >90% (i.e., complete embolization). In the remaining four tumors (nos. 1, 2, 5, and 13), the initial procedure achieved an embolization percentage of 50% to 90% (Table 2). No non-target embolization was found on confirmatory renal arteriography after TAE.
Features of the 18 embolization procedures performed for 16 renal hilar angiomyolipomas in 13 patients.

1Procedures 16, 17, and 18 were retreatments for tumors 2, 5, and 10, respectively. 2In patients 6 and 12, who had high serum creatinine concentrations before initial embolization, serum creatinine concentrations at the endpoint observations were higher than the upper limit of normal (70.7 μmol/L).
TAE, transcatheter arterial embolization; eGFR, estimated glomerular filtration rate.
Follow-up
After the initial embolization, all 16 target AMLs in the 13 patients were followed for 41.6 ± 16.0 months (range, 5–68 months). During follow-up, three (19%) of the tumors underwent re-embolization; the remaining 13 target tumors did not regrow and did not receive retreatment. None of the 16 tumors bled or required surgery.
Tumors no. 2 (in patient 2), 5 (patient 3), and 10 (patient 9) underwent re-embolization 40, 27, and 6 months, respectively, after the initial TAE. Tumor no. 2 was re-embolized because follow-up CT (performed at 39 months) showed mild tumor regrowth (from 61 mL to 62 mL) and the development of an intratumoral aneurysm (diameter, 8 mm) (Table 1, Figure 2). Patient 3 had undergone debulking surgery of two target tumors (nos. 3 and 5) at another hospital approximately 10 years previously. Tumor no. 3 regressed well after initial TAE; however, tumor no. 5 was re-embolized because it regrew (Table 1, Figure 2), and the patient developed left back pain. Twenty-two months after re-TAE, tumor no. 5 had decreased in size, with a volume reduction of 50% compared with its volume before the second TAE (Table 1, Figure 2). Tumor no. 10 showed good volume reduction after the initial TAE (Table 1, Figure 2) but underwent repeat TAE for ablation of minor revascularization. This minor revascularization was observed incidentally and was embolized during the same TAE procedure that targeted the contrarenal AML. Therefore, only two of the three re-embolized tumors (nos. 2 and 5) underwent repeat embolization because of regrowth, and according to the criteria for tumor regrowth, the regrowth rate in our study was 13% (2 of 16). None of the three retreated tumors developed aneurysms that were ≥5 mm in diameter during the follow-up (duration, 3, 21, and 22 months, respectively) after the second TAE.

Trend lines in the volume percentages (y-axis) for each tumor at various time points in the radiologic follow-up (x-axis).
Other than PES, no procedure-related complications occurred during any of the 18 embolization procedures (15 initial and 3 repeat), and in all patients, PES was alleviated through conservative treatment. After all 18 procedures but 1, the patients were discharged in 5 to 8 days; only one patient (no. 11) had a prolonged post-embolization hospitalization time of 20 days (procedure 13, for a right extremely large renal AML [no. 16]), owing to PES, which was treated successfully with medications.
For all 16 target tumors, median tumor volume decreased from 123 mL (interquartile range, 85–216 mL; range, 44–553 mL) before initial TAE to 44 mL (interquartile range, 29–101 mL; range, 25–146 mL) by the final available imaging time point before any re-TAE, which was a mean of 38 months after initial TAE. Furthermore, 1 to 2 months after the initial TAE and before any re-TAE, the 15 AMLs that underwent radiologic examination had a volume reduction ratio (mean [± SD]) of 23% (± 17%). At 3 to 6 months, the 8 evaluated AMLs had a 55% (± 15%) volume reduction; at 7 to 12 months, 9 AMLs had a 55% (± 21%) reduction; at 1 to 2 years, 12 AMLs had a median reduction of 66% (interquartile range, 41%–74%); at 2 to 3 years, 13 AMLs had a median reduction of 67% (interquartile range, 52%–82%); and after 3 years, 10 AMLs had a mean reduction of 54% (± 33%).
Across all 18 TAE procedures for the 16 target tumors, eGFR near the time of discharge (102.4 ± 24 mL min−1 1.73 m−2; median period after procedure, 7 days) was significantly (P = 0.02) higher than the pre-procedure eGFR (94.9 ± 21.8 mL min−1 1.73 m−2). Mean eGFR did not differ between pre-procedure and the first clinical follow-up (median time after procedure, 39 days; eGFR, 95.4 ± 21 mL min−1 1.73 m−2). The median serum creatinine concentration in the 13 patients was 47.7 μmol/L before the initial embolization, and the concentration increased significantly (P = 0.002) to 51.3 μmol/L at the observation endpoint; the mean increase was 6.2 ± 5.3 μmol/L.
Discussion
Among the 13 patients with renal hilar AML, the regrowth rate for the 16 tumors was 13% (2 of 16), and the repeat treatment rate for the 16 tumors was 19% (3 of 16). Results were comparable to previously reported regrowth rates, which ranged from 0% to 40% for AMLs that included renal hilar as well as peripheral tumors. 3 , 11 For all 18 TAE procedures (15 initial and three repeat), the only procedure-related complication was PES; no tumor bled or underwent surgery after embolization. Therefore, prophylactic superselective TAE for renal hilar AML was a safe and minimally invasive procedure that resulted in tumor reduction and prevented bleeding without renal dysfunction, in our patient population.
Previous studies reporting the volume reduction ratios of renal AML after prophylactic embolization vary regarding the method used to measure tumor regression and the follow-up duration. Rimon et al. 18 reported a reduction of 24% in the largest AML diameter in patients followed for a mean of 22.4 months, whereas Patatas et al. 19 showed a 6.5% reduction in the longest axial diameter at 3 months, 16% at 6 months, 24% at 12 months, 26.5% at 2 years, 33.0% at 3 years, and 36.0% at 4 years. Lee et al. 20 reported a short-term (mean duration, 3.4 months) longest-diameter reduction of 33.3% and a long-term (mean duration, 28.2 months) reduction of 43.0%. Regarding reductions in tumor volume, Hocquelet et al. 21 reported a mean volume reduction of 72% at a median follow-up duration of 28 months, and Planche et al. 22 found mean volume reductions of 43% for AMLs followed for 1 to 6 months and 81% for those followed for more than 1 year. Kato et al. 23 assessed the regression of LAM-associated AMLs after superselective TAE and reported a mean reduction in tumor volume of 69% at a mean follow-up of 11 months. Furthermore, 1 year after embolization, sporadic AMLs had a median reduction in tumor diameter of 29.8% and a median reduction in tumor volume of 67.9%; TSC-AMLs yielded equivalent values of 23.6% and 47.7%, respectively. 24
Despite these studies, the regression of renal hilar AMLs after prophylactic embolization has not been addressed specifically in the literature. Given the influence of follow-up duration on regression rates and the fact that large AMLs can be irregular in shape, in this study, renal hilar AMLs at each available imaging time point were assessed retrospectively, and the volume reduction ratio was evaluated. Results revealed reductions in tumor volume (mean or median) of 55% (3–6 months), 55% (7–12 months), 66% (1–2 years), 67% (2–3 years), and 54% (>3 years). These values were within the ranges of volume reduction (43%–81%) reported in previous studies.21–24 However, the volume reduction 1 to 2 months after embolization was as small as 23% in this study, which was less than that previously reported, and likely because our follow-up duration was shorter than those in the aforementioned studies. These results suggest that tumor necrosis occurred after TAE without shrinkage by 1 to 2 months and then absorption of the necrotic area began 3 to 6 months and continued until 2 to 3 years. Tumor regrowth was found more than 3 years after TAE.
Renal function was preserved after TAE, given that the eGFRs soon after TAE of the target hilar tumors (median follow-up, 7 days [near hospital discharge] and 39 days [first clinical follow-up]) were no lower than the pre-procedure eGFRs. Even when post-TAE renal function had deteriorated slightly compared with that before initial embolization, the mean increase in serum creatinine among our 13 patients was as small as 6.2 ± 5.3 µmol/L at the end of the long-term follow-up (mean duration, 41.6 months). In only two patients (nos. 6 and 12), whose pre-embolization serum creatinine concentrations were abnormally high, did the endpoint serum creatinine exceed the upper limit of the normal range. Furthermore, although we focused on renal hilar AMLs, 69% (9 of 13) of this study population had bilateral and multiple AMLs; therefore, for these patients, renal function at the end of long-term follow-up was associated with not only hilar AMLs but also other, non-hilar tumors.
Regarding technical difficulty, advancing the microcatheter can be challenging in feeder vessels that have small diameters or are at a steep angle. 20 In this situation, complete superselective embolization may not be possible if the normal renal parenchyma is to be sufficiently preserved. In this study, the initial embolization was incomplete (embolization percentage, 50%–90%) in four (25%) target tumors (nos. 1, 2, 5, and 13). However, none of these tumors bled after TAE. In addition, two of these tumors (nos. 1 and 13) had no regrowth in terms of volume and were not at risk of bleeding or retreatment. The remaining two tumors (nos. 2 and 5) underwent repeat TAE, after which the tumor volumes decreased, with no risk of bleeding.
This study had several limitations. First, because this was a single-institution study, selection bias was present. Second, given the study’s retrospective nature, the radiographic follow-up time points were difficult to control; not all patients were consistently available for follow-up at the same time points. In addition, because of the small sample size, we could not perform statistical analyses to assess the association between tumor volume reduction ratio and follow-up duration.
In conclusion, this study showed that prophylactic superselective TAE is a safe and kidney-preserving therapy for renal hilar AMLs, with good efficacy in reducing tumor volume and preventing bleeding despite the technical difficulty.
