Abstract
We describe a rare case of late pulmonary recurrence of Wilms tumor, occurring 7 years after initial remission, observed at Saint-Damien Hospital (HSD) in Haiti. The medical team identified a persistent cough in the child and confirmed the diagnosis through chest imaging. The child received chemotherapy and radiotherapy with clinical improvement but without significant radiological response, unfortunately leading to a fatal outcome. This outcome highlights the need for action to improve childhood cancer care in Haiti. It will be challenging to improve pediatric oncology capacity, whether in terms of infrastructure, training, or access to treatment without a clear commitment from health authorities.
Introduction
Wilms’ tumor (WT) accounts for 7% of all pediatric cancers. 1 The annual incidence ranges from 4 to 10 per million in children younger than 15 years, and in the United States, it is higher among black children. Survival rates are more than 90% for early disease and 70% for metastatic disease in high-income countries. However, outcomes remain unsatisfactory in low- and middle-income countries, with a 5-year survival rate of approximately 50%. Despite a good prognosis, relapse of WT under the best conditions can still occur in approximately 15% of patients. Relapses occur within the first 2 years after diagnosis and often affect the lungs, pleura, tumor bed, and liver. Recurrence can occur up to 5–25 years after primary diagnosis, although it is rare according to the literature. 2 Relapse is the appearance of disease at the same site at the time of diagnosis after remission, while recurrence is at a different site at the time of diagnosis after remission. In the event of a relapse or recurrence, it is essential to have a new therapeutic approach. Relapses and recurrences usually occur in advanced stages III and IV. This case highlights the limitations of therapeutic options and the challenges of monitoring in low-resource settings.
Presentation of the case
The patient, a 7-year-old boy, demonstrated remarkable resilience throughout his journey, which is truly admirable. He was diagnosed with stage III left WT at 15 months of age in 2014 at St Damien Hospital in Haiti. At the time, he presented with abdominal distension and difficulty urinating for 15 days. His kidney function was normal, creatinine 0.6 mg/dl (Normal Value: 0.3–0.7), blood urea nitrogen 7 mg/dl (NV: 5–18)—no stigma of genetic malformation. Management included preoperative chemotherapy with vincristine, actinomycin D, and doxorubicin in August 2014 after carrying out an extension workup. Evaluation at week 6 of chemotherapy showed a good response, and the patient underwent left total nephrectomy with loco-regional lymph node sampling and removal of perirenal fat. The pathology described a 10 cm renal tumor with a micro contingent of blastemal, epithelial, and fibroblastic cells; immunohistochemical and cytogenetic tests are unavailable in Haiti. The pathology was conclusive of WT without infiltration of perirenal lymph nodes and renal fat. He completed 24 weeks of chemotherapy without any significant events in February 2015.
During the 2 years following treatment, he was followed monthly in the clinic with a physical examination, abdominal ultrasound, and chest X-ray. This schedule continued quarterly from the third to the fifth year and then every 6 months thereafter. Monitoring ended in December 2019, revealing no signs of disease on abdominal ultrasound and chest X-ray.
At 7 years old, the patient returned to the outpatient oncology clinic in November 2020 with a range of symptoms, including a persistent wet cough, intermittent fevers, vomiting, and severe abdominal pain. These symptoms indicated a potential health issue, particularly the wet cough and fevers. Physical examination revealed a stable patient with adequate height and weight for age, afebrile, nondyspneic, and with decreased left lung sounds and bilateral crackles in the lungs. Laboratory examinations were performed, including COVID-19 polymerase chain reaction (PCR) testing, investigation for tuberculosis, abdominal ultrasound, and chest X-ray. Chest X-ray showed left perihilar opacity and minimal pleural effusion; abdominal Ultrasound Sonography was negative. Investigations for tuberculosis and COVID-19 were negative: tuberculin was 2 mm, GeneXpert Mycobacterium Tuberculosis was not detected, and SARS-CoV-2 PCR was negative.
Antibiotics were administered for 10 days. At the end of antibiotic therapy, a follow-up chest X-ray was performed without much improvement, and a chest computed tomography (CT) scan was ordered. It showed a significant and dense left para-hilar mass measuring 70 × 68 × 66 mm with left basal segmental atelectasis, an air bronchogram, and a pleural effusion (Figure 1). The patient was prepared for a lung biopsy.
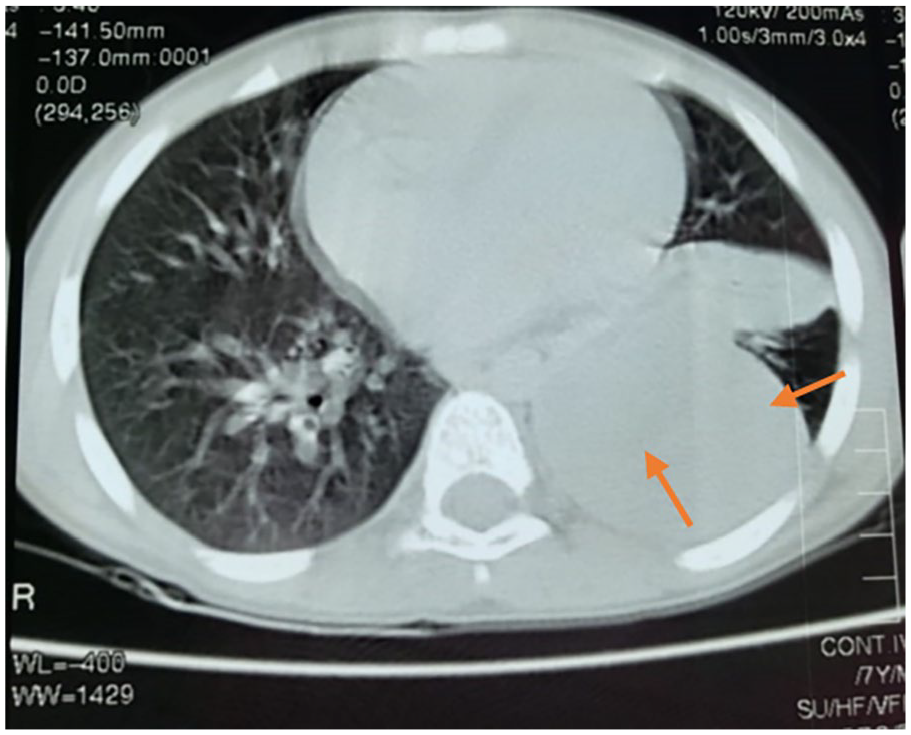
CT scan of the lungs at the time of recurrence diagnosis, large dense left para-hilar mass measuring 70 × 68 × 66 mm with left basal segmental atelectasia, aerial bronchogram.
A left posterior lateral thoracotomy was performed, and a large tumor mass was found, leading to an incisional biopsy and drainage of the left pleural effusion in January 2021. The specimen was sent to the laboratory on January 5, 2021. While awaiting the biopsy result, cyclophosphamide was administered at 200 mg/m² for 5 days to facilitate cytoreduction and maintain patient stability. Given the low number of pathologists in Haiti and the absence of this specialty at our hospital, samples are sent to a private laboratory at the hospital’s expense. This approach highlights the local situation and the importance of cytoreduction for managing patients awaiting pathological diagnoses, underscoring the challenges faced in providing comprehensive care in resource-limited settings.
The pathology report received on February 4, 2021, 4 weeks after the surgical procedure, describes a biopsied mass from the left lung. The mass consists of an elongated brownish tissue fragment measuring 1.5 × 0.6 × 0.2 cm and a beige and brownish budding nodule with a nippled surface measuring 2 × 1.2 × 0.8 cm. Microscopic examination reveals tumor proliferation characterized by large areas of blastema cells with basophilic cytoplasm, slightly nipple-shaped vesicular nuclei, and small nucleoli. The supporting stroma is relatively abundant and fibroblastic, with the periphery showing abundant fibrous tissue consistent with metastatic WT. The patient received the NWTS (National Wilms Tumor Study Group) 4003 chemotherapy protocol based on doxorubicin, vincristine, cyclophosphamide, and etoposide for 24 weeks with radiotherapy sessions in both lungs and boosting the lesion. The total radiation dose was 1950 cGy, administered in 13 fractions.
After treatment, a CT scan revealed a densely lobulated mass measuring 3.3 × 4.5 × 5.6 cm in the left lower lobe; however, the patient remained asymptomatic (Figure 2). He moved to palliative care, and multiple interventions were made with the family, among others; he died 6 months after completing treatment with curative intent. The family’s unwavering support and involvement in the patient’s care were crucial throughout this challenging journey, underscoring the importance of a strong support system in such cases.

CT scan of the after completing treatment including radiotherapy with residual left basal lesion.
Discussion
WT is the most common form of renal cancer in children. Typically, most recurrences of this tumor occur within 2 years of the first diagnosis. It should be noted, however, that relapses occurring 5 years after initial diagnosis are classified as “late recurrence” and are relatively rare. 2 WT is curable through multimodal treatment, which includes chemotherapy, radiotherapy, and surgery (Table 1). Usually, recurrences occur within 2 years of initial diagnosis; our patient recurred 7 years after initial diagnosis despite continued monitoring, which constitutes a significant challenge. This situation is all the more difficult to manage as it occurs after a prolonged period of remission, which may have created a feeling of security for the family. Despite efforts to identify early signs of relapse, the fact that this recurrence occurred so long after initial treatment raises questions about the effectiveness of existing follow-up protocols. It highlights the need to reevaluate surveillance protocols to detect and manage late relapses more effectively.
Description of the different chemotherapy protocols at initial diagnostic and recurrence of the Wilms tumor.

During radiotherapy, chemotherapy is temporarily withheld to reduce cumulative toxicity as both treatments can contribute to it. Upon resumption of chemotherapy, the vincristine dose is increased to intensify treatment, reach the intended cumulative dose, and thereby enhance the patient’s therapeutic response.
Predictors and risk factors
Delays in receiving pathology reports after surgery may compromise the effectiveness of treatment by delaying adjuvant treatment, increasing the risk of disease progression, and thereby reducing the chances of survival. 3 Salvage treatment for a WT recurrence in a child may include a combination of surgery, chemotherapy, and sometimes radiation therapy, depending on factors such as the location and extent of the recurrence, as well as previous response to treatment. Different drugs can be used for high-dose chemotherapy, and an approach such as autologous stem cell rescue can also be considered. 3 Prognostic factors (clinical, histological, and biological) have been proposed for the recurrence of WT: tumor size, patient age, histology, lymph node metastases, and local tumor characteristics. The stage and histology of the tumor are essential factors. Although stage IV and anaplastic histology are associated with adverse outcomes, 4 recurrence beyond 2 years is rare; according to the literature, recurrence can occur up to 25 years after initial diagnosis. There is no specific guideline for relapse. Since 80% of relapses occur within the first 2 years after nephrectomy, it is easier to monitor high-risk patients using imaging. 5 Abdominal USG and chest X-ray are the most commonly used methods. Relapse occurs in approximately 15% of patients with WT, and there is no optimal monitoring method to detect relapse after treatment. 6 According to one study, 5-year overall survival did not differ between patients whose recurrence was detected by CXR (Chest X-Ray)/US (Ultrasound) and CT. 7 Surveillance tests to detect relapse or earlier recurrence are essential but costly, constituting a burden on the health system and families.
Case analysis and recommendations
In our patient’s case, there was a regression of clinical signs, but no significant response to the second-line protocol, including radiotherapy. The most common site of relapse is the lungs. However, relapses in the tumor bed, intra-abdominal, bone, and brain are less frequent. 8 Indicators of a good prognosis for relapsed children include favorable histology, an interval of at least 12 months between nephrectomy and relapse, pulmonary relapse, initial treatment with vincristine and actinomycin D, and absence of prior radiotherapy. 9 Despite the presence of these factors, our patient had a poor outcome. Depending on the risk group, treatment for recurrent WT will include chemotherapy, surgery, radiotherapy, and stem cell transplant. Initiating a new treatment offers hope to the patient and their family. It reflects the team’s continued commitment to exploring all available options to provide the best possible care and marks the start of a new phase in their fight against cancer. The CT scan performed at the end of the second line of treatment showed the persistence of the left basal lung lesion, and in our context, there are no other curative therapeutic options. After no response to the second line of treatment and despite multiple interventions, a transition to palliative care was offered to the family. Unfortunately, the patient died 6 months after completing the second line of treatment. This recurrence was a shock to the parents. Despite this, they sought alternative solutions. Palliative care was painful for them. When a child dies, parents engage in a grieving process filled with pain, a sense of injustice, and unanswered questions. This outcome highlights the challenges encountered in managing this patient, as well as the reality in a country with limited resources. International collaboration can encourage participation in international clinical trials to access promising experimental treatments and to share knowledge and resources. Advocate for health policies that promote investment in the infrastructure and resources needed to manage pediatric cancers. Raise awareness among policymakers of the importance of combating pediatric cancer and the need to support health programs financially.
Conclusion
Recurrences of WT with favorable histology generally have a good prognosis, especially when they appear more than a year after the initial diagnosis. In our setting, despite regular follow-up after treatment, we faced resource limitations. The adaptability of protocols to local contexts, particularly in low-middle income countries, provides us that we can give the best care despite resource limitations. Palliative care must be an option to ensure the child’s dignity and comfort and foster a culture of empathy and compassion.
Early diagnosis of WT is essential for timely access to adequate resources in low-income countries. It is also important to ensure timely access and effective management of recurrences utilizing accessible and available diagnostic and therapeutic methods. In Haiti, the Ministry of Health must prioritize capacity building and the integration of pediatric cancer into a child health strategy; it will require a commitment to ensure the availability of training and resources, a responsibility that we must all share, engaging all of us in the solution.
Footnotes
Acknowledgements
We thank Dr Monika Metzger for her excellent editorial support. Also, we thank all the healthcare providers in the oncology department for their contribution to this work.
Author contributions
Principal author: PYG. designed the study, wrote the manuscript, and coordinated the research.
Co-authors: YA and RA contributed to the writing of the clinical case and the search for relevant bibliography.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data is available subject to approval by the competent authorities. To access the data, permission from the ethics committee, the Ministry of Health, the scientific committee, and the hospital executive director of the hospital is required.
