Abstract
Poland syndrome is indeed a rare congenital malformation that can present with various degrees of thoracic and homolateral upper limb anomalies. The classic features of Poland syndrome include agenesis or hypoplasia of the sternocostal head of the pectoralis major muscle, hypoplasia of the nipple, absence of subcutaneous fat, multiple rib abnormalities, and sometimes Sprengel deformity (elevation of the scapula). Additionally, ipsilateral symbrachydactyly (combination of “short fingers” with cutaneous syndactyly [fused fingers]) may also be observed. However, it’s important to note that not all of these findings are always present in every patient, and the combination of features can vary greatly. Surgical treatment for Poland syndrome primarily focuses on improving pulmonary functions resulting from severe thoracic deformities. However, it is frequently performed with the additional goal of enhancing cosmetic appearance. In recent times, the use of adipose-derived mesenchymal stem cells and fat transfer has shown promising results for correcting chest defects and breast augmentation. In our study, we present a series of 3 cases that were referred to our institution due to thoracic deformities associated with Poland syndrome.
Introduction
Poland syndrome was first described by Alfred Poland in 1841. It is characterized by a congenital absence or underdevelopment of the pectoralis major muscle. In some cases, it is also associated with hypoplasia of the breast, athelia (absence of the nipple), agenesis of ipsilateral costal cartilage (ribs 2, 3, 4, and 5), ipsilateral symbrachydactyly (shortened fingers), and deficiency of subcutaneous fat and axillary hair.1,2
In rare and severe cases, Poland syndrome may involve abnormalities of internal organs such as the lungs, kidneys, and heart. As a result, the severity of the syndrome can vary. 3 Currently, it is believed that Poland syndrome is characterized by the absence of the sternocostal bundle of the pectoralis major muscle.
Poland syndrome is a rare condition, with an incidence ranging from 1 to 3 per 100 000 newborns and a clear male predominance.1,4 The right side is affected in 75% of cases. 1 The exact cause of Poland syndrome remains unknown.1,4 This condition arises from a disruption in the blood supply to the subclavian arteries, vertebral arteries, and/or their branches during the early stages of embryonic development. It is hypothesized that occlusions occur at specific locations within these vessels during or around the sixth week of embryonic development, leading to the emergence of consistent abnormalities. To elucidate the pathogenesis of a group of malformations including the Poland, Klippel-Feil, and Mobius anomalies, the term “Subclavian Artery Supply Disruption Sequence” (SASDS) is proposed.
We present 3 severe cases of chest wall deformity and their clinical and radiological characteristics.
Cases Reports
Case 1
We present a case study of a male infant who is 1.5 years old and was delivered vaginally at full term. The mother’s medical and obstetric history was uneventful, and there were no indications of any thoracic deformities in the parents upon examination. They were not blood relatives.
When the child was 4 months old, the parents observed asymmetry in the thorax and decided to seek advice from a pediatrician.
Upon examination, the infant exhibited left pectoral hypoplasia, with asymmetrical nipples. There were no abnormalities in the fingers or toes, no signs of scoliosis, and no asymmetry in the lower limbs. The cardiac examination yielded normal results, and there were no facial abnormalities (Figure 1).

One and half year boy with Poland syndrome note the flattening of the left pectoral region with displaced nipple.
A standard chest X-ray revealed no presence of extra ribs.
A thoracic CT scan was performed (Figure 2), revealing agenesis of the right pectoral muscle, with intact skin coverage, and underdeveloped intercostal muscles compared to the opposite side.

Axial CT images of the thorax showing hypoplasia of the left pectoralis (white arrow).
Case 2
A 1.5 year old girl, born at full term through vaginal delivery from a closely monitored pregnancy, with no history of consanguinity or maternal drug use during gestation.
When the child was 6 months old, the parents observed asymmetry in the thorax prompting them to seek medical advice from a pediatrician.
Physical examination revealed a depression of the right thoracic wall, resulting in asymmetry between the 2 hemithoraxes (Figure 3).

One and half year girl with Poland syndrome note the chest asymmetry with right anterior chest wall depression and flattening of the right pectoral region with displaced nipple.
The rest of the examination showed no malformation of the upper or lower limbs, cardiac examination was without abnormality.
A thoracic CT scan was performed (Figure 4), which revealed agenesis (absence) of the right pectoralis major and minor muscles, along with hypotrophy (underdevelopment) of the right mammary gland.

Axial CT images of the thorax showing hypoplasia of the right pectoralis (white arrow) hypoplasia of the mammary gland (yellow arrow).
Case 3
We present a case of a 7-year-old boy who was born at full term through vaginal delivery following a pregnancy with inadequate monitoring. There is no history of consanguinity or maternal drug use during gestation. Since birth, the boy has exhibited a deformity of the thoracic cage and has been hospitalized multiple times for respiratory issues.
During the physical examination, a depression of the left thoracic wall was observed, resulting in asymmetry between the 2 hemithoraxes. Additionally, there was an asymmetric elevation of the 2 scapulae (Figure 5).
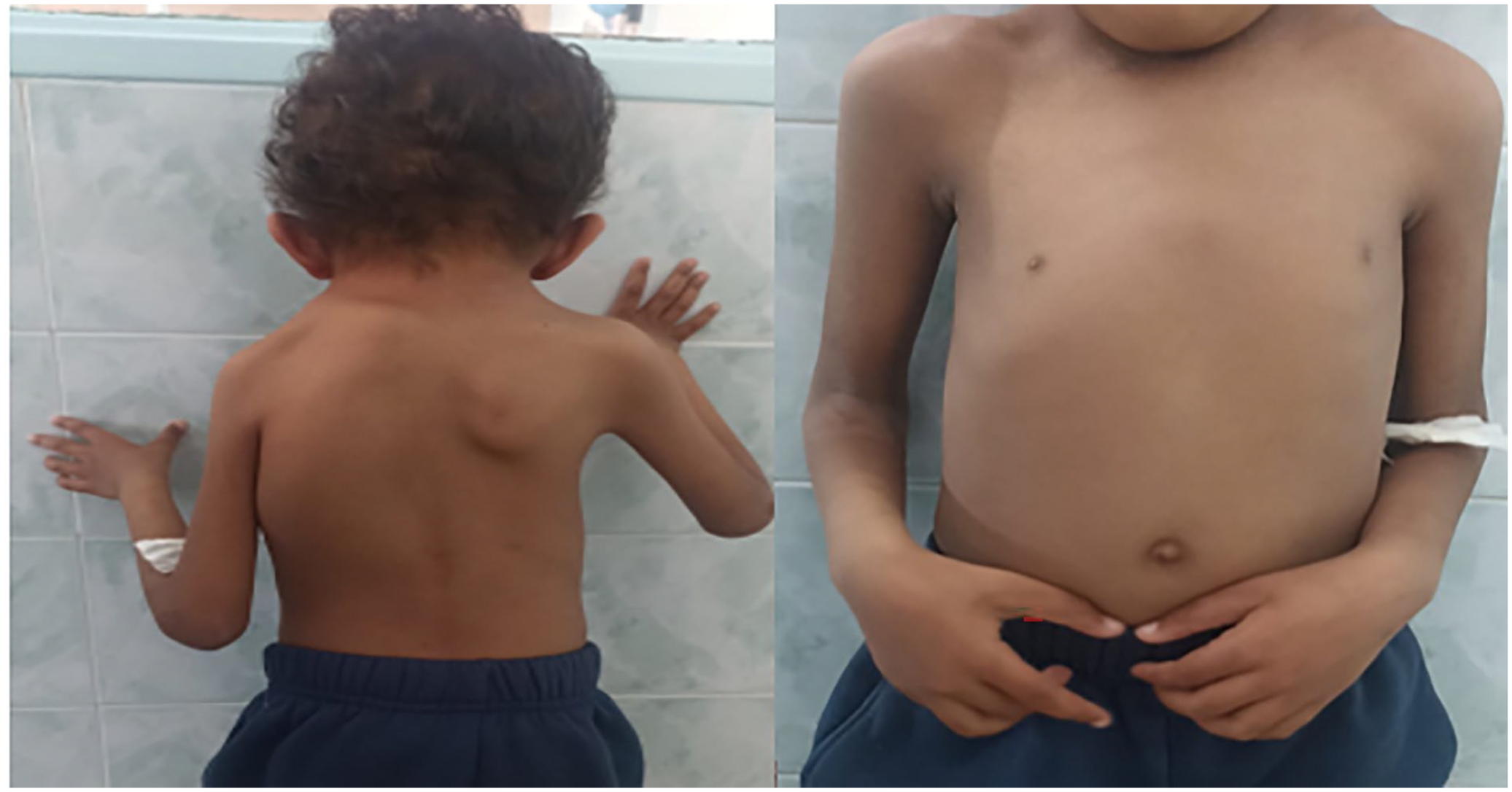
Seven years old boy with Poland syndrome note the chest asymmetry with left anterior chest wall depression and flattening of the left pectoral region with displaced nipple.
A CT scan of the cervical-dorsal spine was performed, revealing the following findings:
A left convex dorsal scoliosis with a vertebral block at C6-C7 and a butterfly vertebra at C8 (Figure 6).
Costal malformations, including fusion of the posterior costal arches of the 1st and 2nd left ribs (Figure 7).
Thinning of the left ribs.
Hypoplasia of the left lung and left pectoral muscle (Figure 8).
Bilateral elevation of the scapulae (Figure 9).
Agenesis of the right kidney (Figure 10).
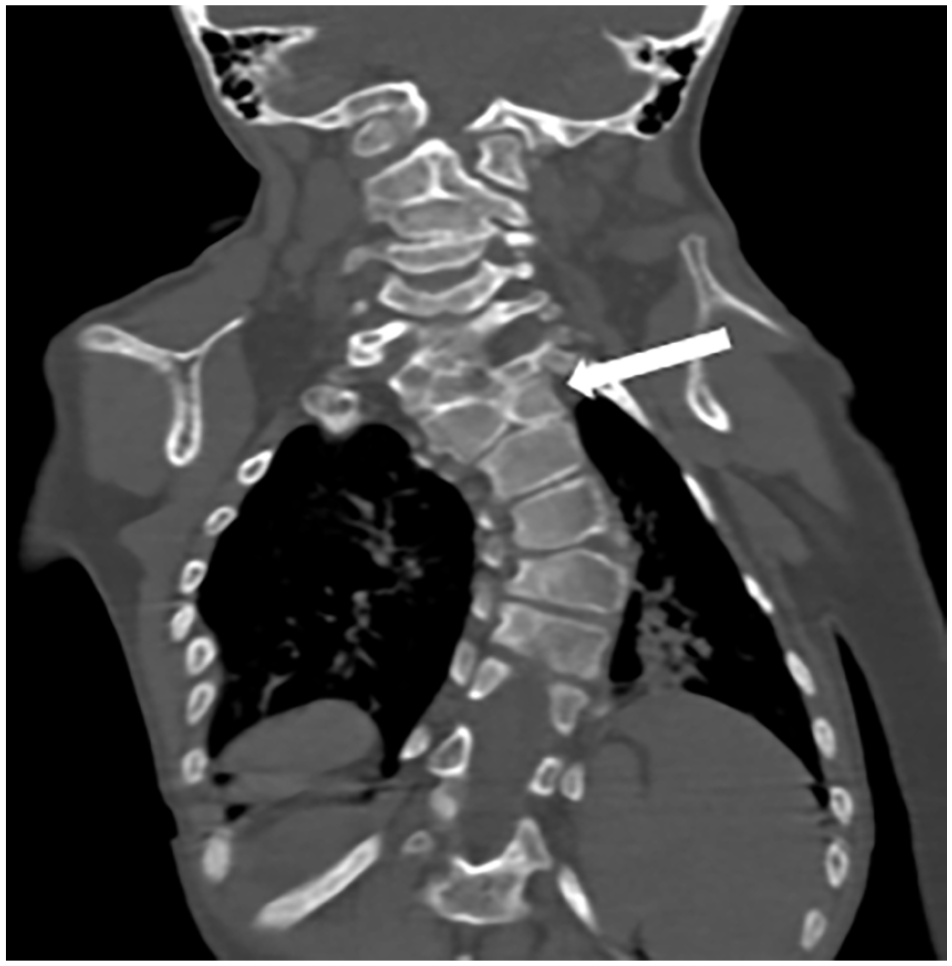
Coronal reconstruction of a CT scan showing dorsal scoliosis with a butterfly vertebra (white arrow).

Sagittal reconstruction of a CT scan showing fusion of the 1st and 2nd left ribs (head of arrow).

Axial CT image of the thorax in the bony windows showing hypoplasia of the left pectoralis major muscle (head of arrow) and the left lung, thinning of the left ribs in comparison with the contralateral side (white arrow).

3D reconstruction of a CT scan showing an elevation of both scapulae.

Axial image of an abdominal CT scan showing an empty left kidney lodge.
Discussion
Poland syndrome is a rare congenital disease characterized by musculoskeletal malformations of the chest wall and the upper limb on the same side of the body. It was first described by Alfred Poland in 1841 during a cadaver dissection. The main hypothesis suggests that the partial or complete blockage of blood flow in the subclavian or vertebral arteries, as well as their branches, occurring during or shortly before horizons 17 and 18, is the cause behind the absence of the pectoralis major muscle, the absence of degeneration in the interdigital tissue, and the absence of segmentation in the cervical vertebrae.5,6
Interrupting the internal thoracic artery leads to the absence of the pectoralis major muscle and underdevelopment of the breast on the same side.
Interrupting the subclavian artery beyond the origin of the internal thoracic artery results in isolated malformations in the distal transverse limb.
Interrupting the subclavian artery before the origin of the internal thoracic artery but after the vertebral artery leads to the Poland anomaly. 6
Additionally, some studies have linked exposure to ergot alkaloids during the first trimester of pregnancy to the development of Poland syndrome. 7 However, it is worth noting that genes involved in the embryonic development of the pectoral girdle may also play a role in this disorder. 8 In addition to its characteristic features, PS has been observed in conjunction with various other conditions and features, including dextrocardia, vertebral abnormalities, and undescended testes. There have been reports of extracorporeal intercostal liver herniation and thoracic myelomeningocele, renal agenesis, and megacalycosis of the ipsilateral kidney.8,9 While associations with malignancies and other rare disorders have been identified, these events are generally considered to be unrelated or not directly correlated.8,9
The incidence of Poland syndrome ranges from 1 to 3 per 100 000 newborns, with a higher prevalence in males.1,4 Based on research findings, the majority of cases are sporadic, where affected children have unaffected parents without any family history of congenital anomalies. However, there have been rare reports of familial cases in the literature, with rates ranging from 4.2% to 8.4%. These familial cases are typically observed in Polish syndrome families, where the index case has PS and at least one parent exhibits normal pectoral muscles but with upper limb and/or thoracic anomalies commonly seen in PS. 10
In sporadic cases, the risk of recurrence, meaning the likelihood of the same couple having another affected child, is low. However, for familial cases, the risk of recurrence is higher compared to the general population. Therefore, it is crucial to clinically evaluate the parents of affected children to rule out the possibility of them having mild, undiagnosed forms of the condition, which could transform an apparently sporadic case into a familial one and increase the risk of recurrence. Genetic counseling is essential in all cases of PS and it should primarily focus on providing information to patients.
The specific pathogenic mechanisms underlying MS are still unknown, and most MS patients present with an isolated form of the condition.
Currently, various transmission models have been suggested, including autosomal recessive (with a 25% risk of recurrence) and autosomal dominant with incomplete penetrance (with a risk of recurrence of up to 50%).
Sporadic cases can be explained by the occurrence of de novo mutations, and non-recurrent genomic rearrangements have been occasionally associated with PS.10,11
Recently, a case was reported where monozygotic twins with MS had a de novo mutation in chromosome 11q12.3. These twins exhibited a heterozygous deletion in genes responsible for cell growth, differentiation, and apoptosis, which the authors believe to be involved in the development of SP. The phenotypic similarity observed between the monozygotic twins emphasizes the role of this mutation in the genesis of PS. 10
In another case, a duplication in the region between 10p13 and 10p14 was identified in a child with congenital hyperinsulinism (CHI) and a PS phenotype. The authors suggested that this duplication, specifically involving UCMA (upper growth plate area and associated cartilage matrix), which regulates the differentiation of fetal cartilage and the cartilage-bone interface, could contribute to the PS phenotype.12,13
Furthermore, there was a report of another de novo duplication, spanning 1.5 Mb in Xp22.31, diagnosed in a child with left Poland syndrome along with multiple ocular, neurological, and renal malformations.
Over time, various variants and associated anomalies have been identified in individuals with Poland syndrome. 14 These include absence of the pectoralis minor or both pectoral muscles, scapula and muscle hypoplasia, rib hypoplasia, and occasionally rare manifestations such as dextrocardia, 3 renal agenesis, pneumothorax of the affected hemithorax, 15 shoulder dislocation, abnormal blood counts, thrombocytopenia, growth hormone deficiency, and facial nerve palsy. 7
The clinical forms of Poland syndrome are highly varied, and a classification based on increasing severity has been proposed 16 :
Stage I: Minor malformation with mild muscular hypoplasia of the pectoralis major muscle and moderate breast hypoplasia in females. The issue presented is a discreet asymmetry of the chest in males and breast asymmetry in females.
Stage II: Moderate malformation with significant aplasia of the pectoralis major muscle, marked breast asymmetry in females, and possible moderate rib malformation. The issue presented is a pronounced asymmetry of the chest.
Stage III: Severe malformation with complete aplasia of both the breast and pectoralis major muscle, along with additional associated muscular aplasia, major thoracic malformation with rib aplasia, and sternal deformity. The issue presented is a major asymmetry of the chest.
Imaging plays a crucial role in confirming the diagnosis of Poland syndrome and can aid surgeons in assessing the severity of muscle hypoplasia or absence, thus guiding the surgical approach. 7 In our study, we present 3 cases that meet the criteria for Poland syndrome. All patients underwent chest CT scans to confirm the clinical suspicion and provide a comprehensive assessment of associated malformations.
Among our patients, 2 were male and 1 was female. The syndrome was present on the left side in 2 patients and on the right side in the third. Two patients exhibited complete absence of the pectoralis major and minor muscles, along with hypoplasia of the ipsilateral mammary gland, without any additional malformations. The third patient showed hypoplasia of the pectoralis major and minor muscles, along with spinal abnormalities such as dorsal scoliosis and cervical vertebral malformations. This patient also displayed elevation of both scapulae and rib abnormalities, including fusion of the posterior costal arches of the 1st and 2nd left ribs. Furthermore, the patient had hypoplasia of the left lung, which led to frequent hospitalizations for respiratory issues, as well as agenesis of the right kidney.
From the existing literature, it can be concluded that Poland syndrome exhibits diverse clinical presentations. This can make it challenging for clinicians to diagnose the syndrome accurately and for plastic surgeons to categorize patients and determine the most appropriate surgical treatment for each individual. In 2011, a clinical and radiological classification of thoracic musculoskeletal anomalies associated with Poland syndrome was proposed after studying a series of 10 patients. 17
This classification consists of 4 categories:
1st degree: Hypoplasia of the pectoralis muscles.
2nd degree: Absence of the sternocostal head of the major pectoralis muscle.
3rd degree: Total absence of the major pectoralis muscle or both pectoralis muscles.
4th degree: Hypoplasia or absence of the pectoralis muscles combined with skeletal anomalies of the thoracic bones (sternum or rib cage).
The treatment of Poland syndrome varies depending on the extent of the malformation and the patient’s gender. Respiratory rehabilitation may be recommended for children with respiratory problems resulting from chest deformities. Surgery is rarely necessary but may be indicated in cases of paradoxical chest wall movement, breast hypoplasia or aplasia in females, or esthetic concerns in females with completed breast development. 18
Poland syndrome patients often grow into childhood and adulthood with relevant defects which can be addressed through reconstructive surgery. Some of the commonly used options to this day include custom-made silicone implants and/or flap based techniques which can improve the appearance of the chest wall in men, and the breast in women. 19
While surgical practices vary from surgeon to surgeon, indeed some use axillary incisions when harvesting the latissimus dorsi flap while others only use a dorsal one to harvest the flap, transposing it through a subcutaneous channel. 20
Typically, surgical reconstruction is based on utilizing the latissimus dorsi muscle and should be performed before the age of 13 in males who have intact latissimus dorsi muscles. 7 The latissimus dorsi muscle is usually harvested through a small incision in the back and sometimes in th axilla and then transposed to fill the void caused by the absence of the pectoralis major muscle. Prosthetic reconstructions have been described for males, but the results are generally unsatisfactory. 17
Conclusion
Poland syndrome is a rare congenital malformation involving thoracic and upper limb anomalies on the same side of the body. The diagnosis is typically made based on clinical suspicion, and imaging is important for confirming the diagnosis, assessing the extent of the malformation, and developing an appropriate treatment strategy. Treatment approaches should be tailored based on the patient’s sex, age, and the severity of the condition.
Footnotes
Acknowledgements
I would like to express my gratitude to my professors and all the colleagues who participated in the completion of this work.
Author Contributions
FC: Contributed to conception and design; Contributed to analysis; Drafted the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy. NB and RC: Contributed to conception and design; Contributed to analysis; Agrees to be accountable for all aspects of work ensuring integrity and accuracy. NMA: Critically revised the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy. FB, ZEA and TEM: Contributed to conception and design; Contributed to analysis; Agrees to be accountable for all aspects of work ensuring integrity and accuracy. NA and SEH: critically revised the manuscript; Gave final approval; Agrees to be accountable for all aspects of work ensuring integrity and accuracy. LC: Contributed to acquisition, analysis, or interpretation; Critically revised the manuscript; Gave final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the parents of patients for the publication of this case report.
Guarantor of Submission
The corresponding author is the guarantor of submission
