Abstract
Infantile colic is the most frequent reason of infant and parental distress. An open-label single-group clinical study was conducted primarily to evaluate the effect of 14-day colic relief remedy administration (1.25 ml orally during colic episode) on average daily crying time in infants compared to baseline. In addition, the percentage of responders, sleep pattern, frequency and severity of gastrointestinal symptoms, tolerability of the product and percentage population with relapse of symptoms post product discontinuation were evaluated. Thirty infants aged 3 to 16 weeks with no significant clinical illness were enrolled in the study with their caregivers. Daily inconsolable crying time and flatulence were significantly reduced (P < .05) within a week of intervention and a sustained effect was observed after discontinuation of the product. The findings of the present study indicate that this product effectively reduced abdominal distension and pain, resulting in a significant decrease in the daily crying of the infants.
Introduction
Infant crying varies significantly with age. It is estimated that colic affects around 10% to 40% of infants globally. 1 Infantile colic is a paroxysmal, spontaneous, inconsolable, and unpredictable crying without an identifiable cause in an otherwise healthy newborn infant.2,3 Although infant colic usually resolves itself after the first 3 to 4 months after birth, it is associated with significant parental distress and if it persists beyond 3 months it can pose a risk of adverse developmental, emotional, and behavioral outcomes. 4
The systematic literature reviews have shown that to date no single effective treatment exists for infantile colic. 5 Unfortunately, the conventional treatments have shown contradictory results, and some of them have potential side effects, including breathing difficulties and coma.5,6 However, parental support, reassurance, education, and recently probiotics have shown some promise in alleviating infantile colic2,6 but the evidence still needs a more robust scientific approach to validate their efficacy. Hence, a practical, effective, and acceptable intervention is required for infantile colic.
The colic relief remedy (COLIC CALM®) investigated in the present study is a proprietary natural formula proposed to alleviate infantile colic. It contains activated charcoal as the major ingredient that has been reported to prevent flatus events and increase breath hydrogen after meals. 7 Its gastrointestinal benefits have been attributed to adsorbing ingested toxins within the gastrointestinal tract, thereby preventing the systemic absorption of such toxins. 8 Chamomile has anti-inflammatory and antispasmodic properties that make it an effective herb for flatulence. 9 Fennel has a mild carminative action and has been reported helpful in infantile colic and flatulence.10,11 Furthermore, Caraway has been proposed to improve digestive function, modulate intestinal flora, and alleviate flatulence and abdominal pain. 12 Prunus spinosa fruits are considered digestive and used to manage minor gastrointestinal complaints. 13 Ginger has well-established antipyretic, analgesic, anti-inflammatory, antispasmodic, sedative, and antibiotic effects, among other properties. 14 Based on the therapeutic potential of individual ingredients this clinical study was aimed to determine whether colic relief remedy benefits infants aged 3 to 16 weeks with colic by reducing the daily duration of infant crying due to colic.
Methods
Study Design
The present study was an open-label single-group clinical study conducted between March 2021 and August 2021 at 4 pediatric clinics in Maharashtra, India. The primary objective of the study was to evaluate the effect of colic relief remedy administration on average daily crying time in infants at day 7 and day 14 from baseline (day 0). The secondary objectives were to evaluate the effect of colic relief remedy administration on the percentage of responders, sleeping pattern, frequency and severity of gastrointestinal symptoms, tolerability of the investigational product (IP) related to gastric symptoms, and percentage of the population with relapse of colic post IP discontinuation.
Participants
Thirty-two healthy full-term 3 to 16 weeks aged infants weighing 2500 to 4000 g and their caregivers were screened for the eligibility criteria as provided in Table 1. All the mothers/caregivers who were found eligible and gave written informed consent to participate in the study were recruited.
Eligibility Criteria for Participation in the Study.

Intervention
The colic relief remedy used in the present study is a completely natural alternative for gripe water comprising Carbo vegetabilis (vegetable charcoal), Prunus spinosa (Blackthorne), Carum carvi (Caraway), Matricaria chamomilla (Chamomile), Foeniculum vulgare (Fennel), Zingiber officinale (Ginger), Melissa officinalis (Lemon Balm), and Mentha piperita (Peppermint). It is non-habit-forming, sugarless, and devoid of any chemicals or artificial flavors and colors. In a previously unpublished pilot study, the efficacy and safety of the product at a dose of 1.25 mg orally were investigated in infantile colic and it was found safe in infants. Based on this preliminary evidence, all the recruited participants were administered colic relief remedy at the same dose on inconsolable crying which was to be repeated once every 120 minutes, if required. It was recommended not to exceed 6 doses in 24 hours. The product was dispensed in a 60 ml bottle along with a graduated syringe for easy administration of the product.
Study Procedures
The infants and their caregivers had weekly assessments till day 21. Data collection was performed virtually on day 7 and day 21 while the day 14 visit was conducted at the clinics. The caregivers were provided with questionnaires and a diary to record their responses regarding the infant’s behavior during the study.
Outcomes
Daily crying time
The primary outcome of the study was the effect of colic relief remedy consumption on the reduction in average crying time of infants on day 7 and day 14 from baseline, as evaluated by modified Barr’s diary. For the present study, the original Barr’s diary 15 was modified. It consisted of a series of time rulers to be marked by caregivers for 14 consecutive days. Each ruler reflects 6 hours, with the smallest division on each ruler being 5 minutes. Within each ruler, parents were required to mark the appropriate time for the infant’s behavior specifically crying and sleeping. Parents/caregivers were also encouraged to express their views on the infant’s behavior for a particular day, whether it was typical or atypical with reason. The diary also captured the duration for which the infant was awake fussy, type of feeding, feeding frequency, and feeding duration.
Responder analysis
In the present study, responders were defined as infants having a reduction of ≥50% in their average daily crying time compared to the baseline. The percentage population of responders and non-responders was reported at the end of the study.
Sleep pattern
Sleep pattern varies considerably throughout infancy. 16 Several studies have associated colicky symptoms with lesser sleep duration. 5 Therefore, the present study evaluated the effect of the study product on sleep duration. The sleep duration was captured in the daily modified Barr’s diary and the average sleep duration was calculated for the first week and the second week consecutively.
Gastrointestinal symptoms
Recent research has suggested that change in gut microbiota results in excessive gas production and hypermotility of the gastrointestinal tract leading to infantile colic.17,18 For evaluating the frequency and severity of gastrointestinal symptoms in colicky infants on day 7 and day 14 a validated IGSQ was used. The IGSQ is a short, 13-item measure of gastrointestinal symptoms in infants that allows parents to describe the frequency and intensity of their infant’s gastrointestinal signs and symptoms for the previous 7 days. 19 Higher scores on IGSQ indicate more gastrointestinal symptoms.20,21
Percentage population with relapse of symptoms
For assessing the sustained effect of the product, the relapse of colic in infants during the post-intervention period of 7 days was evaluated for the present study. Relapse was defined as any episode of colic (as per Rome IV criteria) post IP discontinuation. In the case where relapse occurred, the investigator advised further treatment to the caregivers.
Safety
The tolerability of the study product was evaluated using a 4-point verbal rating scale (very good, good, satisfactory, unsatisfactory). 22 A higher score depicts better tolerability. The tolerability scale was administered on day 14. Also, a diary was dispensed to record the gastrointestinal symptoms that occurred during the study.
Statistical Analysis
A minimum sample size of 30 participants was considered appropriate for this study as this is a single group first-ever clinical study of the product. All the continuous variables were compared to their baseline value on day 7 and day 14 of the intervention by using paired t-test. Categorical variables such as the percentage of responders and participants having relapse were compared using the Chi-square test. The P-value of .05 was considered significant.
Results
Total of 32 participants were screened for the study, out of which 30 were found eligible and completed the study. The enrolled infants were predominantly males with an average age of almost 10 weeks. The other demographic and baseline characteristics are provided in Table 2.
Summary of Demographic and Baseline Characteristics.

Abbreviation: N, number of participants.
Values are presented as mean ± SD (standard deviation) or number of participants (%).
Percentages were calculated using the respective column header count as the denominator.
Outcomes
Daily crying time
The study results showed that the average inconsolable cry time was reduced to >50% of the baseline after 1 week of colic relief remedy administration which further improved (56.34%) during the second week which was clinically and statistically significant (P < .0001) at both the time points (Table 3). This shows that the colic relief remedy effectively reduced the daily crying time after a week of administration which further improved by the end of week 2.
Inconsolable Crying Time Per Day (Minutes/Day).

∆, absolute change calculated as baseline value– day7/14 value. P-value was calculated using paired t-test.
Abbreviations: N, number of participants; SD, standard deviation.
Responder analysis
Responders were defined as the number of participants with a reduction of ≥50% in average inconsolable crying time from baseline after IP administration. By day 7, the majority (N = 22, 73.33%) of the participants responded to colic relief remedy, which further increased to 80% at the end of the intervention period.
Sleep pattern
Sleep pattern was assessed using the modified Barr’s diary. A positive trend was noticed in the sleep duration however it was not statistically significant compared to the baseline sleep duration (P-value > .05) (Table 4).
Average Sleep Duration Per Day (Minutes/Day).

∆, absolute change calculated as baseline value– day7/14 value. P-value was calculated using paired t-test.
Abbreviations: N, number of participants; SD, standard deviation.
Gastrointestinal symptoms
The frequency and severity of gastrointestinal symptoms were assessed by evaluating changes in 13-item IGSQ total and item-wise scores on day 7 and day 14. No statistically significant change was observed in total and the item-wise scores except for the amount of crying time per day and the number of times the baby passed a lot of gas (P < .05) (Table 5) which indicates that colic relief remedy might have reduced the amount of crying time per day by alleviating abdominal distension.
Infant Gastrointestinal Symptom Questionnaire (IGSQ) Score.
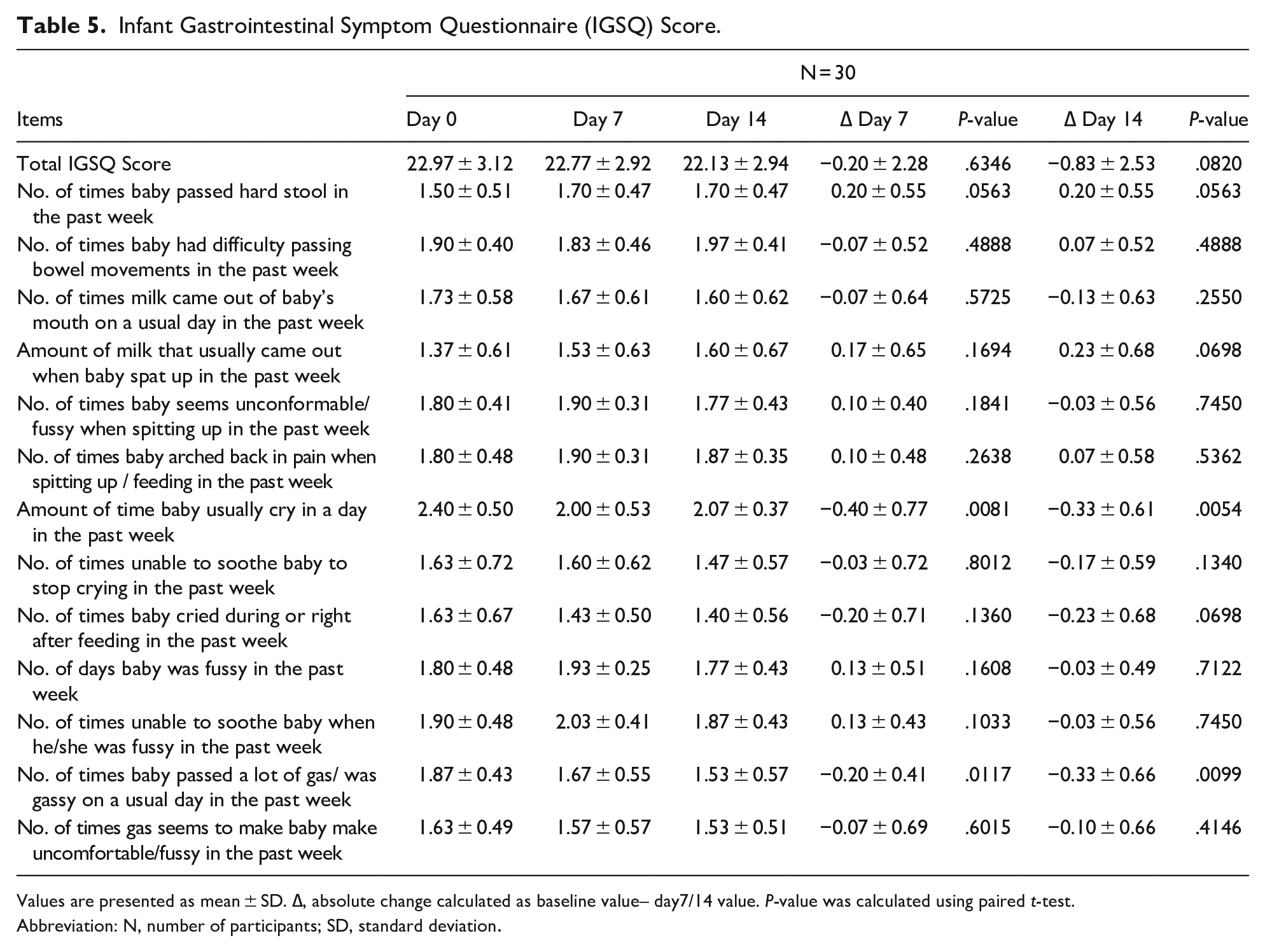
Values are presented as mean ± SD. ∆, absolute change calculated as baseline value– day7/14 value. P-value was calculated using paired t-test.
Abbreviation: N, number of participants; SD, standard deviation.
Percentage population with relapse of symptoms
Only 12 (40%) infants had a relapse of colic symptoms during the 7-day follow-up after stopping the intervention.
Safety
The majority of caregivers rated “good tolerability” (mean score = 3) for colic relief remedy. No significant change in any vital signs was observed during the study. Only one adverse event (fever, N = 1) was reported during the study which was resolved without any consequences.
Discussion
In the present study, the colic relief remedy effectively reduced the daily crying time to a significant extent in infants with colic. In addition, most of the infants responded to the colic relief remedy within the first week of its administration. There was a significant decrease in the flatulence, as indicated by a decrease in the number of times the baby passed a lot of gas/was gassy on a usual day reported in IGSQ. The effect of the colic relief remedy on gastrointestinal symptoms lasted for 7 days after stopping its administration. The majority of the infants did not report any relapse during the follow-up period. In addition, it exhibited an excellent tolerability index as reported by the caregivers of the infants. However, it could not improve sleep duration and other gastrointestinal symptoms. These findings suggest that the colic relief remedy was able to reduce excessive flatus formation and hence abdominal distension and pain, resulting in a significant decrease in daily crying of the infants.
Several over-the-counter medications are available for relieving the symptoms of infantile colic. The use of conventional medications is either controversial or prohibited in infants less than 6 months of age due to serious side effects. The efficacy of the colic relief remedy demonstrated in the present study is equivalent to or better than some of the probiotics23-25 and dietary interventions. 26 A recently conducted clinical study on a synbiotic preparation compared to Simethicone reported 85% versus 39% responders at day 21 respectively for average crying time per day. 27 Colic relief remedy achieved similar results (80%) to that of synbiotic and Simethicone in 2 weeks. In another study, Simethicone showed a 43.2% and 53.5% of reduction in average crying time per day after 2 weeks and 3 weeks of treatment respectively which is lower than the reduction observed in the present study (56.34%) at the same time point. 28 The mechanism of action of colic relief remedy can be explained based on the activities of its ingredients on gastrointestinal disorders. The fennel oil emulsion successfully alleviated infantile colic defined by Wessel criteria in 65% of the enrolled infants which was significantly better than the placebo (P < .01). 29 Arikan et al 30 reported a significant effect of Fennel tea on daily crying time in infants (P < .001) after administration for 1 week. A proprietary formula containing Chamomile, Fennel, and Lemon Balm demonstrated statistically better results in daily crying time and percentage of responders compared to the control group. 31 Another study observed that herbal tea containing Fennel, Chamomile, Liquorice, Vervain, and Lemon Balm effectively reduced colic in 57% of infants compared to placebo (26%) (P < .01). 32 A double-blind crossover study on 30 infants with colic showed the promising effect of Peppermint in relieving the symptoms compared to Simethicone. 33 The present study is a piece of additional evidence supporting the synergistic and safe use of all these ingredients in infantile colic.
Conclusion
This is the pioneer study that demonstrated a highly significant effect of colic relief remedy with a good tolerability index in reducing the daily inconsolable crying time of infants when administered for 14 days.
Footnotes
Acknowledgements
The authors are thankful to all the participants who volunteered for the study and the research team who executed the study. They are grateful to Calm Co., USA for providing the study products.
Author Contributions
Both the authors were involved in the conceptualization of the study. They have reviewed and edited the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the CalmCo., Sarasota, Florida, USA.
Availability of Data and Materials
The data analyzed in the study are not publicly available due to a confidentiality agreement with the sponsor. Data are however available from the authors upon reasonable request and with permission of the sponsor.
Compliance With Ethical Standards
The study was conducted in compliance with the Declaration of Helsinki (Ethical Principles for Medical Research Involving Human Subjects, revised by the WMA General Assembly, Seoul 2008), International Conference on Harmonization (ICH) recommendation on Good Clinical Practice (GCP) − 2016, and Ethical guidelines for biomedical research on human participants 2006, issued by Indian Council of Medical Research (ICMR).
Ethics Approval and Consent Form
The study was approved and monitored by “ACEAS – Institutional Ethics Committee,” India (Reg. No. ECR/281/Indt/GJ/2017/RR-21) to safeguard the rights, safety, and well-being of all trial participants. Only participants who gave written informed consent forms were included in the study.
Consent to Participate
Written informed consent was obtained from the mothers or caretakers of the infants included in the study.
