Abstract
Objectives
We aimed to evaluate the use of intravenous levetiracetam as the first-line treatment of neonatal seizures compared with phenobarbital.
Methods
The study was conducted on 104 neonates (0-28 days) with clinical seizures after inclusion criteria. They were assigned in equal ratio into 2 groups; 1 included neonates who received phenobarbitone, and the other included neonates who received levetiracetam. Neonates were loaded with 20 mg/kg of intravenous drug-A (phenobarbitone) or drug-B (levetiracetam). In persistent seizures, a second loading dose of the same drug was given. Crossover to other drugs occurred if seizures persisted after the second dose of the same drug. The proportion of neonates who achieved cessation of seizures following the first or second loading dose of either drug-A or drug-B (PB or LEV) was the main outcome measure provided that they remained free of seizure for the following 24 hours.
Results
After 1 or 2 doses of Levetiracatam or Phenobarbitone, clinical seizures stopped (and remained seizure-free for 24 hours) in 41 (78.84%) and 34 (65.38%) patients, respectively (P = .01). Neonates in the LEV group showed better seizure control than neonates in the PB group (RR = 0.57; 95% CI (0.17, 0.80). We did not report any adverse drug reactions in the LEV group. However, 12 (23.07%) neonates developed adverse drug reactions in the PB Group.
Conclusion
Levetiracetam is considered an effective and safe drug as a first-line AED in neonatal seizures.
Introduction
Neonatal seizures are common presentation of brain insult with a diagnostic and therapeutic challenge in neonatal care settings. 1 Neonatal seizures affect 1.5 to 5 per 1000 live births in term infants and 57.5 to 132 per 1000 live births in premature infants. 2 Prolonged seizures are associated with a higher rate of neonatal morbidity and mortality. 3 Rapid identification and proper management of neonatal seizures are essential in reducing negative impacts on the developing brain. 4 Among different etiologies, hypoxic-ischemic encephalopathy (HIE) is considered the most common cause of symptomatic neonatal seizures, which affects 1 to 2/100 live births.5,6 Sometimes neonatal seizures do not pose a definitive clinical presentation and are under-or overestimated. 7 Continuous electroencephalography (cEEG) is the gold standard test for the diagnosis and classification of seizures in neonates. However, its scarcity limit its use in many facilities.3,4 Phenobarbital (PB) is well known anti-epileptic drug (AED) in neonatal seizure and is applied as a first-line treatment. 8 It can control neonatal seizures and decrease the metabolic rate of brain cells. PB can control electrical seizures with abnormal EEG in 43% to 80% of cases. 9 The combined use of phenobarbital and phenytoin can control clinical neonatal seizures in 50% to 70% of newborns. 9 The short-term side effects of PB include lowering of blood pressure and depression of respiratory functions. However, impairment of cognitive functions can develop later in infants and children. Also, it was found that PB has a significant role in accelerating neuronal apoptosis in the immature brain. 8 Levetiracetam (LEV) has better efficacy and safety profile compared with other anticonvulsant medications, as no neuronal apoptosis was reported in animal studies. In addition, LEV may have neuroprotective effects on the developing brain.10-13 The recent availability of an intravenous (IV) preparation of LEV has led to its abundant use in the management of neonatal seizures. 14 Recently, it was reported that the use of LEV in neonatal seizures achieved seizure cessation in 77% of neonates compared to 46% in the PB group.3,14,15
There is evidence that LEV may be an alternative to PB in neonatal seizures. 15 However, most previous studies examined LEV as a second-line antiepileptic drug. 15 The use of levetiracetam in neonatal seizures has limited studies with a few number of randomized controlled trials.3,15 The evidence of using LEV as a first line therapy in neonatal seizures is still inconclusive. 15
In this study, we aimed to compare the efficacy and safety profile of LEV compared to PB in the treatment of neonatal seizures.
Patients and Methods
This prospective, double-blind randomized controlled trial was carried out in the neonatal intensive care unit (NICU) level III of a tertiary-care center in Benha University Hospital, Benha, Egypt, over a period of 2 years (March 2020-March 2022). An independent research board (IRB) of Faculty of medicine, Benha University, Benha, Egypt reviewed and approved the study protocol. We followed all ethical guidelines in the 1964 Declaration of Helsinki and its later amendments.
Formal consent was obtained from the parents as soon as possible after being assessed for eligibility. We included 104 neonates (aged 0-28 days) with clinical seizures.
Clinical neonatal seizure was identified as sudden abnormal, stereotyped, and paroxysmal brain dysfunction within the first 28 days of life in full-term infants or before 48 weeks of gestation in preterm infants. 16 Neonatal seizures attributed to electrolyte disturbances (eg, hypoglycemia, correctable hypocalcemia, and hypomagnesemia), inborn errors of metabolism (eg, non-ketotic hyperglycinemia, pyridoxine deficiency), or opioid withdrawal syndrome were excluded because these seizures responded to the correction of the underlying abnormality. Also, neonates who received anticonvulsant drugs prior to the study were excluded.
All neonates enrolled in the study were subjected to history taking, clinical examination, type of seizure, and antiepileptic drug (type, sequence, dose, timing, and duration).
The detection of clinical seizures was dependent on many factors. Parental report of abnormal movements of their babies before assessment for enrolment into the study. There was a continuous qualified nurse at bedside watching the neonates for any abnormal movements, Also, there were neonatal ICU physicians intermittently clinically monitoring the neonates. The EEG recordings were performed bedside in the NICU; 10 or 21 cerebral electrodes, depending on the size of the head, were applied according to the 10-20 International System. The EEG recording was routinely performed for at least 60 min within 1-2 h from the clinical episode and reported by an experienced pediatric neurologist.
Laboratory investigations included random blood sugar, serum electrolytes (calcium, sodium, and magnesium), complete blood count, C-reactive protein (CRP), renal and liver function tests, arterial blood gas analysis, serum ammonia, blood lactate, metabolic screening, and genetic testing.
Radiological investigations included cranial ultrasonography, brain imaging, and electroencephalography (EEG).
Blood sugar and serum calcium were measured after initial stabilization and management of newborns to ensure patent airway, adequate breathing, and circulation. Neonates with persistent seizures after correction of hypoglycemia and hypocalcemia were assigned into 2 groups in a random fashion.
Sealed opaque envelopes with sequential numbers were used for allocation. Neonates with seizures were randomly allocated in a double-blind fashion in a 1:1 ratio into group-A or group-B via a table of random numbers.
Group A
Included neonates with seizures who received the standard drug therapy phenobarbitone (ampoules in the composition of 200 mg/ml or syrup in the composition of 30 mg/5 ml).
Group B
Included neonates who had seizures and were given the experimental drug Levetiracetam (100 mg/ml ampoules or 100 mg/ml syrup).
For any eligible neonate to be enrolled in the study, the envelope was opened by an investigator who was not involved in the study. The neonates were randomized and loaded with 20 mg/kg of intravenous drug-A (phenobarbitone) or drug-B (Levetiracetam) according to their respective allocations. Medications were given by a member of the nursing staff under cardiorespiratory monitoring. The response to medications was monitored by the principal investigator.
Response to the drug was defined as the clinical cessation of seizures within 20 minutes of the intravenous drug therapy. If seizures continued after 20 minutes from the initiation of the intravenous drug therapy, another loading dose of 10 mg/kg of the same drug was given with observation for another 20 minutes. Also, the clinical cessation of seizures as observed during this additional period was considered as a response to the drug.
Treatment failure was considered if the seizure did not stop within the total time period of 40 minutes whenever crossing over with the other drug within the study was done in the same sequence.
Intravenous phenytoin (20 mg/kg diluted in 20 ml of normal saline solution given over 20 minutes) was initiated in cases of treatment failure after administration of drug-A and drug-B.
All cases with treatment failure were managed with intravenous phenytoin initially and then shifted gradually to a maintenance dose of 5 mg/kg/day.
In Group-B
If seizures terminated, LEV was continued as maintenance therapy. If seizures continued, another loading dose of LEV was given, and if seizures still persisted, the patient was switched over to receiving PB.
In Group-A
If seizures were terminated, PB was continued as maintenance therapy. If seizures continued, another loading dose of PB was administered, and if seizures still persisted after 2 loading doses, the patient was switched over to receiving LEV.
The maintenance dose of Levetiracetam was 20 mg/kg/day in 2 equal doses, and that for Phenobarbitone was 5 mg/kg/day given once daily. Initially, these drugs were given intravenously and then shifted to an oral route.
The main primary outcome measure was the proportion of neonates who achieved cessation of seizures following the first or second loading dose of either drug-A or drug-B (PB or LEV) and remained seizure-free for the following 24 hours. However, the secondary outcome measure was the proportion of neonates who experienced adverse events.
Clinical termination of seizure was detected if there were no abnormal movements, eyeball deviation, or nystagmus, and no change in cardiorespiratory state or autonomic function.
We recorded the adverse effects occurring within 2 hours of drug administration. It included hypotension, depression in respiratory functions, abnormalities in heart rate, poor feeding, irritability, increased requirement for ventilator support, fluctuations in heart rate, or blood pressure by more than 10% compared to the previous 2 hours. However, hypotension and respiratory depression were the most common adverse drug reactions.
Calculation of Sample Size
The sample size was calculated using G*power software version 3.1.9.2 based on an expected efficacy of 86% and 62% for LEV and PB, respectively. The minimum sample size needed to detect such an effect size is 104 patients (52 per group). 17 Alpha and power were adjusted at .05 and 0.8, respectively.
Statistical analyses
The analysis of data and variables was done using the Statistical Package for Social Sciences (SPSS), version 16.0 (Chicago, IL, USA). Comparison between the 2 groups regarding continuous variables was done using an independent samples t-test. However, termination of seizures and the development of adverse events were compared using the Chi-square test. The effect size and 95% CI were computed for the primary and secondary outcomes. P-value of less than .05 was considered significant.
Results
One hundred and thirty six neonates with clinical seizures were checked for eligibility for inclusion in our study. Thirty-two neonates did not meet the criteria and were excluded. One hundred and four neonates were randomized equally into 2 groups (Figure 1).

Flow of studied groups in the study.
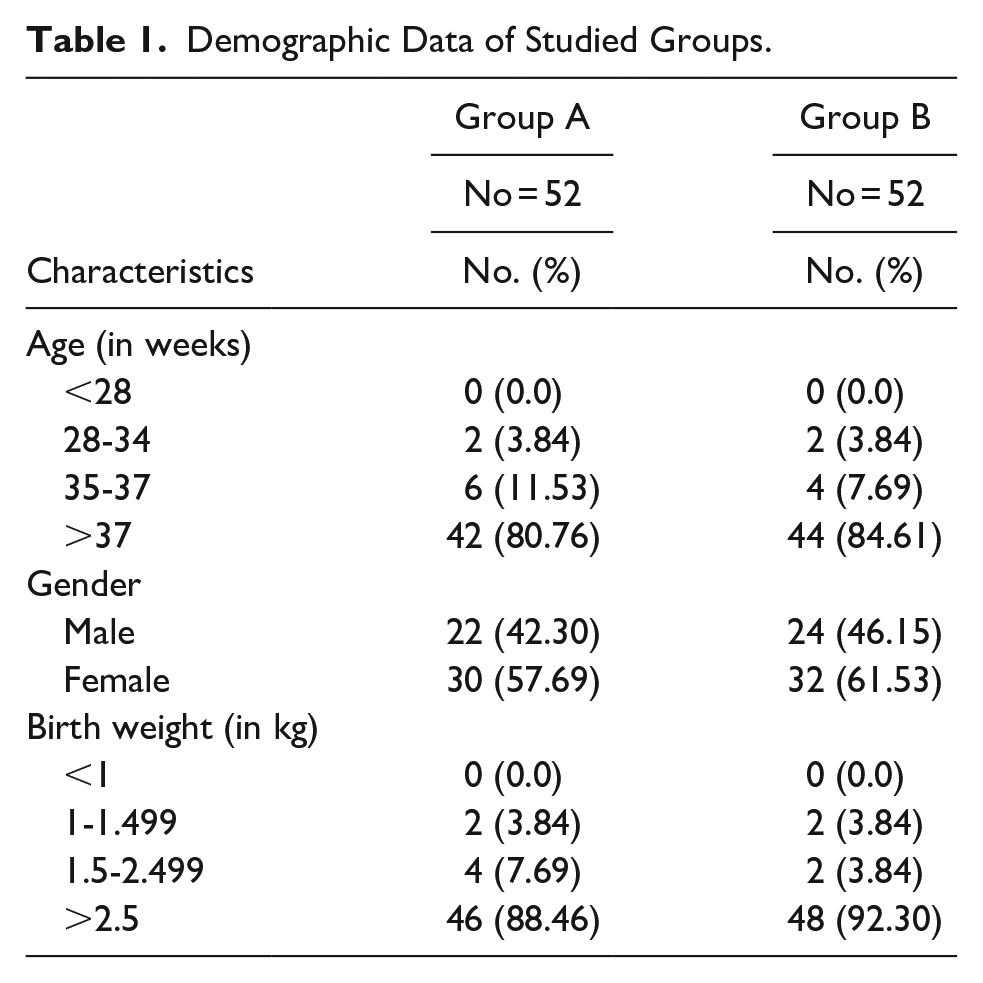
The study groups included 52 neonates assigned to Group A (PB) and 52 neonates assigned to Group B (LEV). No significant differences were found between the 2 groups regarding demographic data and clinical characteristics. In both groups, the majority of neonates were >37 weeks old. Most babies weighed over 2.5 kg (88.46% and 90.30.5% in Group A and Group B, respectively) (Table 1).
Demographic Data of Studied Groups.
Prematurity was reported at (19.23%) and (15.38%) in Group A and Group B, respectively. Gestational diabetes mellitus was the most important risk factor in Group A (19.23%), while hypertension was the main factor in Group B (15.38%). The association was statistically significant (P = .05).
Neonates were delivered via cesarean section in Group A (25.00%) and Group B (23.08%), respectively. Perinatal asphyxia was reported in 28.84% and 32.69 % in Group A and Group B, respectively. This difference was statistically significant (P = .05; Table 2).
Risk Factors of Neonatal Seizure.
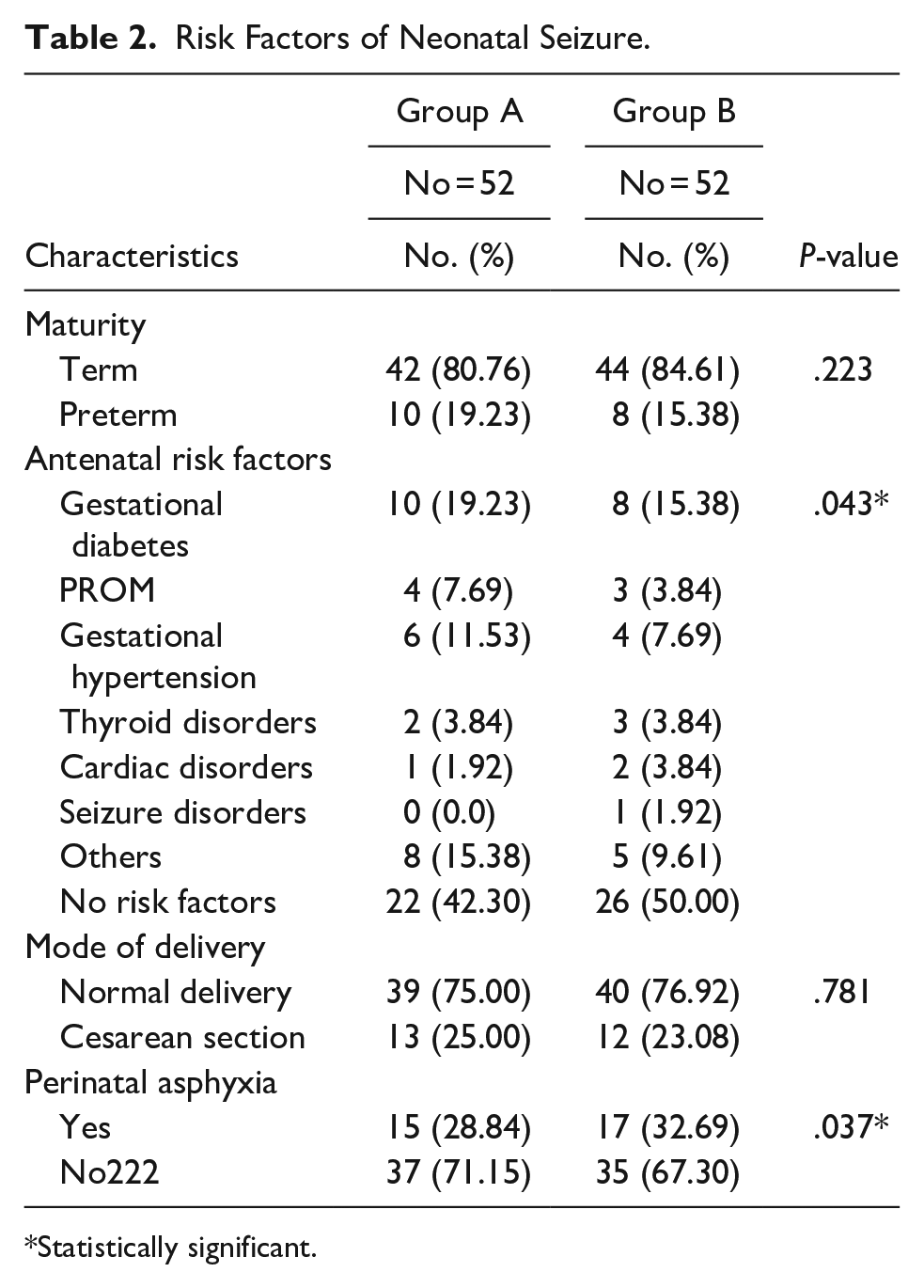
Statistically significant.
In all participants, the most common cause of seizures was hypdoxic-ischemic encephalopathy (HIE). Meanwhile, tonic seizures were the most common type of seizure, followed by clonic seizures (Table 3). After the first dose of medication, seizure cessation was reported in 35(67.30) and 24 (46.15) neonates in the LEV group and PB group, respectively.
Characteristics of Neonatal Seizure Among Studied Groups.
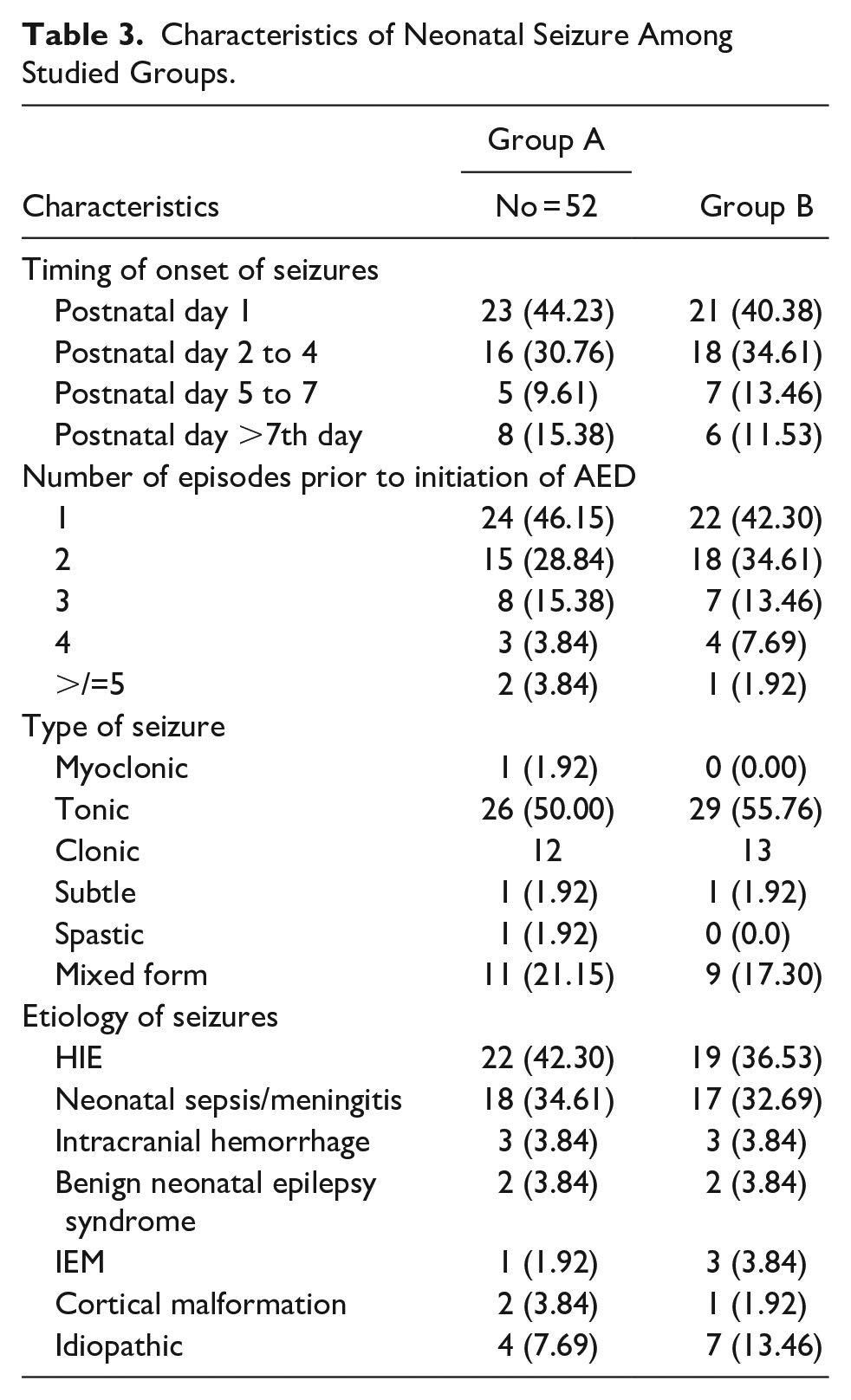
In the LEV group, clinical seizures stopped (and remained free of seizure at 24 hours) in 41 (78.84%), and in the PB group, it was 34 (65.38%) after 1 or 2 doses (P = .01).
Neonates in the LEV group showed better seizure control than neonates in the PB group (RR = 0.53; 95% CI (0.13, 0.80). Neonates who received LEV did not develop any adverse drug reactions. However, 12 (23.07%) neonates developed adverse drug reactions in the PB Group. Adverse events found in the PB group were decrease in blood pressure in 7 neonates, decrease in heart rate in 2 neonates, requirement of mechanical ventilation in 2 neonates, and change of ventilator setting in 1 neonate.
Discussion
Management of neonatal seizures depends mainly on the use of AEDs. According to the guidelines of World Health Organization (WHO), PB is the first-line treatment of neonatal seizures. PB can control neonatal seizures and reduce the metabolism of brain cells.6,18 However, PB has many adverse effects, such as lowering of blood pressure and depression of respiratory functions.19,20 As reported in previous studies, PB may also cause damage of the developing brain, enhance neuronal apoptosis, and cause cognitive impairment. So, its safety and impact on neurodevelopment require further evaluation. In general, LEV is more advantageous than PB regarding side effects and later neurodevelopmental harm. 14 Therefore, the use of LEV becomes abundant in the setting of neonatal intensive care units. However, most studies in neonatal medicine examine it as a second-line agent. 21 In our study, we aimed to compare the efficacy and adverse effects of LEV and PB as first-line treatments of neonatal seizures.
In our study, we found better efficacy and safety of LEV compared to PB as a first-line AED in the treatment of neonatal seizures. Neonates in the LEV group showed a higher percentage of seizure cessation compared to neonates in the PB group. In the current study, neonates who received LEV did not show any adverse drug reactions.
Among 52 neonates in the PB group, 12 neonates developed adverse drug reactions.
Ramantani et al 22 conducted their study on 38 cases with neonatal seizures after exclusion of electrolyte disorders and inborn errors of metabolism (IEM). LEV was used as a first-line AED and was given intravenously. They reported that LEV is more effective than PB, without any reported side effects. Khan et al 23 studied 22 neonates with seizures treated with intravenous LEV.
Seizure cessation was achieved in 19 (86%) of cases within 1 hour of administration and they were discharged later on oral LEV.
In analysis of a systematic review of LEV as a first-line drug in neonatal seizures regarding its efficacy, it was found that neonates in the LEV arm achieved seizure cessation in 37/48 (77%). However, neonates the PB arm achieved seizure cessation in 24/52 (46%).
These results show the efficacy of LEV is the same as PB as a first-line drug in the control of neonatal seizures. 14 Ragunathan and Chandrasekhar, 24 studied 66 neonates with seizures who fulfilled the inclusion criteria and were treated either with PB or LEV as a first-line agent. They found that neonates in the LEV arm achieved seizure control in 64.7% compared to 31.2% in the PB arm (P = .05). Prolonged sedation was reported with PB, but no significant adverse effects were seen with LEV. They concluded that LEV is considered a good alternative as a first-line AED in neonatal seizures.
In a recent study that was conducted on 25 neonates monitored with video electroencephalography (EEG) comparing the efficacy of initial treatment with LEV vs. PB for maintaining low seizure burden, it was found that 11 of 17 (65%) patients sustained seizure burden 10% following initial treatment with LEV compared to 5 of 8 (63%) with PB. A 13 (76%) patients treated with LEV had a sustained seizure burden of 20% compared with 6 (75%) treated with PB. 25
Neonates who received PB showed more reduction in the seizure burden in the hours pre-and post-treatment (24.3 vs 14.2 minutes/hour). However, 6 of 17 (35%) neonates who received LEV remained seizure free after initial treatment, compared with 2 of 8 (25%) neonates who received PB. LEV therapy was associated with a shorter average time to seizure freedom (15 vs 21 hours). Although these results were statistically non-significant, LEV remains a promising first-line agent for neonatal seizures. 25
NEOLEV2 is the first RCT comparing the efficacy, dose-escalation, and adverse effects of LEV to PB as first line treatment in neonatal seizures detected by EEG. PB was found to be a better anticonvulsant in controlling electrographic seizures after analyzing clinical and electrographic cessations of seizures. 26 Although statistically non-significant, LEV had fewer adverse effects compared with PB. This study has the limitation that only neonates with cEEG confirmed seizures were included. However, neonates with clinical seizures of short duration that have terminated prior to the application of cEEG might be excluded. 26
Sharma et al 3 suggested a better safety of LEV compared with PB. Gowda et al 17 reported increased efficacy of LEV compared with PB in the management of clinical seizures. However, they did not account for electrographic-uncoupling. Contrary to our findings, LEV was associated with seizure cessation in 8 (35%) of 23 neonates. Poor response to LEV could be attributed to the use of LEV as a first-line AED in only 1 neonate in this study. 27 No serious adverse effects have been reported with the use of LEV in neonatal seizures.22,27,28 Also, the safety of LEV in neonates has been reported in previous studies.29,30
The limitation of our study is that it was a short-term study with a relatively small sample size. The objective was to obtain the prospective efficacy and safety of LEV in neonates. So, in our study it was acceptable to use seizure cessation as the primary endpoint. Taking into consideration that the main concern in neonatal seizure is the long-term neurodevelopmental sequel and outcome.
A drug that is less effective but leads to a better neurodevelopmental outcome may be the preferred treatment. In addition, the resources in our facility did not allow for EEG monitoring of every high-risk infant for seizures, this is a significant limitation of our study. Also, it is very likely that the infants in this study with clinical seizures continued to have ongoing electrographic-only seizures following treatment.
Lastly, the lack of therapeutic drug levels of PB and LEV. Despite these limitations, the generalizability of our results is reasonable.
In conclusion, we can conclude that LEV has been effective in the control of neonatal seizures. PB showed more adverse events. So, LEV is an effective, reliable, and safer alternative to PB as a first-line AED in neonatal seizures. Further studies on a larger sample of patients with long-term outcome measures are recommended.
Footnotes
Acknowledgements
All authors offer many thanks to all nurses and residents in NICU, Pediatric department, Benha University hospitals for their effort in this work.
Author’s Note
Naglaa M. Kamal is is now affiliated to Professor of Pediatrics and Pediatric Hepatology, Faculty of medicine, Cairo University, Cairo, Egypt.
Author Contributions
HHE conceived the study, NEA, HAS, AHA, AOI, NMK, KAN, AAA, AMA, MKE, OMA, and MGF participated in its design and coordination. NEA, HAS, AHA, AOI, NMK, MKE, and KAN provided key technical guidance. AAA, AMA, OMA, and MGF drafted the manuscript, and HHE, NEA, HAS, AHA, AOI, NMK, MKE, and KAN critically revised the manuscript for important intellectual content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
