Abstract
Objective. The Children’s Hospitals in Africa Mapping Project survey was developed and implemented to assess the readiness of hospitals in sub-Saharan African to respond to emergencies, epidemics, and pandemics affecting children. Methods. This sub-study analyzed 56 survey questions that characterized the hospitals and assessed resources for diagnosis, treatment, and prevention of community-acquired infections and healthcare-associated infections (HAIs). Results. Twenty-four sites were recruited and 20 (from 15 countries) completed the survey in 2018 to 2019. Overall, 90% to 100% of sites reported that diagnostic assays were available for malaria, tuberculosis, and HIV. Periodic shortages of antibiotics, antimalarial drugs, and vaccines were reported by 45%, 25%, and 20% of sites, respectively. Sites reported formal programs for infection prevention and control (79%), monitoring HAIs (17%), and antimicrobial stewardship (53%). Conclusions. Opportunities to improve care for hospitalized African children were identified. These included resources for HAI surveillance and antimicrobial stewardship which can facilitate epidemic and pandemic preparedness.
Keywords
Introduction
In efforts to reduce morbidity and mortality from public health threats, the Joint External Evaluation (JEE) tool was developed and adopted by the WHO in 2016 to help countries assess their level of preparedness to unforeseen public health threats including emerging infectious diseases, epidemics, and pandemics. 1 However, issues specially related to children’s health were not included in the JEE evaluation tool. 1 Thus, we developed and implemented the Children’s Hospitals in Africa Mapping Project (CHAMP) survey for sub-Saharan African hospitals serving children.1,2 The survey goal was to assess the capacity and infrastructure of hospitals to gauge their readiness to deal with public health emergencies and disasters including epidemics and pandemics affecting children.
The survey sought to assess available resources for the diagnosis, treatment, and prevention of community-acquired infections as well as the prevention, surveillance, and management of healthcare-associated in-fections (HAIs).3 -5 Further knowledge of these factors could contribute to strategies to reduce morbidity and mortality in pediatric populations. We also hoped that individual hospitals would use their survey responses to assess their own preparedness and, if appropriate, advocate for support to address the gaps identified.
The aims of this sub-study were to analyze the CHAMP survey responses to questions that assessed the diagnosis, treatment, and prevention of infectious diseases in children in efforts to better understand existing resources and identify opportunities to improve care.
Methods
Study Sites, Site Investigators, and Ethics Review
We sought to recruit hospitals located in sub-Saharan Africa with the expertise to care for substantial numbers of children. Specifically, we sought hospitals that caredexclusively for children or had ward(s) dedicated to the care of neonates, infants, and children. Participating hospitals provided a broad array of pediatric services delivered by health care professionals with formal pediatric training. Potentially eligible hospitals were identified by African leaders, who were heads of pediatric departments or had leadership roles in African pediatric professional societies, and the ELMA Foundation (which funded the study).
English-speaking site investigators who were heads of their departments were contacted by the principal investigators. Those who agreed to participate contacted their local ethics committees to determine if review and signed consent were needed as per local and national guidance. None of the ethics committees deemed this study to be human subject research. Five sites required a formal ethics review. None of the participating sites required written informed consent from site investigators. No patients were included or recruited to participate at any site in this study.
The Columbia University Irving Medical Center (CUIMC) institutional review board (IRB) deemed that this study did not meet the criteria for human subjects research and was exempt from further CUIMC IRB review.
CHAMP Survey Development and Implementation
Investigators from the United States and Africa developed the CHAMP survey. The survey included yes/no questions, follow-up questions to further characterize yes responses, multiple choice questions, and free text. Responses were not forced. The final survey consisted of 958 questions assessing available resources and infrastructure such as bed-size, laboratory tests, clinical staff, staff training, waste removal, hospital-based programs, and committees.
Survey responses were entered by each site’s investigator or their designee into a password-protected REDCap database managed by investigators at CUIMC. Site investigators could ask other administrators at their hospital for assistance answering survey questions, for example, asking the director of clinical laboratories about available molecular diagnostic tests or components of the antimicrobial stewardship program (ASP).
Sub-study Survey Questions
In this sub-study, we extracted questions and corresponding responses for 56 questions related to diagnosis, treatment, and prevention of community-acquired infections and HAIs (Supplemental Table 1). These questions included availability of specific diagnostic tests within the hospital, including the opportunity to add tests not listed on the survey, and their source of payment (eg, government or family), availability of radiographic studies, pharmacy resources, potential shortages of antimicrobial agents and vaccines, as well as availability and characteristics of programs, policies, and procedures for infection prevention and control (IP&C), HAI surveillance, and ASP.
Statistical Analysis
The percentages of positive responses for each survey question were calculated. As responses were not forced, not all sites responded to every survey question. Thus, the percentage of positive responses was determined for each question using the number of overall responses for that question. Microsoft Excel was used for quantitative data analysis. We calculated means, frequencies, ranges and median when applicable. Optional free text res-ponses further describing IP&C, HAI, and ASP activities were extracted and reviewed to further understand hospitals’ perspectives on these activities and potentially identify additional challenges.
Results
Study Sites
Twenty-four hospitals were recruited from September 2018 through June 2019 to participate in the CHAMP study of which 20 (from 15 countries) completed the survey from December 2018 through November 2019 (Figure 1). The average daily census at these 20 hospitals was 116 children (standard deviation: 89). Additional characteristics of the hospitals, including the types of hospitals, are shown in Table 1.

Locations of participating sub-Saharan African hospitals caring for children. Hospitals were located in the following countries: Cameroon, Ethiopia, Ghana, Kenya, Lesotho, Liberia, Malawi, Nigeria, Rwanda, Sierra Leone, South Africa, Tanzania, Uganda, Zambia, and Zimbabwe. Two hospital sites in Ethiopia, Kenya, Liberia, Nigeria and Zambia; one hospital site in the other listed locations. Figure created with mapchart.net.
Characteristics of Participating Hospitals.

Abbreviation: IQR, interquartile range.
n = positive responses and N = number of hospitals responding to survey question.
Diagnostic Testing
Microscopic tests for malaria and tuberculosis (TB) were available at 95% (19/20) and 90% (18/20) of hospitals, respectively (Table 2). Of the 10 hospitals that performed mycobacterial cultures, all performed anti-mycobacterial susceptibility testing. Cultures of body fluids and stool were available in 89% (17/19) and 84% (16/19) of hospitals, respectively. Of the 17 hospitals that performed body fluid cultures, all performed Gram stains and antibiotic susceptibility testing. Governments funded 59% (10/17) to 83% (15/18) of selected diagnostic assays while patients’ families funded 22% (4/18) to 71% (12/17) (Figure 2).
Diagnostic Assays for Infectious Diseases Available at Sub-Saharan African Hospitals Caring for Children.

Abbreviations: HRP2, histidine-rich protein 2; pLDH, parasite lactate dehydrogenase; PCR, polymerase chain reaction.
n = positive responses and N = number of hospitals responding to survey question.
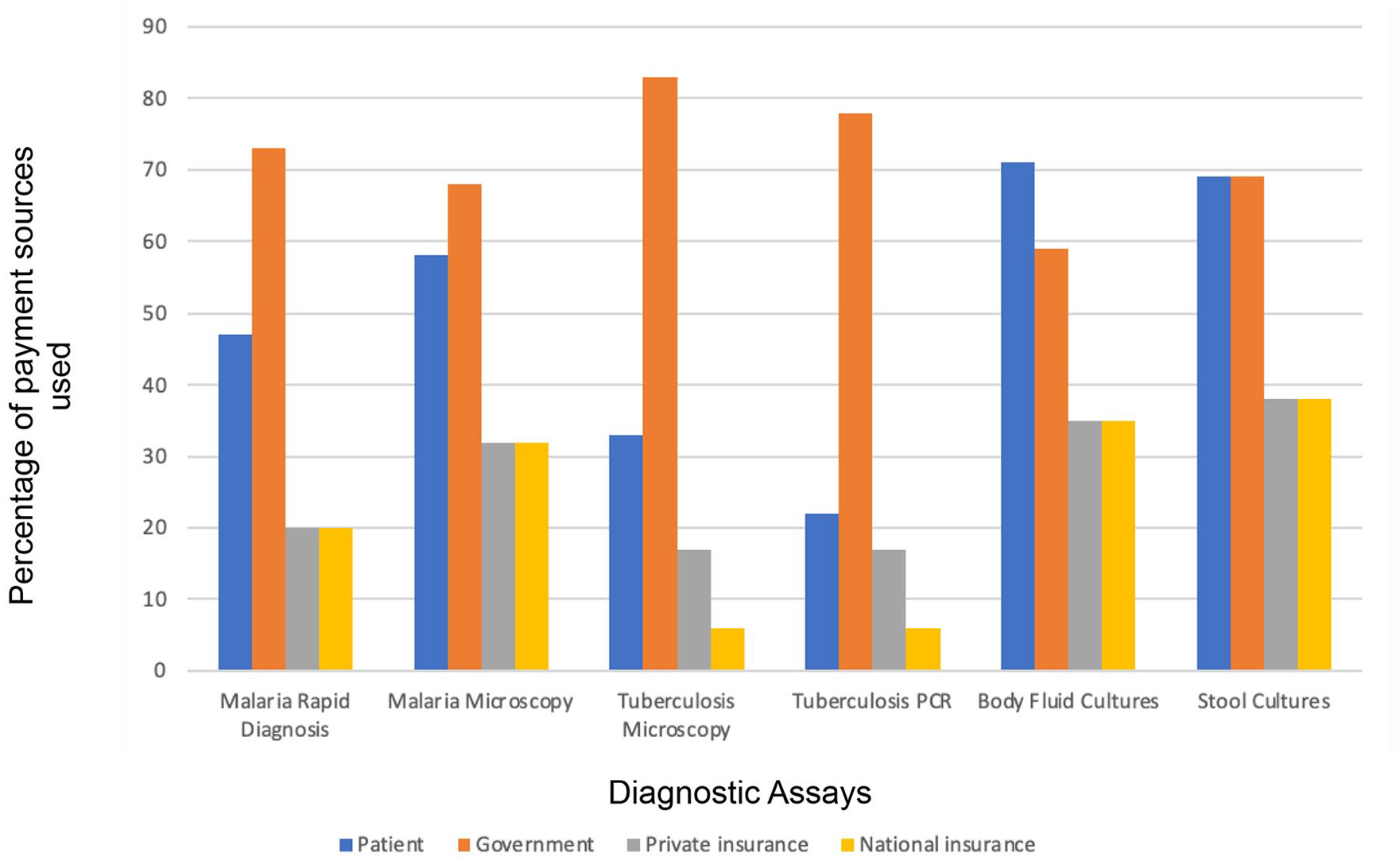
Payment sources for selected diagnostic tests used in sub-Saharan African hospitals caring for children. Selected diagnostic tests are provided on the X axis. The percentage of 4 potential payment sources for each test is shown on the Y axis and calculated as the number of respondents indicating that source of payment was used divided by the number of respondents (range 15-19) who answered the survey item. Respondents could indicate more than one payment source for a diagnostic test.
HIV serology was available at 100% (17/17) of responding hospitals and HIV1 qualitative DNA PCR testing was available at 63% (12/19) (Table 2). A minority reported availability of molecular diagnostic studies for hepatitis B or C, cytomegalovirus (CMV), varicella zoster virus (VZV), or herpes simplex virus (HSV). Three hospitals (16%, 3/19) reported availability of meningitis/encephalitis PCR panels, one had a tropical fever panel, and one had a human papillomavirus assay. None had PCR panels for gastrointestinal (0/19) or respiratory (0/19) pathogens.
All 20 hospitals had ultrasounds and x-rays while computed tomography (CT) scans and magnetic resonance imaging scans were available in 70% (14/20) and 50% (10/20) of hospitals, respectively.
Pharmacy Resources and Antimicrobial and Vaccine Shortages
Overall, 95% (19/20) had a dispensing pharmacy and 55% (11/20) had a compounding pharmacy (Table 3). Shortages of commonly prescribed antibiotics and anti-malarial medications in the past year were reported by 45% (9/20) and 25% (5/20) of hospitals, respectively. Twenty percent (4/20) experienced shortages in recommended pediatric vaccines.
Pharmacy Resources and Antimicrobial and Vaccine Shortages at Sub-Saharan Africa Hospitals Caring for Children.

n = positive responses and N = number of hospitals responding to survey question.
Infection Prevention and Control and Antimicrobial Stewardship Programs
Formal IP&C programs were available in 79% (15/19) of hospitals and 84% (16/19) had written IP&C policies and procedures (Table 4). Among the 19 hospitals that responded to this question, doctors and nurses were part of the IP&C team at 68% (13/19) and 79% (15/19) of hospitals, respectively. Some hospitals also had pharmacists, microbiologists and representatives from the department of health on their IP&C teams. Overall, 84% (16/19) of responding hospitals reported hand hygiene training programs and of those hospitals, 75% (12/16) had formal training annually. In all, 50% of hospitals experienced shortages in sterile gloves (10/20) and 58% (11/19) had water supply interruptions which occurred daily (45%, 5/11), weekly (18%, 2/11), monthly (9%, 1/11), or infrequently (18%, 2/11); one hospital did not provide the frequency of water supply interruptions.
Infection Prevention and Control and Antimicrobial Stewardship Programs, Policies, and Practices at Sub-Saharan Africa Hospitals Caring for Children.

n = positive response for each item and N = number of hospitals responding to each survey item.
Of the 12 responding hospitals, 17% (2/12) had a formal program to monitor HAIs. Several had systems to identify patients with multidrug-resistant organisms (MDROs) upon readmission (47%, 9/19) and to identify the infectious status and isolation needs of patients prior to accepting them from other facilities (50%, 9/18) or transferring them to other facilities (75%, 12/16). Fifty-three percent of hospitals (10/19) had formal ASP, 58% (11/19) had guidelines for antibiotic use, and 63% (12/19) monitored antibiotic use (Table 4).
In addition, all 20 hospitals screened blood products for HIV (20/20) and hepatitis B (20/20), 95% (19/20) screened for hepatitis C, 70% (14/20) screened for syphilis, 20% (4/20) screened for CMV, and 10% (2/20) screened for human T-lymphotropic virus (HTLV). Two hospitals also screened blood products for malaria.
Twelve hospitals provided free text responses further describing their IP&C programs, HAI surveillance, and ASP (Supplemental Table 2). These responses described challenges such as poor integration with other programs and lack of uniform and consistent training for hand hygiene and use of personal protective equipment (PPE). Notably, some site investigators cited their hospital’s unique response to Ebola which was not sustained nor applied to other ongoing IP&C needs.
Discussion
This multicenter survey assessed the diagnosis, treatment, and prevention resources for community-acquired infections and HAIs in hospitals serving children in sub-Saharan Africa. Consistent with high rates of malaria, TB, and HIV in African children and evidence-based treatment, almost all hospitals had diagnostic tests for these infections. The Global Fund, in conjunction with the United Nations Foundation, allocates millions of dollars yearly to combat malaria, TB, and HIV/AIDS in sub-Saharan Africa.6,7 Furthermore, governmental funding, as confirmed by site investigators, is available for diagnosing these community-acquired infections. However, hospitals reported relatively less availability of the HIV 1 qualitative DNA PCR test as this test was only available at 63% of hospitals. Since this assay is used to detect early HIV infections in neonates, lack of its availability can delay diagnosis of perinatal transmission. 8
Hospitals had fewer diagnostic assays available for other viral infections including hepatitis B, hepatitis C, HSV, CMV, and respiratory viruses. Improved diagnostic testing for viruses could further support antimicrobial stewardship efforts as accurate and timely diagnosis of respiratory viruses has decreased antibiotic use in both pediatric and adult patient populations.9,10
Hospitals reported potential challenges to treating infections. Nearly half reported periodic shortages of commonly prescribed antibacterial agents, which could lead to use of more broad-spectrum antibacterial agents, which in turn increases the risk of resistance as previously described.11,12 While nearly all hospitals had a dispensing pharmacy, fewer had compounding pharmacies which can provide antibiotic formulations not commercially available.13,14 Compounding pharmacies are able to prepare liquid formulations of medications for children which optimize dosing and palatability, but they require resources and oversight, which make implementation challenging. 15
Recognition of the importance of HAIs in children is increasing in sub-Saharan Africa,16,17 as confirmed by the survey results. Most hospitals reported formal IP&C programs, including programs for hand hygiene. For decades, the WHO has promoted evidence-based practices for hand hygiene in efforts to reduce HAIs18,19 and these guidelines have been disseminated in sub-Saharan Africa. 20 However, implementation challenges to hand hygiene programs have been described.21,22 Similar challenges were noted by hospitals and in-cluded inadequate monitoring of hand hygiene practices, limited compliance, inadequate staff education, lack of hand hygiene supplies, and water supply in-terruptions. 22
Hospitals also noted opportunities to enhance the effectiveness of their IP&C programs. For example, some hospitals lacked competency-based programs for donning and doffing PPE and several lacked annual PPE training for staff; these training strategies are thought to be integral in reducing HAIs 23 and crucial to protect staff during epidemics and pandemics. Furthermore, free-text responses indicated that better integration, consistent leadership, and administrative buy-in were needed to improve their programs; WHO considers these factors to be core components for IP&C programs worldwide. 24 Not all hospitals had systems to identify patients with MDROs nor systems to communicate the infectious status and isolation needs of their patients. Such communication can reduce the transmission of MDROs in hospitalized patients, including children, by facilitating isolation precautions.25,26
Few hospitals reported that they monitored HAIs which is a key component of HAI reduction strategies worldwide.26 -28 Large-scale HAI surveillance programs in the U.S., supported by the Centers for Disease Control and Prevention, are often mandated by the Centers for Medicare and Medicaid Services and/or state health departments. These programs have generally been associated with reductions in HAIs but increases occurred during the COVID-19 pandemic likely due to increased hospitalizations, crowding, shortages in equipment, staff shortages and/or deployment of staff unfamiliar with local IP&C policies and procedures.29,30 Studies in Africa have also endorsed the importance of HAI surveillance, but noted the challenges implementing surveillance systems.31 -33 Hospitals noted another potential challenge to MDROs surveillance; families at participating hospitals bore most of the cost burden for performing body fluid and stool cultures which likely results in decreased identification of the burden of specific pathogens as well as susceptibility testing. Notably, the WHO Global An-timicrobial Resistance and Use Surveillance System (GLASS) has been working to improve surveillance of antimicrobial resistance worldwide. 33
Half of the hospitals reported the presence of an antimicrobial stewardship program, guidelines for antimicrobial use, and/or monitored antimicrobial use. How-ever, as few hospitals monitored HAIs, including those caused by MDROs, successful antimicrobial stewardship efforts could be impeded. Furthermore, reduced clinical microbiology resources may jeopardize patient treatment, accurate surveillance for antimicrobial resistance, and antimicrobial stewardship. Sustained antimicrobial susceptibility testing, based on existing training and accreditation resources, has been successfully implemented in Ethiopia. 34
This study had limitations. The findings may not be generalizable because only hospitals serving large number of children with English-speaking investigators were recruited. The survey was not assessed for reliability and validity. There were missing responses as the survey did not utilize a forced response format. We did not inquire about the types of vaccines for which shortages existed nor the definition of MDROs. As we did not receive written IP&C and ASP policies from the hospitals, we could not assess specific policies. Finally, survey responses may not reflect actual practices nor consistent availability of specific re-sources.
Conclusions
In conclusion, hospitals reported substantial resources for the diagnosis, treatment, and prevention of infectious diseases for hospitalized children in sub-Saharan Africa. Nearly all hospitals had diagnostic assays for malaria, TB, and HIV and had dispensing pharmacies. The majority had IP&C programs and hand hygiene training. In addition, survey results provided insights into potential areas for improvement. These included enhanced diagnostic testing for viruses, establishing annual competencies for hand hygiene and PPE use, improved HAI surveillance, and resources to monitor antibiotic use and antimicrobial resistance. This sub-study provided a unique needs assessment of resources for managing infectious diseases, including those causing epidemics and pandemics, and could assist other hospitals assess their resources. Follow-up studies
Supplemental Material
sj-docx-1-gph-10.1177_2333794X231210661 – Supplemental material for Diagnosis, Treatment, and Prevention Resources for Infectious Diseases in Sub-Saharan African Hospitals Caring for Children
Supplemental material, sj-docx-1-gph-10.1177_2333794X231210661 for Diagnosis, Treatment, and Prevention Resources for Infectious Diseases in Sub-Saharan African Hospitals Caring for Children by Samiksha Tarun, Workeabeba Abebe, Maitry Mahida, Lisa Saiman, Wilmot James, Philip LaRussa and Lawrence R. Stanberry in Global Pediatric Health
Footnotes
Acknowledgements
The authors gratefully acknowledge the Children’s Hospital of Africa Mapping Project (CHAMP) and the CHAMP Leadership Group. Members of the CHAMP Leadership Group include: Dr. One Bayani, Princess Marina Hospital, Botswana; Dr. Nellie V. T. Bell, Ola During Children’s Hospital, Sierra Leone; Dr. Heloise Buys, Red Cross Children’s Hospital, South Africa; Dr. Joseph Tawanda Chava, Harare Children’s Hospital, Zimbabwe; Dr. Ashraf Coovadia, University of the Witwatersrand, South Africa; Dr. Joel Dipesalema, Princess Marina Hospital, Botswana; Dr Eki-Udoko Fidelis Ewenitie, University of Benin Teaching Hospital, Nigeria; Dr Aimable Kanyamuhunga, University Teaching Hospital of Kigali, Rwanda; Professor IkeOluwa Laganju, University College Hospital Ibadan, Nigeria; Dr. Irene Marete, Moi Teaching and Referral Hospital/Shoe4Africa Children’s Hospital, Kenya; Dr. Sam Miti, Arthur Davison Children’s Hospital, Zambia; Dr. Thembisile Mosalakatane, Princess Marina Hospital, Bo-tswana; Dr. Mildred Anyango Mudany, Pediatrician/Country Director—Jhpiego, Kenya; Dr. Hilda Mujuru, Harare Children’s Hospital, Zimbabwe; Dr. Ezekiel Mupere, Mulago National Referral Hospital, Uganda; Dr. David Musorowegomo, Harare Children’s Hospital, Zimbabwe; Dr Jonathan Kaunda Mwansa, Arthur Davison Children’s Hospital, Zambia; Dr. Ruth Nduati, University of Nairobi, Kenya; Dr. Nancy Biyeah Yang Ngum, New Partnership for Africa Development, South Africa; Dr. Crispen Ngwenya, Parirenyatwa group of hospitals, Zimbabwe; Prof. Augustine Omoigberale, President of Paediatric Association of Nigeria, Nigeria; Dr. Atnafu Mekonnen Tekleab, St Paul Hospital, Ethiopia; Prof Pauline Samia, Aga Khan University Hospital, Kenya.
Authors’ Note
Previous presentation: Part of this work was presented as a poster at Global Health Security, June 18-20, 2019, Sydney, Australia
Author Contributions
Samiksha Tarun contributed to conception or design, contributed to acquisition, analysis, or interpretation, Drafted the manuscript, critically revised the manuscript and Gave final approval. Workeabeba Abebe contributed to acquisition, analysis, or interpretation, critically revised the manuscript and gave final approval. Maitry Mahida contributed to acquisition, analysis, or interpretation, Drafted the manuscript, critically revised the manuscript and gave final approval. Lisa Saiman contributed to conception or design, contributed to acquisition, analysis, or interpretation, Drafted the manuscript, critically revised the manuscript and Gave final approval. Wilmot James contributed to acquisition, analysis, or interpretation, critically revised the manuscript and gave final approval. Philip LaRussa ontributed to acquisition, analysis, or interpretation, critically revised the manuscript and gave final approval. Lawrence R Stanberry contributed to conception or design, contributed to acquisition, analysis, or interpretation, critically revised the manuscript and Gave final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the EMLA Foundation (institutional grant to Philip LaRussa, Wilmot James, and Lawrence Stanberry) which had no role in data collection or analysis. Samiksha Tarun is supported by the training grant “Training in Pediatric Infectious Diseases” (National Institute of Allergy and Infectious Diseases T32AI007531 institutional grant, PI: Lisa Saiman).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
